Submitted:
01 May 2026
Posted:
05 May 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Sites
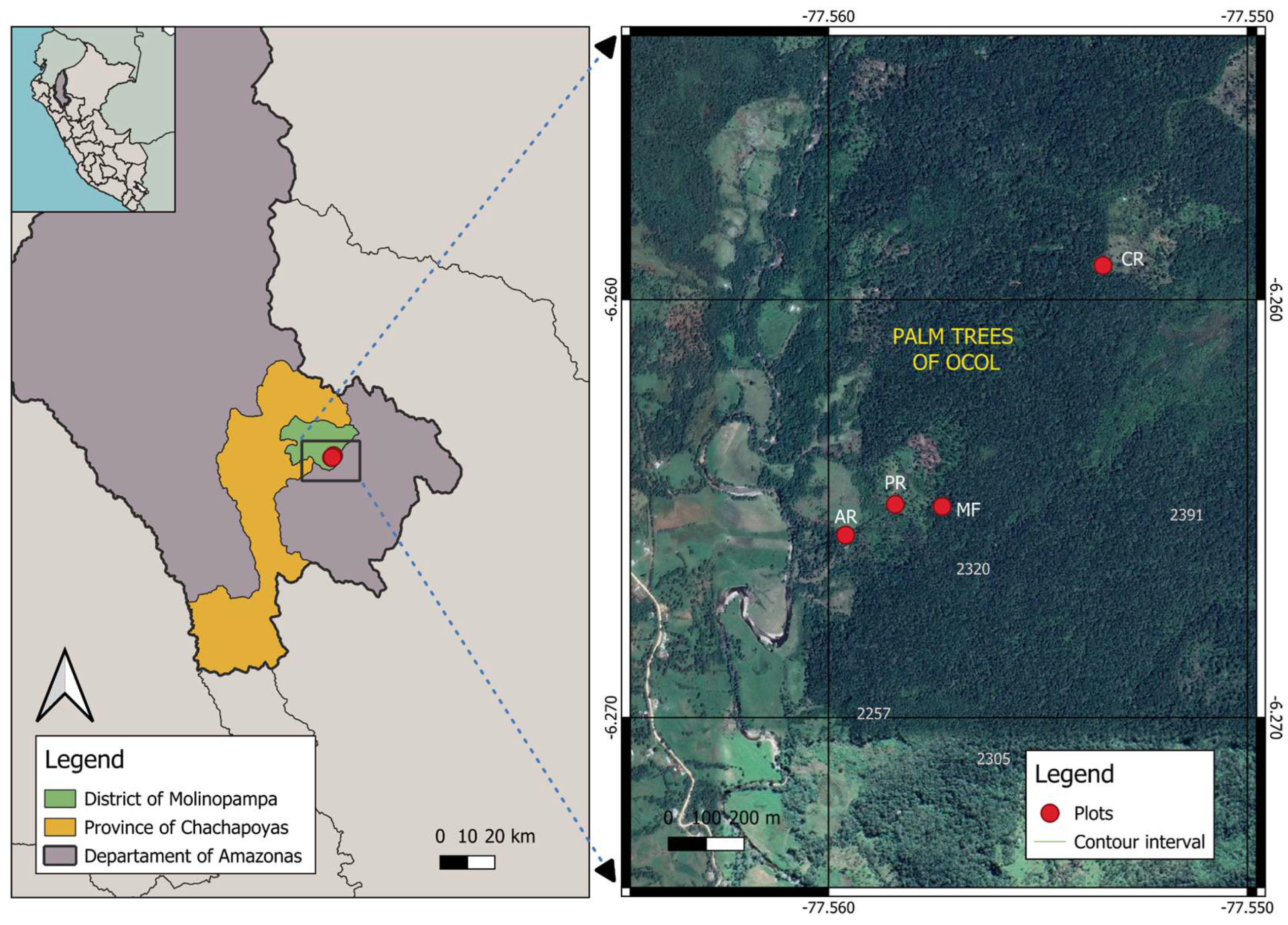

2.2. Sonic Tomography

2.3. Data Analysis
3. Results
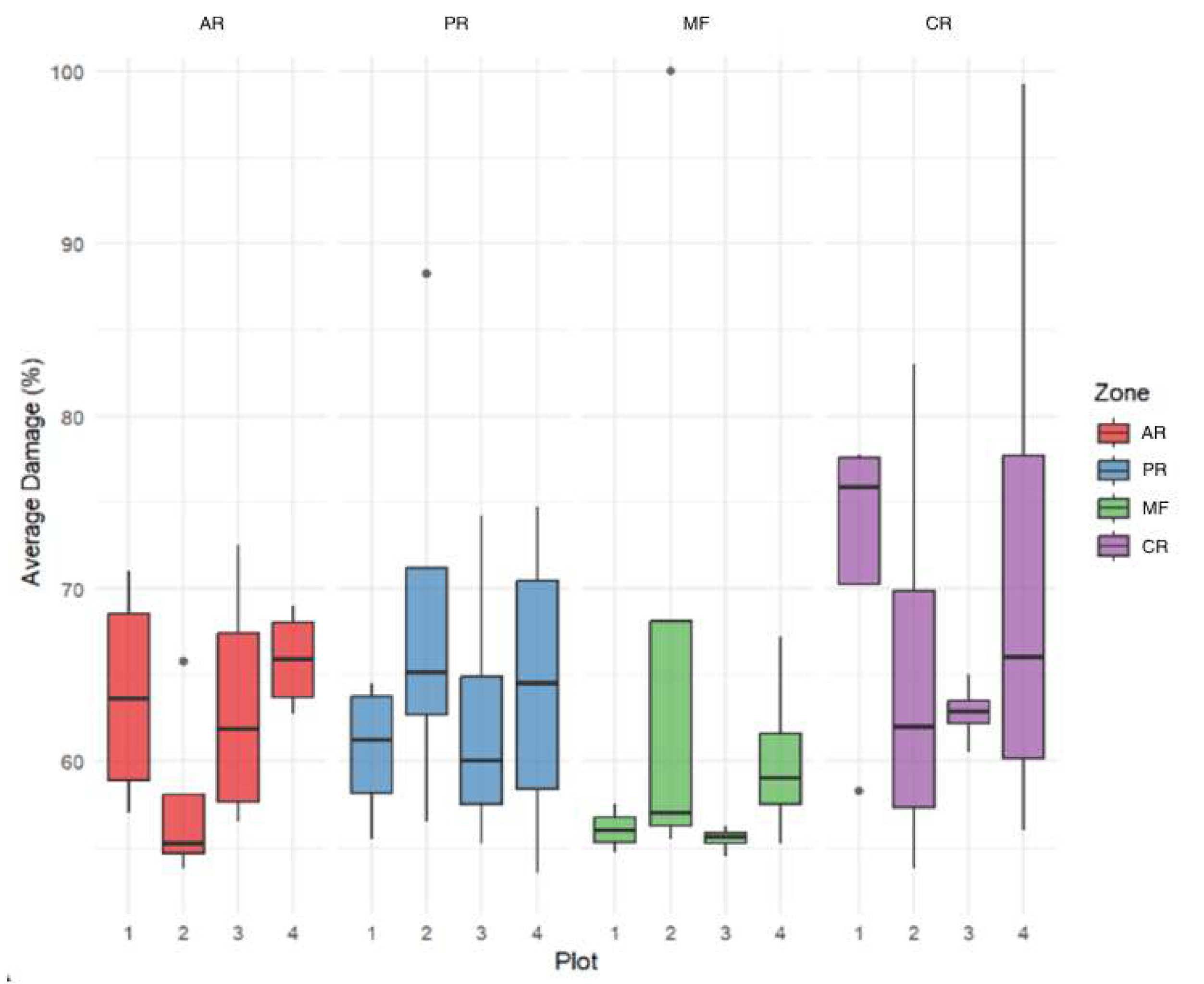
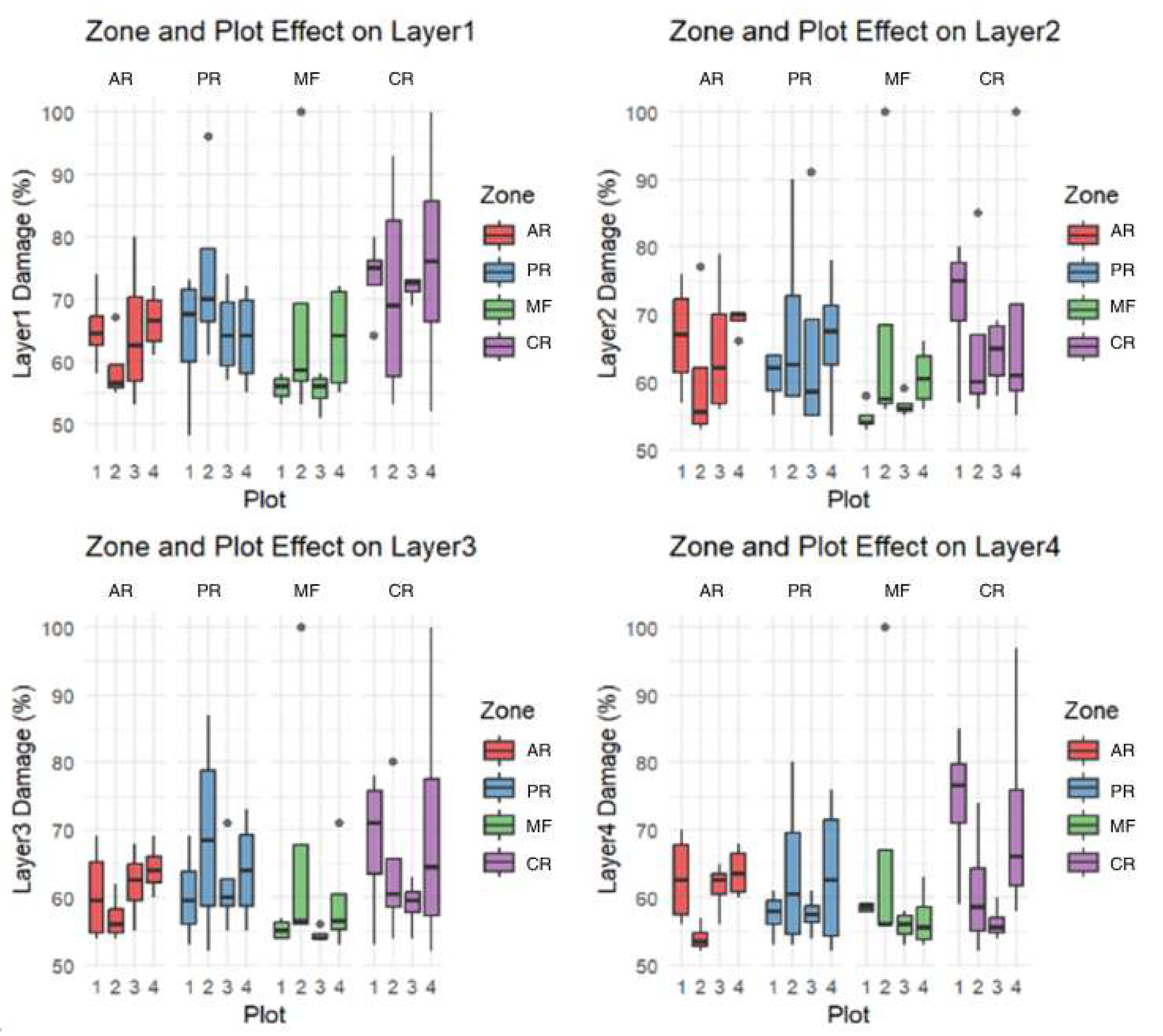
3.1. Tomograms and Level of Wood Decay
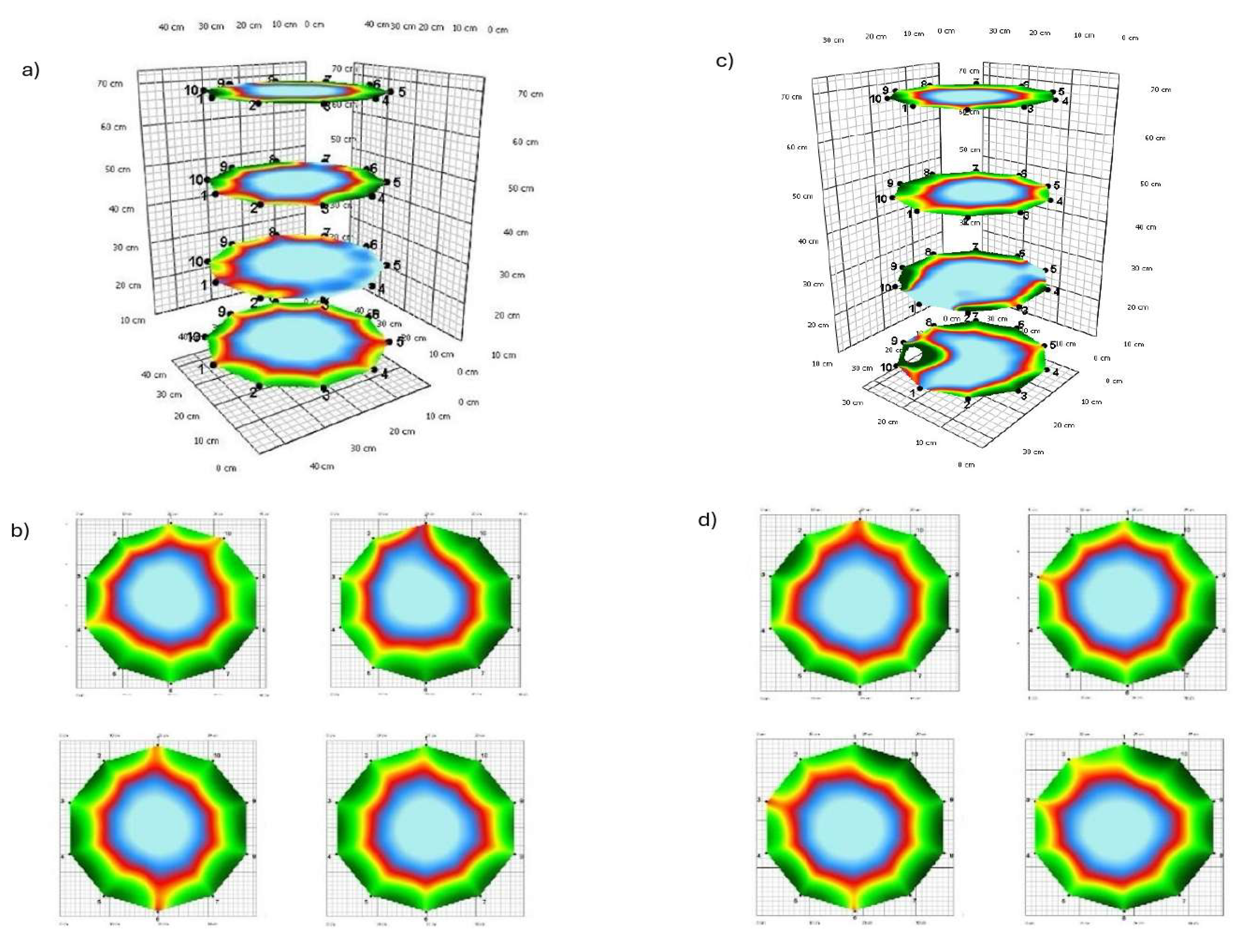
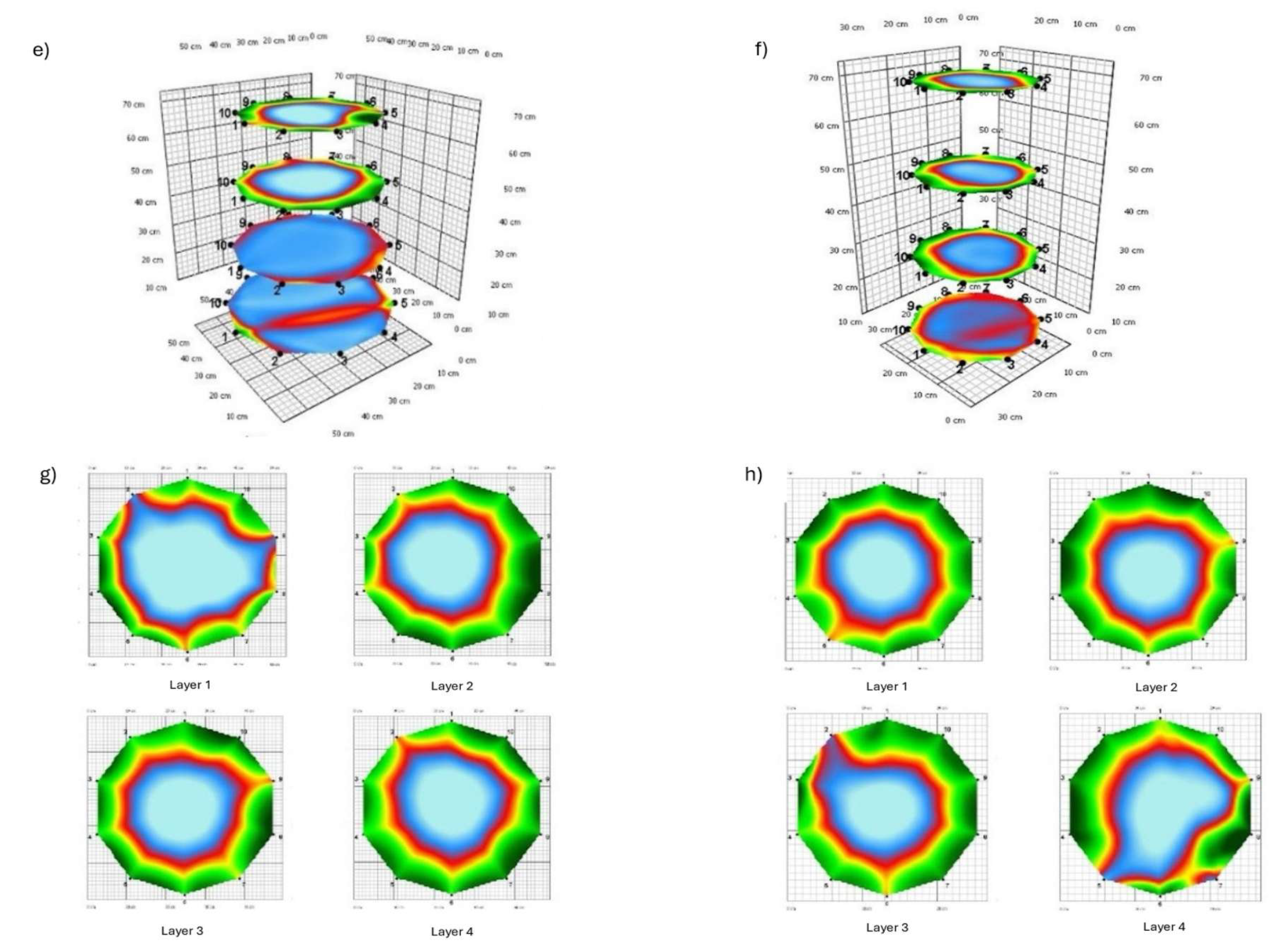
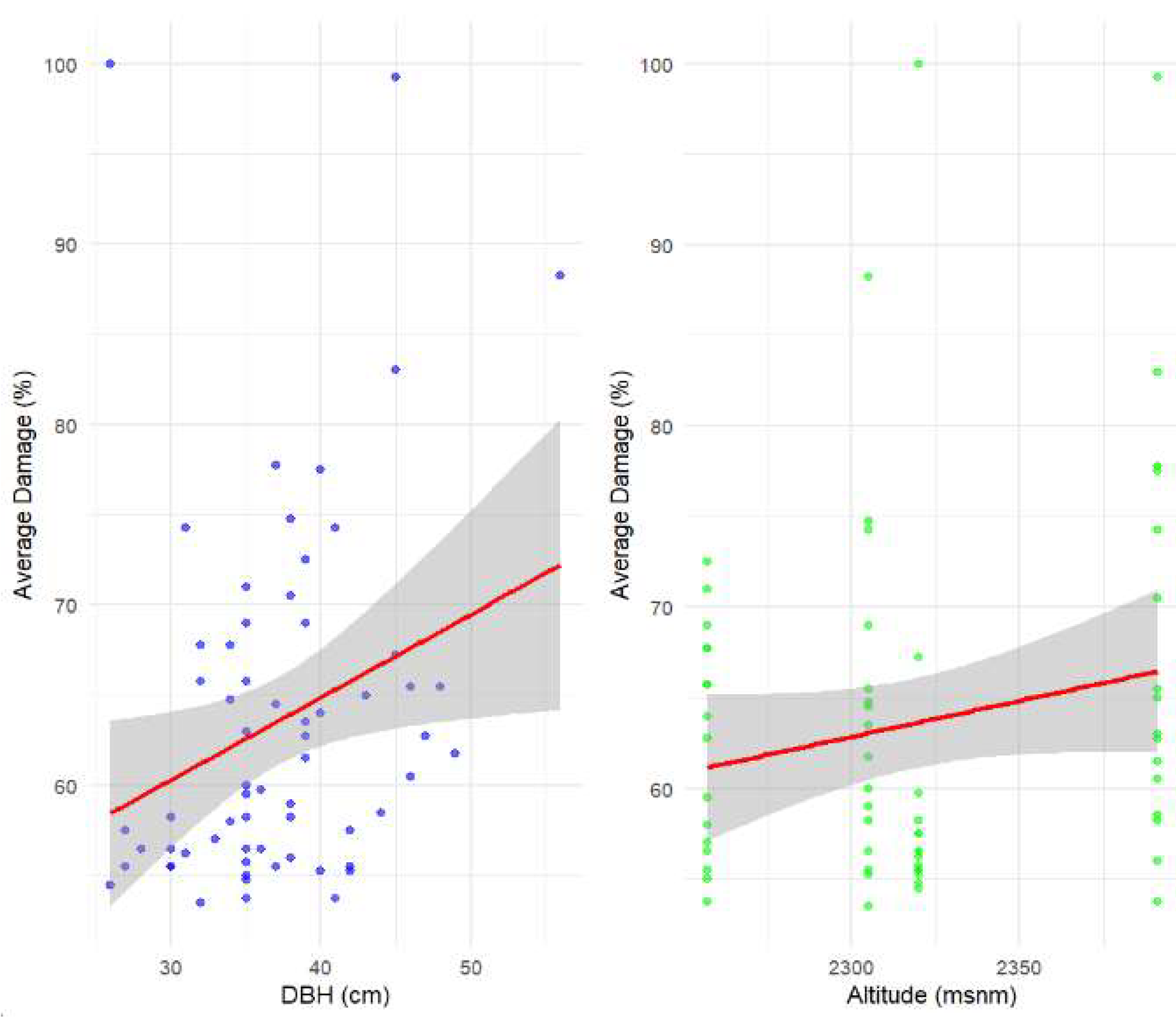
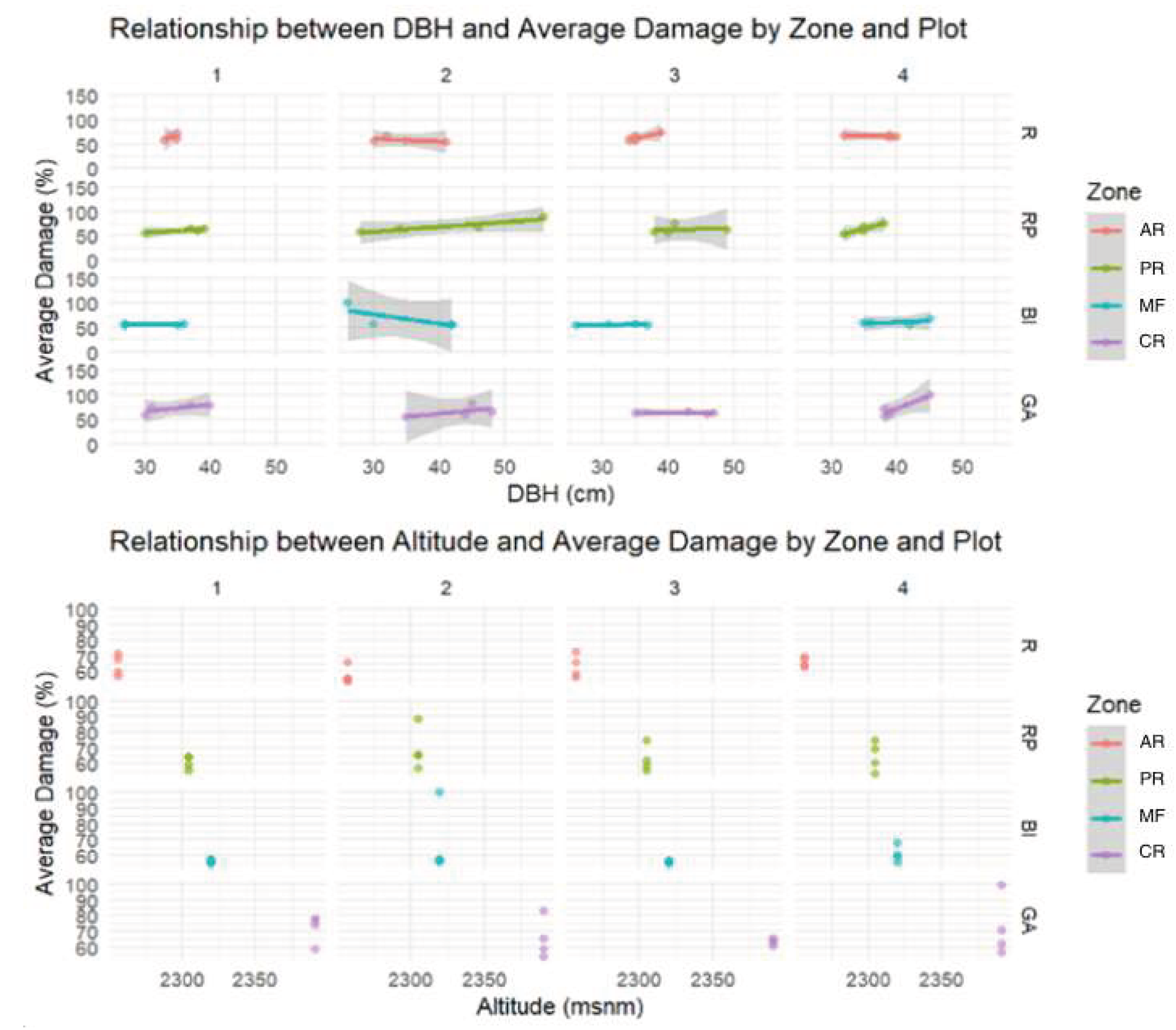
3.2. Analysis Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Baker, W.J.; Dransfield, J. Beyond Genera Palmarum: Progress and prospects in palm systematics. Bot. J. Linn. Soc. 2016, 182, 207–233. [Google Scholar] [CrossRef]
- Pintaud, J.C.; Galeano, G.; Balslev, H.; Bernal, R.; Borchsenius, F.; Ferreira, E.; de Granville, J.J.; Mejía, K.; Millán, B.; Moraes, M.; et al. Las palmeras de América del Sur: Diversidad, distribución e historia evolutiva. Rev. Peru. Biol. 2008, 15, 7–29. [Google Scholar] [CrossRef]
- Quinteros-Gómez, Y.; Zarco-González, M.; Ticerán, D.; Agramont, A.; Monroy-Vilchis, O. Effects of human disturbance on above-ground carbon stocks in north-west Amazonian Mauritia flexuosa peat swamp forests. Mires Peat 2023, 29, 5. [Google Scholar] [CrossRef]
- Muscarella, R.; Emilio, T.; Phillips, O.L.; Lewis, S.L.; Slik, F.; Baker, W.J.; Couvreur, T.L.P.; Eiserhardt, W.L.; Svenning, J.C.; Affum-Baffoe, K.; et al. The global abundance of tree palms. Glob. Ecol. Biogeogr. 2020, 29, 1495–1514. [Google Scholar] [CrossRef]
- Dransfield, J.; Uhl, N.W.; Asmussen, C.B.; Baker, W.J.; Harley, M.M.; Lewis, C.E. Genera Palmarum: The Evolution and Classification of Palms; Kew Publishing: Richmond, UK, 2008. [Google Scholar]
- Eiserhardt, W.L.; Svenning, J.C.; Kissling, W.D.; Balslev, H. Geographical ecology of the palms (Arecaceae): Determinants of diversity and distributions across spatial scales. Ann. Bot. 2011, 108, 1391–1416. [Google Scholar] [CrossRef]
- Henderson, A. Evolution and Ecology of Palms; New York Botanical Garden Press: New York, NY, USA, 2002. [Google Scholar]
