This image suggests that the synergy of three mechanisms dictates chemical, biological, and social evolution. The three mechanisms include natural selection and two other enigmatic mechanisms revealed by this article.
Why did simple carbon-based materials (CBMs) on Earth evolve into complex, orderly, and diverse organisms and social organizations? Extensive research in physics, chemistry, and biology has yet to explicitly and comprehensively answer this fundamental question in science [
1,
2,
3,
4,
5,
6,
7,
8,
9,
10,
11,
12,
13,
14,
15]. For instance, physics’ answers are often inexplicit because they mainly rely on certain elusive and abstract concepts associated with entropy, such as negative entropy, the theory of dissipative structures, and the maximum entropy hypothesis [
2,
3,
4]. Chemistry’s answers focus on the concept of chemical evolution [
5,
6,
7,
8,
9,
10,
11,
12,
13], which cannot address biological evolution and social evolution. Biology’s answers have moved from Darwin’s theory of natural selection to its updated version, the Modern Synthesis. Darwin’s definition of natural selection as “survival of the fittest” is literally confusing because some individuals less fit can also survive. The Modern Synthesis reinforces the importance of natural selection and reinterprets natural selection as gradual changes in gene frequencies of populations because those individuals carrying adaptive mutations are more reproductively successful [
14]. While these two biological theories can well explain many evolutionary phenomena, they can explain neither macroevolutionary issues (e.g., the origin of life) nor social evolution, which emphasizes both competition and inclusiveness, both selfishness and altruism, and both conflicts and collaboration [
15]. Here, we propose the Carbon-Based Evolutionary Theory (CBET), aiming to provide the first explicit, evidence-based, and holistic explanation for the entire evolution of CBMs on Earth.
1. Methods and Definitions
CBET is established not on new experiments, discoveries, principles, or hypotheses, but rather through the integration of diverse well-established knowledge from physics, chemistry, biology, and social sciences. The integration is based on the following definitions.
Driving force: the energy-associated power that makes a specific change.
Mechanism: the reason for the change of things.
Carbon-containing molecules (CCMs): those molecules containing carbon atoms.
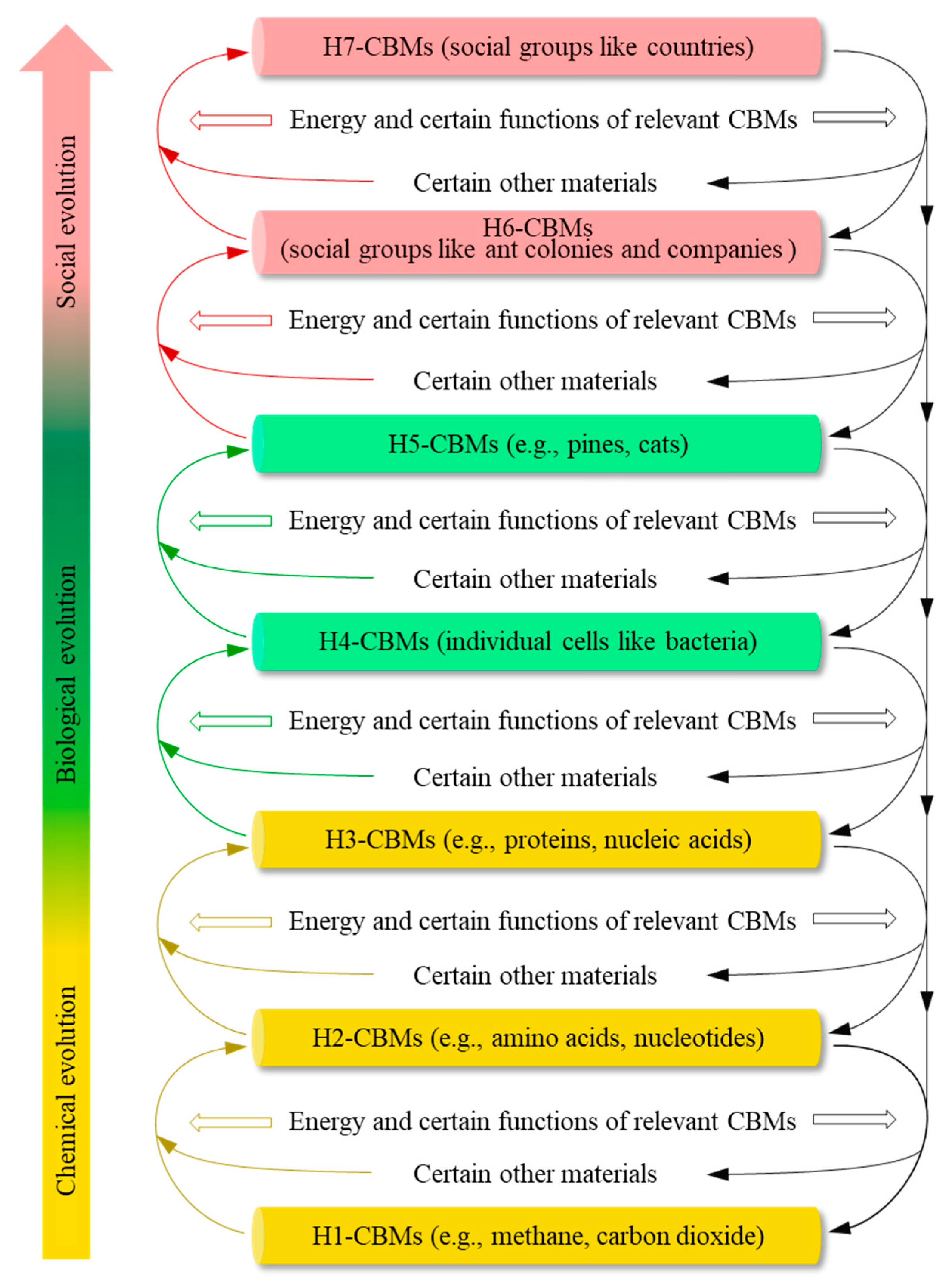
CBMs: those substances in which carbon atoms play a major role at the atomic level. CBMs are classified into the following eight hierarchies.
H0-CBMs: carbon atoms.
H1-CBMs: small CCMs, such as CH4, CO2, and HCN.
H2-CBMs: middle CCMs, such as lysine and glucose.
H3-CBMs: large CCMs, such as proteins and nucleic acids.
H4-CBMs: individual cells, such as bacteria and composed of numerous H3-CBMs and other molecules with internal collaboration.
H5-CBMs: multicellular organisms, such as pines and rabbits.
H6-CBMs: social groups with only one hierarchy in management, such as ant colonies, bee colonies, and many small companies, which are composed of some animal or human individuals that closely cooperate for the social groups with different duties for the social groups.
H7-CBMs: human social groups that have multiple hierarchies in management, and low-hierarchy social groups closely cooperate with different duties for high-hierarchy social groups. Universities, armies, and countries are examples of H7-CBMs.
High-hierarchy CBMs (HHCBMs) and low-hierarchy CBMs (LHCBMs) are defined by comparing their hierarchies. For instance, H4-CBMs are HHCBMs compared to H3-CBMs, but they are LHCBMs compared to H5-CBMs.
Simple CBMs and complex CBMs are defined by comparing their structural complexity. For instance, eukaryotic paramecia are complex CBMs compared to prokaryotic staphylococci, but they are simple CBMs compared with multi-cellular ants.
Multi-molecule aggregates (MMAs): phospholipid bilayers, virions, ribosomes, chloroplasts, and other relatively stable structures composed of multiple H3-CBMs with or without other molecules.
