Submitted:
01 May 2026
Posted:
05 May 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Metabolic Adaptations of Cancer Stem Cells
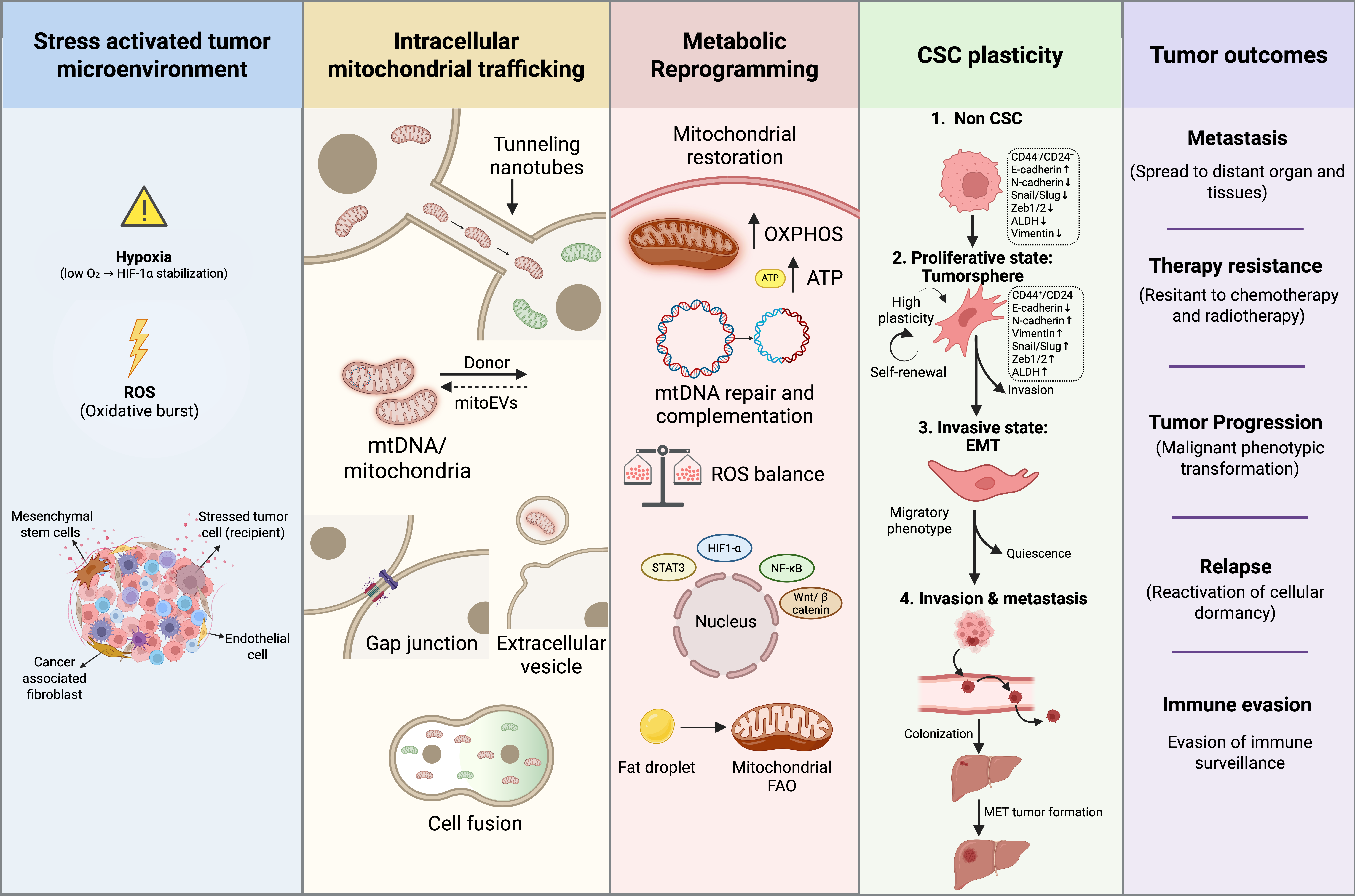
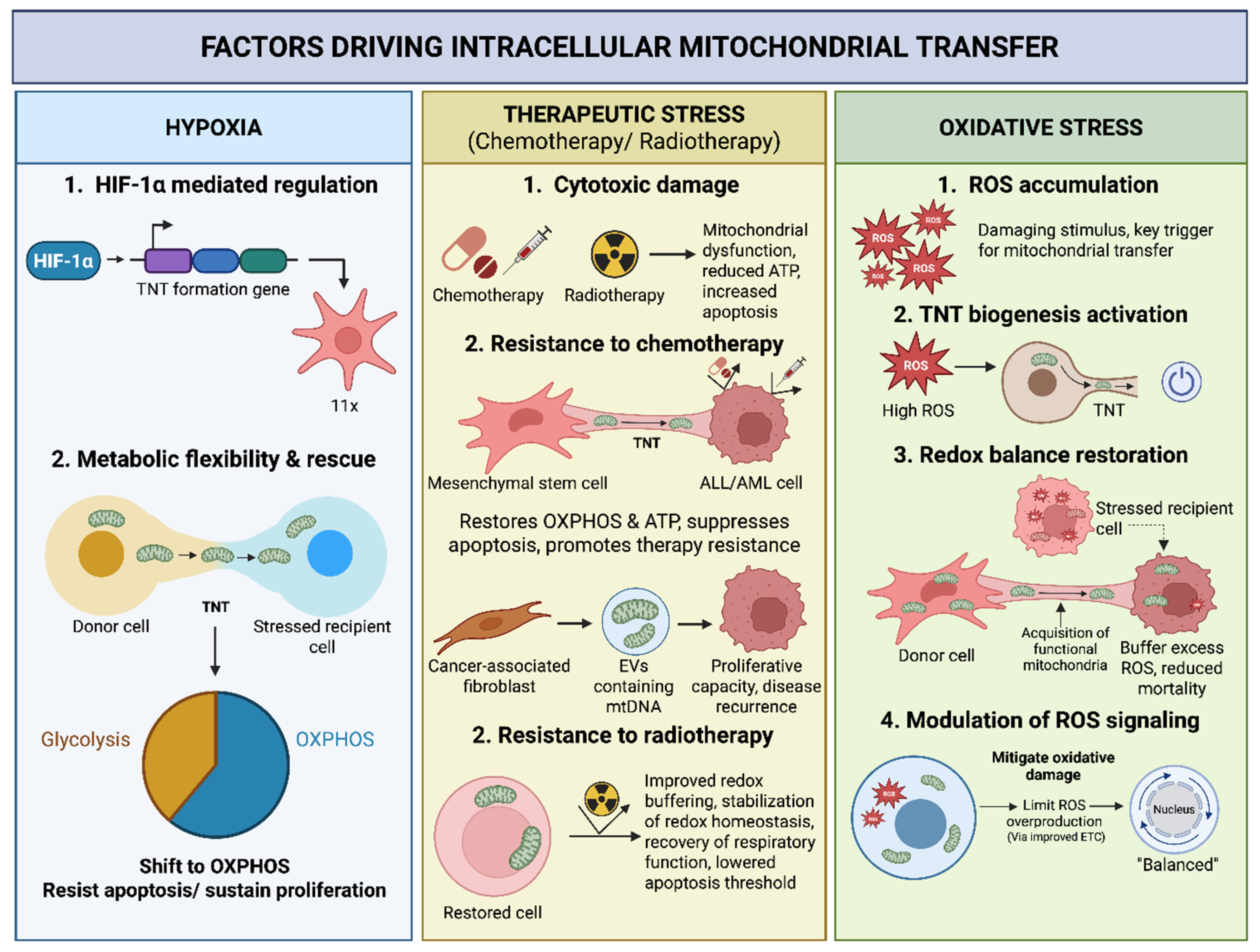
3. Tumor Microenvironment as a Driver of Mitochondrial Transfer
3.1. Hypoxia
3.2. Chemotherapy and Radiation Stress
3.3. Oxidative Stress
3.4. Metabolic Crisis in Tumor Cells
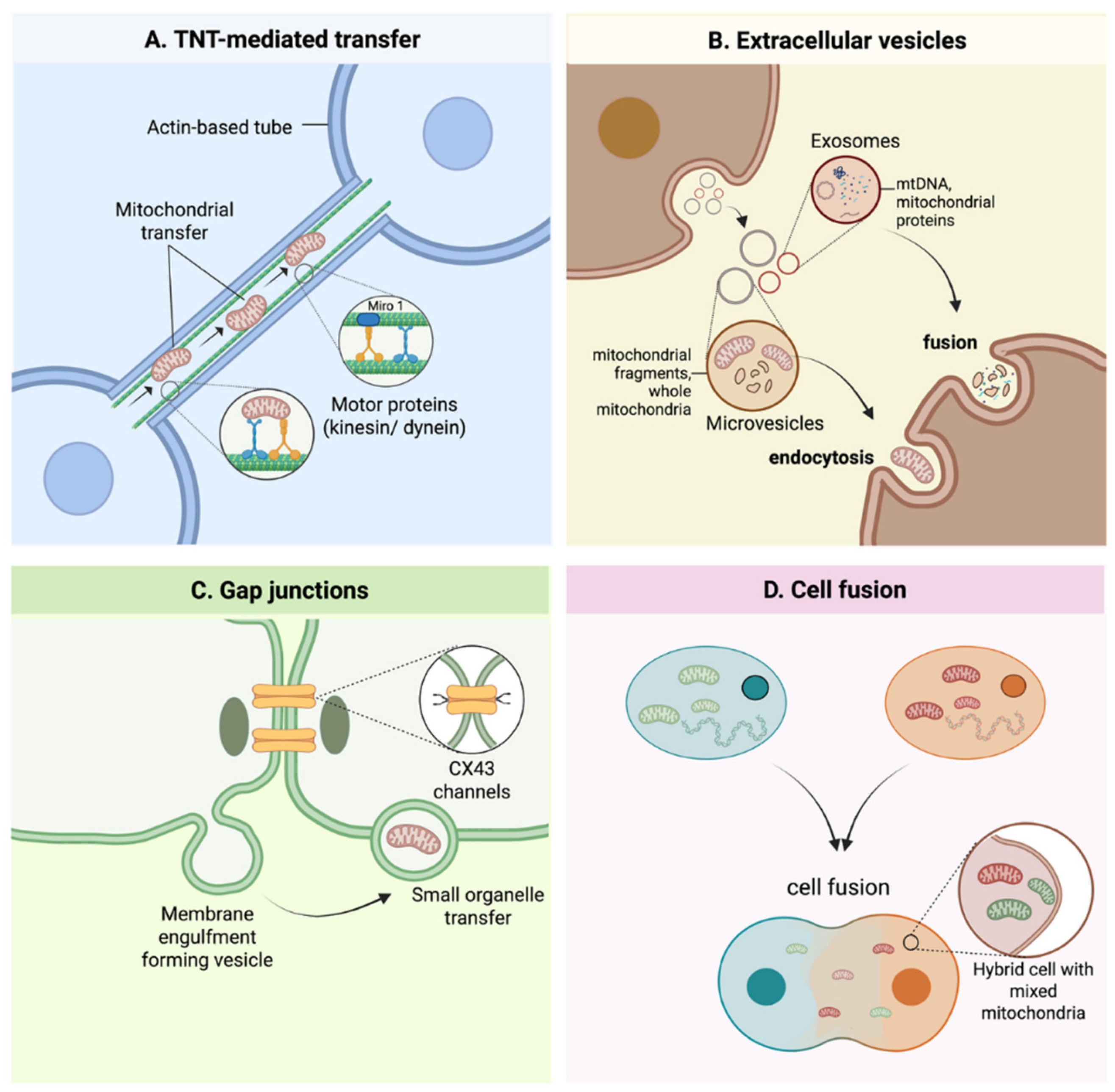
4. Modes of Mitochondrial Transfer
4.1. Tunnelling Nanotubes (TNTs)
4.2. Extracellular Vesicles (EVs)
4.3. Gap Junctions (GJs)
4.4. Cell Fusion
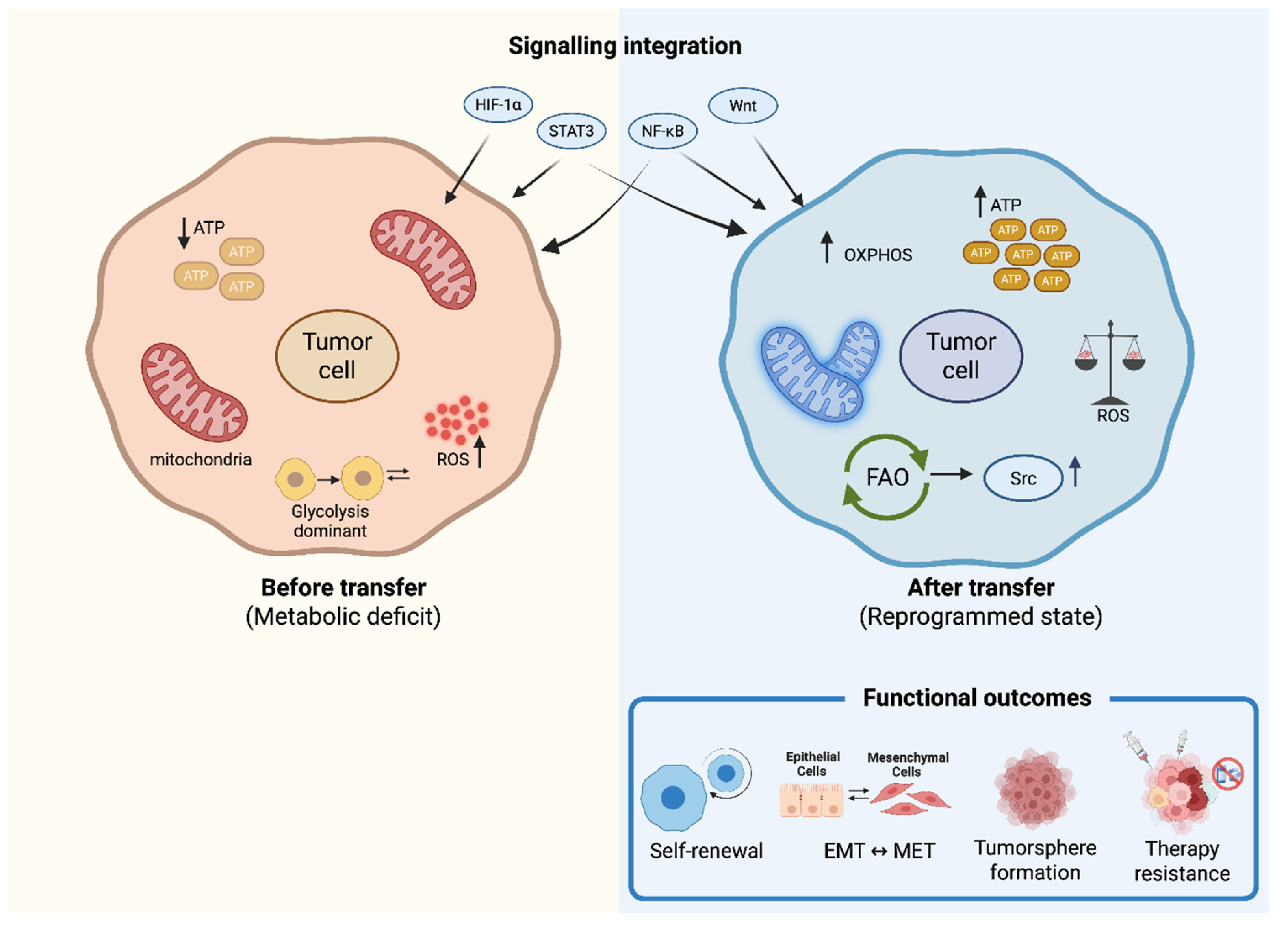
5. Metabolic Reprogramming After Mitochondrial Transfer
5.1. Restoration of Oxidative Phosphorylation and ATP Production
5.2. Metabolic Flexibility Through Integration of Glycolysis and OXPHOS
5.3. mtDNA Transfer and Repair of Mitochondrial Defects
5.4. Redox Recalibration and ROS Signalling
5.5. Fatty Acid Oxidation Couples Mitochondrial Transfer to Sarcoma (Src) Tyrosine Kinase Driven Oncogenic Signalling