- Zona, S.; Henderson, A. A review of animal-mediated seed dispersal of palms. Selbyana 1989, 11, 6–21. [Google Scholar]
- Agostini-Costa, T.S. Bioactive compounds and health benefits of some palm species traditionally used in Africa and the Americas - A review. J. Ethnopharmacol. 2018, 224, 202–229. [Google Scholar] [CrossRef]
- Quinteros-Gómez, Y.M.; Monroy-Vilchis, O.; Zarco-González, M.M.; Endara-Agramont, A.R.; Pacheco, X.P. Floristic composition, structure and species conservation status of Mauritia flexuosa palm swamps in Andean-Amazonian piedmont in the Department of San Martín, Peru. Rev. Mex. Biodivers. 2021, 92, e923186. [Google Scholar] [CrossRef]
- Sanín, M.J.; Galeano, G. A revision of the Andean wax palms, Ceroxylon (Arecaceae). Phytotaxa 2011, 34, 1–64. [Google Scholar] [CrossRef]
- Valdivia, M.I.V.; Gamarra, L.V. A new species of Ceroxylon (Arecaceae) from Cordillera Azul National Park—Peru. Phytotaxa 2021, 483, 267–276. [Google Scholar] [CrossRef]
- Santa Cruz, L.; Pintaud, J.-C.; Rojas-Fox, V.; Ramírez, R.; Rodríguez Rodríguez, E.F. Inventario de las palmeras de la vertiente occidental del Perú. Arnaldoa 2018, 25, 857–876. [Google Scholar] [CrossRef]
- Suárez, D.; Mejía, A.; Marín, O.; Agudelo, L.D. Proposal for nutritional management in Palma of cera (Ceroxylon quindiuense wendl.) low growth condition in vehicular high flow vehicle mesh. J. Res. Eng. Sci. 2016, 1, 163. [Google Scholar] [CrossRef]
- Benítez, Á.; Ramón, P.; Sarango, M.; Torracchi-Carrasco, E. Spatial patterns of Ceroxylon parvifrons (Engel) H. Wendl of the montane forests in Southern Ecuador. Int. J. For. Res. 2022, 2022, 5707906. [Google Scholar] [CrossRef]
- Huisman, S.N.; Raczka, M.F.; McMichael, C.N.H. Palm phytoliths of mid-elevation Andean forests. Front. Ecol. Evol. 2018, 6, 193. [Google Scholar] [CrossRef]
- Sanín, M.J.; Kissling, W.D.; Bacon, C.D.; Borchsenius, F.; Galeano, G.; Svenning, J.C.; de Granville, J.J.; Mejía, K.; Pintaud, J.C. The Neogene rise of the tropical Andes facilitated diversification of wax palms (Ceroxylon: Arecaceae) through geographical colonization and climatic niche separation. Bot. J. Linn. Soc. 2016, 182, 303–317. [Google Scholar] [CrossRef]