Notably, no clear lines separate the above CBM hierarchies. For instance, some peptides are between H2-CBMs and H3-CBMs, some MMAs are between H3-CBMs and H4-CBMs, and lion groups are between H5-CBMs and H6-CBMs.
The closely cooperative social groups of H6-CBMs and H7-CBMs can be viewed as superorganisms [
16], while multicellular organisms and unicellular can be viewed as social groups of cells and molecules, respectively.
Chemical evolution: the evolution from small CCMs to middle and large CCMs, or from H1-CBMs to H2-CBMs and H3-CBMs.
Biological evolution: the origin and evolution of life, namely H4-CBMs and H5-CBMs.
Social evolution: the origin and evolution of animal or human social groups, namely H6-CBMs and H7-CBMs.
The overall performance of the formation and maintenance (OPFM) of a CBM: the holistic or general ability for the CBM to be formed and maintained.
Autocatalysis or allocatalysis: the catalysis of a molecule catalyzing the synthesis or degradation of itself (autocatalysis) or other molecules (allocatalysis).
2. Results
2.1. The Driving Force Mechanism
2.1.1. Origin of the Driving Force Mechanism
In a car, it is the intricate functions of the automotive engine and the powertrain system that transform the energy derived from fuel into the driving force necessary for the car’s running. Similarly, as detailed below in
Section 2.1.2 and outlined in
Figure 1, certain functions of the relevant CBMs transform certain energy on Earth into the driving force of the evolution of CBMs. At each time-point, this driving force constitutes the energy-associated power that forms and degrades complex CBMs.
2.1.2. Effects of the Driving Force Mechanism
(1) Formation of H2-CBMs. Earth possesses abundant heat sources, including sunlight, lightning, geothermal energy, volcanic activity, fires, and the decomposition of organic matter [
17]. The heat energy released from these sources is adjusted by the extensive water, which amounts to approximately 1.35×10
18 tons, as well as the atmosphere, which is over 1,000 km thick, on Earth [
17]. The adjustment helps to maintain a moderate, widespread, and persistent distribution of heat energy on Earth and renders Earth a rare habitable planet [
20]. Under the second law of thermodynamics [
21], many H1-CBMs, such as CH
4, CO
2, and HCN, and other substances (e.g., H
2O) spontaneously absorb heat from these heat sources. Chemically, certain functions of H1-CBMs transform the absorbed heat energy into the driving force to transform many H1-CBMs into diverse distinct H2-CBMs through heat-absorbing organic synthesis reactions [
5,
6,
7,
8,
9,
10,
11,
12,
13]. The prebiotic chemical synthesis of various H2-CBMs, such as amino acids, nucleotides, and monosaccharaides, has been experimentally validated [
5,
6,
7,
8,
9,
10,
11,
12,
13], and supported by the fact that myriad distinct H2-CBMs have been identified from meteorites [
22]. Certain functions of H1-CBMs, such as the reactivity of CO
2 with H
2O and the reactivity of CH
4 with NH
3, are critical for the formation of H2-CBMs.
(2) Formation of H3-CBMs. Many H2-CBMs generated through the above processes can further spontaneously absorb heat under the second law of thermodynamics from abundant heat sources on Earth. Chemically, certain functions of H2-CBMs, such as the dehydration condensation reactions between some function groups of some H2-CBMs, transform the absorbed heat energy into the driving force to transform many H2-CBMs into myriad distinct H3-CBMs through heat-absorbing organic chemical reactions. Studies have shown that high pressure, some inorganic molecules like boric acids, and certain H2-CBMs, such as N-phosphoryl amino acids, can facilitate the abiotic synthesis of some H3-CBMs, such as proteins, nucleic acids, and lipids [
5,
6,
7,
9,
10,
11,
12,
13,
14,
18].
Among all atoms, only carbon atoms can serve as the backbone to bond with other atoms, such as hydrogen, oxygen, nitrogen, sulfur, phosphorus, halogens, metals, and various functional groups, to form long, complex, and water-soluble large molecules [
18], which are essential for the construction of organisms. Consequently, only CBMs have the potential to naturally evolve from simple structures into life [
18].
(3) Formation of H4-CBMs. Myriad H3-CBMs, such as proteins, nucleic acids, and lipids, generated through the above processes, along with other materials (e.g., H
2O, H1-CBMs, and H2-CBMs), can form diverse MMAs. In physics and chemistry, certain functions of H3-CBMs, such as the interaction among proteins through hydrogen bonds, transform the energy provided by the wind, rain, lightning, and solar evaporation into the driving force for the formation of myriad MMAs [
5,
9,
10,
11,
12]. A very few MMAs that emerged occasionally can actively absorb water, organic molecules, and energy to construct or reproduce themselves. The emergence of these MMAs represented the origin of life or the first batch of H4-CBMs. The energy, which is supplied under the laws of physics and chemistry, is critical to the formation and maintenance of H4-CBMs, which involve energy-absorbing synthesis of diverse H3-CBMs. Meanwhile, some functions of certain H3-CBMs, such as the one of nucleic acids in storing genetic information and the catalysis activity of many proteins, are also critical for the formation of H4-CBMs. Consequently, certain functions of diverse H3-CBMs transform certain energy into the driving force of the formation of H4-CBMs, which, for example, is embodied in the exponential growth of
Escherichia coli cultured in broth within a warm incubator. The first generations of H4-CBMs might originate at sea beds near hydrothermal vents [
23]. One of these H4-CBMs survived natural selection and became the Last Universal Common Ancestor (LUCA) of all living things. LUCA could possess hundreds of genes [
24]. The experiments on the abiotic synthesis of viruses and H4-CBMs have been successful, which supports the abiogenesis of LUCA from extremely vast MMAs [
25,
26], although the precise steps by which LUCA originated, particularly the ones involving the origin of codons for protein synthesis [
27], remains unknown.
(4) Formation of H5-CBMs. Myriad variants of prokaryotic H4-CBMs could accumulate on Earth in billions of years after its origin. Some variants became eukaryotic H4-CBMs during the early Proterozoic eon [
14,
28]. Myriad variants of eukaryotic H4-CBMs could accumulate on Earth in the following hundreds of million years. Some variants of H4-CBMs became eukaryotic multicellular organisms (H5-CBMs) during the middle Proterozoic eon [
14,
29]. Certain functions of the relevant H4-CBMs, such as the abilities of the fertilized eggs of elephants or trees to absorb energy and materials, gene regulation, and cell differentiation, transform certain energy on Earth into the driving force for the formation and maintenance of H5-CBMs, driving fertilized eggs and related materials to develop into adult elephants and other H5-CBMs. The driving force is embodied in the spring and summer, when many plants sprout and grow vigorously. It is also embodied in tropical rainforests, which are rich in biodiversity. By contrast, glacial cold periods with less energy on Earth can lead to mass extinction of H5-CBMs [
14].
(5) Formation of H6-CBMs. Multicellular animals possibly emerged on Earth 800 million years ago [
30]. Possibly 100 million years ago, some insects established their social groups (H6-CBMs) [
31]. Sociality is widespread in
Hymenoptera (ants, bees, and wasps),
Blattodea (termites), and
Hemiptera (aphids) [
16,
32,
33]. A typical animal social collective has a queen and a few reproductive males, who take on the roles of the sole reproducers, and other individuals act as soldiers and workers who collaborate to create a living situation favorable for the brood. Some small companies can also be taken as H6-CBMs. Some functions of the relevant animals, such as their complexity in gene regulation, chemical communications, and internal collaboration, are critical for establishing social groups [
31,
32,
33,
34]. These functions transform certain energy on Earth, such as the chemical energy stored in honey collected by honeybees, into the driving force for the formation of the relevant H6-CBMs.
