Submitted:
22 February 2023
Posted:
22 February 2023
Read the latest preprint version here
Abstract
Keywords:
Introduction
Systemic extravasation of bioengineered carrier macrophages:
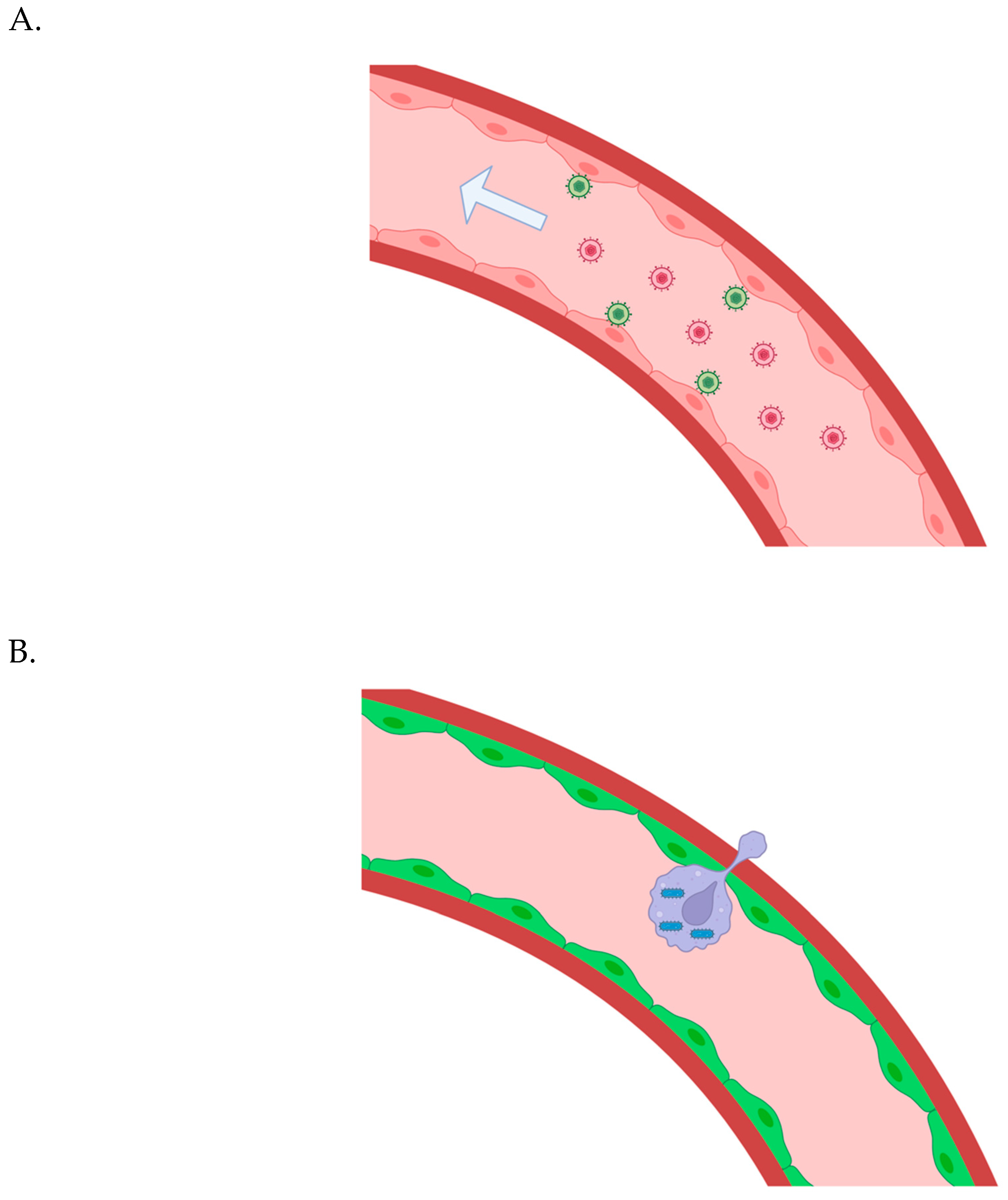
Two remaining issues in the near future:
Anti-aging gene therapy:
Senescent cells:
Cancer:
Conclusion:
Funding
Authors' contributions
Ethics approval and consent to participate
Consent for publication
Availability of data and material
Acknowledgments
Competing interests
References
- Terman A and Brunk UT. Lipofuscin. The International Journal of Biochemistry & Cell Biology 2004;36(8):1400–1404. [CrossRef]
- Zealley B and de Grey ADNJ. Strategies for Engineered Negligible Senescence. Gerontology 2013;59(2):183–189. [CrossRef]
- López-Otín C, Blasco MA, Partridge L, et al. The Hallmarks of Aging. Cell 2013;153(6):1194–1217. [CrossRef]
- Aufschnaiter R, Zamir EA, Little CD, et al. In vivo imaging of basement membrane movement: ECM patterning shapes Hydra polyps. Journal of Cell Science 2011;124(23):4027–4038. [CrossRef]
- Birch J, Gil J. Senescence and the SASP: many therapeutic avenues. Genes Dev 2020;34(23–24):1565–1576. [CrossRef]
- Kabacik S, Lowe D, Fransen L, et al. The Relationship between Epigenetic Age and the Hallmarks of Aging in Human Cells. Nat Aging 2022;2(6):484–493. [CrossRef]
- Lu Y, Brommer B, Tian X, et al. Reprogramming to recover youthful epigenetic information and restore vision. Nature 2020;588(7836):124–129. [CrossRef]
- Wang S, Xia P, Ye B, et al. Transient activation of autophagy via Sox2-mediated suppression of mTOR is an important early step in reprogramming to pluripotency. Cell Stem Cell 2013;13(5):617–625. [CrossRef]
- Kang Y-K, Min B, Eom J, et al. Different Phases of Aging in Mouse Old Skeletal Muscle. Aging 2022;14(1):143–160. [CrossRef]
- Renteln M. Lipofuscin as the main driving force of current age-related disease: justification and strategies for removal. Molecular Biology Reports (Under Review) Preprint →. [CrossRef]
- Johnson M, Karanikolas BDW, Priceman SJ, et al. Titration of Variant HSV1-tk Gene Expression to Determine the Sensitivity of 18F-FHBG PET Imaging in a Prostate Tumor. J Nucl Med 2009;50(5):757–764. [CrossRef]
- Nikitas G, Deschamps C, Disson O, et al. Transcytosis of Listeria monocytogenes across the intestinal barrier upon specific targeting of goblet cell accessible E-cadherin. J Exp Med 2011;208(11):2263–2277. [CrossRef]
- Le Guennec L, Coureuil M, Nassif X, et al. Strategies used by bacterial pathogens to cross the blood–brain barrier. Cellular Microbiology 2020;22(1):e13132. [CrossRef]
- Ley K, Laudanna C, Cybulsky MI, et al. Getting to the site of inflammation: the leukocyte adhesion cascade updated. Nat Rev Immunol 2007;7(9):678–689. [CrossRef]
- Chi J-T, Chang HY, Haraldsen G, Jahnsen FL, Troyanskaya OG, Chang DS, Wang Z, Rockson SG, van de Rijn M, Botstein D, et al. Endothelial cell diversity revealed by global expression profiling. Proceedings of the National Academy of Sciences (2003) 100:10623–10628. [CrossRef]