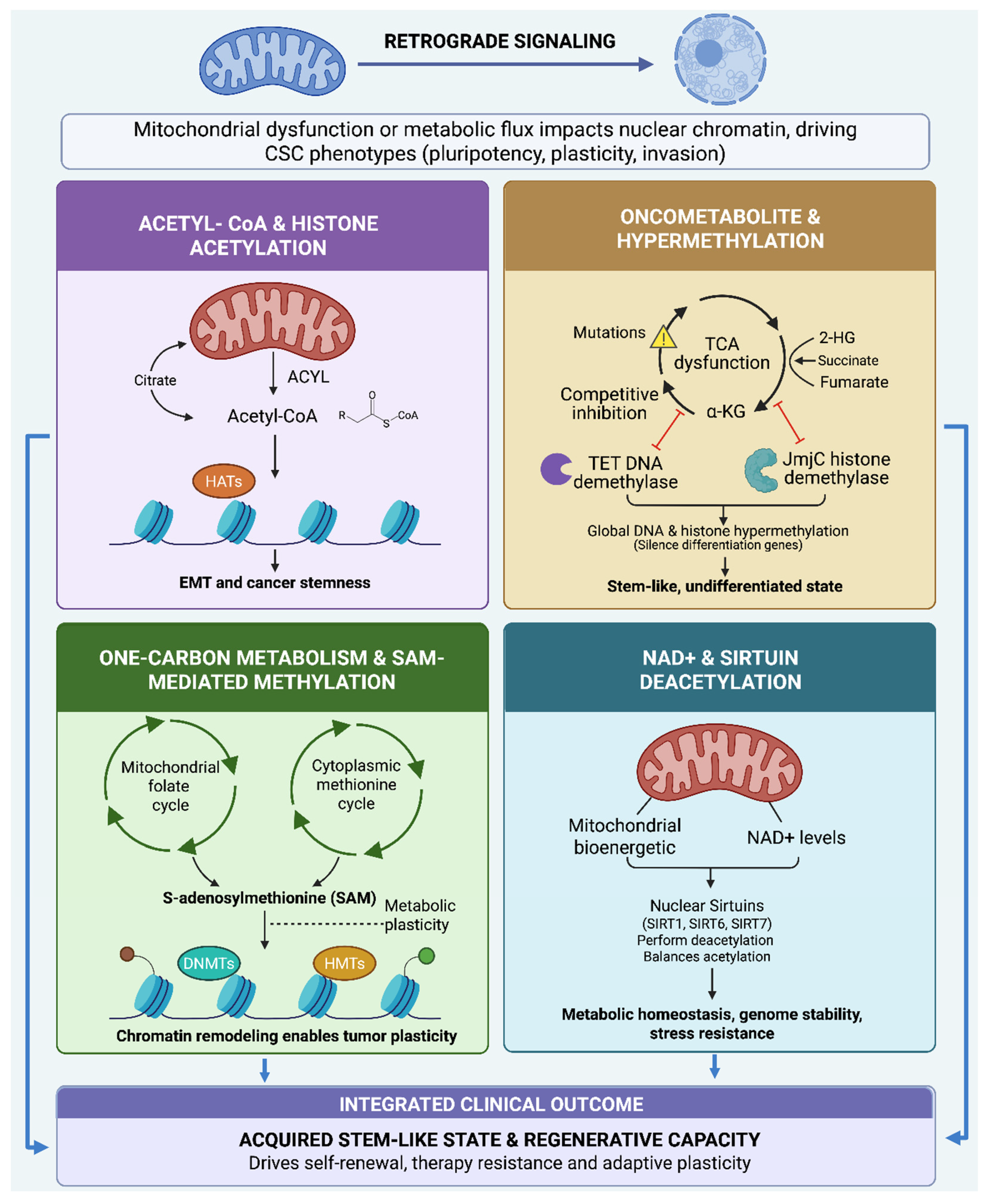
6. Mitochondria Derived Metabolites as Regulators of Nuclear Signaling and Epigenetic Reprogramming
7. Mitochondrial Transfer in Csc Plasticity and Tumor Progression
7.1. Epithelial to Mesenchymal Transition (EMT) Driven by Mitochondrial Transfer
7.2. Migration, Invasion, and Cytoskeletal Dynamics
7.3. Metastatic Colonization and Niche Formation
7.4. Intratumoral Heterogeneity and Clonal Selection
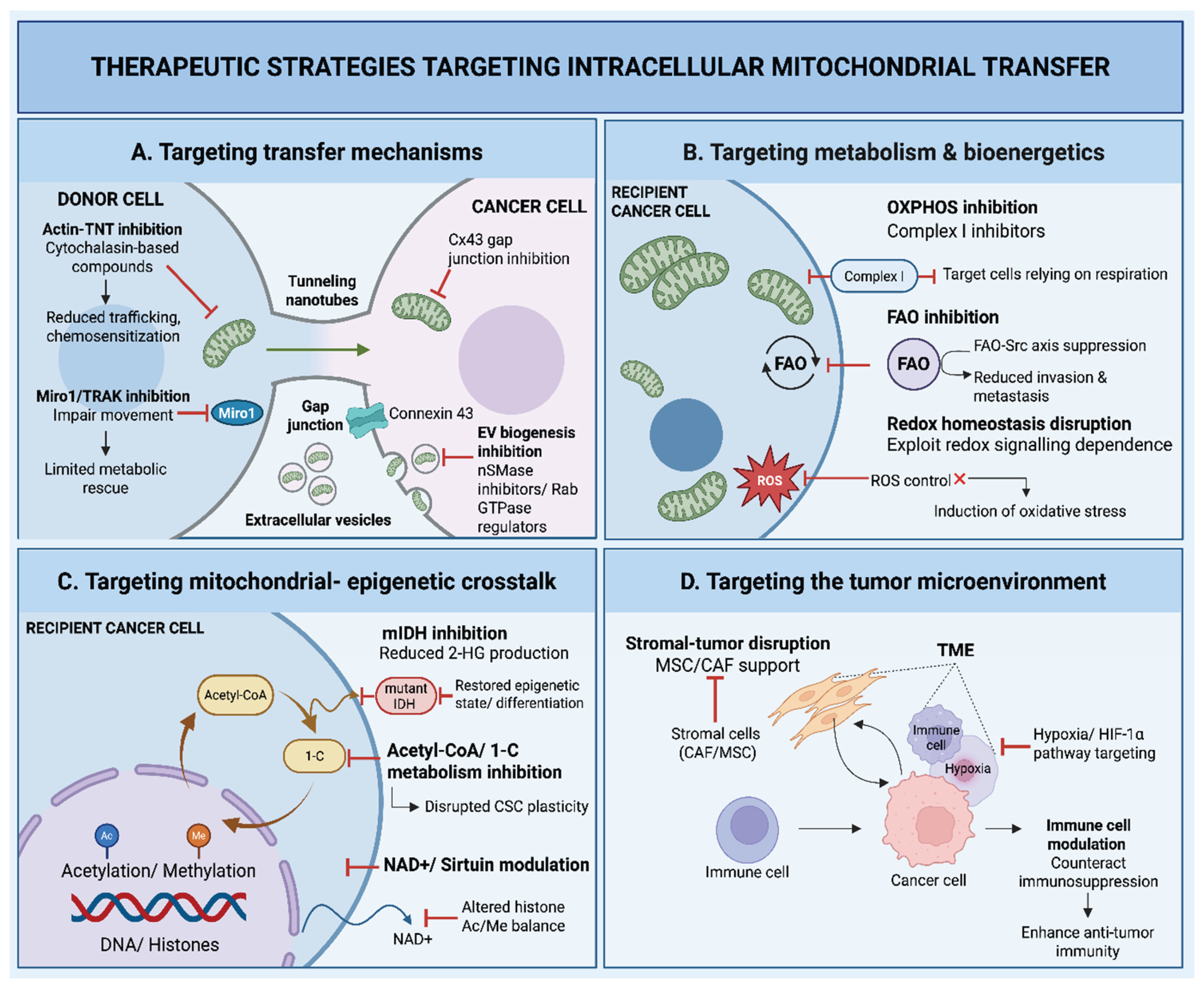
8. Therapeutic Implications and Targeting of Mitochondrial Transfer
8.1. Targeting Mitochondrial Transfer Mechanisms
8.2. Targeting Mitochondrial Metabolism and Bioenergetics
8.3. Targeting Mitochondrial Epigenetic Crosstalk
8.4. Targeting the Tumor Microenvironment
8.5. Challenges and Future Perspectives
9. Discussion
10. Conclusions
11. Future Directions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Siegel RL, Giaquinto AN, Jemal A. Cancer statistics, 2024. CA Cancer J Clin 2024; 74: 12-49. [CrossRef]
