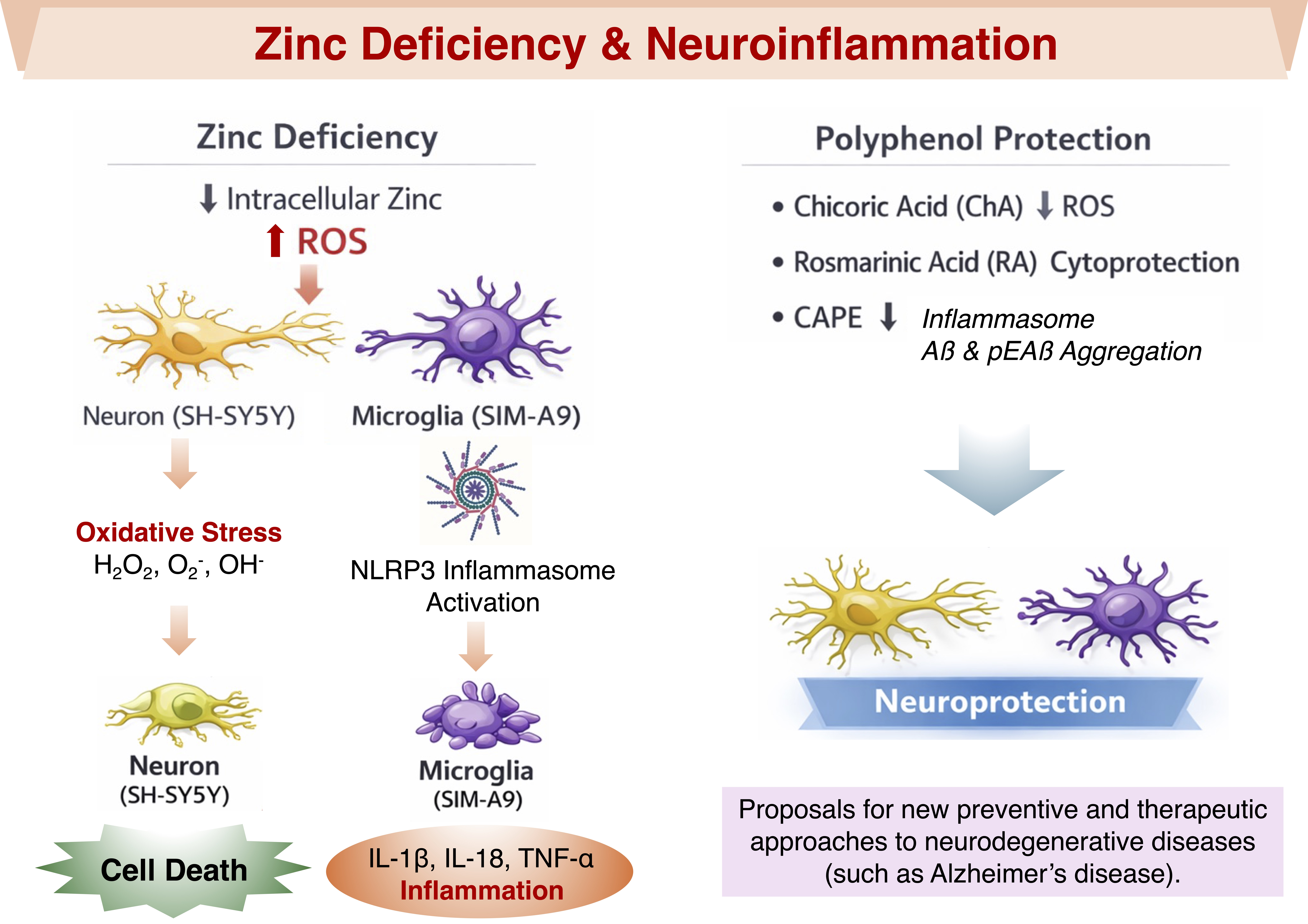
Zinc deficiency is increasingly recognized as a risk factor for neurodegenerative diseases, yet the underlying molecular mechanisms remain incompletely understood. In this study, we investigated the impact of intracellular zinc depletion on oxidative stress and in-flammasome activation in microglial (SIM-A9) and neuronal (SH-SY5Y) cell models, and evaluated the protective effects of polyphenolic compounds. Intracellular zinc chelation with the membrane-permeable chelator TPEN markedly increased reactive oxygen species (ROS) production, reduced cell viability, and upregulated the mRNA expression of NLRP3 inflammasome–related genes and pro-inflammatory cytokines. In contrast, extracellular zinc chelation had no effect, highlighting the critical role of intracellular zinc homeostasis in maintaining redox balance. Zinc supplementation significantly attenuated these responses. Among 32 polyphenols screened by DPPH radical scavenging assay, caffeic acid derivatives—chicoric acid (ChA), rosmarinic acid (RA), and caffeic acid phenethyl ester (CAPE)—exhibited the most potent antioxidant activity, surpassing that of edaravone. These compounds suppressed ROS production and differentially protected against zinc deficiency–induced cellular damage. ChA showed the strongest ROS in-hibitory activity (IC50: 1.9 µM in SIM-A9), RA provided robust cytoprotection even at low concentrations, and CAPE most effectively suppressed inflammasome-related gene ex-pression and inhibited aggregation of both Aβ1–42 and the highly neurotoxic py-roglutamate-modified variant pEAβ3–42. These findings demonstrate that intracellular zinc deficiency drives ROS-dependent NLRP3 inflammasome activation, and suggest that caffeic acid derivative polyphenols may serve as complementary agents for mitigating neuroinflammatory and amyloidogenic processes relevant to Alzheimer's disease.