(6) Formation of H7-CBMs. H7-CBMs are human social groups that have multiple hierarchies in management. Certain functions of some human social groups, such as their advanced technologies, their advanced management systems, and/or their strong willing to expand their group sizes, transform certain energy on Earth associated with food, hydropower, animal power, human power, coal, electricity, nuclear energy, and/or solar energy into the driving force for the formation of H7-CBMs.
(7) Degradation of complex CBMs. Due to certain functions of CBMs, such as that carbon-carbon bonds are relatively fragile, the energy from fires, collisions, radiation, and other sources can degrade some complex CBMs [
14,
15,
18]. Sometimes the energy of asteroid collision and volcanism can directly or indirectly lead to mass extinctions of complex CBMs [
14]. The degradation process usually releases energy (
Figure 1). Complex CBMs can be established hierarchy by hierarchy, and they can be directly degraded into CBMs one or more hierarchies lower (
Figure 1). For instance, a living grass can be directly decomposed into molecules and atoms in a steelmaking furnace.
2.1.3. Repeated Cycles of the Formation and Degradation of Complex CBMs
The above processes lead to repeated cycles of the formation and degradation of complex CBMs. The repeated cycles align with the carbon cycling on Earth, which influences the carbon contents in the atmosphere, land, oceans, biosphere, and human society [
18,
37]. The CBMs in these repeated cycles hold the following two features.
(1) Variability. Regenerated complex CBMs often exhibit some structural changes, which exhibit myriad possibilities. Consequently, myriad distinct complex CBMs, from H2-CBMs to H7-CBMs, have been produced naturally.
(2) Relative stability. For example, some proteins have been well preserved in fossils for millions of years [
35], and various H2-CBMs could have been preserved billions of years in meteorites [
36], and humans can typically live for decades.
The repeated cycles of the formation and degradation of H4-CBMs and H5-CBMs involve the formation and degradation of many H2-CBMs and H3-CBMs. Consequently, biological evolution is the natural extension of chemical evolution.
The repeated cycles of the formation and degradation of H6-CBMs and H7-CBMs involve the birth and death of cells and animal individuals within H6-CBMs or H7-CBMs. Consequently, social evolution is the natural extension of biological evolution.
2.2. The Structure-Function Mechanism
2.2.1. Origin of the Structure-Function Mechanism
Fact 1: The hierarchical escalation of the complexity of CBMs arising from the driving force mechanism can generate new functions that LHCBMs do not have. This aligns with the core principle of systems theory: the whole is greater than the sum of its parts [
38]. For instance, bird cells cannot fly, but birds can. When a group of individuals transform into a basketball team, the team generates certain new functions, such as competing in games to attract sponsorships, which, in turn, supports the maintenance of the team.
Fact 2: During the repeated cycles of the formation and degradation of HHCBMs, some HHCBMs can acquire new functions due to their structural changes in the cycles. For instance, genomic mutations can alter the running speeds of goats, the chemical communication within insect colonies, and the intelligence of humans.
The above two facts stem from the logic that structural changes can lead to new functions. They constitute the structure-function mechanism of the evolution of CBMs, which generates some new functions that can be beneficial, harmful, or neutral to the formation and maintenance of CBMs from H2-CBMs to H7-CBMs. Many functions of relevant CBMs mentioned in
Figure 1 are generated through this mechanism, as elucidated below.
2.2.2. Effects of the Structure-Function Mechanism
Many H2-CBMs obtain novel functions through the structure-function mechanism. Some H2-CBMs, such as glucose and adenosine triphosphate, can store energy. Some H2-CBMs can catalyze certain chemical reactions [
8,
9,
39,
40,
41], including the synthesis of diverse chiral compounds. Some H2-CBMs have two or more active function groups and can thus be polymerized. Many H2-CBMs (e.g., gentamicin and metformin) have the effects to cure human diseases, which cannot be cured by H1-CBMs, and they are thus beneficial to the maintenance of the H5-CBMs of humans. Meanwhile, certain structural changes of H2-CBMs can significantly change the functions of the H2-CBMs, which have been applied frequently in drug optimization.
Many H3-CBMs obtain novel functions through the structure-function mechanism. Some proteins have the functions of ion channels. Many proteins can catalyze the synthesis of certain H2-CBMs or H3-CBMs. The H3-CBMs of nucleic acids (DNA or RNA) can serve as the template to direct the precise synthesis of DNA and RNA. The H3-CBMs of phospholipids are amphiphilic molecules and can thus self-assemble into a barrier-like bilayer. The H3-CBMs of many antibodies can block the infections of viruses. These functions of H3-CMBs cannot be realized by the H2-CBMs that form the relevant H3-CBMs. Meanwhile, certain structural changes of H3-CBMs (e.g., proteins) can significantly change the functions of the H3-CBMs, which constitute the genetic bases of certain human genetic diseases. It has been known
Many MMAs obtain novel functions through the structure-function mechanism. Some chlorophyll-like MMAs composed of certain proteins can transform CO2 to sugar. Some MMAs composed of certain proteins and DNA molecules can reproduce the DNA molecules through the DNA polymerase activity of the proteins. Some MMAs composed of the bilayer of phospholipids can safeguard water-soluble H2-CBMs and H3-CBMs from degradation by enzymes. The above functions of MMAs facilitate the formation and maintenance of H2-CBMs and H3-CBMs, which, in turn, facilitate the formation and maintenance of MMAs. Furthermore, a very few MMAs can actively absorb water, organic molecules, and solar energy to construct themselves. The emergence of these MMAs represents the origin of life or H4-CBMs, which possess the critical function of life: reproduction. Reproduction of H4-CBMs can efficiently facilitate the formation and maintenance of H2-CBMs, H3-CBMs, and MMAs within H4-CBMs. Meanwhile, certain structural changes (e.g., genomic changes) of H4-CBMs can significantly change the functions of the H4-CBMs.
Many H5-CBMs obtain novel functions through the structure-function mechanism. For instance, cat cells can neither ran quickly nor catch rats, but cats can. Meanwhile, structural changes of H5-CBMs can significantly change the functions of the H5-CBMs. For instance, inflammatory changes leading to necrosis of heart, brain, and vascular cells kill many humans annually.
Many H6 -CBMs obtain novel functions through the structure-function mechanism. For instance, similar to the breeding of cows by humans for milk, European red ants (
Formica rufa) cultivate various species of aphids for collecting the honeydew of aphids, which cannot be realized by a single ant [
42]. Meanwhile, certain structural changes H6-CBMs can significantly change the functions of H6-CBMs. For instance, changes of the leader of a company can significantly change the functions of the company.
Many H7-CBMs obtain novel functions through the structure-function mechanism. For instance, many agricultural companies can reduce overall operating costs, produce better agricultural products (i.e., certain CBMs) through technological complementarity, and obtain a higher market share through market complementarity, after they merge into large agricultural companies, which are HHCBMs, and their production involve the formation and maintenance of CBMs from H2-CBMs to H7-CBMs, including themselves. Meanwhile, certain structural changes H7-CBMs (e.g., changes of the leaders of a country) can significantly change the functions of H6-CBMs. For instance, changes of the leaders of a university or a country can significantly change the functions of the university or the country.
