Submitted:
05 May 2026
Posted:
05 May 2026
You are already at the latest version
Abstract
Keywords:
- This study simultaneously quantified matrix metalloproteinase (MMP) activity and glycooxidation-related protein modification in visceral and subcutaneous adipose tissue, as well as in plasma, from individuals with and without obesity.
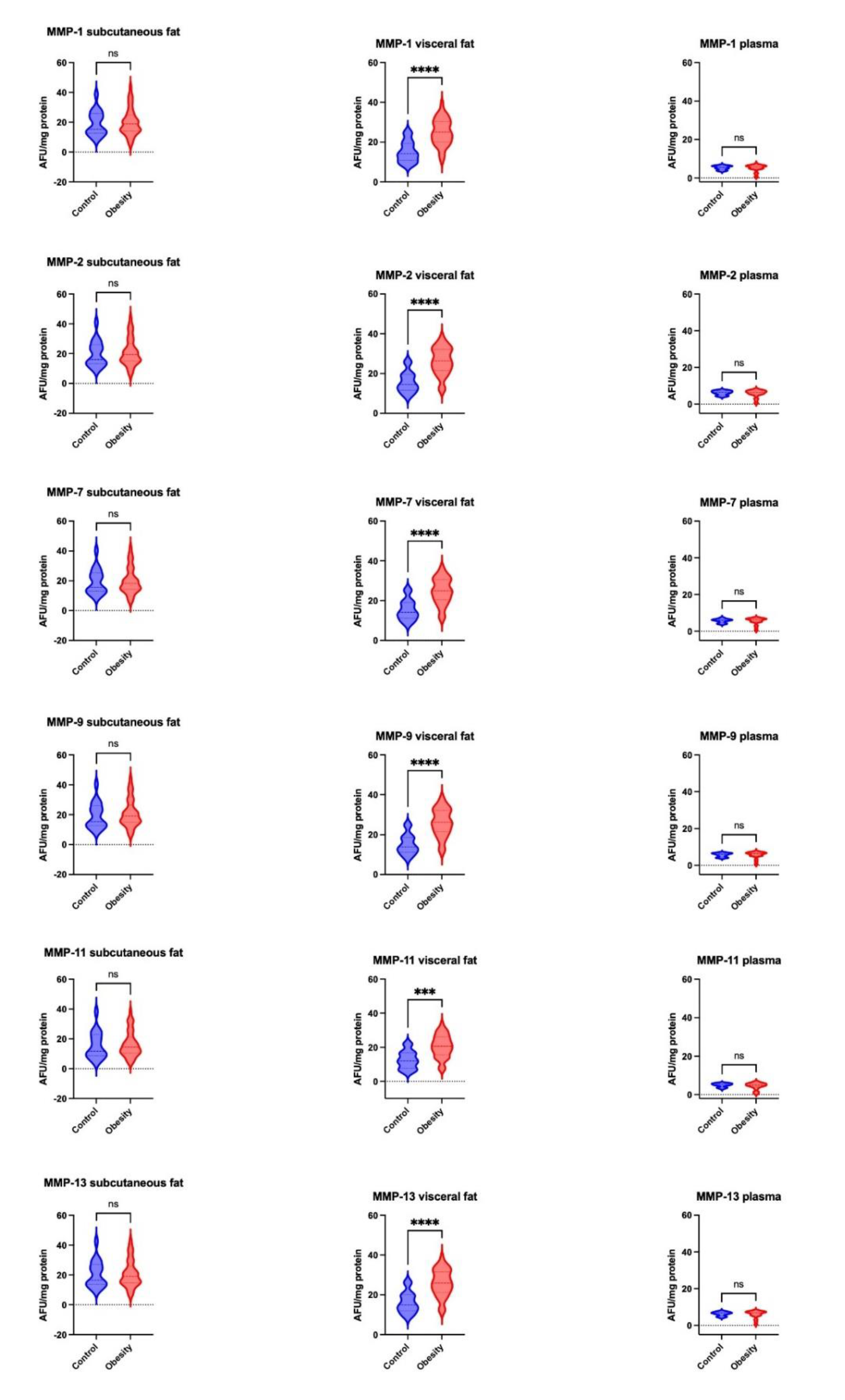
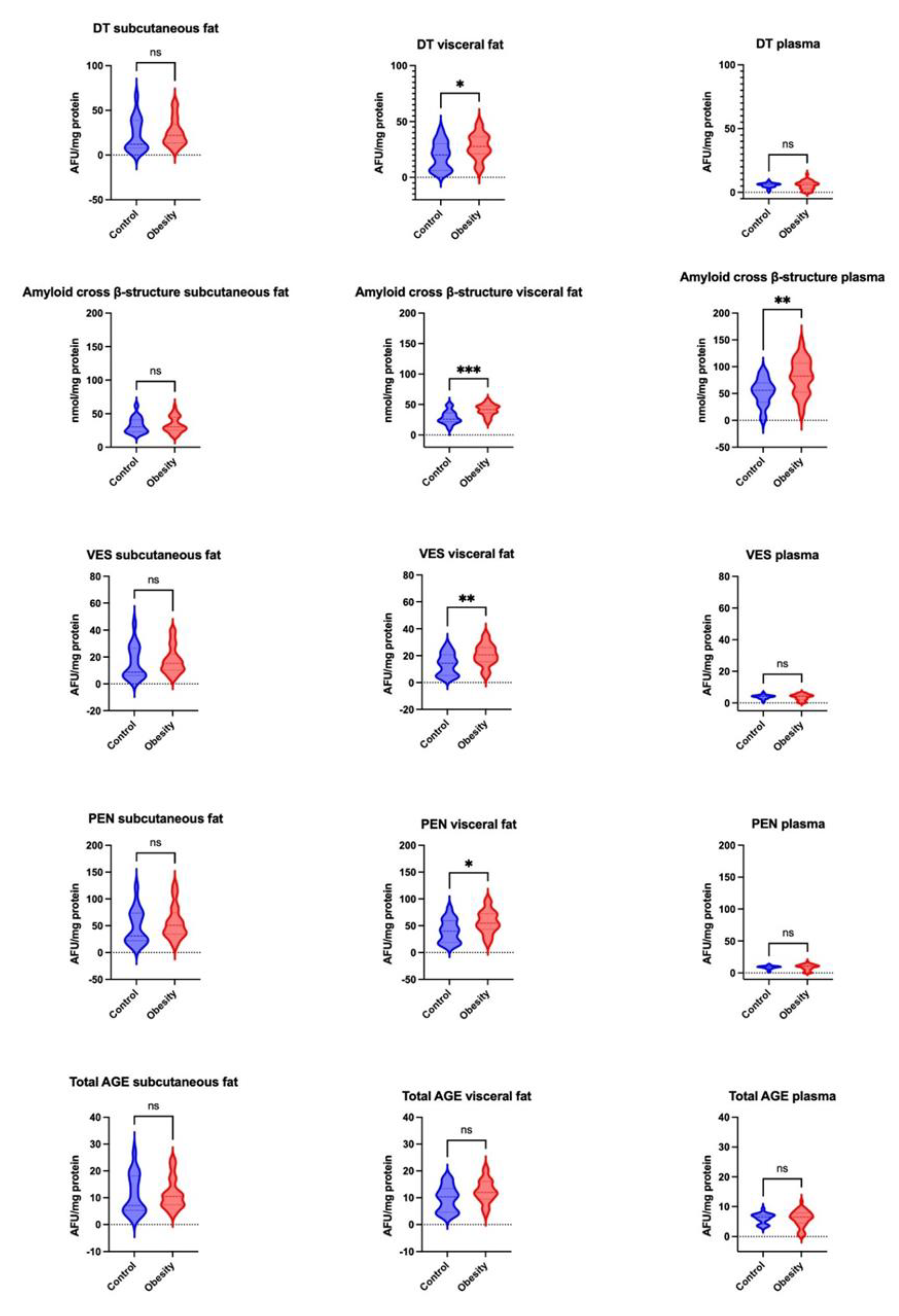
- Across all examined MMP isoforms and AGE-related markers (i.e., indicators of advanced glycation end-products—non-enzymatic protein modifications formed under conditions of oxidative and carbonyl stress), molecular alterations were most pronounced in visceral adipose tissue.
