Submitted:
04 May 2026
Posted:
05 May 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Search Strategy and Scope of the Review
3. Defining Neuroendocrine Differentiation: A Diagnostic and Biological Spectrum
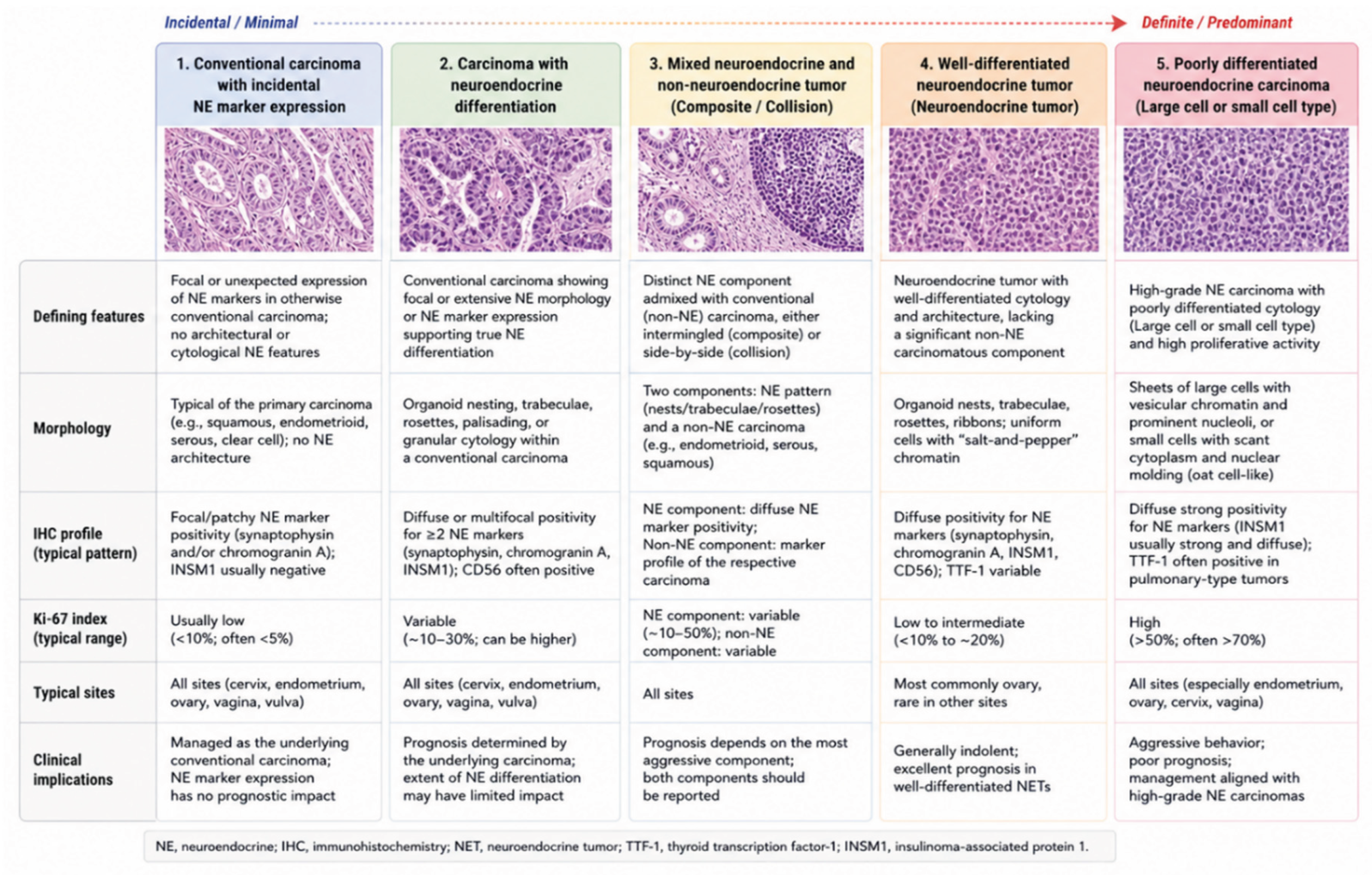
4. Classification and Diagnostic Terminology
4.1. From Marker Expression to Diagnostic Category
4.2. Well-Differentiated Neuroendocrine Tumors and Poorly Differentiated Neuroendocrine Carcinomas
4.3. Mixed Neuroendocrine and Non-Neuroendocrine Tumors
4.4. Practical Terminology Across Gynecologic Sites
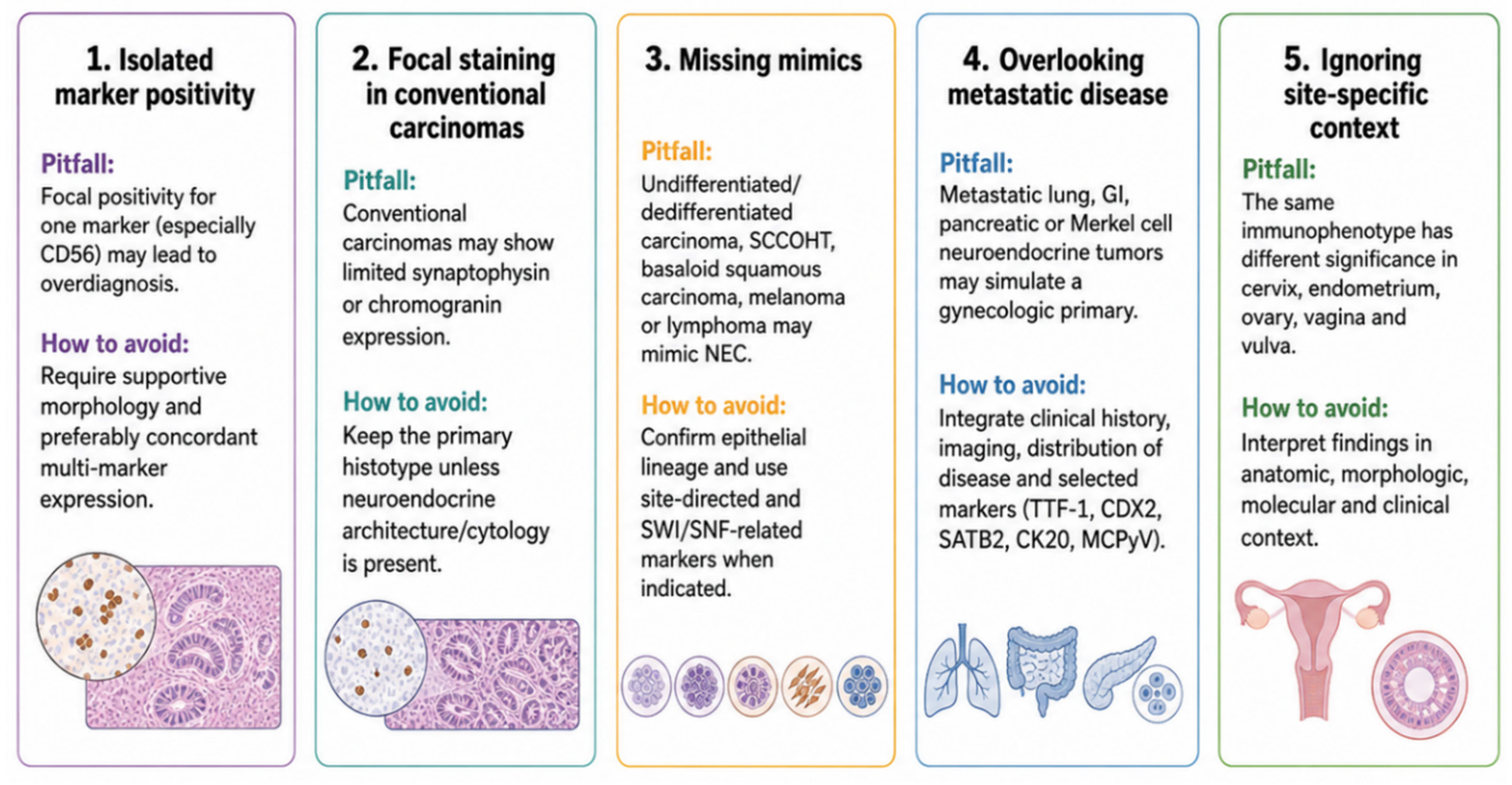
5. Immunohistochemical Assessment: Markers, Interpretation and Diagnostic Pitfalls
5.1. Core Neuroendocrine Markers
5.2. Proliferation, Epithelial Lineage and Site-Directed Markers
5.3. Molecularly Relevant Immunohistochemical Markers
5.4. Practical Interpretation of Staining Patterns
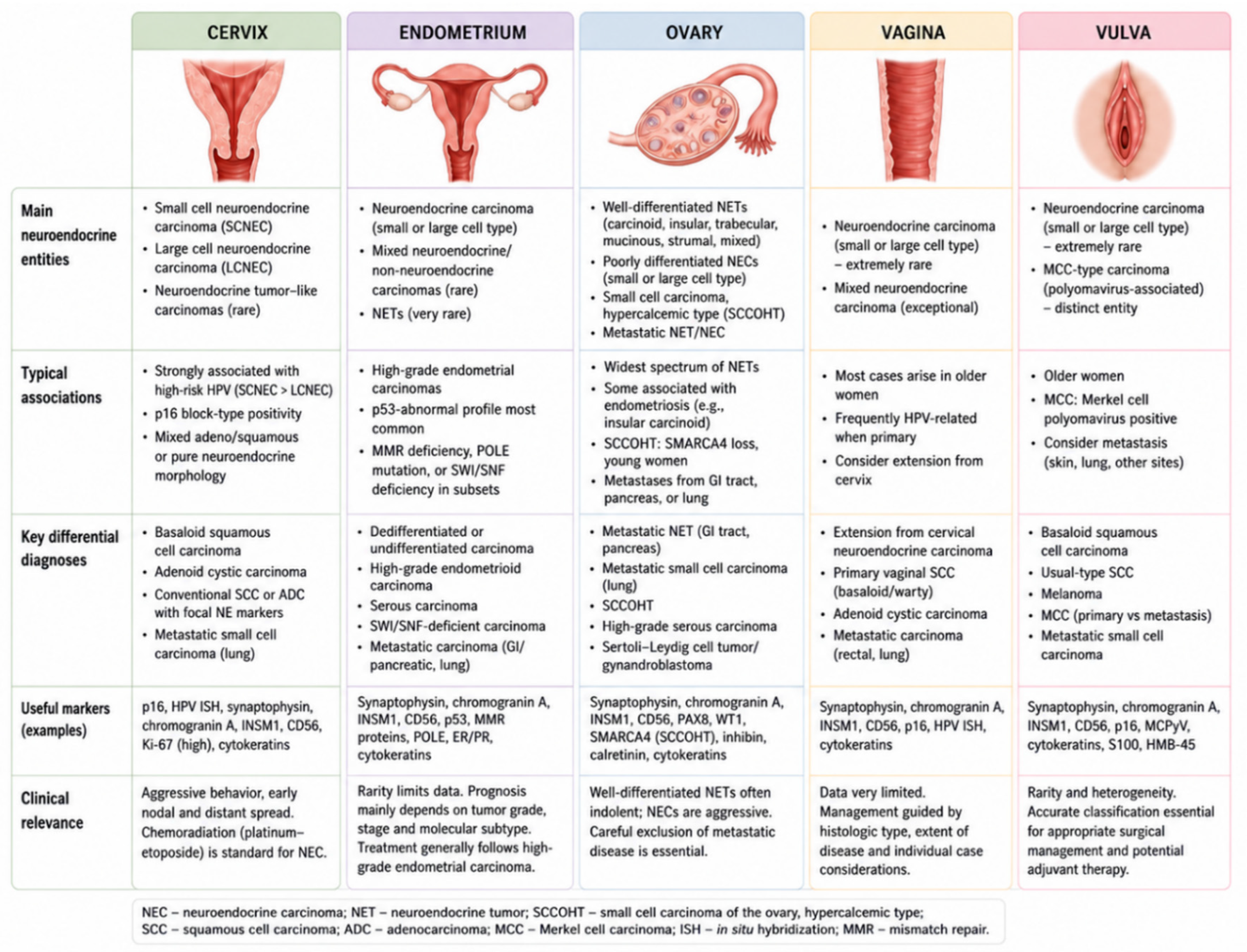
6. Site-Specific Patterns of Neuroendocrine Differentiation in the Female Genital Tract
6.1. Cervix
6.2. Endometrium
6.3. Ovary
6.4. Vagina and Vulva
6.5. Integrating Site-Specific Interpretation
7. The Gray Zone: Incidental Immunophenotype Versus Clinically Meaningful Neuroendocrine Differentiation
7.1. Incidental Marker Expression and Partial Neuroendocrine Differentiation
7.2. Criteria Favoring Clinically Meaningful Differentiation
7.3. Reporting Gray-Zone Cases
8. Mixed Neuroendocrine and Non-Neuroendocrine Tumors
8.1. Definition and Patterns Across Gynecologic Sites
8.2. Biological and Clinical Significance
8.3. Reporting Mixed Tumors
9. Differential Diagnosis and Metastatic Mimics
9.1. Conventional Gynecologic Carcinomas and High-Grade Epithelial Mimics
9.2. Undifferentiated Tumors and Small Round Cell Mimics
9.3. Metastatic Neuroendocrine Neoplasms
10. Molecular Features and Biological Insights
10.1. Site-Specific Molecular Contexts
10.2. SWI/SNF-Deficient Mimics and Diagnostic Refinement
10.3. Mixed Tumors, Clonal Relationships and Tumor Plasticity
10.4. Therapeutic Relevance and Limitations of Molecular Data
11. Prognostic Significance
11.1. Poorly Differentiated Neuroendocrine Carcinoma
11.2. Well-Differentiated Neuroendocrine Tumors
11.3. Mixed Tumors and Focal Marker Expression
11.4. Determinants and Limitations of Prognostic Assessment
12. Therapeutic Implications
12.1. Poorly Differentiated Neuroendocrine Carcinoma
12.2. Well-Differentiated Neuroendocrine Tumors and Metastatic NETs
12.3. Mixed Tumors and Conventional Carcinomas with Focal Marker Expression
12.4. Molecularly Informed Therapy and Future Directions
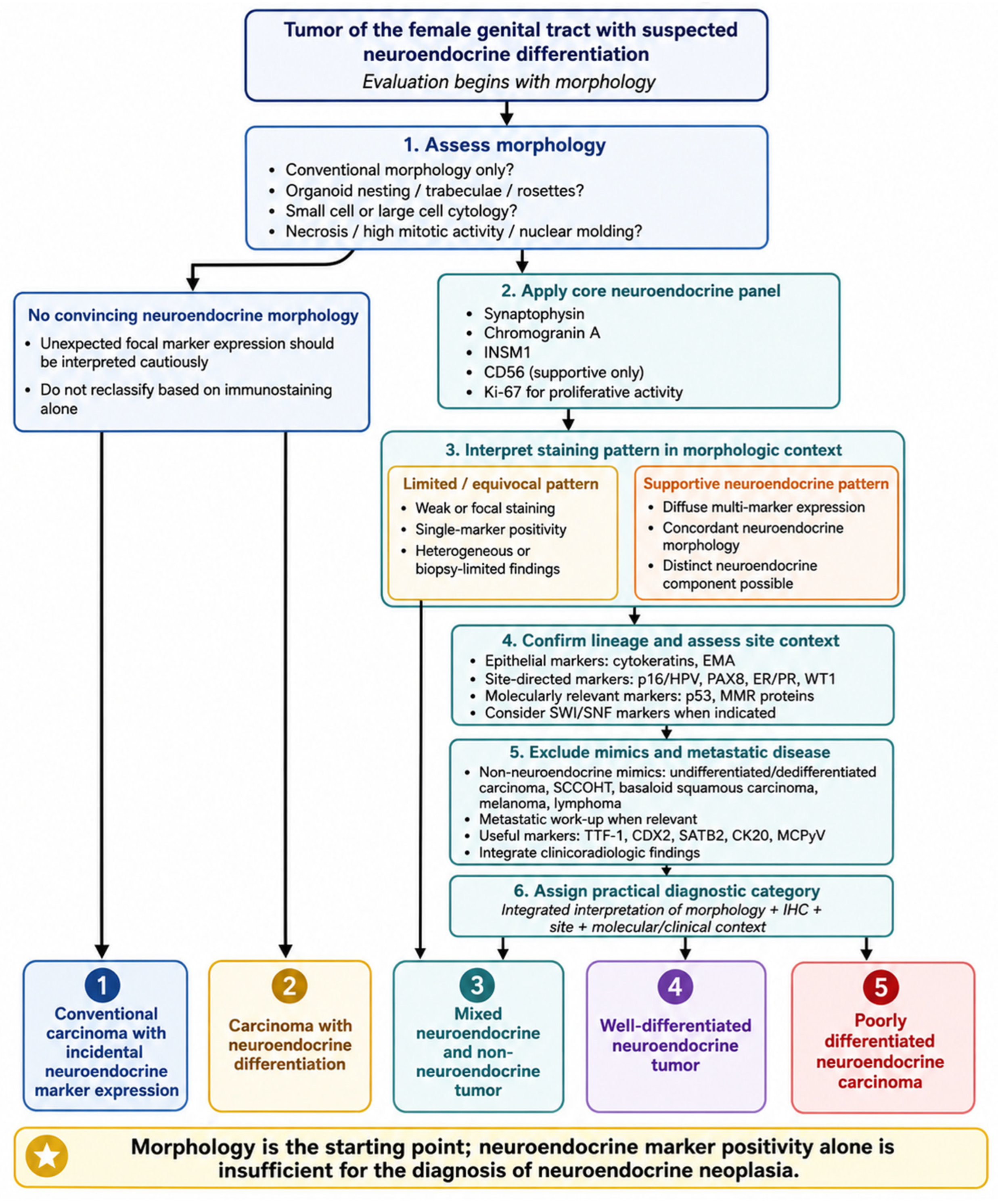
13. Proposed Integrated Diagnostic–Clinical Algorithm
14. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Howitt, B.E.; Kelly, P.; McCluggage, W.G. Pathology of Neuroendocrine Tumours of the Female Genital Tract. Curr. Oncol. Rep. 2017, 19, 59. [Google Scholar] [CrossRef] [PubMed]
- Chun, Y.K. Neuroendocrine Tumors of the Female Reproductive Tract: A Literature Review. J. Pathol. Transl. Med. 2015, 49, 450–461. [Google Scholar] [CrossRef]
- Talia, K.L.; Ganesan, R. Neuroendocrine Neoplasia of the Female Genital Tract. Surg. Pathol. Clin. 2022, 15, 407–420. [Google Scholar] [CrossRef]