2.3. The Natural Selection Mechanism
2.3.1. Origin and Features of the Natural Selection Mechanism
As elucidated in
Section 2.1.2, there are cycles of the formation and degradation of complex CBMs, and many complex CBMs undergo structural and functional changes during these cycles. These cycles hold the following three mathematical logics.
Logic 1: If the total formation of a complex CBM exceeds its total degradation, this complex CBM persists on Earth.
Logic 2: If the ratio of the formation to degradation of a complex CBM exceeds 1 (or falls below 1), the quantity of this complex CBM is increasing (or decreasing) during the relevant period.
Logic 3: If the ratio of the formation to degradation of complex CBM A is greater than that of complex CBM B, the ratio of CBM A versus CBM B in quantity is increasing during the relevant period.
These three mathematical logics constitute the natural selection mechanism of the evolution of CBMs, which has the following features:
(1) Natural selection stems from mathematical logic, which is the essence of its operation. Natural selection in previous theories stems from the survival competition of organisms [
14], which is a biological phenomenon governed by the mathematical logic.
(2) Natural selection applies to biological evolution, as validated by previous theories [
14], and it also applies to chemical evolution and social evolution, as suggested by the above three logics.
(3) The OPFM of a complex CBM determines its fate in natural selection according to the three mathematical logics. The OPFM itself is determined not solely by any single component, trait, or hierarchy of the complex CBM, but collectively by all components, traits, and hierarchies of the complex CBM. This suggests that all components, all traits, and all hierarchies of a complex CBM are subject to natural selection. Moreover, certain traits of complex CBMs, such as the running ability of antelopes, the cooperative behavior of wolves, or human individual expertise, can significantly enhance a CBM’s OPFM. Consequently, natural selection underscores both all-round development and specialized development.
(4) Natural selection underscores both competition and collaboration, as suggested by Logics 2 and 3 stated above. The competition results in the accumulation of the structural changes (positive selection) that enhance the OPFM of complex CBMs and the elimination of the structural changes (negative selection) that impair the OPFM of complex CBMs. The synergy of positive and negative selection leads to a continuous process of optimizing the internal structures, internal collaboration, and/or internal orderliness of CBMs from H2-CBMs to H7-CBMs. In other words, the advantages of a complex CBM in natural selection usually rely on its internal collaboration. For ants, bees, humans, and other animals living in social groups (H6-CBMs and H7-CBMs), their collaboration is vital for the existence of the relevant social groups.
(5) Natural selection underscores both selfishness and altruism. Selfishness is essential for animals to obtain adequate energy, materials, suitable environments, and mating opportunities to ensure their total formation exceeds their total degradation. Meanwhile, altruism is also widespread throughout evolution. For instance, many molecules act as catalyzers, energy-providers, or constituent materials for the synthesis of other molecules in organisms. Many molecules in H4-CBMs (e.g., bacteria) facilitate the passage of nucleic acids to the next generation. Many cells in H5-CBMs (e.g., rats) facilitate the passage of reproductive cells to the next generation. Many animal individuals in H6-CBMs (e.g., ant colonies) support the reproduction of a few individuals in the social groups, and many human soldiers, policemen, and firefighters in H7-CBMs sacrifice themselves for the benefit of their social groups or other individuals.
(6) Natural selection in CBET underscores inclusiveness. According to Logic 1 stated above, a complex CBM can persist on Earth if its total formation exceeds its total degradation, even if its OPFM is lower than that of its predecessor or other CBMs. Consequently, natural selection permits organisms to have some disadvantages. For instance, many people carry gene mutations linked to genetic diseases, and human neonates are more vulnerable than those of some other mammals. This suggests that some biological traits, such as the long necks of giraffes, can be beneficial, neutral, or detrimental in natural selection. The inclusiveness of natural selection aligns with the prevalence of neutral mutations in genomes [
14], as these mutations have minimal impacts on the OPFM of the relevant organisms.
(7) Natural selection underscores both inheritable and non-inheritable factors, as they can both significantly affect the OPFM of relevant organisms. For instance, the non-heritable factor of vaccination can increase animals’ OPFM, and education can increase humans’ OPFM. Sexual selection, no matter whether inheritable and non-inheritable, which is distinct from natural selection and a function and behavior of many animals, highly affects the evolution of animals.
(8) Natural selection underscores a balance between resource exploitation and environmental protection. Firstly, whether an HHCBM has sufficient OPFM in natural selection depends not only on its own characteristics but also on the environment, which provides energy and constituent materials for HHCBMs. Secondly, major environmental changes, such as asteroid impacts, can destroy enormous HHCBMs [
14,
16,
32].
(9) Natural selection of many CBMs escalates over time, particularly for intra-species competition and those interacting species, such as predators and prey, parasites and hosts, and plants and herbivores. The improvements in the adaptations of one population or species are often offset by the simultaneous evolution of other populations or species. Even if a population or species is currently dominant, ceasing to evolve could result in being outpaced by other evolving populations or species [
43].
(10) Natural selection of LHCBMs is frequently affected by HHCBMs, which is detailed below in
Section 2.3.2.
2.3.2. Other Effects of the Natural Selection Mechanism
According to the three mathematical logics stated above, natural selection have the following effects on the evolution of CBMs that have not been elucidated above.
(1) The natural selection of many H2-CBMs is affected by certain H3-CBMs (e.g., some enzymes, which can catalyze the production of certain H2-CBMs), H4-CBMs (e.g., bacteria, which can degrade many H3-CBMs into H2-CBMs through catabolism), and H5-CBMs (e.g., trees, which can produce many H2-CBMs through photosynthesis and other chemical reactions). Consequently, nowadays those H2-CBMs involved in the formation of organisms, such as amino acids and nucleotides, or the development of human societies, such as gentamicin and many other chemical drugs, have been prevalent on Earth, and many H2-CBMs not having been involved in the formation of organisms could have been rare on Earth.
(2). The natural selection of many H3-CBMs is affected by certain H4-CBMs (e.g., bacteria, which can degrade many H3-CBMs into H2-CBMs through catabolism and synthesize many H3-CBMs through anabolism), H5-CBMs (e.g., trees, which can produce many H3-CBMs like celluloses, proteins, and nucleic acids), and H6-CBMs (e.g., some companies produce diverse H3-CBMs, which are employed for producing clothes and shoes). Consequently, nowadays those H3-CBMs involved in the formation of organisms, such as proteins and nucleic acids, or the development of human societies, such as polyethylene, polypropylene, and polyvinyl chloride, have been prevalent on Earth, and many H3-CBMs not having been involved in the formation of organisms could have been rare on Earth.
(3) The natural selection of many H4-CBMs is affected by certain H5-CBMs (e.g., rats, which can produce many new cells and kill many old cells every day in their bodies) and H6-CBMs (e.g., some ant colonies, whose development involves the formation and degradation of their cells). Consequently, nowadays those H4-CBMs (e.g., certain fungi, bacteria, and cells of H5-CBMs) involved in the formation of H5-CBMs or the maintenance of human societies (e.g., the production of yogurt or antibiotics) have been prevalent on Earth, and many H4-CBMs harmful to human societies, such as Vibrio cholera and Yersinia pestis, have been controlled on Earth.
