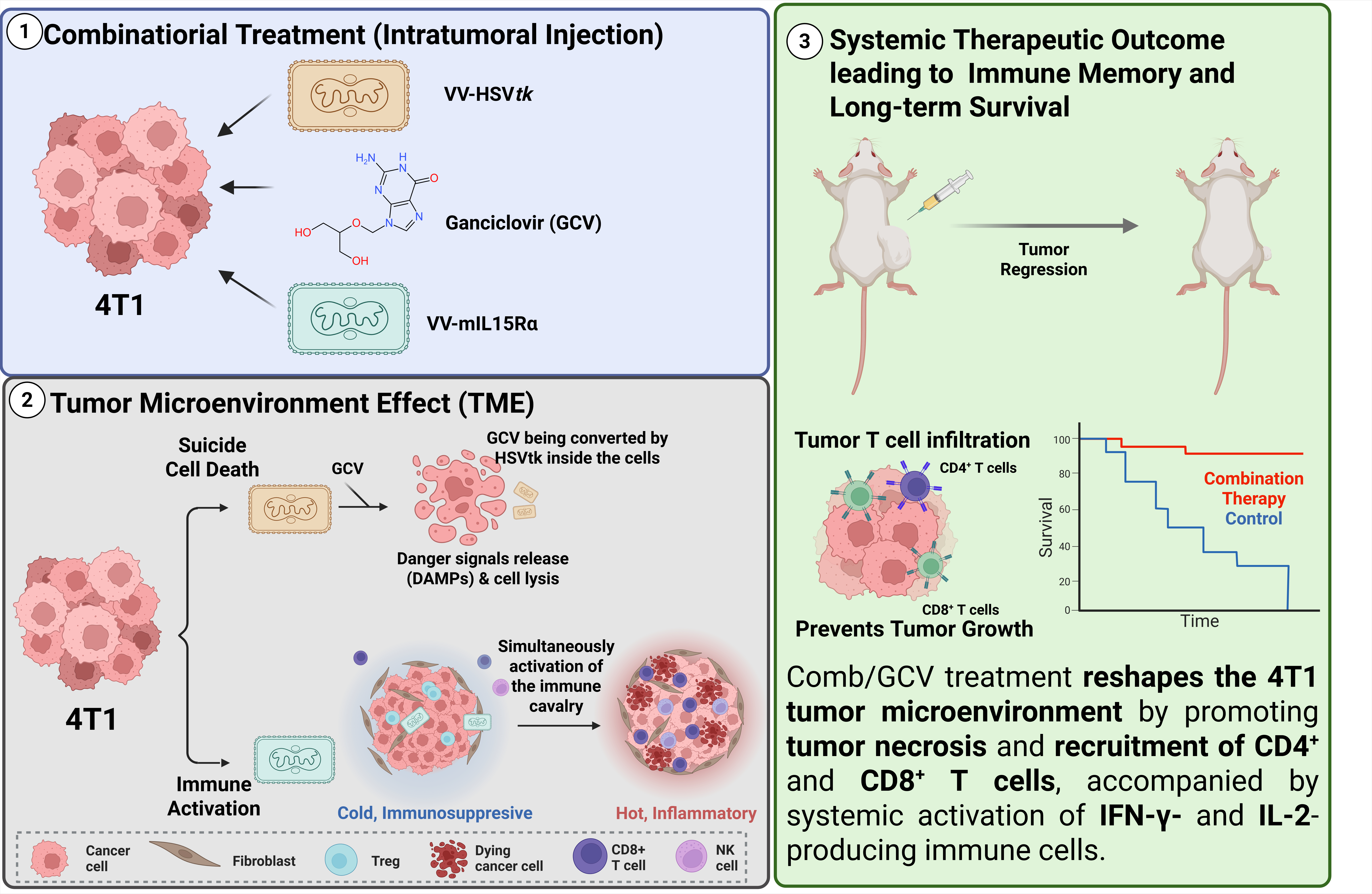
Oncolytic virotherapy offers a promising avenue for solid tumor treatment, yet single-agent approaches are frequently limited by insufficient tumor lysis and inadequate immune activation. Here we report that combined therapy with two recombinant variants of the oncolytic vaccinia virus, armed with either herpes simplex virus thymidine kinase (VV-HSVtk) or the interleukin 15 receptor subunit alpha (VV-mIL15Rα), induces enhanced cytotoxicity and immune stimulation in a murine mammary adenocarcinoma model (4T1). In vitro, VV-HSVtk exhibited dose-dependent cytotoxicity markedly potentiated by ganciclovir (GCV) through HSVtk-mediated phosphorylation into a cytotoxic nucleoside analogue, and co-culture of VV-infected tumor cells with donor-derived NK cells further amplified oncolytic efficiency. In vivo, combined treatment with VV-HSVtk, VV-mIL15Rα, and GCV resulted in significant tumor regression and extended survival relative to monotherapy controls in 4T1 syngeneic mice. Histological examination revealed robust lymphocytic infiltration at tumor sites and absence of hepatic or splenic toxicity. Mechanistically, VV-mIL15Rα-driven IL-15 trans-presentation amplified immune cell activation, while VV-HSVtk/GCV provided targeted tumor debulking and immunogenic cell death, collectively reshaping the immunosuppressive tumor microenvironment. These findings establish a multimodal oncolytic platform combining direct viral cytotoxicity, suicide gene therapy, and cytokine-mediated immune co-stimulation as a translatable strategy for treating solid tumors.