- Winer, I.; Kim, C.; Gehrig, P. Neuroendocrine Tumors of the Gynecologic Tract Update. Gynecol. Oncol. 2021, 162, 210–219. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Li, M.; Zhang, G.; Shen, D. Neuroendocrine Carcinoma of the Cervix: A Comprehensive Clinicopathologic Study and Literature Review. gocm 2023, 3. [Google Scholar] [CrossRef]
- Moritz, A.W.; Schlumbrecht, M.P.; Nadji, M.; Pinto, A. Expression of Neuroendocrine Markers in Non-Neuroendocrine Endometrial Carcinomas. Pathology 2019, 51, 369–374. [Google Scholar] [CrossRef]
- Kuji, S.; Watanabe, R.; Sato, Y.; Iwata, T.; Hirashima, Y.; Takekuma, M.; Ito, I.; Abe, M.; Nagashio, R.; Omae, K.; et al. A New Marker, Insulinoma-Associated Protein 1 (INSM1), for High-Grade Neuroendocrine Carcinoma of the Uterine Cervix: Analysis of 37 Cases. Gynecol. Oncol. 2017, 144, 384–390. [Google Scholar] [CrossRef]
- Moreno-Moreno, E.; Caniego-Casas, T.; Carretero-Barrio, I.; Cortés, A.; Muriel, A.; Domínguez-Rullán, J.A.; Martín-Gromaz, C.; Moreno-Bueno, G.; Matías-Guiu, X.; Palacios, J.; et al. Histologic and Molecular Type Changes in Endometrial Cancer Recurrences in Comparison With Their Corresponding Primary Tumors. Am. J. Surg. Pathol. 2024, 48, 1580. [Google Scholar] [CrossRef]
- Wei, K.-N.; Fu, X.-D.; Wang, M.-Y.; Wang, L.-X. Two Cases of Mixed Large Cell Neuroendocrine Carcinoma and Adenocarcinoma of the Cervix: Case Report and Review of the Literature. Diagn. Pathol. 2024, 19, 166. [Google Scholar] [CrossRef]
- Tessier-Cloutier, B.; Kang, E.-Y.; Alex, D.; Stewart, C.J.R.; McCluggage, W.G.; Köbel, M.; Lee, C.-H. Endometrial Neuroendocrine Carcinoma and Undifferentiated Carcinoma Are Distinct Entities with Overlap in Neuroendocrine Marker Expression. Histopathology 2022, 81, 44–54. [Google Scholar] [CrossRef] [PubMed]
- Di Vito, V.; Veroi, G.; Rizza, L.; Rota, F.; Baiocchini, A.; Macciomei, M.C.; Lubrano, C.; La Salvia, A.; Lania, A.; Grillo, L.R.; et al. Ovarian Neuroendocrine Neoplasms: Challenges and Future Perspectives. J. Clin. Med. 2025, 14, 8833. [Google Scholar] [CrossRef]
- Stumpo, S.; Formelli, M.G.; Persano, I.; Parlagreco, E.; Lauricella, E.; Rodriquenz, M.G.; Guerrera, L.P.; Zurlo, I.V.; Campana, D.; Brizzi, M.P.; et al. Extrapulmonary Neuroendocrine Carcinomas: Current Management and Future Perspectives. J. Clin. Med. 2023, 12. [Google Scholar] [CrossRef]
- Virarkar, M.; Vulasala, S.S.; Morani, A.C.; Waters, R.; Gopireddy, D.R.; Kumar, S.; Bhosale, P.; Lall, C. Neuroendocrine Neoplasms of the Gynecologic Tract. Cancers 2022, 14, 1835. [Google Scholar] [CrossRef] [PubMed]
- Maragliano, R.; Libera, L.; Carnevali, I.; Pensotti, V.; De Vecchi, G.; Testa, M.; Amaglio, C.; Leoni, E.; Formenti, G.; Sessa, F.; et al. Mixed Neuroendocrine/Non-Neuroendocrine Neoplasm (MiNEN) of the Ovary Arising from Endometriosis: Molecular Pathology Analysis in Support of a Pathogenetic Paradigm. Endocr. Pathol. 2022, 33, 400–410. [Google Scholar] [CrossRef]