- van Buul JD, Hordijk PL. Signaling in Leukocyte Transendothelial Migration. Arteriosclerosis, Thrombosis, and Vascular Biology (2004) 24:824–833. [CrossRef]
- Gupta M, Shin D-M, Ramakrishna L, et al. IRF8 directs stress-induced autophagy in macrophages and promotes clearance of Listeria monocytogenes. Nature Communications 2015;6:6379. [CrossRef]
- Miller ST, Xavier KB, Campagna SR, Taga ME, Semmelhack MF, Bassler BL, Hughson FM. Salmonella typhimurium recognizes a chemically distinct form of the bacterial quorum-sensing signal AI-2. Mol Cell (2004) 15:677–687. [CrossRef]
- Lipinski DM, Reid CA, Boye SL, et al. Systemic Vascular Transduction by Capsid Mutant Adeno-Associated Virus After Intravenous Injection. Hum Gene Ther 2015;26(11):767–776. [CrossRef]
- Krolak T, Chan KY, Kaplan L, et al. A High-Efficiency AAV for Endothelial Cell Transduction throughout the Central Nervous System. Nat Cardiovasc Res 2022;1(4):389–400. [CrossRef]
- Lu ZH, Kaliberov S, Zhang J, et al. The Myeloid-Binding Peptide Adenoviral Vector Enables Multi-Organ Vascular Endothelial Gene Targeting. Lab Invest 2014;94(8):881–892. [CrossRef]
- Zahr A, Alcaide P, Yang J, et al. Endomucin prevents leukocyte–endothelial cell adhesion and has a critical role under resting and inflammatory conditions. Nat Commun 2016;7(1):10363. [CrossRef]
- Qiu Y, Tong S, Zhang L, et al. Magnetic forces enable controlled drug delivery by disrupting endothelial cell-cell junctions. Nat Commun 2017;8(1):15594. [CrossRef]
- Zhang G, Yang X, Gao R. Research progress on the structure and function of endomucin. Animal Model Exp Med 2021;3(4):325–329. [CrossRef]
- Hunter M, Fusco D. Superinfection exclusion: A viral strategy with short-term benefits and long-term drawbacks. PLoS Comput Biol 2022;18(5):e1010125. [CrossRef]
- Caussinus E, Kanca O, Affolter M. Fluorescent fusion protein knockout mediated by anti-GFP nanobody. Nature Structural & Molecular Biology (2012) 19:117–121. [CrossRef]
- Horan KA, Hansen K, Jakobsen MR, Holm CK, Søby S, Unterholzner L, Thompson M, West JA, Iversen MB, Rasmussen SB, et al. Proteasomal degradation of herpes simplex virus capsids in macrophages releases DNA to the cytosol for recognition by DNA sensors. J Immunol (2013) 190:2311–2319. [CrossRef]
- Acar M, Pando BF, Arnold FH, Elowitz MB, van Oudenaarden A. A general mechanism for network-dosage compensation in gene circuits. Science (2010) 329:1656–1660. [CrossRef]
- Montezano, Augusto C., Karla B. Neves, Rheure A. M. Lopes, and Francisco Rios. “Isolation and Culture of Endothelial Cells from Large Vessels.” In Hypertension: Methods and Protocols, edited by Rhian M. Touyz and Ernesto L. Schiffrin, 345–48. Methods in Molecular Biology. New York, NY: Springer, 2017. [CrossRef]
