Submitted:
04 May 2026
Posted:
05 May 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
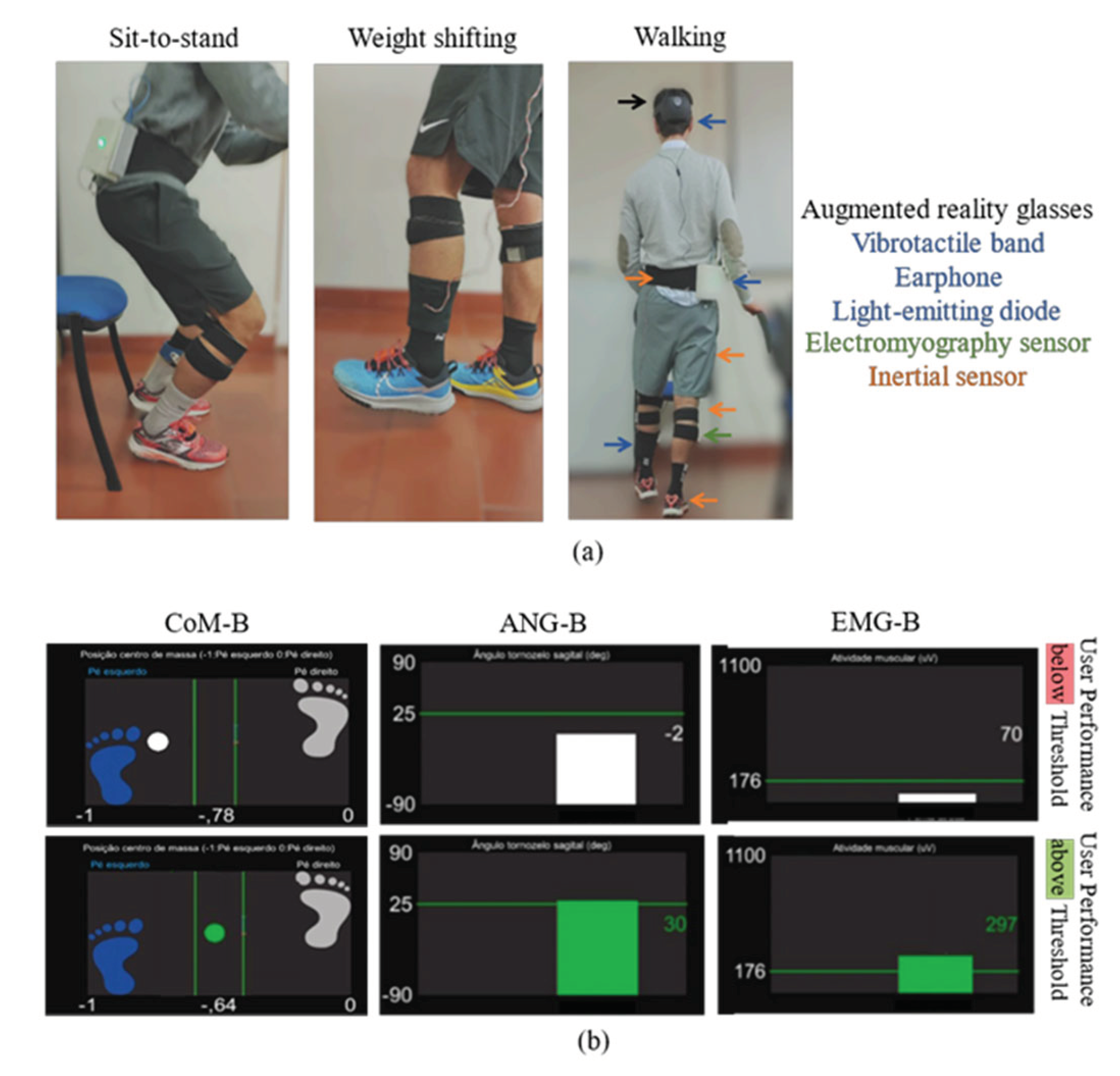
2.1. Wearable Multimodal Technology
2.2. Biofeedback
2.3. Post-Stroke Participant
2.4. Biofeedback Parameters and Favourable Motor Performance
2.5. Physical Rehabilitation Complemented by Biofeedback
2.6. Outcomes
3. Results
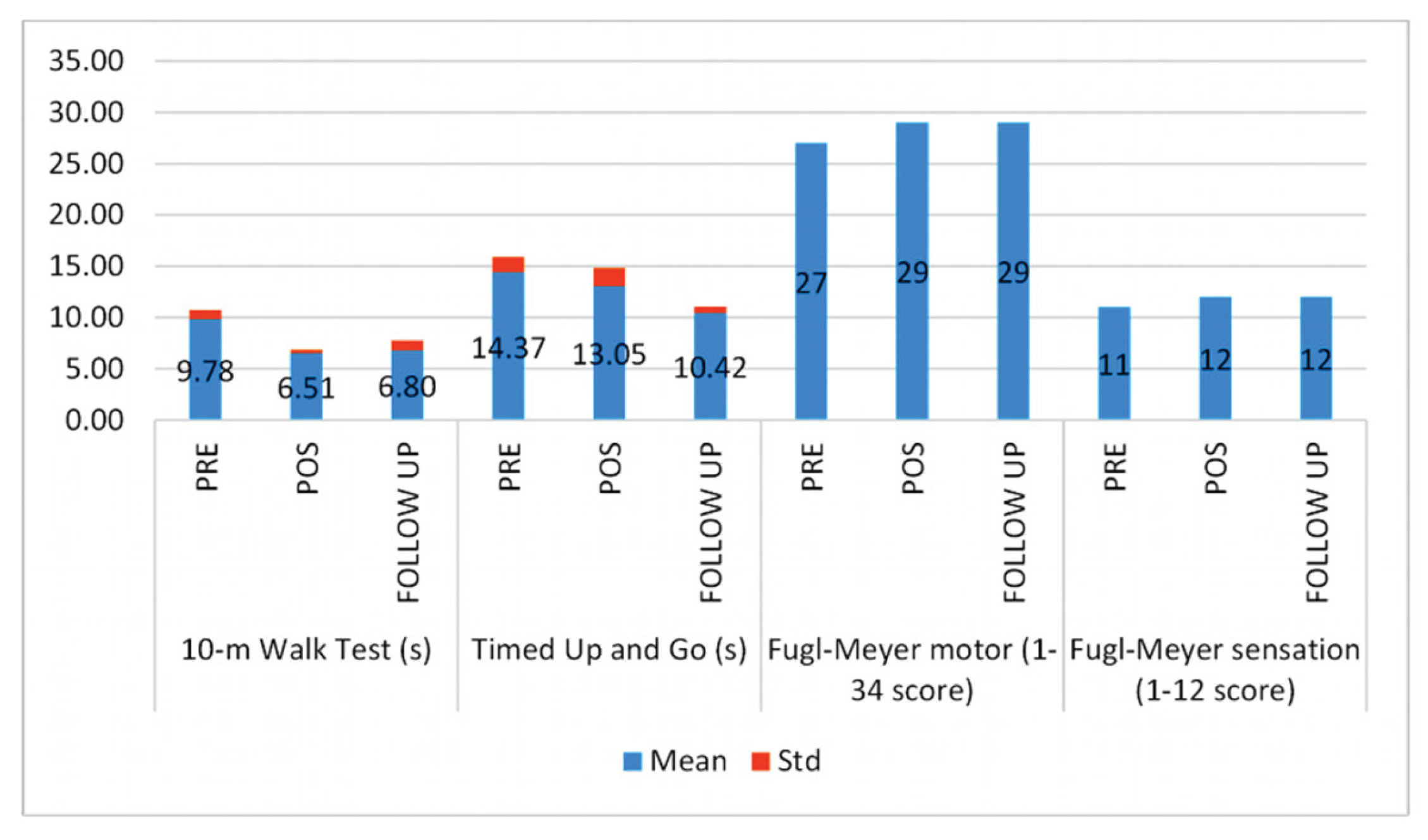
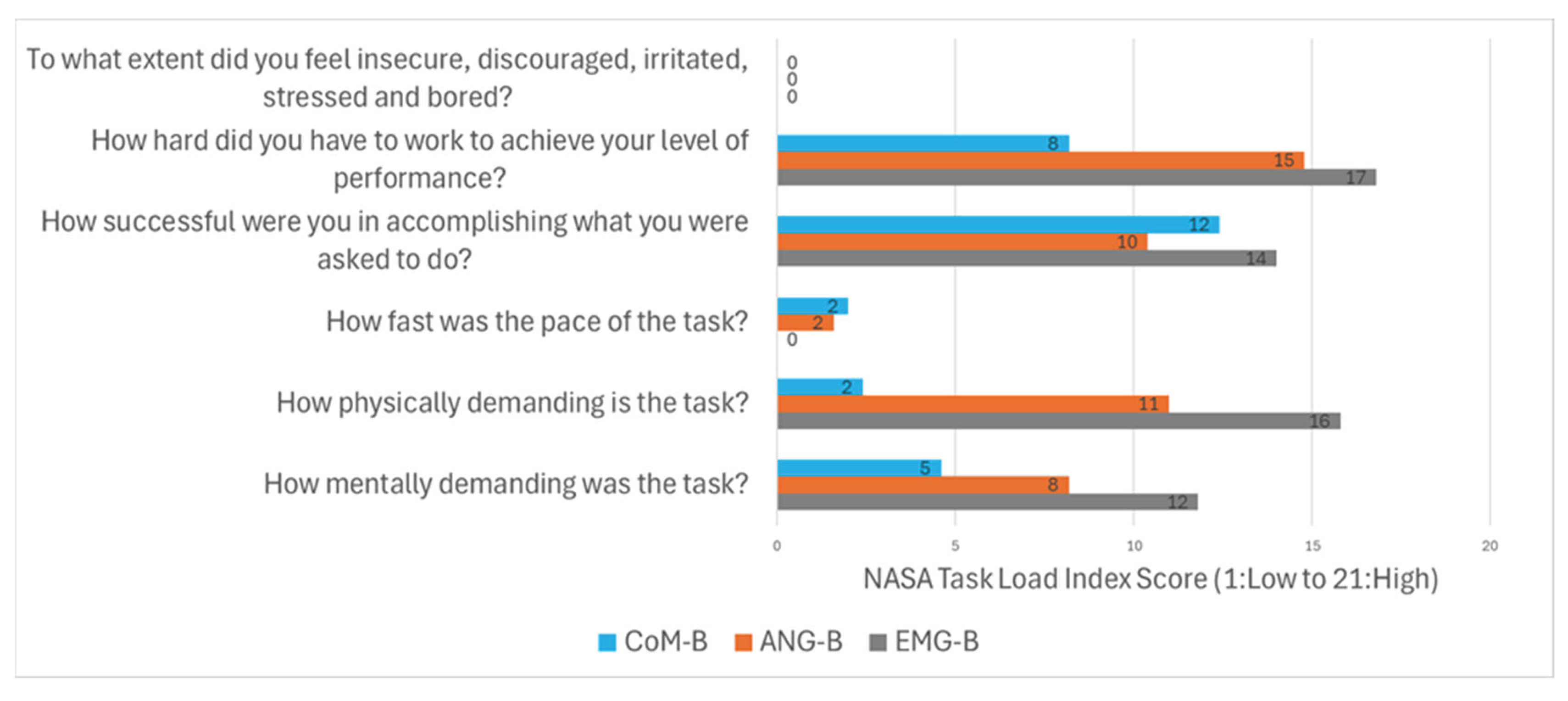
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ANG | Angle |
| CoM | Center of mass |
| EMG | Electromyographic |
References
- Huang, H.; Wolf, S. L.; He, J. Recent developments in biofeedback for neuromotor rehabilitation. J. Neuroeng. Rehabil. 2006, vol. 3, 11. [Google Scholar] [CrossRef]
- Wolf, S. L. Electromyographic Biofeedback Applications to Stroke Patients. Phys. Ther. 1983, vol. 63(no. 9), 1448–1459. [Google Scholar] [CrossRef]
- Dost Sürücü, G.; Tezen, Ö. The effect of EMG biofeedback on lower extremity functions in hemiplegic patients. Acta Neurol. Belg. 2021, vol. 121(no. 1), 113–118. [Google Scholar] [CrossRef]
- Bowman, T.; et al. Wearable Devices for Biofeedback Rehabilitation: A Systematic Review and Meta-Analysis to Design Application Rules and Estimate the Effectiveness on Balance and Gait Outcomes in Neurological Diseases. Sensors 2021, vol. 21(no. 10), 3444. [Google Scholar] [CrossRef]