- Montúfar, R.; Anthelme, F.; Pintaud, J.C.; Balslev, H. Disturbance and resilience in tropical American palm populations and communities. Bot. Rev. 2011, 77, 426–461. [Google Scholar] [CrossRef]
- Galeano, G.; Bernal, R. Palmas (familia Arecaceae o Palmae). In Libro Rojo de Plantas de Colombia; Calderón, E., Galeano, G., García, N., Eds.; Instituto de Ciencias Naturales-Universidad Nacional de Colombia: Bogotá, Colombia, 2005; Volume 2, pp. 59–224. [Google Scholar]
- Anthelme, F.; Lincango, J.; Gully, C.; Duarte, N.; Montúfar, R. How anthropogenic disturbances affect the resilience of a keystone palm tree in the threatened Andean cloud forest? Biol. Conserv. 2011, 144, 1059–1067. [Google Scholar] [CrossRef]
- Balslev, H.; Copete, J.C.; Pedersen, D.; Bernal, R.; Galeano, G.; Duque, Á.; Sanín, M.J. Palm diversity and abundance in the Colombian Amazon. In Forest Structure, Function and Dynamics in Western Amazonia; Mistretta, P., Ed.; Wiley-Blackwell: Hoboken, NJ, USA, 2015; pp. 101–123. [Google Scholar]
- Barmpoutis, P.; Papaioannou, P.; Dimitropoulos, K.; Grammalidis, N. A Review on Early Forest Fire Detection Systems Using Optical Remote Sensing. Sensors 2020, 20, 6442. [Google Scholar] [CrossRef]
- Gao, B.; Jia, W.; Wang, Q.; Yang, G. All-weather forest fire automatic monitoring and early warning application based on multi-source remote sensing data: Case study of Yunnan. Fire 2025, 8, 344. [Google Scholar] [CrossRef]
- Quinteros-Gómez, Y.; Salinas-Inga, A.; Macedo-Bedoya, J.; Peralta-Alcantara, E.; La Rosa-Sánchez, M.; Camones-Gonzales, F.; Yamunaque, A.; Angeles-Alvarez, F.; Gómez-Ticerán, D.; Solano-Dávila, O.L. Noninvasive sonic tomography for the detection of internal defects in relict woodlands of Polylepis in Peru. Forests 2025, 16, 957. [Google Scholar] [CrossRef]
- Kurbanov, E.; Vorobev, O.; Lezhnin, S.; Sha, J.; Wang, J.; Li, X.; Cole, J.; Dergunov, D.; Wang, Y. Remote sensing of forest burnt area, burn severity, and post-fire recovery: A review. Remote Sens. 2022, 14, 4714. [Google Scholar] [CrossRef]