- Chu X, Tian W, Ning J, Xiao G, Zhou Y, Wang Z, Zhai Z, Tanzhu G, Yang J, Zhou R. Cancer stem cells: advances in knowledge and implications for cancer therapy. Signal Transduct Target Ther 2024; 9: 170. [CrossRef]
- Bonnet D, Dick JE. Human acute myeloid leukemia is organized as a hierarchy that originates from a primitive hematopoietic cell. Nat Med 1997; 3: 730-737. [CrossRef]
- Ailles LE, Weissman IL. Cancer stem cells in solid tumors. Curr Opin Biotechnol 2007; 18: 460-466. [CrossRef]
- Batlle E, Clevers H. Cancer stem cells revisited. Nat Med 2017; 23: 1124-1134. [CrossRef]
- Mathan SV, Singh RP. Cancer Stem Cells Connecting to Immunotherapy: Key Insights, Challenges, and Potential Treatment Opportunities. Cancers (Basel) 2025; 17. [CrossRef]
- Xie Y, Ma S, Tong M. Metabolic Plasticity of Cancer Stem Cells in Response to Microenvironmental Cues. Cancers (Basel) 2022; 14. [CrossRef]
- Selvaraj NR, Nandan D, Nair BG, Nair VA, Venugopal P, Aradhya R. Oxidative Stress and Redox Imbalance: Common Mechanisms in Cancer Stem Cells and Neurodegenerative Diseases. Cells 2025; 14. [CrossRef]
- Nakano T, Irie K, Matsuo K, Mishima K, Nakamura Y. Molecular and cellular mechanisms of mitochondria transfer in models of central nervous system disease. J Cereb Blood Flow Metab 2026; 46: 269-288. [CrossRef]
- Dong LF, Kovarova J, Bajzikova M, Bezawork-Geleta A, Svec D, Endaya B, Sachaphibulkij K, Coelho AR, Sebkova N, Ruzickova A, Tan AS, Kluckova K, Judasova K, Zamecnikova K, Rychtarcikova Z, Gopalan V, Andera L, Sobol M, Yan B, Pattnaik B, Bhatraju N, Truksa J, Stopka P, Hozak P, Lam AK, Sedlacek R, Oliveira PJ, Kubista M, Agrawal A, Dvorakova-Hortova K, Rohlena J, Berridge MV, Neuzil J. Horizontal transfer of whole mitochondria restores tumorigenic potential in mitochondrial DNA-deficient cancer cells. Elife 2017; 6. [CrossRef]