- Jung, K.-S.; Bang, H.; In, T.-S.; Cho, H.-Y. Gait training with auditory feedback improves trunk control, muscle activation and dynamic balance in patients with hemiparetic stroke: A randomized controlled pilot study. J. Back. Musculoskelet. Rehabil. 2020, vol. 33(no. 1), 1–6. [Google Scholar] [CrossRef]
- Byl, N.; Zhang, W.; Coo, S.; Tomizuka, M. Clinical impact of gait training enhanced with visual kinematic biofeedback: Patients with Parkinson’s disease and patients stable post stroke. Neuropsychologia 2015, vol. 79, 332–343. [Google Scholar] [CrossRef] [PubMed]
- Hankinson, K.; Shaykevich, A.; Vallence, A.-M.; Rodger, J.; Rosenberg, M.; Etherton-Beer, C. A Tailored Music-Motor Therapy and Real-Time Biofeedback Mobile Phone App (‘GotRhythm’) to Promote Rehabilitation Following Stroke: A Pilot Study. Neurosci. Insights 2022, vol. 17, 263310552211005. [Google Scholar] [CrossRef] [PubMed]
- Arpa, S.; Ozcakir, S. Does electromyographic biofeedback improve exercise effects in hemiplegic patients? A pilot randomized controlled trial. J. Rehabil. Med. 2019, vol. 51(no. 2), 109–112. [Google Scholar] [CrossRef]
- Mottaz, et al. Modulating functional connectivity after stroke with neurofeedback: Effect on motor deficits in a controlled cross-over study. NeuroImage Clin. 2018, vol. 20, no. July, 336–346. [Google Scholar] [CrossRef]