- Vasilakos, C.; Tsekouras, G.E.; Kavroudakis, D. LSTM-based prediction of Mediterranean vegetation dynamics using NDVI time-series data. Land 2022, 11, 923. [Google Scholar] [CrossRef]
- Adão, T.; Hruška, J.; Pádua, L.; Bessa, J.; Peres, E.; Morais, R.; Sousa, J.J. Hyperspectral imaging: A review on UAV-based sensors, data processing and applications for agriculture and forestry. Remote Sens. 2017, 9, 1110. [Google Scholar] [CrossRef]
- Liu, J.; Feng, Z.; Mannan, A.; Khan, T.U.; Cheng, Z. Comparing non-destructive methods to estimate volume of three tree taxa in Beijing, China. Forests 2019, 10, 92. [Google Scholar] [CrossRef]
- Lee, B.J.; Son, S.; Jung, J.K.; Park, Y. Non-invasive assessment of the internal condition of urban trees infested by two cerambycid beetles, Aromia bungii and Massicus raddei, using sonic tomography. Forests 2024, 15, 1231. [Google Scholar] [CrossRef]
- Montoya-Rojas, R. Análisis multitemporal de cambio de uso del suelo y cobertura vegetal en el área de conservación privada bosques de palmera, Ocol - Amazonas, período 1987-2017. Undergraduate Thesis, Universidad Nacional Toribio Rodríguez de Mendoza de Amazonas, Chachapoyas, Peru, 2020. [Google Scholar]
- Bruijnzeel, L.A.; Scatena, F.N.; Hamilton, L.S. Tropical Montane Cloud Forests: Science for Conservation and Management; Cambridge University Press: Cambridge, UK, 2011. [Google Scholar]
- Galeano, G.; Bernal, R. Palms of the Americas; Princeton University Press: Princeton, NJ, USA, 2010. [Google Scholar]
- Luis. Falta referencia; 2020. [Google Scholar]
- Young, K.R.; León, B. Peru’s Humid Eastern Montane Forests: An Overview of Their Physical Settings, Biological Diversity, Human Use and Settlement, and Conservation Needs; DIVA Technical Report 5; Centre for Research on the Cultural and Biological Diversity of Andean Rainforests (DIVA): Rønde, Denmark, 1999. [Google Scholar]
- Burcham, D.C.; Brazee, N.J.; Marra, R.E.; Kane, B. Geometry matters for sonic tomography of trees. Trees 2023, 37, 335–347. [Google Scholar] [CrossRef]
- Ross, R.J. Nondestructive Evaluation of Wood: Second Edition; General Technical Report FPL-GTR-238; USDA Forest Service, Forest Products Laboratory: Madison, WI, USA, 2015. [Google Scholar]