- Tan AS, Baty JW, Dong LF, Bezawork-Geleta A, Endaya B, Goodwin J, Bajzikova M, Kovarova J, Peterka M, Yan B, Pesdar EA, Sobol M, Filimonenko A, Stuart S, Vondrusova M, Kluckova K, Sachaphibulkij K, Rohlena J, Hozak P, Truksa J, Eccles D, Haupt LM, Griffiths LR, Neuzil J, Berridge MV. Mitochondrial genome acquisition restores respiratory function and tumorigenic potential of cancer cells without mitochondrial DNA. Cell Metab 2015; 21: 81-94. [CrossRef]
- Paul R, Dorsey JF, Fan Y. Cell plasticity, senescence, and quiescence in cancer stem cells: Biological and therapeutic implications. Pharmacol Ther 2022; 231: 107985. [CrossRef]
- Chen W, Zhao H, Li Y. Mitochondrial dynamics in health and disease: mechanisms and potential targets. Signal Transduct Target Ther 2023; 8: 333. [CrossRef]
- Martinez-Reyes I, Chandel NS. Mitochondrial TCA cycle metabolites control physiology and disease. Nat Commun 2020; 11: 102. [CrossRef]
- O’Malley J, Kumar R, Inigo J, Yadava N, Chandra D. Mitochondrial Stress Response and Cancer. Trends Cancer 2020; 6: 688-701.