- Pinheiro; Figueiredo, J.; Cerqueira, J.; Santos, C. P. Robotic Biofeedback for Post-Stroke Gait Rehabilitation: A Scoping Review. Sensors 2022, vol. 22(no. 19), 7197. [Google Scholar] [CrossRef] [PubMed]
- Walker, E. R.; Hyngstrom, A. S.; Schmit, B. D. Influence of visual feedback on dynamic balance control in chronic stroke survivors. J. Biomech. 2016, vol. 49(no. 5), 698–703. [Google Scholar] [CrossRef]
- Held, J. P. O.; et al. Augmented Reality–Based Rehabilitation of Gait Impairments: Case Report. JMIR mHealth uHealth 2020, vol. 8(no. 5), e17804. [Google Scholar] [CrossRef] [PubMed]
- Phan, H. L.; Le, T. H.; Lim, J. M.; Hwang, C. H.; Koo, K. Effectiveness of Augmented Reality in Stroke Rehabilitation: A Meta-Analysis. Appl. Sci. 2022, vol. 12(no. 4), 1848. [Google Scholar] [CrossRef]
- Pinheiro, C.; Figueiredo, J.; Pereira, T.; Santos, C. P. Design and Usability Assessment of Multimodal Augmented Reality System for Gait Training. in Robot 2023: Sixth Iberian Robotics Conference, 2024; pp. 437–449. [Google Scholar]
- Kinsella, S.; Moran, K. Gait pattern categorization of stroke participants with equinus deformity of the foot. Gait Posture 2008, vol. 27(no. 1), 144–151. [Google Scholar] [CrossRef] [PubMed]
- Balaban, B.; Tok, F. Gait Disturbances in Patients With Stroke. PM R 2014, vol. 6(no. 7), 635–642. [Google Scholar] [CrossRef] [PubMed]