- Mohsin, M.; Dube, R.; Hamza, D.; Ali, M.; Garg, H. Primary Ovarian Neuroendocrine Neoplasia with Concurrent Large Epithelial Borderline Ovarian Tumor, Coexistent with Non-Malignant Pleural Effusion and Multiple Uterine Fibroids: A Case Report and Review of the Literature. J. Med. Case Rep. 2025, 19, 125. [Google Scholar] [CrossRef]
- Dai, X.; Chen, S.; Yang, S. Clinical, Pathological Characteristics, and Therapeutic Outcomes of Primary Ovarian Carcinoid Tumors: A Case Series of 15 Cases. World J. Surg. Onc 2025, 23, 82. [Google Scholar] [CrossRef] [PubMed]
- Shen, L.-P.; Yang, A.-Q.; Jin, L. Strumal Carcinoid Tumor of the Ovary: Report of Rare Occurrence with Review of Literature. Diagnostics 2022, 12. [Google Scholar] [CrossRef]
- Georgescu, T.-A.; Bohiltea, R.E.; Munteanu, O.; Furtunescu, F.; Lisievici, A.-C.; Grigoriu, C.; Gherghiceanu, F.; Vlădăreanu, E.M.; Berceanu, C.; Ducu, I.; et al. Emerging Therapeutic Concepts and Latest Diagnostic Advancements Regarding Neuroendocrine Tumors of the Gynecologic Tract. Medicina 2021, 57. [Google Scholar] [CrossRef] [PubMed]
- Yin, Y.; Han, L.; Chen, Y.; Ruan, J.; Zheng, A. Vulvar Neuroendocrine Carcinoma: A Case Report and Literature Review. Gynecol. Pelvic Med. 2022, 5. [Google Scholar] [CrossRef]
- Saglam, O. Uncommon Morphologic Types of Endometrial Cancer and Their Mimickers: How Much Does Molecular Classification Improve the Practice for Challenging Cases? Life 2024, 14. [Google Scholar] [CrossRef]
- Möller, K.; Uhlig, R.; Gorbokon, N.; Dum, D.; Menz, A.; Büscheck, F.; Luebke, A.M.; Hube-Magg, C.; Hinsch, A.; Höflmayer, D.; et al. Comparison of INSM1 Immunostaining with Established Neuroendocrine Markers Synaptophysin and Chromogranin A in over 14,000 Neuroendocrine and Non-Neuroendocrine Tumors. Mol. Cell. Endocrinol. 2024, 581, 112106. [Google Scholar] [CrossRef]
- Zou, Q.; Zhang, L.; Cheng, Z.; Guo, X.; Cao, D. INSM1 Is Less Sensitive But More Specific Than Synaptophysin in Gynecologic High-Grade Neuroendocrine Carcinomas: An Immunohistochemical Study of 75 Cases With Specificity Test and Literature Review. Am. J. Surg. Pathol. 2021, 45, 147–159. [Google Scholar] [CrossRef]
- Gardner, G.J.; Reidy-Lagunes, D.; Gehrig, P.A. Neuroendocrine Tumors of the Gynecologic Tract: A Society of Gynecologic Oncology (SGO) Clinical Document. Gynecol. Oncol. 2011, 122, 190–198. [Google Scholar] [CrossRef]
- Salvo, G.; Gonzalez Martin, A.; Gonzales, N.R.; Frumovitz, M. Updates and Management Algorithm for Neuroendocrine Tumors of the Uterine Cervix. Int. J. Gynecol. Cancer 2019, 29, 986–995. [Google Scholar] [CrossRef] [PubMed]
- Kendall Bártů, M.; Němejcová, K.; Michálková, R.; Bui, Q.H.; Drozenová, J.; Fabian, P.; Fadare, O.; Hausnerová, J.; Laco, J.; Matěj, R.; et al. Neuroendocrine Marker Expression in Primary Non-Neuroendocrine Epithelial Tumors of the Ovary: A Study of 551 Cases. Int. J. Gynecol. Pathol. 2024, 43, 123–133. [Google Scholar] [CrossRef]
- Gupta, S.; Ahuja, S.; Kalwaniya, D.S. The Evolving Landscape of Immunohistochemistry in Cervical and Uterine Carcinoma in Gynecologic Oncology: Current Status and Future Directions. Obstet. Gynecol. Sci. 2024, 67, 449–466. [Google Scholar] [CrossRef]