- Gilbert, E.A.; Smiley, E.T. Picus sonic tomography for the quantification of decay in white oak (Quercus alba) and hickory (Carya spp.). J. Arboric. 2004, 30, 277–281. [Google Scholar] [CrossRef]
- Lin, C.J.; Chang, T.T.; Juan, M.Y. Detecting deterioration in royal palm (Roystonea regia) using ultrasonic tomographic and resistance microdrilling techniques. J. Trop. For. Sci. 2011, 23, 37–45. [Google Scholar]
- Wang, X. Acoustic measurements on trees and logs: A review and analysis. Wood Sci. Technol. 2013, 47, 965–975. [Google Scholar] [CrossRef]
- Gilbert, G.S.; Ballesteros, J.O.; Barrios-Rodriguez, C.A.; Bonadies, E.F.; Cedeño-Sánchez, M.L.; Fossatti-Caballero, N.J.; Trejos-Rodríguez, M.M.; Pérez-Suñiga, J.M.; Holub-Young, K.S.; Henn, L.A.W.; et al. Use of sonic tomography to detect and quantify wood decay in living trees. Appl. Plant Sci. 2016, 4, 1600060. [Google Scholar] [CrossRef] [PubMed]
- Divos, F.; Divos, P. Resolution of stress wave based acoustic tomography. In Proceedings of the 14th International Symposium on Nondestructive Testing of Wood, Hannover, Germany, 28–30 September 2005; pp. 309–314. [Google Scholar]
- Wang, X.; Allison, R.B. Decay detection in red oak trees using a combination of visual inspection, acoustic testing, and resistance microdrilling. Arboric. Urban For. 2008, 34, 1–4. [Google Scholar] [CrossRef]
- Helmanto, H.; Kristiati, E.; Wardhani, F.F.; Zulkarnaen, R.N.; Sahromi; Mujahidin; Rachmadiyanto, A.N.; Abdurachman. Tree health assessment of Agathis borneensis Warb. in Bogor Botanical Garden using arborsonic. IOP Conf. Ser. Earth Environ. Sci. 2018, 203, 012032. [Google Scholar] [CrossRef]
- Bates, D.; Mächler, M.; Bolker, B.; Walker, S. Fitting Linear Mixed-Effects Models Using lme4. J. Stat. Softw. 2015, 67, 1–48. [Google Scholar] [CrossRef]
- Wickham, H. ggplot2: Elegant Graphics for Data Analysis; Springer: New York, NY, USA, 2016; ISBN 978-3-319-24277-4. [Google Scholar]
- Wickham, H.; François, R.; Henry, L.; Müller, K.; Vaughan, D. dplyr: A Grammar of Data Manipulation . R package version 1.1.4. 2023. Available online: https://CRAN.R-project.org/package=dplyr.
