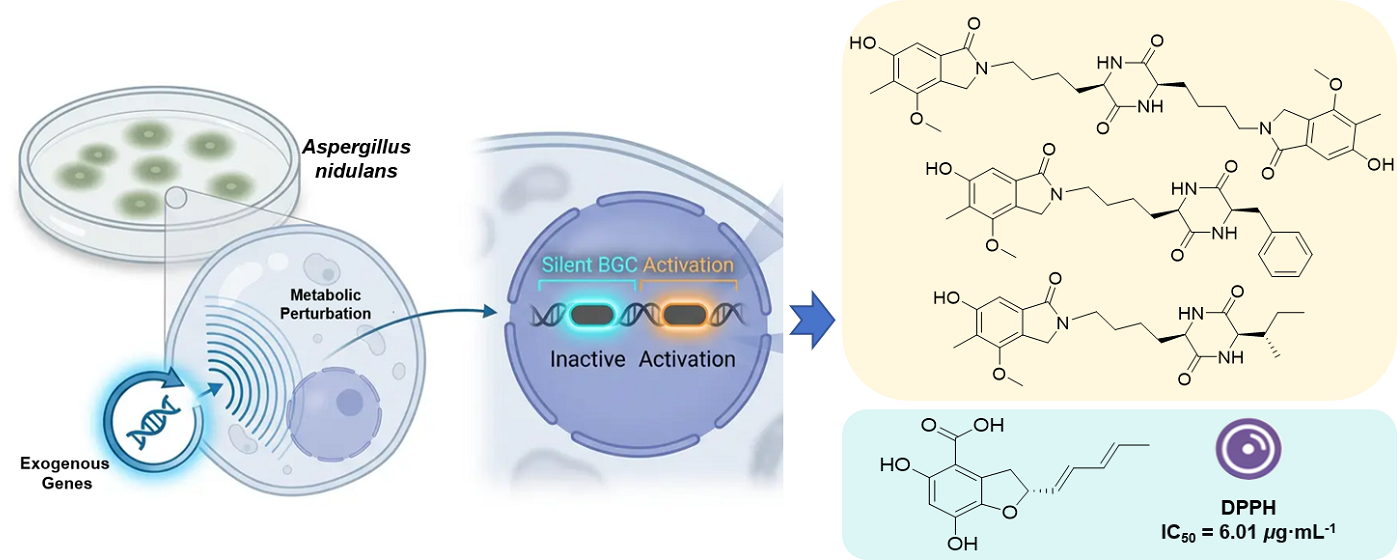
Aspergillus nidulans, a model filamentous fungus endowed with well-established genetic tools and a repertoire of cryptic secondary metabolite biosynthetic gene clusters (BGCs), is extensively exploited as a microbial chassis for heterologous biosynthesis. Mining of its secondary metabolites facilitates the discovery of novel bioactive compounds and the development and application of chassis cells. In the course of heterologous expression of exogenous genes in A. nidulans, we unexpectedly observed the activation of cryptic host BGCs, which resulted in substantial alterations to its secondary metabolic profile. Four previously undescribed compounds (1–4), together with six known analogs (5–10), were isolated from three recombinant A. nidulans strains. Notably, compounds 1–3 are the first naturally occurring examples of diketopiperazine-isoindolinone hybrid alkaloids, while compound 4 is a previously unreported benzofuran carboxylic acid derivative. Their structures and absolute configurations were assigned by interpretation of a combination of spectroscopic data and electronic circular dichroism calculations. Compounds 4 and 5 exhibited potent DPPH radical scavenging activity (IC50, 6.01 and 7.00 μg·mL-1, respectively). This study uncovers a "metabolic perturbation" effect on the host metabolic network during heterologous expression and offers a new strategy for activating silent gene clusters and discovering novel natural products through genetic manipulation.