- In the context of existing literature, the observed pattern is consistent with visceral adipose tissue representing a key site of tissue-level molecular alterations in obesity.
- The results highlight the importance of considering tissue-compartment–specific biology when investigating metabolic risk in obesity.
1. Introduction
2. Results
2.1. Study Population
2.2. Matrix Metalloproteinase Activity
2.3. Protein Modification and Glycooxidation Markers
2.4. Comparison Between Obese Participants with and Without Metabolic Syndrome
2.5. Correlation Analyses
3. Discussion
4. Materials and Methods
4.1. Study Population and Clinical Data Collection
4.2. Adipose Tissue and Plasma Sample Collection
4.3. Ethical Approval and Informed Consent
4.4. Assessment of Matrix Metalloproteinase Activity
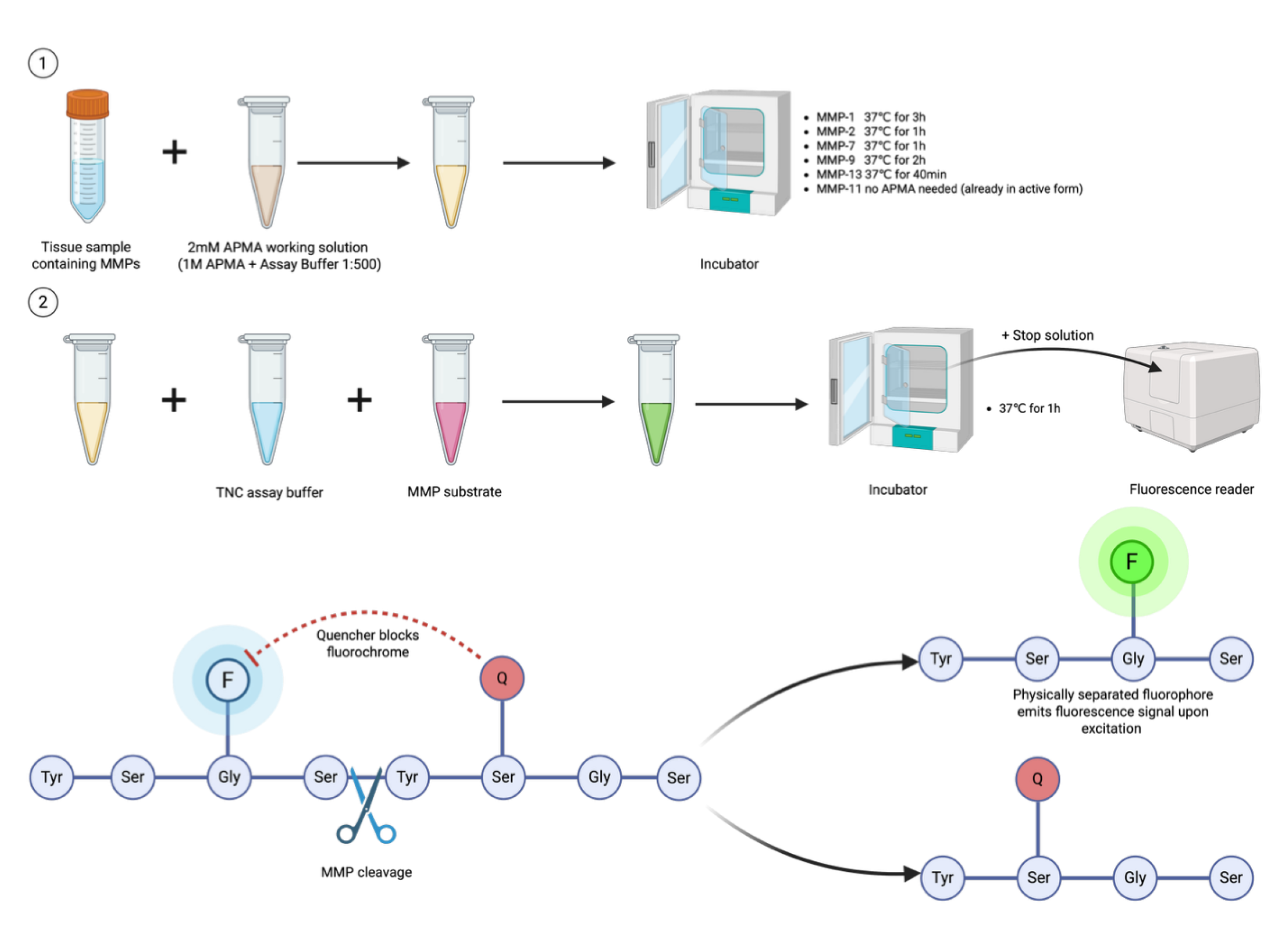
4.5. Determination of Advanced Glycation End-Products and Glycooxidation Markers
4.6. Statistical Analysis
5. Limitations
6. Conclusion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Elmaleh-Sachs, A.; Schwartz, J.; Bramante, C.; Nicklas, J.; Gudzune, K.; Jay, M. Obesity Management in Adults: A Review. JAMA 2023, 330, 2000–2015. [Google Scholar] [CrossRef] [PubMed]
- Rubino, F.; Batterham, R.L.; Koch, M.; Mingrone, G.; le Roux, C.W.; Farooqi, I.S.; Farpour-Lambert, N.; Gregg, E.W.; Cummings, D.E. Lancet Diabetes &. Lancet Diabetes Endocrinol. 2023, 11, 226–228. [Google Scholar] [CrossRef]
- Henning, R. Obesity and Obesity-Induced Inflammatory Disease Contribute to Atherosclerosis: A Review of the Pathophysiology and Treatment of Obesity. Am. J. Cardiovasc. Dis. 2021, 11, 504–529. [Google Scholar] [PubMed]
- Sun, K.; Kusminski, C.M.; Scherer, P.E. Adipose Tissue Remodeling and Obesity. J. Clin. Investig. 2011, 121, 2094–2101. [Google Scholar] [CrossRef]
- Kaess, B.M.; Pedley, A.; Massaro, J.M.; Murabito, J.; Hoffmann, U.; Fox, C.S. The Ratio of Visceral to Subcutaneous Fat, a Metric of Body Fat Distribution, Is a Unique Correlate of Cardiometabolic Risk. Diabetologia 2012, 55, 2622–2630. [Google Scholar] [CrossRef]
- Oka, R.; Miura, K.; Sakurai, M.; Nakamura, K.; Yagi, K.; Miyamoto, S.; Moriuchi, T.; Mabuchi, H.; Koizumi, J.; Nomura, H.; et al. Impacts of Visceral Adipose Tissue and Subcutaneous Adipose Tissue on Metabolic Risk Factors in Middle--aged Japanese. Obesity 2010, 18, 153–160. [Google Scholar] [CrossRef] [PubMed]