- Kuznetsova, A.; Brockhoff, P.B.; Christensen, R.H.B. lmerTest Package: Tests in Linear Mixed Effects Models. J. Stat. Softw. 2017, 84, 1–26. [Google Scholar] [CrossRef]
- Tomlinson, P.B.; Horn, J.W.; Fisher, J.B. The Anatomy of Palms: Arecaceae—Palmae; Oxford University Press: Oxford, UK, 2011. [Google Scholar]
- Parthsarathy, M.V.; Klotz, L.H. Palm wood I. Anatomical aspects. Wood Sci. Technol. 1976, 10, 215–229. [Google Scholar] [CrossRef]
- Sainz-Resendiz, B.A.; Estrada-Ruiz, E.; Mateo-Cid, L.E.; Porras-Múzquiz, H. Primer registro de un estípite de Coryphoideae: Palmoxylon kikaapoa de la Formación Olmos del Cretácico Superior, Coahuila, México. Rev. Mex. Biodivers. 2015, 86, 872–881. [Google Scholar] [CrossRef]
- Zimmermann, M.H.; Tomlinson, P.B. Analysis of the complex vascular system of palms: Arecaceae. Science 1966, 152, 72–73. [Google Scholar] [CrossRef] [PubMed]
- Najmie, M.; Khalid, K.; Aziz, S.; Jusoh, A. Density and ultrasonic characterization of oil palm trunk infected by Ganoderma boninense disease. Meas. Sci. Rev. 2011, 11, 160–164. [Google Scholar] [CrossRef]
- Taban, E.; Khavanin, A.; Ohadi, A.; Putra, A.; Jafari, A.; Faridan, M.; Soleimanian, A. Study on the acoustic characteristics of natural date palm fibres: Experimental and theoretical approaches. Build. Environ. 2019. [Google Scholar] [CrossRef]
- Smiley, E.T.; Matheny, N.; Lilly, S. Best management practices: tree risk assessment; International Society of Arboriculture: Champaign (IL), 2011. [Google Scholar]
- Costa, M.W.; et al. Detection of internal decay in palm trees using sonic tomography. Arboric. Urban For. 2019, 45, 110–122. [Google Scholar]
- Schubert, S.; Gsell, D.; Dual, J.; Motavalli, M.; Niemz, P. Acoustic wood tomography on trees and the challenge of wood heterogeneity. Holzforschung 2009, 63, 107–112. [Google Scholar] [CrossRef]
- Alves, R.C.; Mantilla, J.N.; Bremer, C.F.; Carrasco, E.V. Application of acoustic tomography and ultrasonic waves to estimate stiffness constants of muiracatiara Brazilian wood. BioResources 2015, 10(1), 1845–56. [Google Scholar] [CrossRef]
- Anthelme, F.; Lincango, J.; Gully, C.; Duarte, N.; Montúfar, R. How anthropogenic disturbances affect the resilience of a keystone palm tree in the threatened Andean cloud forest? Forests 2022, 13(3), 1–14. [Google Scholar] [CrossRef]
- Agualzaca Caisaguano, D.O.; Llumitasig Quinatoa, M.S.; Cusquillo Quispillo, B.D.; Carranza Patiño, M.; Jiménez Romero, E.M. Propuesta de plan de manejo integral para Ceroxylon echinulatum basado en criterios ecológicos en la comunidad Mocata, Pallatanga (Ecuador). Multidiscip. Collab. J. 2025, 3, 227–245. [Google Scholar] [CrossRef]
- Bernal, R.; Sanín, M.J. The palm stands of Ceroxylon quindiuense (Arecaceae) in the Cocora Valley, Quindío: Perspectives on an iconic Colombian landscape. Colomb. For. 2013, 16, 67–79. [Google Scholar] [CrossRef]
- Bennett, A.C.; McDowell, N.G.; Allen, C.D.; Anderson-Teixeira, K.J. Larger trees suffer most during drought in forests worldwide. Nat. Plants 2015, 1, 15139. [Google Scholar] [CrossRef]
- Lindenmayer, D.B.; Laurance, W.F.; Franklin, J.F. Global decline in large old trees. Science 2012, 338, 1305–1306. [Google Scholar] [CrossRef]
- Sanín, M.J.; Anthelme, F.; Pintaud, J.C.; Galeano, G.; Bernal, R. Juvenile resilience and adult longevity explain residual populations of the Andean wax palm Ceroxylon quindiuense after deforestation. PLoS ONE 2013, 8, e74139. [Google Scholar] [CrossRef]
- Chacón-Vargas, K.; García-Merchán, V.H.; Sanín, M.J. From keystone species to conservation: Conservation genetics of wax palm Ceroxylon quindiuense in the largest wild populations of Colombia and selected neighboring ex situ plant collections. Biodivers. Conserv. 2020, 29, 283–302. [Google Scholar] [CrossRef]