- Sancho P, Barneda D, Heeschen C. Hallmarks of cancer stem cell metabolism. Br J Cancer 2016; 114: 1305-1312. [CrossRef]
- Bai F, Liu S, Liu X, Hollern DP, Scott A, Wang C, Zhang L, Fan C, Fu L, Perou CM, Zhu WG, Pei XH. PDGFRbeta is an essential therapeutic target for BRCA1-deficient mammary tumors. Breast Cancer Res 2021; 23: 10. [CrossRef]
- Jin X, Zhang N, Yan T, Wei J, Hao L, Sun C, Zhao H, Jiang S. Lactate-mediated metabolic reprogramming of tumor-associated macrophages: implications for tumor progression and therapeutic potential. Front Immunol 2025; 16: 1573039. [CrossRef]
- Sriramkumar S, Sood R, Huntington TD, Ghobashi AH, Vuong TT, Metcalfe TX, Wang W, Nephew KP, O’Hagan HM. Platinum-induced mitochondrial OXPHOS contributes to cancer stem cell enrichment in ovarian cancer. J Transl Med 2022; 20: 246. [CrossRef]
- Lou E, Zhai E, Sarkari A, Desir S, Wong P, Iizuka Y, Yang J, Subramanian S, McCarthy J, Bazzaro M, Steer CJ. Cellular and Molecular Networking Within the Ecosystem of Cancer Cell Communication via Tunneling Nanotubes. Front Cell Dev Biol 2018; 6: 95. [CrossRef]
- Valdebenito S, Malik S, Luu R, Loudig O, Mitchell M, Okafo G, Bhat K, Prideaux B, Eugenin EA. Tunneling nanotubes, TNT, communicate glioblastoma with surrounding non-tumor astrocytes to adapt them to hypoxic and metabolic tumor conditions. Sci Rep 2021; 11: 14556. [CrossRef]
- Raghavan A, Rao P, Neuzil J, Pountney DL, Nath S. Oxidative stress and Rho GTPases in the biogenesis of tunnelling nanotubes: implications in disease and therapy. Cell Mol Life Sci 2021; 79: 36. [CrossRef]
- Piwocka O, Piotrowski I, Suchorska WM, Kulcenty K. Dynamic interactions in the tumor niche: how the cross-talk between CAFs and the tumor microenvironment impacts resistance to therapy. Front Mol Biosci 2024; 11: 1343523. [CrossRef]
- Chun S, An J, Kim MS. Mitochondrial Transfer Between Cancer and T Cells: Implications for Immune Evasion. Antioxidants (Basel) 2025; 14. [CrossRef]
- Guan F, Wu X, Zhou J, Lin Y, He Y, Fan C, Zeng Z, Xiong W. Mitochondrial transfer in tunneling nanotubes-a new target for cancer therapy. J Exp Clin Cancer Res 2024; 43: 147. [CrossRef]
- Jin P, Jiang J, Zhou L, Huang Z, Nice EC, Huang C, Fu L. Mitochondrial adaptation in cancer drug resistance: prevalence, mechanisms, and management. J Hematol Oncol 2022; 15: 97. [CrossRef]
- Dong LF, Rohlena J, Zobalova R, Nahacka Z, Rodriguez AM, Berridge MV, Neuzil J. Mitochondria on the move: Horizontal mitochondrial transfer in disease and health. J Cell Biol 2023; 222. [CrossRef]
- Goncalves AC, Richiardone E, Jorge J, Polonia B, Xavier CPR, Salaroglio IC, Riganti C, Vasconcelos MH, Corbet C, Sarmento-Ribeiro AB. Impact of cancer metabolism on therapy resistance - Clinical implications. Drug Resist Updat 2021; 59: 100797. [CrossRef]
- Sheehan C, Muir A. The requirement for mitochondrial respiration in cancer varies with disease stage. PLoS Biol 2022; 20: e3001800. [CrossRef]
- Neuzil J, Berridge MV. Mitochondria break through cellular boundaries. Aging (Albany NY) 2019; 11: 4308-4309. [CrossRef]
- Liu D, Gao Y, Liu J, Huang Y, Yin J, Feng Y, Shi L, Meloni BP, Zhang C, Zheng M, Gao J. Intercellular mitochondrial transfer as a means of tissue revitalization. Signal Transduct Target Ther 2021; 6: 65. [CrossRef]
- Zhang H, Menzies KJ, Auwerx J. The role of mitochondria in stem cell fate and aging. Development 2018; 145. [CrossRef]
- Rustom A, Saffrich R, Markovic I, Walther P, Gerdes HH. Nanotubular highways for intercellular organelle transport. Science 2004; 303: 1007-1010. [CrossRef]
- Qin Y, Jiang X, Yang Q, Zhao J, Zhou Q, Zhou Y. The Functions, Methods, and Mobility of Mitochondrial Transfer Between Cells. Front Oncol 2021; 11: 672781. [CrossRef]
- Cangkrama M, Liu H, Wu X, Yates J, Whipman J, Gabelein CG, Matsushita M, Ferrarese L, Sander S, Castro-Giner F, Asawa S, Sznurkowska MK, Kopf M, Dengjel J, Boeva V, Aceto N, Vorholt JA, Werner S. MIRO2-mediated mitochondrial transfer from cancer cells induces cancer-associated fibroblast differentiation. Nat Cancer 2025; 6: 1714-1733. [CrossRef]