(4) The natural selection of many H5-CBMs is affected by certain H6-CBMs (e.g., European red ants, which cultivate various species of aphids for collecting the honeydew of aphids [
42]) and H7-CBMs (e.g., countries, whose management affects the lives of many humans). Consequently, nowadays those H5-CBMs involved in the development of human societies, such as rice, wheat, potatoes, pigs, cows, sheep, and chickens, have been prevalent on Earth, and many H5-CBMs harmful to human societies, such as tigers and opium poppies, have been controlled on Earth.
(5) The natural selection of many H6-CBMs is affected by certain H7-CBMs. For instance, the laws of a country affect the fortunes of many companies in the country. Furthermore, natural selection can lead to the improvement in the management of H6-CBMs. Well managed H6-CBMs have some advantages in natural selection as they can significantly reduce internal competition and struggles, as well as utilize collective advantages to obtain energy and relevant materials to reproduce and maintain them and fight against predators. For instance, certain ants, such as
Atta colombica, can cultivate fungi as a primary food source [
44], which cannot be realized by a single ant. Consequently, although sometimes social animals require individuals to sacrifice their freedom and even their lives for the benefit of others, social animals typically have significantly longer lifespans compared to their counterparts without sociality within the same taxa [
32,
33]. For instance, the naked mole rat (
Heterocephalus glaber) living in society has a lifespan of up to 30 years, several times longer than that of other rodents [
45]. On the other hand, the natural selection advantages of animal social groups also lead to sometimes intense competition or conflicts between animal social groups. For instance, battles between ant colonies can result in ant massacres [
46].
(6) Natural selection can lead to the improvement of the management and internal collaboration of H7-CBMs through competition, which underscores the balance between the interests of human individuals and human social groups. Well managed H7-CBMs have some advantages over poorly managed H7-CBMs in natural selection because well managed H7-CBMs can better reduce internal competition and struggles, better utilize collective advantages to obtain energy and relevant materials to maintain themselves, and better compete with other H7-CBMs and fight against enemies. H7-CBMs tend to expand their sizes because usually large, well-managed H7-CBMs have more natural selection advantages than small, well-managed H7-CBMs. Consequently, human social groups have evolved over millennia from clans to tribes, tribal alliances, nations, and national alliances. This evolutionary trend argues for the unification of all nations into a single, harmonious entity. The advantages of well-managed H7-CBMs in natural selection have also escalated competition and conflicts among them, encompassing international wars. In 2022, global military expenditure topped
$2.2 trillion, with 238,000 lives lost to warfare [
47]. Technological advancements, like nuclear weaponry and artificial intelligence, have drastically enhanced the potential for devastation in international wars, jeopardizing both humanity and Earth. This further underscores the critical need for all nations to be unified into a single, harmonious entity, which can avoid the staggering military costs, fatalities, and threats to humanity and Earth.
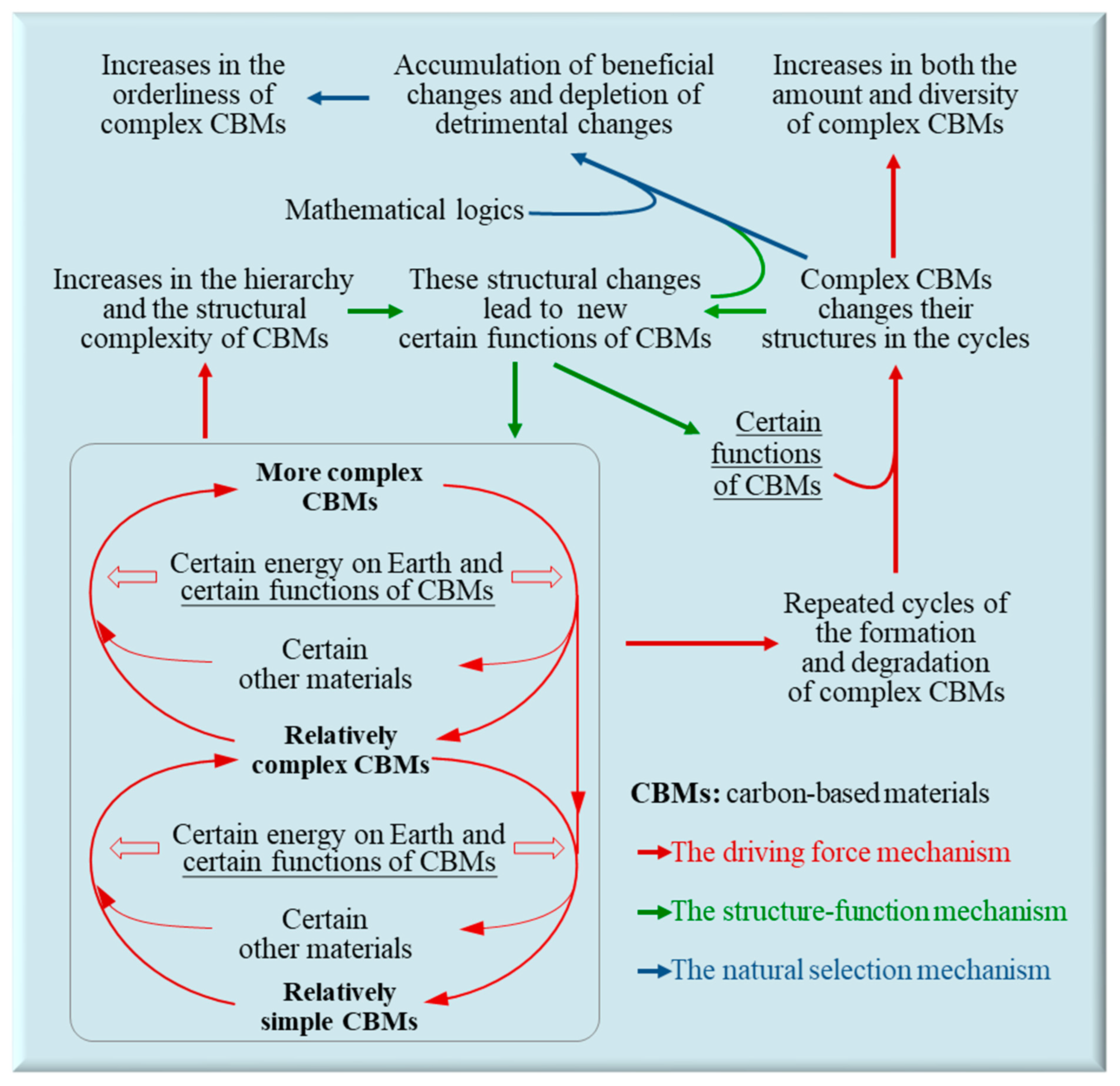
2.4. The Synergy of the Three Mechanisms Leads to Evolution
As outlined in
Figure 2, the synergy of the above three mechanisms hierarchically escalates the complexity of CBMs, increases the abundance, diversity, and orderliness of complex CBMs, leading to chemical, biological, and social evolution on Earth. Firstly, the driving force mechanism, which arises from the combination of energy and certain functions of relevant CBMs, hierarchically escalates the complexity of CBMs and leads to the repeated cycles of the formation and degradation of complex CBMs (
Section 2.1). Secondly, the hierarchical escalation in the complexity of CBMs and structural changes of complex CBMs during the repeated cycles can generate new functions, which can be beneficial, harmful, or neutral to enhance the OPFM of complex CBMs. This constitutes the structure-function mechanism (
Section 2.2). Thirdly, in mathematics, those functions beneficial to enhance the OPFM of certain complex CBMs, along with the relevant structural changes, will be relatively more abundant on Earth, which constitutes the natural selection mechanism, leading to continuous structural and functional optimization of complex CBMs (
Section 2.3).