- Neeland, I.J.; Ayers, C.R.; Rohatgi, A.K.; Turer, A.T.; Berry, J.D.; Das, S.R.; Vega, G.L.; Khera, A.; McGuire, D.K.; Grundy, S.M.; et al. Associations of Visceral and Abdominal Subcutaneous Adipose Tissue with Markers of Cardiac and Metabolic Risk in Obese Adults. Obesity 2013, 21. [Google Scholar] [CrossRef]
- Ibrahim, M.M. Subcutaneous and Visceral Adipose Tissue: Structural and Functional Differences. Obes. Rev. 2010, 11, 11–18. [Google Scholar] [CrossRef]
- Wajchenberg, B.L. Subcutaneous and Visceral Adipose Tissue: Their Relation to the Metabolic Syndrome. Endocr. Rev. 2000, 21, 697–738. [Google Scholar] [CrossRef]
- Ruiz-Ojeda, F.J.; Méndez-Gutiérrez, A.; Aguilera, C.M.; Plaza-Díaz, J. Extracellular Matrix Remodeling of Adipose Tissue in Obesity and Metabolic Diseases. Int. J. Mol. Sci. 2019, 20. [Google Scholar] [CrossRef]
- DeBari, M.K.; Abbott, R.D. Adipose Tissue Fibrosis: Mechanisms, Models, and Importance. Int. J. Mol. Sci. 2020, 21, 6030. [Google Scholar] [CrossRef]
- Gliniak, C.M.; Pedersen, L.; Scherer, P.E. Adipose Tissue Fibrosis: The Unwanted Houseguest Invited by Obesity. J. Endocrinol. 2023, 259. [Google Scholar] [CrossRef]
- Zitka, O.; Kukacka, J.; Krizkova, S.; Huska, D.; Adam, V.; Masarik, M.; Prusa, R.; Kizek, R. Matrix Metalloproteinases. Curr. Med. Chem. 2010, 17, 3751–3768. [Google Scholar] [CrossRef]
- Cabral-Pacheco, G.A.; Garza-Veloz, I.; Castruita-De la Rosa, C.; Ramirez-Acuña, J.M.; Perez-Romero, B.A.; Guerrero-Rodriguez, J.F.; Martinez-Avila, N.; Martinez-Fierro, M.L. The Roles of Matrix Metalloproteinases and Their Inhibitors in Human Diseases. Int. J. Mol. Sci. 2020, 21, 9739. [Google Scholar] [CrossRef]
- Li, X.; Zhao, Y.; Chen, C.; Yang, L.; Lee, H.; Wang, Z.; Zhang, N.; Kolonin, M.G.; An, Z.; Ge, X.; et al. Critical Role of Matrix Metalloproteinase 14 in Adipose Tissue Remodeling during Obesity. Mol. Cell. Biol. 2020, 40, e00564-19. [Google Scholar] [CrossRef] [PubMed]
- Mirica, R.M.; Ionescu, M.; Mırıca, A.; Gınghına, O.; Iosıfescu, R.; Vacarasu, A.-B.; Cıotarla, D.-C.; Rosca, A.; Zagrean, L.; Iordache, N. Pathophysiology of Metalloproteinase Matrix in Relation to Morbid Obesity and Associated Pathologies. Eur. Res. J. 2022, 8, 411–419. [Google Scholar] [CrossRef]