- Watson DC, Bayik D, Storevik S, Moreino SS, Sprowls SA, Han J, Augustsson MT, Lauko A, Sravya P, Rosland GV, Troike K, Tronstad KJ, Wang S, Sarnow K, Kay K, Lunavat TR, Silver DJ, Dayal S, Joseph JV, Mulkearns-Hubert E, Ystaas LAR, Deshpande G, Guyon J, Zhou Y, Magaut CR, Seder J, Neises L, Williford SE, Meiser J, Scott AJ, Sajjakulnukit P, Mears JA, Bjerkvig R, Chakraborty A, Daubon T, Cheng F, Lyssiotis CA, Wahl DR, Hjelmeland AB, Hossain JA, Miletic H, Lathia JD. GAP43-dependent mitochondria transfer from astrocytes enhances glioblastoma tumorigenicity. Nat Cancer 2023; 4: 648-664. [CrossRef]
- Ahmad T, Mukherjee S, Pattnaik B, Kumar M, Singh S, Kumar M, Rehman R, Tiwari BK, Jha KA, Barhanpurkar AP, Wani MR, Roy SS, Mabalirajan U, Ghosh B, Agrawal A. Miro1 regulates intercellular mitochondrial transport & enhances mesenchymal stem cell rescue efficacy. EMBO J 2014; 33: 994-1010. [CrossRef]
- Frisbie L, Pressimone C, Dyer E, Baruwal R, Garcia G, St Croix C, Watkins S, Calderone M, Gorecki G, Javed Z, Atiya HI, Hempel N, Pearson A, Coffman LG. Carcinoma-associated mesenchymal stem cells promote ovarian cancer heterogeneity and metastasis through mitochondrial transfer. Cell Rep 2024; 43: 114551. [CrossRef]
- Wang C, Xie C. Unveiling the power of mitochondrial transfer in cancer progression: a perspective in ovarian cancer. J Ovarian Res 2024; 17: 233. [CrossRef]
- Rickard BP, Overchuk M, Obaid G, Ruhi MK, Demirci U, Fenton SE, Santos JH, Kessel D, Rizvi I. Photochemical Targeting of Mitochondria to Overcome Chemoresistance in Ovarian Cancer (dagger). Photochem Photobiol 2023; 99: 448-468. [CrossRef]
- Guo X, Can C, Liu W, Wei Y, Yang X, Liu J, Jia H, Jia W, Wu H, Ma D. Mitochondrial transfer in hematological malignancies. Biomark Res 2023; 11: 89. [CrossRef]
- Lu J, Zheng X, Li F, Yu Y, Chen Z, Liu Z, Wang Z, Xu H, Yang W. Tunneling nanotubes promote intercellular mitochondria transfer followed by increased invasiveness in bladder cancer cells. Oncotarget 2017; 8: 15539-15552. [CrossRef]
- Ikeda H, Kawase K, Nishi T, Watanabe T, Takenaga K, Inozume T, Ishino T, Aki S, Lin J, Kawashima S, Nagasaki J, Ueda Y, Suzuki S, Makinoshima H, Itami M, Nakamura Y, Tatsumi Y, Suenaga Y, Morinaga T, Honobe-Tabuchi A, Ohnuma T, Kawamura T, Umeda Y, Nakamura Y, Kiniwa Y, Ichihara E, Hayashi H, Ikeda JI, Hanazawa T, Toyooka S, Mano H, Suzuki T, Osawa T, Kawazu M, Togashi Y. Immune evasion through mitochondrial transfer in the tumour microenvironment. Nature 2025; 638: 225-236. [CrossRef]
- Ahmadian S, Lindsey PJ, Smeets HJM, van Tienen FHJ, van Zandvoort M. Spinning Disk Confocal Microscopy for Optimized and Quantified Live Imaging of 3D Mitochondrial Network. Int J Mol Sci 2024; 25. [CrossRef]
- Lambert C, Schmidt K, Karger M, Stadler M, Stradal TEB, Rottner K. Cytochalasans and Their Impact on Actin Filament Remodeling. Biomolecules 2023; 13. [CrossRef]
- Novak J, Nahacka Z, Oliveira GL, Brisudova P, Dubisova M, Dvorakova S, Miklovicova S, Dalecka M, Puttrich V, Grycova L, Magalhaes-Novais S, Correia CM, Levoux J, Stepanek L, Prochazka J, Svec D, Reguera DP, Lopez-Domenech G, Zobalova R, Sedlacek R, Terp MG, Gammage PA, Lansky Z, Kittler J, Oliveira PJ, Ditzel HJ, Berridge MV, Rodriguez AM, Boukalova S, Rohlena J, Neuzil J. The adaptor protein Miro1 modulates horizontal transfer of mitochondria in mouse melanoma models. Cell Rep 2025; 44: 115154. [CrossRef]
- Allelein S, Medina-Perez P, Lopes ALH, Rau S, Hause G, Kolsch A, Kuhlmeier D. Potential and challenges of specifically isolating extracellular vesicles from heterogeneous populations. Sci Rep 2021; 11: 11585. [CrossRef]
- Jaiswal R, Sedger LM. Intercellular Vesicular Transfer by Exosomes, Microparticles and Oncosomes - Implications for Cancer Biology and Treatments. Front Oncol 2019; 9: 125. [CrossRef]
- Minciacchi VR, Freeman MR, Di Vizio D. Extracellular vesicles in cancer: exosomes, microvesicles and the emerging role of large oncosomes. Semin Cell Dev Biol 2015; 40: 41-51. [CrossRef]
- Majood M, Rawat S, Mohanty S. Delineating the role of extracellular vesicles in cancer metastasis: A comprehensive review. Front Immunol 2022; 13: 966661. [CrossRef]