The natural selection mechanism allows certain complex CBMs to tend to form and maintain their bodies using less energy and materials. Meanwhile, the driving force mechanism and the structure-function mechanism collectively allow certain other complex CBMs to tend to form and maintain their bodies using more energy and materials. For example, many primates have become larger in body sizes than their earliest ancestors, which could be as small as mice [
48].
The evolution of CBMs significantly impacts itself. It stores energy, prepares constituent materials, produces catalyzers, generates novel functions, and escalates the natural selection pressure for its subsequent stages (
Figure 2). Furthermore, the evolution of CBMs substantially modifies Earth’s surface and environment, leading to opportunities, competition, or disasters for certain CBMs [
43,
49]. For instance, the increase in photosynthetic bacteria around 2.5 billion years ago likely resulted in a significant increase in the concentration of oxygen in the air, posing a disaster for anaerobic bacteria and opportunities for aerobic bacteria [
14,
49].
3. Novelties of CBET
CBET carries the following important novelties.
(1) In general science and philosophy, CBET offers the first explicit and holistic explanation for chemical, biological, and social evolution, from both a bird’s-eye view and an introspective view. In contrast, previous studies have yet to provide an explicit and holistic explanation for the entire evolution of CBMs.
(2) In general science and philosophy, CBET recognizes the complexity of evolution and innovatively employs three mechanisms to explain evolution, while previous studies underestimated the complexity of evolution and tried to explain evolution using a single mechanism, such as natural selection in Darwin’s theory [
14], entropy dissipation into the surroundings in Schrödinger’s negative entropy notion [
2], self-organization in Prigogine’s dissipative structure theory [
3], the constructal law [
50], the maximum entropy production hypothesis [
4], the free-energy principle [
51], Eigen’s hypothesis of hypercycles proposed for interpreting the origin of life [
52], and the RNA world hypothesis [
53]. These one-single-mechanism theories or hypotheses are either elusive or incomplete, primarily focusing on specific aspects of chemical, biological, or social evolution using an introspective methodology. Additionally, Eigen’s hypothesis of hypercycles and the RNA world hypothesis both underscored the organic syntheses of a few kinds of special molecules through autocatalysis [
52,
53], while CBET underscores allocatalysis and collaboration of various common CBMs (
Section 2.1,
Section 2.2,
Section 2.3 and
Section 2.4). Autocatalysis is much rarer than allocatalysis both in biosphere and abiotic environments [
8,
39,
40,
41], and should be thus much less important than allocatalysis for evolution.
(3) In general science and philosophy, the three evolutionary mechanisms of CBET innovatively clarify the intricate relationships among the five basic concepts associated with evolution: materials, energy, structures, functions, and orderliness (
Figure 2). In contrast, the relationships of these basic concepts have been frequently entangled with certain elusive notions associated with entropy [
54], as shown in the following paragraph.
(4) In physics, CBET innovatively interprets the relationships between the second law of thermodynamics and evolution. It is widely stated in textbooks and encyclopedias that entropy represents disorder and consequently, the second law of thermodynamics contradicts evolution, as this law seems to validate that many systems tend to increase their entropy and thus become more disordered [
1,
2,
3,
50,
54]. Creationists have exploited this perceived contradiction to argue against evolution [
1,
55]. Some influential theories, such as Schrödinger’s negative entropy theory and Prigogine’s dissipative structure theory, accepted and reconciled this contradiction by suggesting that open systems (like organisms) can gain orderliness not through natural selection but through the dissipation of entropy into the environment [
2,
3]. In contrast, CBET embraces the new notion that entropy does not represent disorder [
54,
56], as entropy is analogous to energy with the unit of joule/kelvin, representing the quotient of heat energy and the corresponding thermodynamic temperature, while disorder refers to chaos, untidiness, or abnormalities, and is not analogous to energy [
54,
56]. The unit of disorder is not joule/kelvin, either. Consequently, CBET supports the new notion that the second law of thermodynamics does not suggest that many systems tend to become more disordered and hence this law does not contradict evolution [
54,
56]. Moreover, CBET clarifies that this thermodynamic law is highly associated with the driving force of evolution (
Section 2.1), which aligns with some other research reports [
50,
55].
(5) In biology, as elucidated in
Section 2.1, CBET reveals the driving force of biological evolution in terms of energy. Previous studies posited that natural selection, mutations, or genetic drift are the driving forces of evolution [
57,
58,
59], but these factors require energy and do not provide energy.
(6) In biology, as elucidated in
Section 2.3, CBET provides novel and comprehensive explanations for the mathematical essence of natural selection, the targets of natural selection, and biological altruism from a bird’s-eye view.
(7) In biology, the inclusiveness of natural selection, along with the driving force mechanism and the structure-function mechanism, innovatively and explicitly interprets the increasing biodiversity on Earth and certain macroevolution events. For instance, organisms, multicellular organisms, and endothermic animals are not inherently fitter than inanimate materials, unicellular organisms, and ectothermic animals, respectively, yet their total formation can all exceed their total degradation due to some functions generated by the structure-function mechanism, the energy supplied by the driving force mechanism, and the materials supported by the environment. In contrast, previous theories emphasized the fierce competition in natural selection, which was expressed as “survival of the fittest” in Darwin’s theory and “gradual replacement of populations with those carrying advantageous mutations” in the Modern Synthesis [
14]. These previous theories could not explain the above macroevolution events [
1].
(8) In biology, neither Darwin’s natural selection nor the Modern Synthesis can incorporate non-random mutations [
60,
61], neutral mutations [
14], epigenetic changes [
62,
63], and non-heritable strengths [
14], which are prevalent in or important for numerous organisms. In contrast, CBET can innovatively incorporate these facts, as non-random mutations and epigenetic changes can be realized through the structure-function mechanism, neutral mutations affect little the OPFM of organisms, and epigenetic changes and certain non-heritable strengths can significantly affect the OPFM of organisms [
4].
(9) In social sciences, CBET innovatively uncovers the natural roots of the balance between multiple crucial and seemingly paradoxical social notions, such as all-round development versus specialized development, inclusiveness versus elimination, collaboration versus competition, altruism versus selfishness, and resource exploitation versus environmental protection (
Section 2.3). Accordingly, CBET advocates for the balanced and harmonious development of human society. Historically, nations that adopted relatively balanced and harmonious development in their social governance often performed powerfully in international competitions [
64]. CBET also suggests that in the future it is better for all nations to be unified into a single, harmonious entity to avoid too expensive and disastrous international wars (
Section 2.3). In contrast, Darwin’s natural selection only emphasizes selfishness, competition, and the elimination of the less advantaged and has been used to rationalize wars, colonialism, slavery, racism, and genocide [
15].
(10) CBET highlights the structural hierarchies of CBMs or organisms using the innovative terminology of the eight hierarchies of CBMs. Although hierarchies have also been highlighted for decades or even hundreds of years in the research of evolutionary theories [
65], previous research has neither systematically categorized CBMs into the eight hierarchies nor utilized structural hierarchies to deduce the driving force and mechanisms of the evolution of CBMs [
65].
4. Credibility of CBET
CBET is credible for the following reasons:
(1) CBET is established through the novel integration of abundant well-established knowledge from diverse disciplines, and the integration utilizes precise logic and novel terminology, such as the eight hierarchies of CBMs, making CBET a novel, credible, and self-consistent theory.
(2) CBET contains neither elusive nor weird concepts. Meanwhile, to our knowledge, CBET does not contradict any established facts across physics, chemistry, biology, geology, astronomy, and social sciences.
