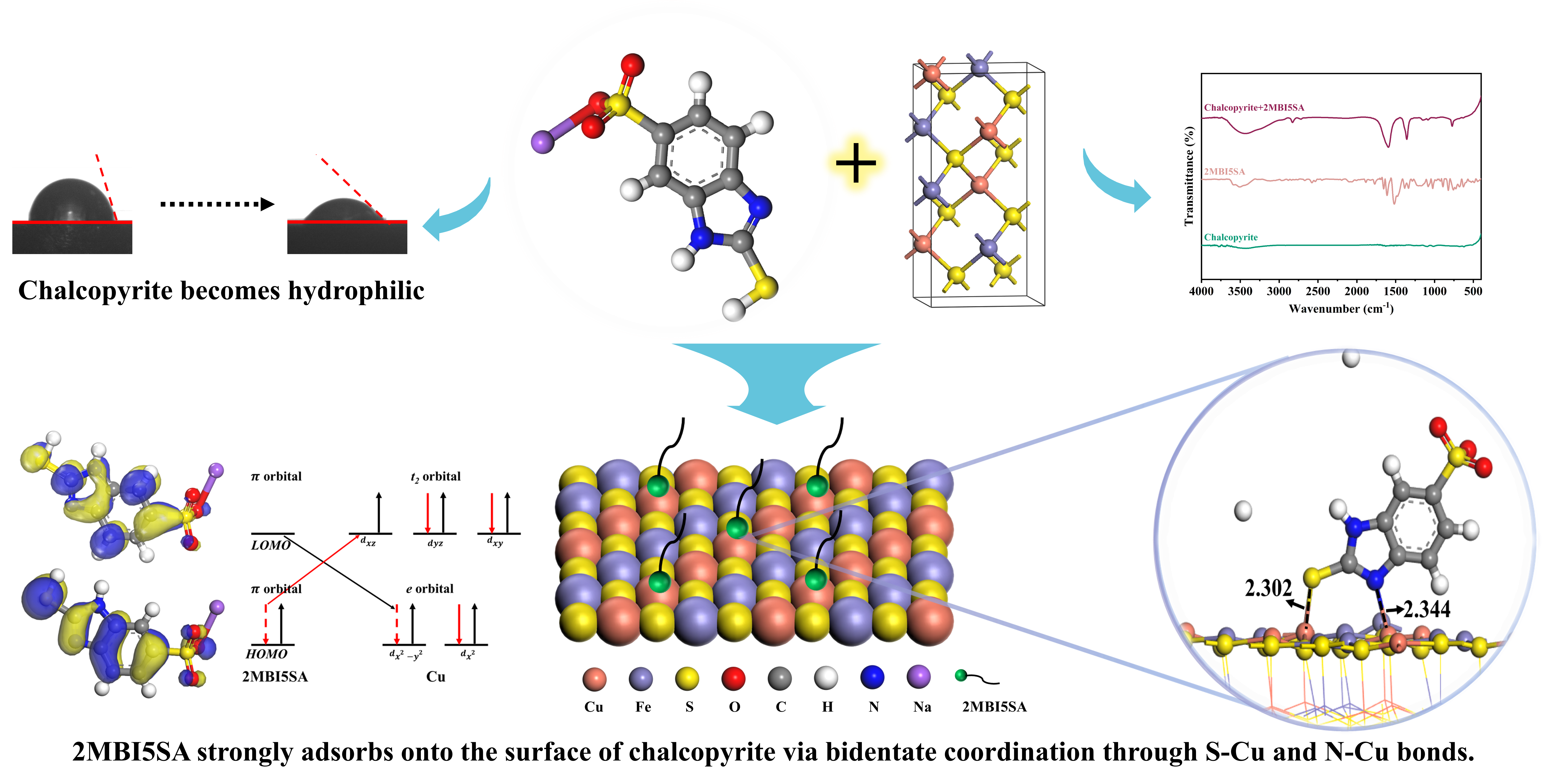
Chalcopyrite and molybdenite exhibit similar surface wettability and high floatability, which has long hindered their efficient and selective separation in mineral processing. In this work, the novel chalcopyrite depressant 2-mercapto-5-benzoimidazole sulfonate dihydrate (2MBI5SA) was investigated for its effect on the flotation behavior of chalcopyrite and molybdenite. Compared with the conventional depressant sodium sulfide (Na2S), 2MBI5SA exhibited stronger selective depression toward chalcopyrite; under conditions yielding Mo recovery of 81.46% and a Mo grade of 4.46%, the Cu recovery decreased to 13.03%. To clarify the origin of this selectivity, interfacial properties were systematically characterized using adsorption measurements, contact angle measurements, zeta potential measurements, FT-IR, XPS, and SEM-EDS, and the adsorption mechanism was further elucidated using SCC-DFTB calculations. The results demonstrate that 2MBI5SA chemisorbs onto the chalcopyrite surface via bidentate coordination, forming a stable adsorption layer that effectively suppresses chalcopyrite flotation. Moreover, structure-function relationship analysis confirmed that introducing hydrophilic and ionizable functional groups into the collector framework can convert a collector into a selective depressant, thereby providing new insight into the design of environmentally benign flotation depressants.