- Kozák, D.; Svitok, M.; Zemlerová, V.; Mikoláš, M.; Lachat, T.; Larrieu, L.; Paillet, Y.; Buechling, A.; Bače, R.; Keeton, W.S.; et al. Importance of conserving large and old trees to continuity of tree-related microhabitats. Conserv. Biol. 2023, 37, e14066. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, G.S.; Faircloth, B.C.; Glenn, T.C.; Ballesteros, J.O.; Barrios-Rodríguez, C.A.; Bonadies, E.; Cedeño-Sánchez, M.L.; Fossatti-Caballero, N.J.; Pérez-Suñiga, J.M.; Trejos-Rodríguez, M.M.; et al. Hidden decay of live trees in a tropical rain forest. Ecology 2025, 106, e70208. [Google Scholar] [CrossRef]
- Deflorio, G.; Fink, S.; Schwarze, F.W.M.R. Detection of incipient decay in tree stems with sonic tomography after wounding and fungal inoculation. Wood Sci. Technol. 2008, 42, 117–132. [Google Scholar] [CrossRef]
- Li, P.; Feng, H.; Zhang, H.; Wang, X. Evaluation of internal decay in standing trees using sonic tomography and stress wave techniques. Sensors 2020, 20, 695. [Google Scholar] [CrossRef]
- Wang, X.; Allison, R.B.; Wang, L.; Ross, R.J. Acoustic Tomography for Decay Detection in Red Oak Trees . In Research Paper FPL-RP-642; USDA Forest Service, Forest Products Laboratory: Madison, WI, USA, 2004. [Google Scholar]
- Bucur, V. Acoustics of Wood, 2nd ed.; Springer: Berlin/Heidelberg, Germany, 2006. [Google Scholar] [CrossRef]
- Zellweger, F.; De Frenne, P.; Lenoir, J.; Vangansbeke, P.; Verheyen, K.; Bernhardt-Römermann, M.; Baeten, L.; Hédl, R.; et al. Forest microclimate dynamics drive plant responses to warming. Science 2020, 368, 772–775. [Google Scholar] [CrossRef]
- Saine, S.; Penttilä, R.; Fukami, T.; Furneaux, B.; Hytönen, T.; Miettinen, O.; Monkhouse, N.; et al. Idiosyncratic responses to biotic and environmental filters in wood-inhabiting fungal communities. Ecology 2025, 106, e70013. [Google Scholar] [CrossRef] [PubMed]
- Brazee, N.J.; Marra, R.E.; Göcke, L.; Van Wassenaer, P. Non-destructive assessment of internal decay in three hardwood species of northeastern North America using sonic and electrical impedance tomography. Forestry 2011, 84, 33–39. [Google Scholar] [CrossRef]
- Renton, K.; Salinas-Melgoza, A.; De Labra-Hernández, M.A.; de la Parra-Martínez, S.M. Resource requirements of parrots: Nest site selectivity and dietary plasticity of Psittaciformes. J. Ornithol. 2015, 156, 73–90. [Google Scholar] [CrossRef]
- Molina, D. Secularise to conserve: The history of the wax palm in Colombia. Plant Perspect. 2024, 1, 1–25. [Google Scholar] [CrossRef]
- Lindenmayer, D.B.; Laurance, W.F. The ecology, distribution, conservation and management of large old trees. Biol. Rev. 2017, 92, 1434–1458. [Google Scholar] [CrossRef] [PubMed]
- Jones, C.G.; Lawton, J.H.; Shachak, M. Organisms as ecosystem engineers. Oikos 1994, 69, 373–386. [Google Scholar] [CrossRef]
- De Frenne, P.; Zellweger, F.; Rodríguez-Sánchez, F.; Scheffers, B.R.; Hylander, K.; Luoto, M.; Fauset, S.; Verheyen, K.; Lenoir, J. Forest microclimates and climate change: Importance, drivers and future perspectives. Glob. Chang. Biol. 2021, 27, 2279–2297. [Google Scholar] [CrossRef]
- Lovejoy, T.E.; Nobre, C. Amazon tipping point. Sci. Adv. 2018, 4, eaat2340. [Google Scholar] [CrossRef] [PubMed]
- Seddon, N.; Smith, A.; Smith, P.; Key, I.; Chausson, A.; Girardin, C.; Cassidy, M.; Turner, B. Getting the message right on nature-based solutions to climate change. Glob. Chang. Biol. 2021, 27, 1518–1546. [Google Scholar] [CrossRef] [PubMed]
- Watson, J.E.M.; Evans, T.; Venter, O.; Williams, B.; Tulloch, A.; Stewart, C.; Thompson, I.; Ray, J.C.; Murray, K.; Salazar, A.; et al. The exceptional value of intact forest ecosystems. Nat. Ecol. Evol. 2018, 2, 599–610. [Google Scholar] [CrossRef]
- Griscom, B.W.; Adams, J.; Ellis, P.W.; Houghton, R.A.; Lomax, G.; Miteva, D.A.; Schlesinger, W.H.; Shoch, D.; Siikamäki, J.V.; Smith, P.; et al. Natural climate solutions. Proc. Natl. Acad. Sci. USA 2017, 114, 11645–11650. [Google Scholar] [CrossRef] [PubMed]
- Pan, Y.; Birdsey, R.A.; Fang, J.; Houghton, R.; Kauppi, P.E.; Kurz, W.A.; Phillips, O.L.; Shvidenko, A.; Lewis, S.L.; Canadell, J.G.; et al. A large and persistent carbon sink in the world’s forests. Science 2011, 333, 988–993. [Google Scholar] [CrossRef]