- Daly, J. J.; Roenigk, K.; Cheng, R.; Ruff, R. L. Abnormal Leg Muscle Latencies and Relationship to Dyscoordination and Walking Disability after Stroke. Rehabil. Res. Pract. 2011, vol. 2011, 1–8. [Google Scholar] [CrossRef]
- Chan, H. S.; Ng, A. W. Y. Finger response times to visual, auditory and tactile modality stimuli. Lect. Notes Eng. Comput. Sci. 2012, vol. 2196, 1449–1454. [Google Scholar]
- Hribernik, M.; Umek, A.; Tomažič, S.; Kos, A. Review of Real-Time Biomechanical Feedback Systems in Sport and Rehabilitation. Sensors 2022, vol. 22(no. 8), 3006. [Google Scholar] [CrossRef]
- Moreira, L.; et al. Study of Gait Cycle Using a Five-Link Inverted Pendulum Model: First Developments. in 2019 IEEE 6th Portuguese Meeting on Bioengineering (ENBENG), Feb. 2019; pp. 1–4. [Google Scholar] [CrossRef]
- Pinheiro, C. Kinematic and kinetic study of sit-to-stand and stand-to-sit movements towards a human-like skeletal model. in 2019 IEEE 6th Portuguese Meeting on Bioengineering (ENBENG), Feb. 2019; pp. 1–4. [Google Scholar] [CrossRef]
- Brooke, J. SUS: A ‘Quick and Dirty’ Usability Scale. Usability Eval. Ind. 1995, 207–212. [Google Scholar] [CrossRef]
- Li, S.; Francisco, G. E.; Zhou, P. Post-stroke Hemiplegic Gait: New Perspective and Insights. Front. Physiol. 2018, vol. 9, no. AUG, 1–8. [Google Scholar] [CrossRef]