- Chen, P.P.; Ramalingam, P.; Alvarado-Cabrero, I.; Euscher, E.D.; Nagarajan, P.; Lawson, B.C.; Malpica, A. High-Grade Neuroendocrine Carcinomas of the Vulva: A Clinicopathologic Study of 16 Cases. Am. J. Surg. Pathol. 2021, 45, 304–316. [Google Scholar] [CrossRef] [PubMed]
- da Mata, S.; Ferreira, J.; Nicolás, I.; Esteves, S.; Esteves, G.; Lérias, S.; Silva, F.; Saco, A.; Cochicho, D.; Cunha, M.; et al. P16 and HPV Genotype Significance in HPV-Associated Cervical Cancer—A Large Cohort of Two Tertiary Referral Centers. Int. J. Mol. Sci. 2021, 22, 2294. [Google Scholar] [CrossRef] [PubMed]
- Bellizzi, A.M. Immunohistochemistry in the Diagnosis and Classification of Neuroendocrine Neoplasms: What Can Brown Do for You? Hum. Pathol. 2020, 96, 8–33. [Google Scholar] [CrossRef] [PubMed]
- Juhlin, C.C.; Zedenius, J.; Höög, A. Metastatic Neuroendocrine Neoplasms of Unknown Primary: Clues from Pathology Workup. Cancers 2022, 14. [Google Scholar] [CrossRef]
- Concin, N.; Matias-Guiu, X.; Cibula, D.; Colombo, N.; Creutzberg, C.L.; Ledermann, J.; Mirza, M.R.; Vergote, I.; Abu-Rustum, N.R.; Bosse, T.; et al. ESGO–ESTRO–ESP Guidelines for the Management of Patients with Endometrial Carcinoma: Update 2025. Policy Rev. [CrossRef]
- Léon-Castillo, A. Update in the Molecular Classification of Endometrial Carcinoma. Int. J. Gynecol. Cancer 2023, 33, 333–342. [Google Scholar] [CrossRef]
- Zheng, W. Molecular Classification of Endometrial Cancer and the 2023 FIGO Staging: Exploring the Challenges and Opportunities for Pathologists. Cancers 2023, 15, 4101. [Google Scholar] [CrossRef] [PubMed]
- Tung, H.-J.; Wu, R.-C.; Lin, C.-Y.; Lai, C.-H. Rare Subtype of Endometrial Cancer: Undifferentiated/Dedifferentiated Endometrial Carcinoma, from Genetic Aspects to Clinical Practice. Int. J. Mol. Sci. 2022, 23. [Google Scholar] [CrossRef] [PubMed]
- McCluggage, W.G.; Stewart, C.J.R. SWI/SNF-Deficient Malignancies of the Female Genital Tract. Semin. Diagn. Pathol. 2021, 38, 199–211. [Google Scholar] [CrossRef]
- WHO Classification Editorial Board. WHO Classification of Tumours: Female Genital Tumours, 5th ed.; International Agency for Research on Cancer: Lyon, France, 2020; Volume 4, ISBN -13. 978-92-832-4504-9. [Google Scholar]
- BacalbaȘa, N.; Stoica, C.; Marcu, M.; Mihalache, D.; Vasilescu, F.; Popa, I.; Mirea, G.; Bălescu, I. Large Cell Neuroendocrine Cervical Tumor Treated by Radical Surgery and Adjuvant Chemotherapy: A Case Report and Literature Review. Oncol. Lett. 2016, 11, 189–193. [Google Scholar] [CrossRef]
- Shamir, E.R.; Devine, W.P.; Pekmezci, M.; Umetsu, S.E.; Krings, G.; Federman, S.; Cho, S.-J.; Saunders, T.A.; Jen, K.-Y.; Bergsland, E.; et al. Identification of High-Risk Human Papillomavirus and Rb/E2F Pathway Genomic Alterations in Mutually Exclusive Subsets of Colorectal Neuroendocrine Carcinoma. Mod. Pathol. 2019, 32, 290–305. [Google Scholar] [CrossRef]
- Stolnicu, S.; Allison, D.; Patrichi, A.; Flynn, J.; Iasonos, A.; Soslow, R.A. Invasive Squamous Cell Carcinoma of the Cervix: A Review of Morphological Appearances Encountered in Human Papillomavirus-Associated and -Independent Tumors and Precursor Lesions. Adv. Anat. Pathol. 2024, 31, 1–14. [Google Scholar] [CrossRef]