- García-Prieto, C.F.; Gil-Ortega, M.; Vega-Martín, E.; Ramiro-Cortijo, D.; Martín-Ramos, M.; Bordiú, E.; Sanchez-Pernaute, A.; Torres, A.; Aránguez, I.; Fernández-Alfonso, M.; et al. Beneficial Effect of Bariatric Surgery on Abnormal MMP-9 and AMPK Activities: Potential Markers of Obesity-Related CV Risk. Front. Physiol. 2019, 10–2019. [Google Scholar] [CrossRef] [PubMed]
- Liberale, L.; Bonaventura, A.; Carbone, F.; Bertolotto, M.; Contini, P.; Scopinaro, N.; Camerini, G.B.; Papadia, F.S.; Cordera, R.; Camici, G.G.; et al. Early Reduction of Matrix Metalloproteinase-8 Serum Levels Is Associated with Leptin Drop and Predicts Diabetes Remission after Bariatric Surgery. Int. J. Cardiol. 2017, 245, 257–262. [Google Scholar] [CrossRef]
- Choromańska, B.; Myśliwiec, P.; Łuba, M.; Wojskowicz, P.; Dadan, J.; Myśliwiec, H.; Choromańska, K.; Zalewska, A.; Maciejczyk, M. A Longitudinal Study of the Antioxidant Barrier and Oxidative Stress in Morbidly Obese Patients after Bariatric Surgery. Does the Metabolic Syndrome Affect the Redox Homeostasis of Obese People? J. Clin. Med. 2020, 9. [Google Scholar] [CrossRef] [PubMed]
- Hawkins, C.L.; Morgan, P.E.; Davies, M.J. Quantification of Protein Modification by Oxidants. Free Radic. Biol. Med. 2009, 46, 965–988. [Google Scholar] [CrossRef]
- Peyroux, J.; Sternberg, M. Advanced Glycation Endproducts (AGEs): Pharmacological Inhibition in Diabetes. Pathol. Biol. 2006, 54, 405–419. [Google Scholar] [CrossRef] [PubMed]
- Lauko, K.K.; Nesterowicz, M.; Trocka, D.; Dańkowska, K.; Żendzian-Piotrowska, M.; Zalewska, A.; Maciejczyk, M. Novel Properties of Old Propranolol─Assessment of Antiglycation Activity through In Vitro and In Silico Approaches. ACS Omega 2024, 9, 27559–27577. [Google Scholar] [CrossRef] [PubMed]
- Strieder-Barboza, C.; Baker, N.A.; Flesher, C.G.; Karmakar, M.; Neeley, C.K.; Polsinelli, D.; Dimick, J.B.; Finks, J.F.; Ghaferi, A.A.; Varban, O.A.; et al. Advanced Glycation End-Products Regulate Extracellular Matrix-Adipocyte Metabolic Crosstalk in Diabetes. Sci. Rep. 2019, 9, 19748. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, A.M.; Stern, D.M. RAGE: A New Target for the Prevention and Treatment of the Vascular and Inflammatory Complications of Diabetes. Trends Endocrinol. Metab. 2000, 11, 368–375. [Google Scholar] [CrossRef]