- Graham GJ, Handel TM, Proudfoot AEI. Leukocyte Adhesion: Reconceptualizing Chemokine Presentation by Glycosaminoglycans. Trends Immunol 2019;40(6):472–481. [CrossRef]
- Stanley SA, Gagner JE, Damanpour S, et al. Radio-Wave Heating of Iron Oxide Nanoparticles Can Regulate Plasma Glucose in Mice. Science 2012;336(6081):604–608. [CrossRef]
- Guidi N, Marka G, Sakk V, et al. An Aged Bone Marrow Niche Restrains Rejuvenated Hematopoietic Stem Cells. STEM CELLS 2021;39(8):1101–1106. [CrossRef]
- Landspersky T, Saçma M, Rivière J, et al. Autophagy in Mesenchymal Progenitors Protects Mice against Bone Marrow Failure after Severe Intermittent Stress. Blood 2022;139(5):690–703. [CrossRef]
- Zhang X, Chen W, Gao Q, et al. Rapamycin Directly Activates Lysosomal Mucolipin TRP Channels Independent of MTOR. PLOS Biology 2019;17(5):e3000252. [CrossRef]
- Ain Q, Schmeer C, Penndorf D, et al. Cell cycle-dependent and -independent telomere shortening accompanies murine brain aging. Aging (Albany NY) 2018;10(11):3397–3420. [CrossRef]
- Tomás-Loba A, Flores I, Fernández-Marcos PJ, et al. Telomerase Reverse Transcriptase Delays Aging in Cancer-Resistant Mice. Cell 2008;135(4):609–622. [CrossRef]
- Bernardes de Jesus B, Vera E, Schneeberger K, et al. Telomerase Gene Therapy in Adult and Old Mice Delays Aging and Increases Longevity without Increasing Cancer. EMBO Mol Med 2012;4(8):691–704. [CrossRef]
- Jaijyan DK, Selariu A, Cruz-Cosme R, et al. New Intranasal and Injectable Gene Therapy for Healthy Life Extension. Proc Natl Acad Sci U S A 2022;119(20):e2121499119. [CrossRef]
- Sewell PE. Systemic Human Htert Aav Gene Transfer Therapy And The Effect On Telomere Length And Biological Age, A Case Report. J Regen Biol Med 2022. [CrossRef]
- Ali M, Devkota S, Roh J-I, et al. Telomerase Reverse Transcriptase Induces Basal and Amino Acid Starvation-Induced Autophagy through MTORC1. Biochemical and Biophysical Research Communications 2016;478(3):1198–1204. [CrossRef]
- Yang C, Przyborski S, Cooke MJ, et al. A Key Role for Telomerase Reverse Transcriptase Unit in Modulating Human Embryonic Stem Cell Proliferation, Cell Cycle Dynamics, and In Vitro Differentiation. Stem Cells 2008;26(4):850–863. [CrossRef]
- Zincarelli C, Soltys S, Rengo G, et al. Analysis of AAV Serotypes 1-9 Mediated Gene Expression and Tropism in Mice after Systemic Injection. Mol Ther 2008;16(6):1073–1080. [CrossRef]
- Gray SJ, Matagne V, Bachaboina L, Yadav S, Ojeda SR, Samulski RJ. Preclinical differences of intravascular AAV9 delivery to neurons and glia: a comparative study of adult mice and nonhuman primates. Mol Ther (2011) 19:1058–1069. [CrossRef]
- Bevan AK, Duque S, Foust KD, Morales PR, Braun L, Schmelzer L, Chan CM, McCrate M, Chicoine LG, Coley BD, et al. Systemic gene delivery in large species for targeting spinal cord, brain, and peripheral tissues for pediatric disorders. Mol Ther (2011) 19:1971–1980. [CrossRef]