- Gordhandas, S.; Schlappe, B.A.; Zhou, Q.; Iasonos, A.; Leitao, M.M.; Park, K.J.; de Brot, L.; Alektiar, K.M.; Sabbatini, P.J.; Aghajanian, C.A.; et al. Small Cell Neuroendocrine Carcinoma of the Cervix: Analysis of Prognostic Factors and Patterns of Metastasis. Gynecol. Oncol. Rep. 2022, 43, 101058. [Google Scholar] [CrossRef] [PubMed]
- Salvo, G.; Meyer, L.A.; Gonzales, N.R.; Frumovitz, M.; Hillman, R.T. Neuroendocrine Cervical Carcinomas: Genomic Insights, Controversies in Treatment Strategies, and Future Directions: A NeCTuR Study. Int. J. Gynecol. Cancer 2025, 35. [Google Scholar] [CrossRef]
- Liu, X.; Tian, Y.; Yan, S.; Fu, H.; Si, L.; Lai, T.; Mao, M.; Wang, Q.; Bai, J.; Li, H.; et al. Neuroendocrine Carcinoma of the Endometrium: A Retrospective Analysis of Data from a Single Center. BMC Cancer 2024, 24, 636. [Google Scholar] [CrossRef]
- Tessier-Cloutier, B.; Coatham, M.; Carey, M.; Nelson, G.S.; Hamilton, S.; Lum, A.; Soslow, R.A.; Stewart, C.J.; Postovit, L.M.; Köbel, M.; et al. SWI/SNF-deficiency Defines Highly Aggressive Undifferentiated Endometrial Carcinoma. J. Pathol. Clin. Res. 2020, 7, 144–153. [Google Scholar] [CrossRef]
- Giordano, G.; Ferioli, E.; Guareschi, D.; Tafuni, A. Dedifferentiated Endometrial Carcinoma: A Rare Aggressive Neoplasm-Clinical, Morphological and Immunohistochemical Features. Cancers 2023, 15. [Google Scholar] [CrossRef]
- Zhai, L.-R.; Zhang, X.-W.; Yu, T.; Jiang, Z.-D.; Huang, D.-W.; Jia, Y.; Cui, M.-H. Primary Ovarian Carcinoid. Medicine 2020, 99, e21109. [Google Scholar] [CrossRef]
- Legarreta, A.F.; Saab, R.; Gonzales, N.R.; Chisholm, G.B.; Westin, S.N.; Hillman, R.T.; Frumovitz, M. Neuroendocrine Neoplasms of the Ovary: A Review of 63 Cases. Int. J. Gynecol. Cancer 2024, 34, 566–573. [Google Scholar] [CrossRef]
- Zhang, X.; Jones, A.; Jenkins, S.M.; Huang, Y. Ki67 Proliferative Index in Carcinoid Tumors Involving Ovary. Endocr. Pathol. 2018, 29, 43–48. [Google Scholar] [CrossRef] [PubMed]
- Tischkowitz, M.; Huang, S.; Banerjee, S.; Hague, J.; Hendricks, W.P.D.; Huntsman, D.G.; Lang, J.D.; Orlando, K.A.; Oza, A.M.; Pautier, P.; et al. Small Cell Carcinoma of the Ovary, Hypercalcaemic Type – Genetics, New Treatment Targets and Current Management Guidelines. Clin. Cancer Res. 2020, 26, 3908–3917. [Google Scholar] [CrossRef] [PubMed]
- Wens, F.S.P.L.; Hulsker, C.C.C.; Fiocco, M.; Zsiros, J.; Smetsers, S.E.; de Krijger, R.R.; van der Steeg, A.F.W.; Zweemer, R.P.; Baas, I.O.; Roes, E.M.; et al. Small Cell Carcinoma of the Ovary, Hypercalcemic Type (SCCOHT): Patient Characteristics, Treatment, and Outcome-A Systematic Review. Cancers 2023, 15, 3794. [Google Scholar] [CrossRef]
- Galanis, I.; Floros, G.; Bompoula, M.S.; Theodoropoulos, C. Mixed Large Cell Neuroendocrine Carcinoma of the Ovary: Report of a Rare Case. Case Rep. Obstet. Gynecol. 2020, 2020, 8896070. [Google Scholar] [CrossRef]
- Barcellini, A.; Golia D’Augè, T.; Mandato, V.D.; Cuccu, I.; Musella, A.; Fruscio, R.; Vitale, M.G.; Martinello, R.; Mangili, G.; Pignata, S.; et al. Primary Neuroendocrine Neoplasms of the Vulva: A Review of the MITO Rare Cancer Group. Crit. Rev. Oncol. Hematol. 2024, 193, 104201. [Google Scholar] [CrossRef] [PubMed]