- Gutowska, K.; Czajkowski, K.; Kuryłowicz, A. Receptor for the Advanced Glycation End Products (RAGE) Pathway in Adipose Tissue Metabolism. Int. J. Mol. Sci. 2023, 24, 10982. [Google Scholar] [CrossRef]
- Feng, Z.; Du, Z.; Shu, X.; Zhu, L.; Wu, J.; Gao, Q.; Wang, L.; Chen, N.; Li, Y.; Luo, M.; et al. Role of RAGE in Obesity-Induced Adipose Tissue Inflammation and Insulin Resistance. Cell Death Discov. 2021, 7, 305. [Google Scholar] [CrossRef]
- Song, F.; Hurtado del Pozo, C.; Rosario, R.; Zou, Y.S.; Ananthakrishnan, R.; Xu, X.; Patel, P.R.; Benoit, V.M.; Yan, S.F.; Li, H.; et al. RAGE Regulates the Metabolic and Inflammatory Response to High-Fat Feeding in Mice. Diabetes 2014, 63, 1948–1965. [Google Scholar] [CrossRef]
- Ouchi, N.; Parker, J.L.; Lugus, J.J.; Walsh, K. Adipokines in Inflammation and Metabolic Disease. Nat. Rev. Immunol. 2011, 11, 85–97. [Google Scholar] [CrossRef]
- Aksoyer Sezgin, S.; Bayoğlu, B.; Ersoz, F.; Sarici, M.; Niyazoğlu, M.; Dirican, A.; Cengiz, M. Downregulation of MMP-2 and MMP-9 Genes in Obesity Patients and Their Relation with Obesity-Related Phenotypes. Turk. J. Biochem. 2022, 47. [Google Scholar] [CrossRef]
- Mazor, R.; Friedmann-Morvinski, D.; Alsaigh, T.; Kleifeld, O.; Kistler, E.B.; Rousso-Noori, L.; Huang, C.; Li, J.B.; Verma, I.M.; Schmid-Schönbein, G.W. Cleavage of the Leptin Receptor by Matrix Metalloproteinase–2 Promotes Leptin Resistance and Obesity in Mice. Sci. Transl. Med. 2018, 10, eaah6324. [Google Scholar] [CrossRef]
- Choromańska, B.; Myśliwiec, P.; Łuba, M.; Wojskowicz, P.; Myśliwiec, H.; Choromańska, K.; Żendzian-Piotrowska, M.; Dadan, J.; Zalewska, A.; Maciejczyk, M. Impact of Weight Loss on the Total Antioxidant/Oxidant Potential in Patients with Morbid Obesity-A Longitudinal Study. Antioxidants 2020, 9. [Google Scholar] [CrossRef]
- Choromańska, B.; Myśliwiec, P.; Łuba, M.; Wojskowicz, P.; Myśliwiec, H.; Choromańska, K.; Dadan, J.; Żendzian-Piotrowska, M.; Zalewska, A.; Maciejczyk, M. Bariatric Surgery Normalizes Protein Glycoxidation and Nitrosative Stress in Morbidly Obese Patients. Antioxidants 2020, 9. [Google Scholar] [CrossRef]