- Hordeaux J, Wang Q, Katz N, Buza EL, Bell P, Wilson JM. The Neurotropic Properties of AAV-PHP.B Are Limited to C57BL/6J Mice. Mol Ther (2018) 26:664–668. [CrossRef]
- Cagan A, Baez-Ortega A, Brzozowska N, et al. Somatic Mutation Rates Scale with Lifespan across Mammals. Nature 2022;604(7906):517–524. [CrossRef]
- Chen X, Burkhardt DB, Hartman AA, et al. MLL-AF9 Initiates Transformation from Fast-Proliferating Myeloid Progenitors. Nat Commun 2019;10(1):5767. [CrossRef]
- Se A, S A, Mk T, et al. Constitutive Telomerase Expression Promotes Mammary Carcinomas in Aging Mice. Proceedings of the National Academy of Sciences of the United States of America 2002;99(12). [CrossRef]
- Weng N. Aging of the Immune System: How Much Can the Adaptive Immune System Adapt? Immunity 2006;24(5):495–499. [CrossRef]
- Torra A, Parent A, Cuadros T, et al. Overexpression of TFEB Drives a Pleiotropic Neurotrophic Effect and Prevents Parkinson’s Disease-Related Neurodegeneration. Mol Ther 2018;26(6):1552–1567. [CrossRef]
- Bredenkamp N, Nowell CS, Blackburn CC. Regeneration of the aged thymus by a single transcription factor. Development 2014;141(8):1627–1637. [CrossRef]
- Zook EC, Krishack PA, Zhang S, et al. Overexpression of Foxn1 attenuates age-associated thymic involution and prevents the expansion of peripheral CD4 memory T cells. Blood 2011;118(22):5723–5731. [CrossRef]
- Uddin MM, Ohigashi I, Motosugi R, et al. Foxn1-β5t transcriptional axis controls CD8+ T-cell production in the thymus. Nat Commun 2017;8(1):14419. [CrossRef]
- Baker DJ, Childs BG, Durik M, et al. Naturally occurring p16Ink4a-positive cells shorten healthy lifespan. Nature 2016;530(7589):184–189. [CrossRef]
- Renteln M. Conditional Replication of Oncolytic Viruses Based on Detection of Oncogenic MRNA. Gene Therapy 2018;25(1):1–3. [CrossRef]
- Renteln M. Promoting oncolytic vector replication with switches that detect ubiquitous mutations. Current Cancer Therapy Reviews (Under Review) Preprint. [CrossRef]
- Muthana M, Kennerley AJ, Hughes R, et al. Directing cell therapy to anatomic target sites in vivo with magnetic resonance targeting. Nature Communications 2015;6(1):8009. [CrossRef]
- Cornel AM, Mimpen IL, Nierkens S. MHC Class I Downregulation in Cancer: Underlying Mechanisms and Potential Targets for Cancer Immunotherapy. Cancers (Basel) 2020;12(7):E1760. [CrossRef]
- Park JH, Rivière I, Gonen M, et al. Long-Term Follow-up of CD19 CAR Therapy in Acute Lymphoblastic Leukemia. N Engl J Med 2018;378(5):449–459. [CrossRef]
- Cercek A, Lumish M, Sinopoli J, et al. PD-1 Blockade in Mismatch Repair–Deficient, Locally Advanced Rectal Cancer. New England Journal of Medicine 2022;0(0):null. [CrossRef]
- Stritzker J, Hill PJ, Gentschev I, et al. Myristoylation negative msbB-mutants of probiotic E. coli Nissle 1917 retain tumor specific colonization properties but show less side effects in immunocompetent mice. Bioeng Bugs 2010;1(2):139–145. [CrossRef]