- Capote, S.; Domènech, M.; Valdivieso, L.; Tuset, V.; Sanchez, M.; Carballas, E.; Teruel, I.; Durany, D.; Moragas, G.; Molina, C.; et al. Small Cell Carcinoma of the Vagina: First Systematic Review of Case Reports and Proposal of a Management Algorithm. J. Low. Genit. Tract. Dis. 2023, 27, 56–67. [Google Scholar] [CrossRef]
- Pongsuvareeyakul, T.; Garcia-Moliner, M.; Lokich, E.; Dizon, D.S.; Singh, K. Small Cell Neuroendocrine Carcinoma of Vagina: Report of a Unique Case with Literature Review. Cancer Treat. Res. Commun. 2022, 33, 100645. [Google Scholar] [CrossRef] [PubMed]
- Hirakawa, T.; Yano, M.; Nishida, H.; Sato, S.; Nasu, K. Vulvar Neuroendocrine Carcinoma That Is Independent of Merkel Cell Polyomavirus and Human Papillomavirus Suggests Endometrial Cancer Recurrence: A Case Report. BMC Endocr. Disord. 2022, 22, 81. [Google Scholar] [CrossRef]
- Bösmüller, H.-C.; Wagner, P.; Pham, D.L.; Fischer, A.K.; Greif, K.; Beschorner, C.; Sipos, B.; Fend, F.; Staebler, A. CD56 (Neural Cell Adhesion Molecule) Expression in Ovarian Carcinomas: Association With High-Grade and Advanced Stage But Not With Neuroendocrine Differentiation. Int. J. Gynecol. Cancer 2017, 27, 239–245. [Google Scholar] [CrossRef] [PubMed]
- Saraei, P.; Heshmati, A.; Hosseini, S. Small-Cell Neuroendocrine Carcinoma of the Female Genital Tract: A Comprehensive Overview. J. Neuroendocrinol. 2024, 36, e13394. [Google Scholar] [CrossRef]
- Čelebić, A.; Harasani, K.; Miladinović, M.; Mandić, A.; Vasiljević, T.; Drusany Starič, K.; Radunović, V.; Tripac, I.; Starič Drusany, A.K. Neuroendocrine Tumors of the Gynecological Tract: A Narrative Literature Review. Eur. J. Surg. Oncol. 2025, 51, 109735. [Google Scholar] [CrossRef] [PubMed]
- Berek, J.S.; Matias-Guiu, X.; Creutzberg, C.; Fotopoulou, C.; Gaffney, D.; Kehoe, S.; Lindemann, K.; Mutch, D.; Concin, N. Endometrial Cancer Staging Subcommittee, FIGO Women’s Cancer Committee FIGO Staging of Endometrial Cancer: 2023. Intl J. Gynecol. Obste 2023, 162, 383–394. [Google Scholar] [CrossRef]
- La Rosa, S.; Sessa, F.; Uccella, S. Mixed Neuroendocrine-Nonneuroendocrine Neoplasms (MiNENs): Unifying the Concept of a Heterogeneous Group of Neoplasms. Endocr. Pathol. 2016, 27, 284–311. [Google Scholar] [CrossRef]
- Guerrera, L.P.; Suarato, G.; Napolitano, R.; Perrone, A.; Caputo, V.; Ventriglia, A.; Martini, G.; Corte, C.M.D.; Orditura, M.; Martinelli, E.; et al. Mixed Neuroendocrine Non-Neuroendocrine Neoplasms of the Gastrointestinal Tract: A Case Series. Healthcare 2022, 10. [Google Scholar] [CrossRef]
- Toor, D.; Loree, J.M.; Gao, Z.-H.; Wang, G.; Zhou, C. Mixed Neuroendocrine-Non-Neuroendocrine Neoplasms of the Digestive System: A Mini-Review. World J. Gastroenterol. 2022, 28, 2076–2087. [Google Scholar] [CrossRef]
- Opalińska, M.; Sowa-Staszczak, A.; Olearska, H.; Ulatowska-Bialas, M.; Gilis-Januszewska, A.; Hubalewska-Dydejczyk, A. Clinical Approach to Neuroendocrine Neoplasm Associated With Ovarian Teratoma. Front. Endocrinol. 2021, 12. [Google Scholar] [CrossRef]
- Cancer Protocol Templates. Available online: https://www.cap.org/protocols-and-guidelines/cancer-reporting-tools/cancer-protocol-templates (accessed on 2 May 2026).
