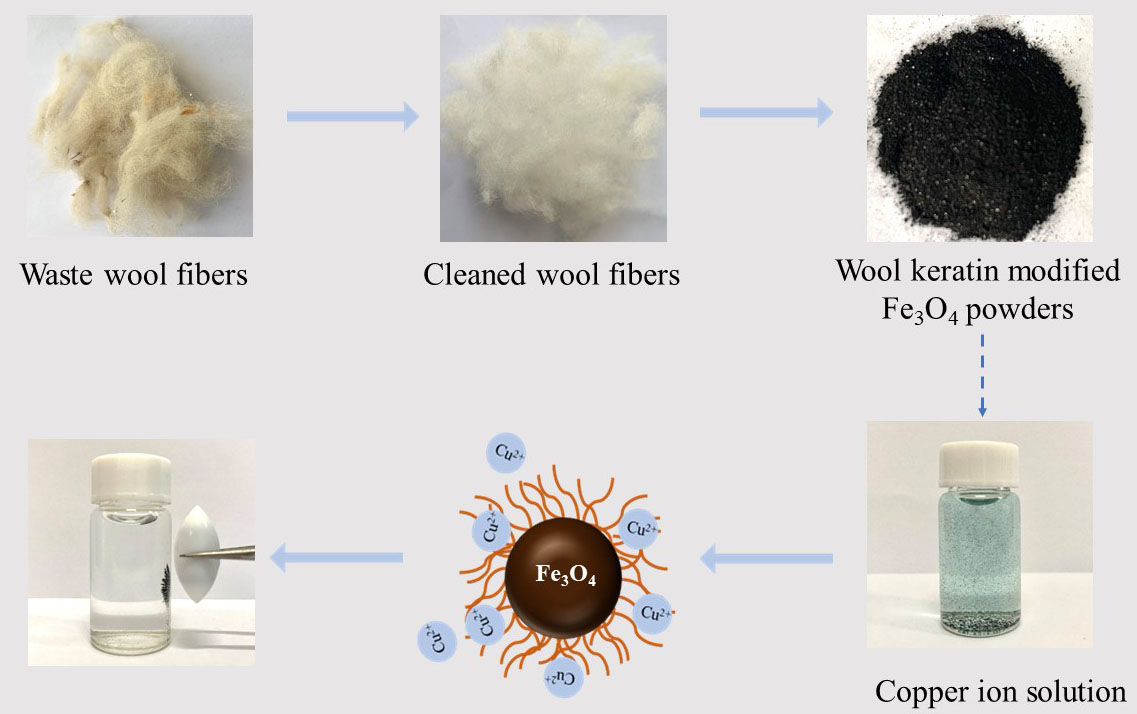
The treatment of wastewater containing heavy metals and the utilization of wool waste are very important for the sustainable development of textile mills. In this study, the wool keratin modified magnetite (Fe3O4) powders were fabricated by using wool waste via a co-precipitation technique for removal of Cu2+ ions from aqueous solutions. The morphology, chemical compositions, crystal structure, microstructure, magnetism properties, organic content, and specific surface area of as-fabricated powders were systematically characterized by various techniques including field emission scanning electron microscopy (FESEM), energy dispersive spectroscopy (EDS), X-ray diffraction (XRD), transmission electron microscopy (TEM), X-ray photoelectron spectroscopy (XPS), vibrating sample magnetometer (VSM), thermogravimetric (TG) analysis, and BET surface area analyzer. The effects of experimental parameters such as the volume of wool keratin hydrolysate, the dosage of powder, the initial Cu2+ ion concentration, and the pH value of solution on the adsorption capacity of Cu2+ ions by the powders were examined. The experimental results indicated that the Cu2+ ion adsorption performance of the wool keratin modified Fe3O4 powders exhibited much better than that of the chitosan modified ones with a maximum Cu2+ adsorption capacity of 29.2 mg/g under favorable conditions (0.05 g powders; 50 mL of 40 mg/L CuSO4; pH 5; temperature 293 K). The high adsorption capacity towards Cu2+ ions on the wool keratin modified Fe3O4 powders was primarily because of the strong surface complexation of -COOH and -NH2 functional groups of wool keratins with Cu2+ ions. The Cu2+ ion adsorption process on the wool keratin modified Fe3O4 powders followed the Langmuir adsorption isotherm model and the pseudo-second-order adsorption kinetic model. After Cu2+ ion removal, the wool keratin modified Fe3O4 powders were easily separated using a magnet from aqueous solution and efficiently regenerated using 0.5 M EDTA-H2SO4 eluting. The wool keratin modified Fe3O4 powders possessed good regenerative performance after five cycles. This study provided a feasible way to utilize waste wool textiles for preparing magnetic biomass-based adsorbents for the removal of heavy metal ions from aqueous solutions.