(3) The rapid advancements in scientific research in physics, chemistry, biology, geology, astronomy, and social sciences over the past two centuries have revealed numerous factors critical for the evolution of CBMs (
Section 2.1,
Section 2.2,
Section 2.3 and
Section 2.4). CBET precisely integrates and aligns with these numerous critical factors. In contrast, previous research typically emphasized only a limited number of critical factors. For instance, the Modern Synthesis did not address the chemical properties of carbon atoms, the laws of thermodynamics, the reactions of organic chemistry, or the inclusiveness of social sciences.
(4) CBET avoids diverse pitfalls, such as the misconceptions that entropy is a measure of disorder and that the second law of thermodynamics contradicts evolution [
1,
54,
56]. While many previous studies fell into certain pitfalls.
(5) As shown in
Section 3, CBET is superior to other theories in explaining multiple evolutionary issues, which reinforces its credibility.
5. Conclusions and Outlook
The evolutionary theory of CBET, as delineated in this article through the synthesis of well-established interdisciplinary knowledge, offers a clear, evidence-supported, and comprehensive explanation for chemical, biological, and social evolution. It clarifies the intricate relationships between materials, energy, structures, functions, and orderliness. Additionally, it reveals the natural roots of multiple crucial social notions. As such, it has the potential to bridge natural sciences and social sciences.
As a novel theory, CBET would benefit from additional validation and refinement. Should it gain widespread acceptance, CBET could be a basic theory of sciences, directing research across a variety of scientific disciplines. Also, CBET can guide the rational development of human society because it advocates for the balanced, harmonious, and peaceful development of human societies.
Conflict of Interest
The authors declare no competing interests.
Acknowledgments
The authors thank Meng Yang and Yiqing Chen for their numerous constructive comments. This work was supported by the High-Level Talent Fund of Foshan University (No. 20210036).
Author Contributions
JMC and JWC conceptualized, designed, and performed this study. JMC wrote this article and JMC and JWC revised this article. JMC acquired the fund.
References
- Xie, P. The aufhebung and breakthrough of the theories on the origin and evolution of life (Science Press, 2014).
- Schrodinger, E. What is life (Cambridge University Press, 2012).
- Prigogine, I. Time, structure and fluctuation. Science 1978, 201, 777–785. [Google Scholar] [CrossRef] [PubMed]
- Dewar, R.C. Maximum entropy production and plant optimization theories. Philos. Trans. R. Soc. Lond. B. Biol. Sci. 2010, 365, 1429–1435. [Google Scholar] [CrossRef] [PubMed]
- Oparin, A.I. Chemistry and the origin of life. R. Inst. Chem. Rev. 1969, 2, 1–12. [Google Scholar] [CrossRef]
- Guo, X.; Fu, S.; Ying, J.; Zhao, Y. Prebiotic chemistry: A review of nucleoside phosphorylation and polymerization. Open Biol. 2023, 13, 220234. [Google Scholar] [CrossRef] [PubMed]
- Sumie, Y.; Sato, K.; Kakegawa, T.; Furukawa, Y. Boron-assisted abiotic polypeptide synthesis. Commun. Chem. 2023, 6, 89. [Google Scholar] [CrossRef]
- de Graaf, R.; De Decker, Y.; Sojo, V.; Hudson, R. Quantifying catalysis at the origin of life. Chemistry 2023, 29, e202301447. [Google Scholar] [CrossRef]
- Nogal, N.; Sanz-Sánchez, M.; Vela-Gallego, S.; Ruiz-Mirazo, K.; de la Escosura, A. The protometabolic nature of prebiotic chemistry. Chem Soc Rev. 2023, 52, 7359–7388. [Google Scholar] [CrossRef]
- Chieffo, C.; Shvetsova, A.; Skorda, F.; Lopez, A.; Fiore, M. The origin and early evolution of life: Homochirality emergence in prebiotic environments. Astrobiology 2023, 23, 1368–1382. [Google Scholar] [CrossRef]
- Fiore, M. Prebiotic chemistry and life’s origin (Royal Society of Chemistry, 2022). [CrossRef]
- Farías-Rico, J.A.; Mourra-Díaz, C.M. A short tale of the origin of proteins and ribosome evolution. Microorganisms 2022, 10, 2115. [Google Scholar] [CrossRef]
- Ershov, B. Natural radioactivity and chemical evolution on the early Earth: Prebiotic chemistry and oxygenation. Molecules 2022, 27, 8584. [Google Scholar] [CrossRef]
- Futuyma, D.J.; Kirkpatrick, M. Evolution (Sinauer Associates, 2017).
- Rudman, L.A.; Saud, L.H. Justifying social inequalities: The role of social Darwinism. Pers. Soc. Psychol. B. 2020, 46, 1139–1155. [Google Scholar] [CrossRef] [PubMed]
- Nowak, M.; Tarnita, C.; Wilson, E. The evolution of eusociality. Nature 2010, 466, 1057–1062. [Google Scholar] [CrossRef] [PubMed]
- Bell, J. The Earth book (Sterling, 2019).
- Razeghi, M. The mystery of carbon: An introduction to carbon materials (Institute of Physics Publishing, 2019).
- Cowan, J. One of the first of the second stars. Nature 2012, 488, 288–289. [Google Scholar] [CrossRef]
- Seager, S. Exoplanet habitability. Science 2013, 340, 577–581. [Google Scholar] [CrossRef]
- Borgnakke, C.; Sonntag, R.E. Fundamentals of thermodynamics (Wiley, 2022).
- Schmitt-Kopplin, P.; Gabelica, Z.; Gougeonm, R.D.; et al. High molecular diversity of extraterrestrial organic matter in Murchison meteorite revealed 40 years after its fall. Proc. Natl. Acad. Sci. U. S. A. 2010, 107, 2763–2768. [Google Scholar] [CrossRef]
- Dodd, M.S.; Papineau, D.; Grenne, T.; et al. Evidence for early life in earth’s oldest hydrothermal vent precipitates. Nature 2017, 543, 60–64. [Google Scholar] [CrossRef]
- Weiss, M.C.; Sousa, F.L.; Mrnjavac, N.; et al. The physiology and habitat of the last universal common ancestor. Nat. Microb. 2016, 1, 16116. [Google Scholar] [CrossRef]
- Stobart, C.C.; Moore, M.L. RNA virus reverse genetics and vaccine design. Viruses 2014, 6, 2531–2550. [Google Scholar] [CrossRef]
- Hutchison, C.A., 3rd; Chuang, R.Y.; Noskov, V.N.; et al. Design and synthesis of a minimal bacterial genome. Science 2016, 351, aad6253. [Google Scholar] [CrossRef]
- Xie, P. Who is the missing “matchmaker” between proteins and nucleic acids? Innovation (Camb) 2021, 2, 100120. [Google Scholar] [CrossRef]
- Han, T.M.; Runnegar, B. Megascopic eukaryotic algae from the 2.1-billion-year-old negaunee iron-formation, Michigan. Science 1992, 257, 232–235. [Google Scholar] [CrossRef] [PubMed]
- Zhu, S.; Zhu, M.; Knoll, A.; et al. Decimetre-scale multicellular eukaryotes from the 1.56-billion-year-old Gaoyuzhuang formation in North China. Nat. Commun. 2016, 7, 11500. [Google Scholar] [CrossRef] [PubMed]
- Anderson, R.P.; Woltz, C.R.; Tosca, N.J.; Porter, S.M.; Briggs, D.E.G. Fossilisation processes and our reading of animal antiquity. Trends Ecol. Evol. 2023, 38, 1060–1071. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Yin, X.; Shih, C.; Gao, T.; Ren, D. Termite colonies from mid-Cretaceous Myanmar demonstrate their early eusocial lifestyle in damp wood. Natl. Sci. Rev. 2020, 7, 381–390. [Google Scholar] [CrossRef]
- Mera-Rodríguez, D.; Jourdan, H.; Ward, P.S.; Shattuck, S.; Cover, S.P.; Wilson, E.O.; Rabeling, C. Biogeography and evolution of social parasitism in Australian Myrmecia bulldog ants revealed by phylogenomics. Mol. Phyogenet. Evol. 2023, 186, 107825. [Google Scholar] [CrossRef]
- Plowers, N. An introduction to eusociality. Nature Education Knowledge 2010, 3, 7. [Google Scholar]
- Majelantle, T.L.; Ganswindt, A.; Hart, D.W.; Hagenah, N.; Ganswindt, S.B.; Bennett, N.C. The dissection of a despotic society: Exploration, dominance and hormonal traits. Proc. Biol. Sci. 2024, 291, 20240371. [Google Scholar] [CrossRef]
- Li, Z.H.; Bailleul, A.M.; Stidham, T.A.; Wang, M.; Teng, T. Exceptional preservation of an extinct ostrich from the Late Miocene Linxia Basin of China. Vertebrata PalAsiatica 2021, 59, 229. [Google Scholar] [CrossRef]
- Heck, P.R.; Greer, J.; Kööp, L.; et al. Lifetimes of interstellar dust from cosmic ray exposure ages of presolar silicon carbide. Proc. Natl. Acad. Sci. USA 2020, 117, 1884–1889. [Google Scholar] [CrossRef]
- Reichlem, D.E. The global carbon cycle and climate change (Elsevier; 2023).