- Höhn, A.K.; Brambs, C.E.; Hiller, G.G.R.; May, D.; Schmoeckel, E.; Horn, L.-C. 2020 WHO Classification of Female Genital Tumors. Geburtshilfe Frauenheilkd. 2021, 81, 1145–1153. [Google Scholar] [CrossRef]
- Ishida, H.; Kasajima, A.; Yamauchi, T.; Akaishi, R.; Ueki, S.; Taniyama, Y.; Fujishima, F.; Koike, T.; Kamei, T.; Lam, A.K.-Y.; et al. A Diagnostic Pitfall; Small Cell Carcinoma-like Features in Basaloid Squamous Cell Carcinoma of the Esophagus. Histol. Histopathol. 2023, 38, 155–163. [Google Scholar] [CrossRef]
- Saha, S.; Ghosh, P.; Mukherjee, G.; Roy, A. Metastatic Primary Neuroendocrine Tumor of Ovary—A Rare Presentation. Indian J. Radiol. Imaging 2022, 32, 270–274. [Google Scholar] [CrossRef]
- Oneda, E.; Zorzi, F.; Gorio, A.; Quaglia, F.; Abeni, C.; Rota, L.; Zaniboni, A. Differential Diagnosis of Small Cell Carcinoma of the Ovary or Ovarian Metastases of Small Cell Carcinoma of the Lung: A Case Report and Review of the Literature. Case Rep. Oncol. 2020, 13, 822–828. [Google Scholar] [CrossRef] [PubMed]
- Agoff, S.N.; Lamps, L.W.; Philip, A.T.; Amin, M.B.; Schmidt, R.A.; True, L.D.; Folpe, A.L. Thyroid Transcription Factor-1 Is Expressed in Extrapulmonary Small Cell Carcinomas but Not in Other Extrapulmonary Neuroendocrine Tumors. Mod. Pathol. 2000, 13, 238–242. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Yuan, J.; Wei, L.; Zhou, L.; Mei, K.; Yue, J.; Gao, H.; Zhang, M.; Jia, L.; Kang, Q.; et al. SATB2 Is a Sensitive Marker for Lower Gastrointestinal Well-Differentiated Neuroendocrine Tumors. Int. J. Clin. Exp. Pathol. 2015, 8, 7072–7082. [Google Scholar] [PubMed]
- Mahdi, H.; Joehlin-Price, A.; Elishaev, E.; Dowlati, A.; Abbas, A. Genomic Analyses of High-grade Neuroendocrine Gynecological Malignancies Reveal a Unique Mutational Landscape and Therapeutic Vulnerabilities. Mol. Oncol. 2021, 15, 3545–3558. [Google Scholar] [CrossRef]
- Addante, F.; d’Amati, A.; Santoro, A.; Angelico, G.; Inzani, F.; Arciuolo, D.; Travaglino, A.; Raffone, A.; D’Alessandris, N.; Scaglione, G.; et al. Mismatch Repair Deficiency as a Predictive and Prognostic Biomarker in Endometrial Cancer: A Review on Immunohistochemistry Staining Patterns and Clinical Implications. Int. J. Mol. Sci. 2024, 25, 1056. [Google Scholar] [CrossRef]
- Kumari, A.; Kumar, H.; Harvey, S.E.; Xing, D.; Li, Z. Molecular Classification of Endometrial Carcinomas: Review and Recent Updates. Cancers 2025, 18, 51. [Google Scholar] [CrossRef]
- Galant, N.; Krawczyk, P.; Monist, M.; Obara, A.; Gajek, Ł.; Grenda, A.; Nicoś, M.; Kalinka, E.; Milanowski, J. Molecular Classification of Endometrial Cancer and Its Impact on Therapy Selection. Int. J. Mol. Sci. 2024, 25. [Google Scholar] [CrossRef]
- Ordulu, Z.; Mino-Kenudson, M.; Young, R.H.; Van de Vijver, K.; Zannoni, G.F.; Félix, A.; Burandt, E.; Wong, A.; Nardi, V.; Oliva, E. Morphologic and Molecular Heterogeneity of Cervical Neuroendocrine Neoplasia: A Report of 14 Cases. Am. J. Surg. Pathol. 2022, 46, 1670–1681. [Google Scholar] [CrossRef] [PubMed]
- Masuda, M.; Iida, K.; Iwabuchi, S.; Tanaka, M.; Kubota, S.; Uematsu, H.; Onuma, K.; Kukita, Y.; Kato, K.; Kamiura, S.; et al. Clonal Origin and Lineage Ambiguity in Mixed Neuroendocrine Carcinoma of the Uterine Cervix. Am. J. Pathol. 2024, 194, 415–429. [Google Scholar] [CrossRef]
- Bellone, S.; Jeong, K.; Halle, M.K.; Krakstad, C.; McNamara, B.; Greenman, M.; Mutlu, L.; Demirkiran, C.; Hartwich, T.M.P.; Yang-Hartwich, Y.; et al. Integrated Mutational Landscape Analysis of Poorly Differentiated High-Grade Neuroendocrine Carcinoma of the Uterine Cervix. Proc. Natl. Acad. Sci. U S A 2024, 121, e2321898121. [Google Scholar] [CrossRef] [PubMed]
- Joshi, A.; Bhaskar, N.; Pearson, J.D. Neuroendocrine Transformation as a Mechanism of Resistance to Targeted Lung Cancer Therapies: Emerging Mechanisms and Their Therapeutic Implications. Cancers 2025, 17. [Google Scholar] [CrossRef]
- Imamura, J.; Ganguly, S.; Muskara, A.; Liao, R.S.; Nguyen, J.K.; Weight, C.; Wee, C.E.; Gupta, S.; Mian, O.Y. Lineage Plasticity and Treatment Resistance in Prostate Cancer: The Intersection of Genetics, Epigenetics, and Evolution. Front Endocrinol. 2023, 14, 1191311. [Google Scholar] [CrossRef]
- Oluloro, A.; Wells, D.L.; Childers, C.K.; Luu, T.; Eaton, K.D.; Urban, R.R.; Konnick, E.Q.; Paulson, V.A.; Banda, K. Revealing Neuroendocrine Transformation in Gynecological Cancers through Genomic Analysis. npj Precis Oncol. 2025, 9, 77. [Google Scholar] [CrossRef]
- Verma, S.; Dubey, H.; Gupta, S.; Ranjan, A.; Goel, H.; Sharma, A. Neuroendocrine Carcinoma of Cervix and Review Literature. Int. J. Surg. Case Rep. 2023, 105, 107982. [Google Scholar] [CrossRef]
- Fernandes, J.; Hodgson, A.; Han, K.; Milosevic, M.; Lukovic, J.; Lheureux, S.; Ferguson, S.E.; Santiago, A.; Croke, J. Clinical Outcomes of Neuroendocrine Carcinoma of the Cervix: Retrospective Review from a Large Academic Cancer Centre. Gynecol. Oncol. Rep. 2025, 61, 101933. [Google Scholar] [CrossRef]
- Salvo, G.; Legarreta, A.F.; Ramalingam, P.; Jhingran, A.; Bhosale, P.; Saab, R.; Gonzales, N.R.; Chisholm, G.B.; Frumovitz, M. Clinicopathologic Characteristics, Oncologic Outcomes, and Prognostic Factors in Neuroendocrine Cervical Carcinoma: A Neuroendocrine Cervical Tumor Registry Study. Int. J. Gynecol. Cancer 2023, 33, 1359–1369. [Google Scholar] [CrossRef] [PubMed]
- Mantilidewi, K.I.; Winarno, G.N.A.; Harsono, A.B.; Suardi, D.; Hidayat, Y.M.; Kurniadi, A.; Salima, S.; Erfiandi, F.; Haniah, A.S.; Kampan, N.C. Management Paradigm for Ovarian Neuroendocrine Carcinoma: A Systematic Review. J. Ovarian Res. 2025, 18, 125. [Google Scholar] [CrossRef]
- Lamarca, A.; Bartsch, D.K.; Caplin, M.; Kos-Kudla, B.; Kjaer, A.; Partelli, S.; Rinke, A.; Janson, E.T.; Thirlwell, C.; van Velthuysen, M.-L.F.; et al. European Neuroendocrine Tumor Society (ENETS) 2024 Guidance Paper for the Management of Well-Differentiated Small Intestine Neuroendocrine Tumours. J. Neuroendocrinol. 2024, 36, e13423. [Google Scholar] [CrossRef] [PubMed]
- Ren, X.; Wu, W.; Li, Q.; Li, W.; Wang, G. Advances in Research, Diagnosis, and Treatment of Neuroendocrine Cervical Carcinoma: A Review. Oncol. Rev. 2023, 17, 11764. [Google Scholar] [CrossRef] [PubMed]
- Eads, J.R.; Halfdanarson, T.R.; Asmis, T.; Bellizzi, A.M.; Bergsland, E.K.; Dasari, A.; El-Haddad, G.; Frumovitz, M.; Meyer, J.; Mittra, E.; et al. Expert Consensus Practice Recommendations of the North American Neuroendocrine Tumor Society for the Management of High Grade Gastroenteropancreatic and Gynecologic Neuroendocrine Neoplasms. Endocr. Relat. Cancer 2023, 30, e220206. [Google Scholar] [CrossRef]
- Pang, L.; Guo, Z. Primary Neuroendocrine Tumors of the Ovary: Management and Outcomes. Cancer Med. 2021, 10, 8558–8569. [Google Scholar] [CrossRef] [PubMed]
| Marker | Typical staining pattern | Main diagnostic value | Key limitations / pitfalls | Practical interpretation |