- Nan, W.; Dias, A. P. B.; Rosa, A. C. Neurofeedback Training for Cognitive and Motor Function Rehabilitation in Chronic Stroke: Two Case Reports. Front. Neurol. 2019, vol. 10, no. JUL, 1–6. [Google Scholar] [CrossRef]
- Bae, S.; Lee, J.; Lee, B.-H. Effect of an EMG–FES Interface on Ankle Joint Training Combined with Real-Time Feedback on Balance and Gait in Patients with Stroke Hemiparesis. Healthcare 2020, vol. 8(no. 3), 292. [Google Scholar] [CrossRef]
- Pandian, S.; Arya, K. N.; Kumar, D. Minimal clinically important difference of the lower-extremity fugl–meyer assessment in chronic-stroke. Top. Stroke Rehabil. 2016, vol. 23(no. 4), 233–239. [Google Scholar] [CrossRef]
| Sensor-based outcome | Motor task | Biofeedback parameter | Favourable change | ||
| CoM-B | ANG-B | EMG-B | |||
| Medio-lateral CoM displacement (0-1) | STS | -0.03 (0.75) | 0.02 (0.09) | -0.02 (0.63) | < 0 |
| WS | 0.00 (0.01) | 0.05 (0.88) | 0.00 (0.04) | > 0 | |
| W | 0.06 (0.47) | 0.05 (0.07) | -0.03 (0.35) | ||
| Sagittal ankle angle (deg) | STS | 1 (0.20) | 1 (0.54) | 0 (0.01) | > 0 |
| WS | 2 (0.40) | 0 (0.11) | -2 (0.49) | ||
| W | 0 (0.02) | 1 (0.74) | 0 (0.00) | ||
| Tibialis anterior muscle activity (uV) | STS | 11 (0.93) | -1 (0.04) | -2 (0.06) | > 0 |
| WS | 3 (0.50) | -1 (0.24) | 5 (0.82) | ||
| W | -6 (0.91) | 3 (0.45) | 2 (0.29) | ||
| STS: stand-to-sit, WS: split-stance weight shifting, W: walking | |||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