- von Bertalanffy, L. General system theory: Foundations, development, applications (George Braziller, 1968).
- Lovinger, G.J.; Sak, M.H.; Jacobsen, E.N. Catalysis of an SN2 pathway by geometric preorganization. Nature 2024, 632, 1052–1059. [Google Scholar] [CrossRef]
- Stone, E.A.; Cutrona, K.J.; Miller, S.J. Asymmetric catalysis upon helically chiral loratadine analogues unveils enantiomer-dependent antihistamine activity. J. Am. Chem. Soc. 2020, 142, 12690–12698. [Google Scholar] [CrossRef] [PubMed]
- Morrell, D.G. Catalysis of organic reactions (CRC Press, 2019).
- Kilpeläinen, J.; Finér, L.; Neuvonen, S.; et al. Does the mutualism between wood ants (Formica rufa group) and Cinara aphids affect Norway spruce growth? Forest Ecology and Management. 2009, 257, 238–243. [Google Scholar] [CrossRef]
- Benton, M.J. The red queen and the Court Jester: Species diversity and the role of biotic and abiotic factors through time. Science 2009, 323, 728–732. [Google Scholar] [CrossRef] [PubMed]
- Suen, G.; Teiling, C.; Li, L.; et al. The genome sequence of the leaf-Cutter Ant Atta cephalotes reveals insights into its obligate symbiotic lifestyle. PLoS Genet. 2011, 7, e1002007. [Google Scholar] [CrossRef]
- Kim, E.; Fang, X.; Fushan, A.; et al. Genome sequencing reveals insights into physiology and longevity of the naked mole rat. Nature 2011, 479, 223–227. [Google Scholar] [CrossRef]
- Moffett, M.W. Adventures among ants (University of California Press, 2010).
-
SIPRI Military Expenditure Database (https://www.sipri.org/databases/milex, accessed 9 April 2024).
- Wilson Mantilla, G.P.; Chester, S.G.B.; Clemens, W.A.; et al. Earliest Palaeocene purgatoriids and the initial radiation of stem primates. R. Soc. Open Sci. 2021, 8, 210050. [Google Scholar] [CrossRef]
- Olejarz, J.; Iwasa, Y.; Knoll, A.H.; et al. The Great Oxygenation Event as a consequence of ecological dynamics modulated by planetary change. Nat. Commun. 2021, 12, 3985. [Google Scholar] [CrossRef]
- Bejan, A. The principle underlying all evolution, biological, geophysical, social and technological. Philos. Trans. A. Math. Phys. Eng. Sci. 2023, 381, 20220288. [Google Scholar] [CrossRef]
- Ramstead, M.J.D.; Badcock, P.B.; Friston, K.J. Answering Schrödinger’s question: A free-energy formulation. Phys. Life Rev. 2018, 24, 1–16. [Google Scholar] [CrossRef]
- Kauffman, S.A. Approaches to the origin of life on the Earth. Life (Basel). 2011, 1, 34–48. [Google Scholar] [CrossRef]
- Robertson, M.P.; Joyce, G.F. The origins of the RNA world. Cold Spring Harb. Perspect. Biol. 2012, 4, a003608. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.M.; Chen, J.W.; Zivieri, R. Systematically challenging three prevailing notions about entropy and life. Qeios 2024. [CrossRef]
- Schreiber, A.; Gimbel, S. Evolution and the second law of thermodynamics: Effectively communicating to non-technicians. Evo. Edu. Outreach. 2010, 3, 99–106. [Google Scholar] [CrossRef]
- Chen, J.M.; Chen, J.W. Root of science?the driving force and mechanisms of the extensive evolution (Science Press, 2000).
- Ma, S.; Guo, Y.; Liu, D.; et al. Genome-wide analysis of the membrane attack complex and perforin genes and their expression pattern under stress in the Solanaceae. Int. J. Mol. Sci. 2023, 24, 13193. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Liu, X.; Wang, C.; et al. The pig pangenome provides insights into the roles of coding structural variations in genetic diversity and adaptation. Genome Res. 2023, 33, 1833–1847. [Google Scholar] [CrossRef]
- Wang, Y.; Li, X.; Feng, Y. Autotetraploid origin of Chinese cherry revealed by chromosomal karyotype and in situ hybridization of seedling progenies. Plants (Basel) 2023, 12, 3116. [Google Scholar] [CrossRef]
- Fitzgerald, D.M.; Rosenberg, S.M. What is mutation? A chapter in the series: How microbes “jeopardize” the modern synthesis. PLoS Genet. 2019, 15, e1007995. [Google Scholar] [CrossRef]
- Olivieri, D.N.; Mirete-Bachiller, S.; Gambón-Deza, F. Insights into the evolution of IG genes in amphibians and reptiles. Dev. Comp. Immunol. 2021, 114, 103868. [Google Scholar] [CrossRef]
- Sabarís, G.; Fitz-James, M.H.; Cavalli, G. Epigenetic inheritance in adaptive evolution. Ann. N. Y. Acad. Sci. 2023, 1524, 22–29. [Google Scholar] [CrossRef]
- Gómez-Schiavon, M.; Buchler, N.E. Epigenetic switching as a strategy for quick adaptation while attenuating biochemical noise. PLoS Comput. Biol. 2019, 15, e1007364. [Google Scholar] [CrossRef]
- McNeill, J.R.; Pomeranz, K. The Cambridge World History (Cambridge University Press, 2015).
- Niles Eldredge, N.; Pievani, T.; Serrelli, E.; Tëmkin, I. Evolutionary theory: A hierarchical perspective (The University of Chicago Press, 2016).
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).