- Harimoto T, Hahn J, Chen Y-Y, et al. A Programmable Encapsulation System Improves Delivery of Therapeutic Bacteria in Mice. Nat Biotechnol 2022;40(8):1259–1269. [CrossRef]
- García-Cao I, García-Cao M, Martín-Caballero J, et al. ‘Super P53’ Mice Exhibit Enhanced DNA Damage Response, Are Tumor Resistant and Age Normally. EMBO J 2002;21(22):6225–6235. [CrossRef]
- Zhao Y, Burikhanov R, Qiu S, et al. Cancer Resistance in Transgenic Mice Expressing the SAC Module of Par-4. Cancer Res 2007;67(19):9276–9285. [CrossRef]
- Garcia-Cao I, Song MS, Hobbs RM, et al. Systemic Elevation of PTEN Induces a Tumor-Suppressive Metabolic State. Cell 2012;149(1):49–62. [CrossRef]
- Grey ADNJ de. Whole-Body Interdiction of Lengthening of Telomeres: A Proposal for Cancer Prevention. Front Biosci 2005;10(1–3):2420. [CrossRef]
- MacKenzie D, Watters AK, To JT, et al. ALT Positivity in Human Cancers: Prevalence and Clinical Insights. Cancers (Basel) 2021;13(10):2384. [CrossRef]
- Streeter MD, Rowan S, Ray J, et al. Generation and Characterization of Anti-Glucosepane Antibodies Enabling Direct Detection of Glucosepane in Retinal Tissue. ACS Chem Biol 2020;15(10):2655–2661. [CrossRef]
- Furber JD. Extracellular glycation crosslinks: prospects for removal. Rejuvenation Res 2006;9(2):274–278. [CrossRef]
- Planque SA, Nishiyama Y, Hara M, et al. Physiological IgM class catalytic antibodies selective for transthyretin amyloid. J Biol Chem 2014;289(19):13243–13258. [CrossRef]
- Bruxel MA, Tavares AMV, Zavarize Neto LD, et al. Chronic whole-body heat treatment relieves atherosclerotic lesions, cardiovascular and metabolic abnormalities, and enhances survival time restoring the anti-inflammatory and anti-senescent heat shock response in mice. Biochimie 2019;156:33–46. [CrossRef]
- Hansen T, Ahlström H, Johansson L. Whole-body screening of atherosclerosis with magnetic resonance angiography. Top Magn Reson Imaging 2007;18(5):329–337. [CrossRef]
- Uebe R, Schüler D. Magnetosome biogenesis in magnetotactic bacteria. Nat Rev Microbiol 2016;14(10):621–637. [CrossRef]
- Shiratori T, Suzuki S, Kakizawa Y, Ishida K. Phagocytosis-like cell engulfment by a planctomycete bacterium. Nat Commun (2019) 10:5529. [CrossRef]
- Marques ARA, Ramos C, Machado-Oliveira G, et al. Lysosome (Dys)Function in Atherosclerosis—A Big Weight on the Shoulders of a Small Organelle. Frontiers in Cell and Developmental Biology 2021;9:658995. [CrossRef]
- Cheng C-W, Adams GB, Perin L, et al. Prolonged Fasting reduces IGF-1/PKA to promote hematopoietic stem cell-based regeneration and reverse immunosuppression. Cell Stem Cell 2014;14(6):810–823. [CrossRef]
- Best BP. Nuclear DNA Damage as a Direct Cause of Aging. Rejuvenation Research 2009;12(3):199–208. [CrossRef]
- Freitas Jr. RA. Progress in Nanomedicine and Medical Nanorobotics. In: Handbook of Theoretical and Computational Nanotechnology. (Rieth M, Schommers W. eds.) American Scientific Publishers: Stevenson Ranch, Calif; 2005.