- Alberti, K.G.M.M.; Zimmet, P.; Shaw, J. The Metabolic Syndrome—a New Worldwide Definition. The Lancet 2005, 366, 1059–1062. [Google Scholar] [CrossRef] [PubMed]
- Hinte, L.C.; Castellano-Castillo, D.; Ghosh, A.; Melrose, K.; Gasser, E.; Noé, F.; Massier, L.; Dong, H.; Sun, W.; Hoffmann, A.; et al. Adipose Tissue Retains an Epigenetic Memory of Obesity after Weight Loss. Nature 2024, 636, 457–465. [Google Scholar] [CrossRef]
- Bel Lassen, P.; Charlotte, F.; Liu, Y.; Bedossa, P.; Le Naour, G.; Tordjman, J.; Poitou, C.; Bouillot, J.-L.; Genser, L.; Zucker, J.-D.; et al. The FAT Score, a Fibrosis Score of Adipose Tissue: Predicting Weight-Loss Outcome After Gastric Bypass. J. Clin. Endocrinol. Metab. 2017, 102, 2443–2453. [Google Scholar] [CrossRef]
- Troeberg, L.; Nagase, H. Monitoring Metalloproteinase Activity Using Synthetic Fluorogenic Substrates. Curr. Protoc. Protein Sci. 2003, 33, 21.16.1–21.16.9. [Google Scholar] [CrossRef] [PubMed]
- Walker, J.M. The Bicinchoninic Acid (BCA) Assay for Protein Quantitation. In Basic Protein and Peptide Protocols; Humana Press: New Jersey; pp. 5–8.
| Parameter | Control (median [min–max]) | Obesity (median [min–max]) | p-value |
| Body weight (kg) | 80.0 (69.0–87.0) | 113.0 (82.0–185.0) | <0.0001 |
| BMI (kg/m2) | 24.51 (20.05–25.96) | 39.24 (32.37–51.79) | <0.0001 |
| Creatinine (mg/dL) | 0.90 (0.68–1.06) | 0.74 (0.52–1.24) | 0.0783 |
| Potassium (mmol/L) | 4.3 (4.0–4.8) | 4.3 (3.7–5.2) | 0.7628 |
| Sodium (mmol/L) | 140 (137–143) | 139 (136–146) | 0.0612 |
| WBC (×103/µL) | 5.67 (4.38–8.40) | 11.19 (5.73–17.92) | <0.0001 |
| RBC (×106/µL) | 4.72 (4.15–6.10) | 4.43 (3.52–5.55) | 0.0016 |
| Hemoglobin (g/dL) | 14.6 (12.6–16.1) | 13.1 (10.4–14.9) | <0.0001 |
| Hematocrit (%) | 43.1 (37.3–47.3) | 39.2 (31.6–46.1) | 0.0009 |
| MCV (fL) | 88.5 (85.0–92.0) | 88.4 (82.0–93.4) | 0.8427 |
| MCH (pg) | 31.2 (30.7–31.7) | 29.4 (25.3–32.6) | 0.1159 |
| MCHC (g/dL) | 34.5 (25.2–47.3) | 33.4 (30.0–35.9) | 0.0094 |
| Platelets (×103/µL) | 261 (87–419) | 243 (123–353) | 0.8991 |
| INR | 1.09 (0.99–1.27) | 1.05 (0.93–1.41) | 0.3611 |
| Systolic blood pressure (mmHg) | 127 (106–142) | 141 (100–168) | 0.0008 |
| Diastolic blood pressure (mmHg) | 76 (65–88) | 95 (78–109) | <0.0001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