- Durlak, W.; Dudkiewicz-Pietrzyk, M.; Szot, P. Historic trees, modern tools: Innovative health assessment of a linden avenue in an urban environment. Sustainability 2025, 17, 9681. [Google Scholar] [CrossRef]
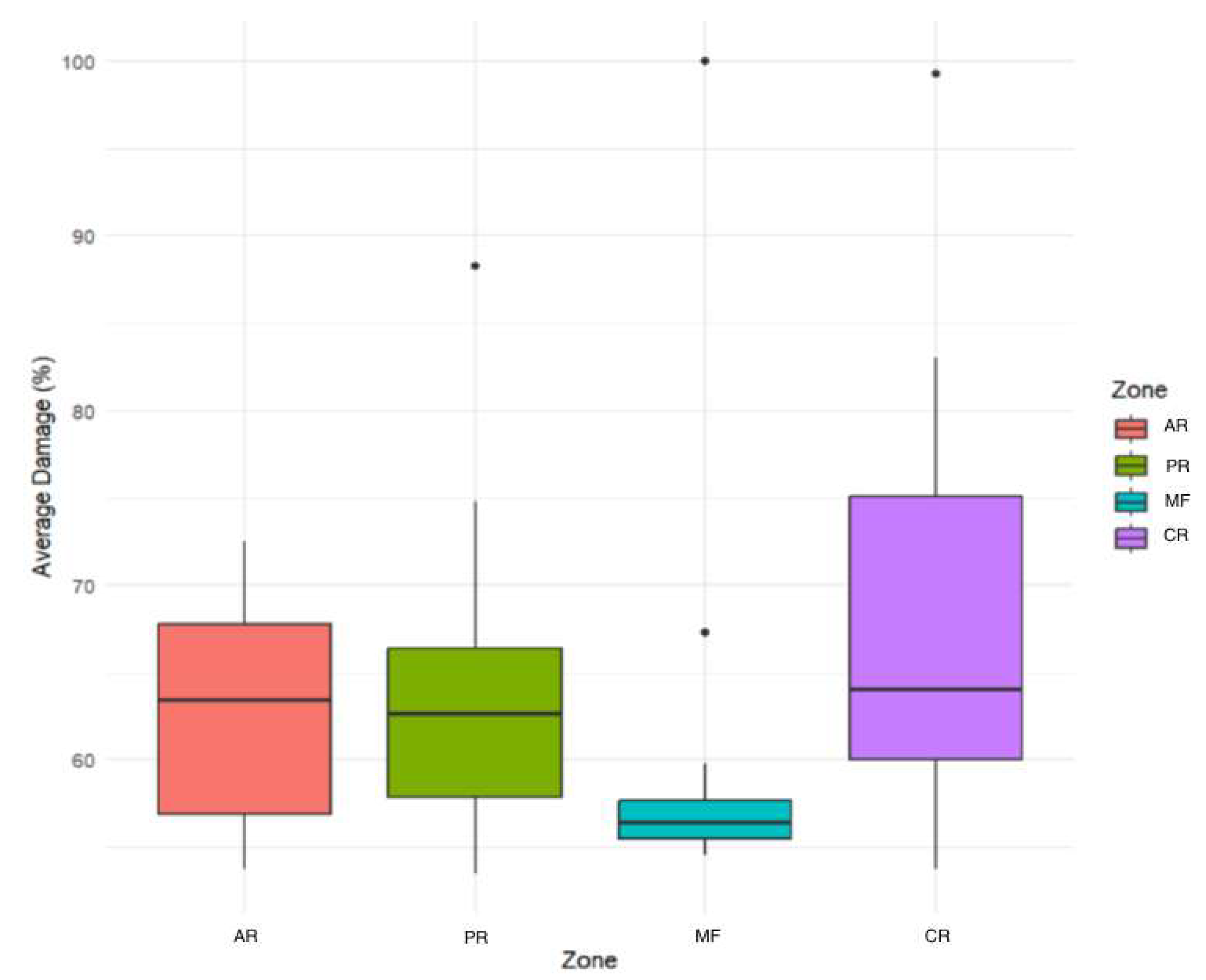
| Zone | Plot | Tree | DBH | Altitude | Layer 1 | Layer 2 | Layer 3 | Layer 4 | Average Damage |
| AR | 1 | 1 | 34 | 2257 | 64 | 76 | 64 | 67 | 67.75 |
| AR | 1 | 2 | 35 | 2257 | 74 | 71 | 69 | 70 | 71.00 |
| AR | 1 | 3 | 33 | 2257 | 58 | 57 | 55 | 58 | 57.00 |
| AR | 1 | 4 | 35 | 2257 | 65 | 63 | 54 | 56 | 59.50 |
| AR | 2 | 1 | 41 | 2257 | 56 | 53 | 54 | 52 | 53.75 |
| AR | 2 | 2 | 30 | 2257 | 57 | 57 | 55 | 53 | 55.50 |
| AR | 2 | 3 | 35 | 2257 | 55 | 54 | 57 | 54 | 55.00 |
| AR | 2 | 4 | 32 | 2257 | 67 | 77 | 62 | 57 | 65.75 |
| AR | 3 | 1 | 35 | 2257 | 58 | 57 | 55 | 56 | 56.50 |
| AR | 3 | 2 | 39 | 2257 | 80 | 79 | 68 | 63 | 72.50 |
| AR | 3 | 3 | 35 | 2257 | 67 | 67 | 64 | 65 | 65.75 |
| AR | 3 | 4 | 34 | 2257 | 53 | 56 | 61 | 62 | 58.00 |
| AR | 4 | 1 | 32 | 2257 | 72 | 70 | 63 | 66 | 67.75 |
| AR | 4 | 2 | 40 | 2257 | 61 | 70 | 65 | 60 | 64.00 |
| AR | 4 | 3 | 39 | 2257 | 64 | 66 | 60 | 61 | 62.75 |
| AR | 4 | 4 | 39 | 2257 | 69 | 70 | 69 | 68 | 69.00 |
| PR | 1 | 1 | 38 | 2305 | 71 | 55 | 57 | 53 | 59.00 |
| PR | 1 | 2 | 39 | 2305 | 73 | 60 | 62 | 59 | 63.50 |
| PR | 1 | 3 | 30 | 2305 | 48 | 64 | 53 | 57 | 55.50 |
| PR | 1 | 4 | 37 | 2305 | 64 | 64 | 69 | 61 | 64.50 |
| PR | 2 | 1 | 56 | 2305 | 96 | 90 | 87 | 80 | 88.25 |
| PR | 2 | 2 | 28 | 2305 | 61 | 58 | 52 | 55 | 56.5 |
| PR | 2 | 3 | 46 | 2305 | 68 | 67 | 61 | 66 | 65.5 |
| PR | 2 | 4 | 34 | 2305 | 72 | 58 | 76 | 53 | 64.75 |
| PR | 3 | 1 | 41 | 2305 | 74 | 91 | 71 | 61 | 74.25 |
| PR | 3 | 2 | 40 | 2305 | 57 | 55 | 55 | 54 | 55.25 |
| PR | 3 | 3 | 38 | 2305 | 60 | 55 | 60 | 58 | 58.25 |
| PR | 3 | 4 | 49 | 2305 | 68 | 62 | 60 | 57 | 61.75 |
| PR | 4 | 1 | 32 | 2305 | 55 | 52 | 55 | 52 | 53.50 |
| PR | 4 | 2 | 35 | 2305 | 59 | 66 | 60 | 55 | 60.00 |
| PR | 4 | 3 | 35 | 2305 | 69 | 69 | 68 | 70 | 69.00 |
| PR | 4 | 4 | 38 | 2305 | 72 | 78 | 73 | 76 | 74.75 |
| MF | 1 | 1 | 27 | 2320 | 55 | 54 | 54 | 59 | 55.50 |
| MF | 1 | 2 | 36 | 2320 | 58 | 54 | 56 | 58 | 56.50 |
| MF | 1 | 3 | 35 | 2320 | 53 | 53 | 54 | 59 | 54.75 |
| MF | 1 | 4 | 27 | 2320 | 57 | 58 | 57 | 58 | 57.50 |
| MF | 2 | 1 | 42 | 2320 | 59 | 58 | 57 | 56 | 57.50 |
| MF | 2 | 2 | 42 | 2320 | 53 | 57 | 56 | 56 | 55.50 |
| MF | 2 | 3 | 30 | 2320 | 58 | 56 | 56 | 56 | 56.50 |
| MF | 2 | 4 | 26 | 2320 | 100 | 100 | 100 | 100 | 100.00 |
| MF | 3 | 1 | 35 | 2320 | 58 | 56 | 54 | 55 | 55.75 |
| MF | 3 | 2 | 37 | 2320 | 57 | 56 | 56 | 53 | 55.50 |
| MF | 3 | 3 | 26 | 2320 | 51 | 55 | 54 | 58 | 54.50 |
| MF | 3 | 4 | 31 | 2320 | 55 | 59 | 54 | 57 | 56.25 |
| MF | 4 | 1 | 36 | 2320 | 71 | 58 | 57 | 53 | 59.75 |
| MF | 4 | 2 | 42 | 2320 | 55 | 56 | 53 | 57 | 55.25 |
| MF | 4 | 3 | 45 | 2320 | 72 | 63 | 71 | 63 | 67.25 |
| MF | 4 | 4 | 35 | 2320 | 57 | 66 | 56 | 54 | 58.25 |
| CR | 1 | 1 | 30 | 2391 | 64 | 57 | 53 | 59 | 58.25 |
| CR | 1 | 2 | 31 | 2391 | 75 | 80 | 67 | 75 | 74.25 |
| CR | 1 | 3 | 37 | 2391 | 75 | 73 | 78 | 85 | 77.75 |
| CR | 1 | 4 | 40 | 2391 | 80 | 77 | 75 | 78 | 77.50 |
| CR | 2 | 1 | 44 | 2391 | 59 | 59 | 60 | 56 | 58.50 |
| CR | 2 | 2 | 35 | 2391 | 53 | 56 | 54 | 52 | 53.75 |
| CR | 2 | 3 | 48 | 2391 | 79 | 61 | 61 | 61 | 65.50 |
| CR | 2 | 4 | 45 | 2391 | 93 | 85 | 80 | 74 | 83.00 |
| CR | 3 | 1 | 46 | 2391 | 72 | 62 | 54 | 54 | 60.50 |
| CR | 3 | 2 | 47 | 2391 | 73 | 58 | 60 | 60 | 62.75 |
| CR | 3 | 3 | 43 | 2391 | 73 | 68 | 63 | 56 | 65 |
| CR | 3 | 4 | 35 | 2391 | 69 | 69 | 59 | 55 | 63 |
| CR | 4 | 1 | 39 | 2391 | 71 | 60 | 52 | 63 | 61.50 |
| CR | 4 | 2 | 45 | 2391 | 100 | 100 | 100 | 97 | 99.25 |
| CR | 4 | 3 | 38 | 2391 | 52 | 55 | 59 | 58 | 56.00 |
| CR | 4 | 4 | 38 | 2391 | 81 | 62 | 70 | 69 | 70.50 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