- Anonymous. Nebula Genomics - DNA Testing. n.d. Available from: https://nebula.org/whole-genome-sequencing-dna-test/ [Last accessed: 11/16/2022].
- Stewart JB, Chinnery PF. Extreme heterogeneity of human mitochondrial DNA from organelles to populations. Nat Rev Genet 2021;22(2):106–118. [CrossRef]
- Yoon YG, Koob MD. Transformation of isolated mammalian mitochondria by bacterial conjugation. Nucleic Acids Res 2005;33(16):e139. [CrossRef]
- Yoon YG, Koob MD. Nonreplicating intracellular bacterial vector for conjugative DNA transfer into mitochondria. Pharm Res 2012;29(4):1040–1045. [CrossRef]
- Latorre-Pellicer A, Moreno-Loshuertos R, Lechuga-Vieco AV, et al. Mitochondrial and nuclear DNA matching shapes metabolism and healthy ageing. Nature 2016;535(7613):561–565. [CrossRef]
- Hirose M, Schilf P, Gupta Y, et al. Low-level mitochondrial heteroplasmy modulates DNA replication, glucose metabolism and lifespan in mice. Sci Rep 2018;8(1):5872. [CrossRef]
- Lechuga-Vieco AV, Latorre-Pellicer A, Calvo E, et al. Heteroplasmy of Wild-Type Mitochondrial DNA Variants in Mice Causes Metabolic Heart Disease With Pulmonary Hypertension and Frailty. Circulation 2022;145(14):1084–1101. [CrossRef]
- Keogh M, Chinnery PF. Hereditary mtDNA Heteroplasmy: A Baseline for Aging? Cell Metabolism 2013;18(4):463–464. [CrossRef]
- Shokolenko IN, Wilson GL, Alexeyev MF. Aging: A mitochondrial DNA perspective, critical analysis and an update. World J Exp Med 2014;4(4):46–57.
- Barroca V, Fouchet P. Germline Stem Cells: The First Guards of Heredity. Cell Stem Cell 2008;2(2):108–110. [CrossRef]
- Silvestris E, Cafforio P, Felici C, et al. Ddx4+ Oogonial Stem Cells in Postmenopausal Women’s Ovaries: A Controversial, Undefined Role. Cells 2019;8(7):650. [CrossRef]
- Stringer JM, Winship A, Zerafa N, et al. Oocytes can efficiently repair DNA double-strand breaks to restore genetic integrity and protect offspring health. Proceedings of the National Academy of Sciences 2020;117(21):11513–11522. [CrossRef]
- St. John JC. Mitochondria and Female Germline Stem Cells—A Mitochondrial DNA Perspective. Cells 2019;8(8):852. [CrossRef]
- Franco I, Johansson A, Olsson K, et al. Somatic mutagenesis in satellite cells associates with human skeletal muscle aging. Nat Commun 2018;9(1):800. [CrossRef]
- Abascal F, Harvey LMR, Mitchell E, et al. Somatic mutation landscapes at single-molecule resolution. Nature 2021;593(7859):405–410. [CrossRef]
- Blokzijl F, Ligt J de, Jager M, et al. Tissue-specific mutation accumulation in human adult stem cells during life. Nature 2016;538(7624):260. [CrossRef]
- Manders F, van Boxtel R, Middelkamp S. The Dynamics of Somatic Mutagenesis During Life in Humans. Frontiers in Aging 2021;2.
- Cagan A, Baez-Ortega A, Brzozowska N, et al. Somatic mutation rates scale with lifespan across mammals. Nature 2022;604(7906):517–524. [CrossRef]
- Han X, Chen M, Wang F, et al. Forebrain engraftment by human glial progenitor cells enhances synaptic plasticity and learning in adult mice. Cell Stem Cell 2013;12(3):342–353. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