| Synaptophysin | Cytoplasmic, often diffuse in true neuroendocrine tumors and carcinomas | Sensitive marker of neuroendocrine differentiation; useful as part of the initial panel | Not entirely specific; focal or patchy expression may occur in conventional gynecologic carcinomas | Strongest when diffuse and concordant with neuroendocrine morphology |
| Chromogranin A | Cytoplasmic granular staining | More specific marker; particularly useful in well-differentiated neuroendocrine tumors | Less sensitive in poorly differentiated neuroendocrine carcinomas; may be weak or negative | Negative staining does not exclude neuroendocrine carcinoma if morphology and other markers support the diagnosis |
| CD56 | Membranous and/or cytoplasmic staining | Sensitive supportive marker | Low specificity; isolated positivity is a common diagnostic pitfall | Should not be used alone to diagnose neuroendocrine differentiation or neuroendocrine carcinoma |
| INSM1 | Nuclear staining | Useful marker, especially in poorly differentiated neuroendocrine carcinoma; easier to interpret in crushed or small biopsy material | Not completely specific; must still be interpreted in morphologic context | Strong nuclear staining supports neuroendocrine differentiation when morphology is appropriate |
| Ki-67 | Nuclear proliferation index | Helps assess proliferative activity and supports distinction between well-differentiated NET and poorly differentiated NEC in the appropriate context | Does not establish neuroendocrine lineage | Should be interpreted as a proliferation marker, not as evidence of neuroendocrine differentiation |
| Cytokeratins / EMA | Cytoplasmic and/or membranous epithelial staining | Confirm epithelial lineage in poorly differentiated tumors | May be reduced or focal in some high-grade tumors | Essential before diagnosing poorly differentiated neuroendocrine carcinoma, especially in small round cell tumors |
| p16 / HPV testing | p16 block-type staining; HPV detected by ISH or molecular methods | Supports HPV-associated cervical or lower genital tract origin when morphology and clinical context fit | p16 is not site-specific and can be positive in non-cervical high-grade tumors | Most useful in cervical tumors and selected vaginal/vulvar tumors |
| PAX8, ER, PR, WT1 | Nuclear staining, depending on marker | Supports Müllerian, endometrial or ovarian lineage in appropriate settings | None is definitive alone; expression may vary by tumor type and differentiation | Used as site-directed markers within a broader panel |
| p53 / MMR proteins | p53 wild-type or abnormal pattern; retained or lost MMR protein expression | Provides molecular context, especially in endometrial tumors | Does not define neuroendocrine differentiation | Useful for classifying high-grade endometrial tumors and identifying therapeutically relevant subgroups |
| SMARCA4 / SMARCB1 / SWI/SNF markers | Retained or lost nuclear expression | Helps identify SWI/SNF-deficient mimics such as dedifferentiated carcinoma or SCCOHT | Should be applied selectively based on morphology and clinical context | Loss of expression may redirect diagnosis away from neuroendocrine carcinoma |
| TTF-1, CDX2, SATB2, CK20, MCPyV | Site-dependent nuclear, cytoplasmic or membranous patterns | Helps evaluate metastatic pulmonary, gastrointestinal, pancreatic or Merkel cell origin | Expression overlap may occur; no marker proves origin in isolation | Must be interpreted with clinical history, imaging and disease distribution |
| Scenario | Findings | Preferred diagnostic wording | Suggested comment | Diagnostic rationale |
| Conventional carcinoma with focal neuroendocrine marker expression | Conventional morphology; focal or weak staining for one or more neuroendocrine markers; no neuroendocrine architecture or cytology | “Conventional carcinoma, with focal neuroendocrine marker expression” | “The findings do not support a diagnosis of neuroendocrine carcinoma.” | Avoids overdiagnosis and inappropriate treatment intensification |
| Isolated CD56 positivity | Conventional morphology; CD56 positive; synaptophysin, chromogranin A and/or INSM1 negative or non-supportive | Do not diagnose neuroendocrine differentiation based on CD56 alone | “CD56 expression is nonspecific and, in isolation, is insufficient for neuroendocrine classification.” | Prevents misclassification based on a low-specificity marker |
| Partial morphology and partial IHC support | Focal organoid/trabecular growth, rosettes or nuclear molding; limited or heterogeneous neuroendocrine marker expression | “Carcinoma with neuroendocrine differentiation” | “The neuroendocrine features are focal/limited and insufficient for definitive classification as neuroendocrine carcinoma.” | Communicates uncertainty while acknowledging a potentially relevant phenotype |
| Limited biopsy with high-grade neuroendocrine features | Small biopsy shows high-grade carcinoma with neuroendocrine morphology and supportive markers, but tumor extent cannot be assessed | “High-grade carcinoma with neuroendocrine differentiation” or “Poorly differentiated neuroendocrine carcinoma, if supported by morphology and diffuse markers” | “Definitive classification and assessment of any associated non-neuroendocrine component may require evaluation of the resection specimen.” | Avoids overquantification and recognizes sampling limitations |
| Distinct neuroendocrine and non-neuroendocrine components | Separate morphologic components, each supported by appropriate IHC | “Mixed neuroendocrine and non-neuroendocrine tumor” | “Both components should be reported, with approximate proportions when possible.” | Ensures that the clinically aggressive component is not missed |
| Poorly differentiated neuroendocrine carcinoma | Classic small cell or large cell morphology; necrosis; high mitotic activity; diffuse multimarker neuroendocrine expression | “Poorly differentiated neuroendocrine carcinoma, small cell type” or “large cell type” | “Correlation with site-specific findings and exclusion of metastatic disease are recommended when clinically indicated.” | Identifies a high-risk tumor category with therapeutic implications |
| Well-differentiated ovarian neuroendocrine tumor | Well-differentiated neuroendocrine morphology; supportive markers; ovarian setting, often with teratomatous or mucinous elements | “Well-differentiated neuroendocrine tumor of the ovary” | “Features supporting primary ovarian origin versus metastasis should be documented.” | Separates indolent primary ovarian NETs from aggressive NECs and metastases |
| Possible metastatic neuroendocrine neoplasm | Neuroendocrine morphology and markers; atypical site, bilateral ovarian disease, extraovarian disease or clinical history of extragenital NET/NEC | “Neuroendocrine neoplasm involving [site], favor metastatic origin” or “primary site uncertain” | “Clinicoradiologic correlation is required to determine the primary site.” | Prevents unsupported assignment of a gynecologic primary |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