- Maas SLN, Breakefield XO, Weaver AM. Extracellular Vesicles: Unique Intercellular Delivery Vehicles. Trends Cell Biol 2017; 27: 172-188. [CrossRef]
- Wang Y, Yu HY, Yi ZJ, Qi LY, Yang JS, Xie HX, Zhao M, Liu NH, Chen JQ, Zhou TJ, Xing L, Cheng XW, Jiang HL. Super mitochondria-enriched extracellular vesicles enable enhanced mitochondria transfer. Nat Commun 2025; 16: 9448. [CrossRef]
- Xia K, Zhang S, Peng H, Chen H, Yang C, Yu J, Luo P, Lu Q, Chen H, Huang L, Xiong Y, Zhao L, Jia L, Li L, Qiu Y, Guo Y, Liu C, Fan H, Dai Z, Liu G, Ke Q, Wang T, Li W, Chen L, Deng C, Xiao H, Xiang AP. An extracellular vesicle-mediated mitochondrial transfer network critical for testosterone synthesis. Nat Cell Biol 2026. [CrossRef]
- Zhou L, Zhang W, Hu X, Wang D, Tang D. Metabolic Reprogramming of Cancer-Associated Fibroblast in the Tumor Microenvironment: From Basics to Clinic. Clin Med Insights Oncol 2024; 18: 11795549241287058. [CrossRef]
- Rahmati S, Moeinafshar A, Rezaei N. The multifaceted role of extracellular vesicles (EVs) in colorectal cancer: metastasis, immune suppression, therapy resistance, and autophagy crosstalk. J Transl Med 2024; 22: 452. [CrossRef]
- Xiong L, Wei Y, Jia Q, Chen J, Chen T, Yuan J, Pi C, Liu H, Tang J, Yin S, Zuo Y, Zhang X, Liu F, Yang H, Zhao L. The application of extracellular vesicles in colorectal cancer metastasis and drug resistance: recent advances and trends. J Nanobiotechnology 2023; 21: 143. [CrossRef]
- Chen Y, Kleeff J, Sunami Y. Pancreatic cancer cell- and cancer-associated fibroblast-derived exosomes in disease progression, metastasis, and therapy. Discov Oncol 2024; 15: 253. [CrossRef]
- Zhou P, Du X, Jia W, Feng K, Zhang Y. Engineered extracellular vesicles for targeted reprogramming of cancer-associated fibroblasts to potentiate therapy of pancreatic cancer. Signal Transduct Target Ther 2024; 9: 151. [CrossRef]
- Aasen T, Mesnil M, Naus CC, Lampe PD, Laird DW. Gap junctions and cancer: communicating for 50 years. Nat Rev Cancer 2016; 16: 775-788. [CrossRef]
- Ruch R. Gap Junctions and Connexins in Cancer Formation, Progression, and Therapy. Cancers (Basel) 2020; 12. [CrossRef]
- Fu H, Xie X, Zhai L, Liu Y, Tang Y, He S, Li J, Xiao Q, Xu G, Yang Z, Zhang X, Liu Y. CX43-mediated mitochondrial transfer maintains stemness of KG-1a leukemia stem cells through metabolic remodeling. Stem Cell Res Ther 2024; 15: 460. [CrossRef]
- Singh AK, Prasad P, Cancelas JA. Mesenchymal stromal cells, metabolism, and mitochondrial transfer in bone marrow normal and malignant hematopoiesis. Front Cell Dev Biol 2023; 11: 1325291. [CrossRef]
- Goodenough DA, Paul DL. Gap junctions. Cold Spring Harb Perspect Biol 2009; 1: a002576. [CrossRef]
- Kidder GM, Vanderhyden BC. Bidirectional communication between oocytes and follicle cells: ensuring oocyte developmental competence. Can J Physiol Pharmacol 2010; 88: 399-413. [CrossRef]
- Rapani A, Nikiforaki D, Karagkouni D, Sfakianoudis K, Tsioulou P, Grigoriadis S, Maziotis E, Pantou A, Voutsina A, Pantou A, Koutsilieris M, Hatzigeorgiou A, Pantos K, Simopoulou M. Reporting on the Role of miRNAs and Affected Pathways on the Molecular Backbone of Ovarian Insufficiency: A Systematic Review and Critical Analysis Mapping of Future Research. Front Cell Dev Biol 2020; 8: 590106. [CrossRef]
- Luo M, Luo Y, Mao N, Huang G, Teng C, Wang H, Wu J, Liao X, Yang J. Cancer-Associated Fibroblasts Accelerate Malignant Progression of Non-Small Cell Lung Cancer via Connexin 43-Formed Unidirectional Gap Junctional Intercellular Communication. Cell Physiol Biochem 2018; 51: 315-336. [CrossRef]
- Harris AL. Emerging issues of connexin channels: biophysics fills the gap. Q Rev Biophys 2001; 34: 325-472. [CrossRef]
- Su J, Song Y, Zhu Z, Huang X, Fan J, Qiao J, Mao F. Cell-cell communication: new insights and clinical implications. Signal Transduct Target Ther 2024; 9: 196. [CrossRef]
- Zhou X, Merchak K, Lee W, Grande JP, Cascalho M, Platt JL. Cell Fusion Connects Oncogenesis with Tumor Evolution. Am J Pathol 2015; 185: 2049-2060. [CrossRef]
- Dittmar T, Hass R. Intrinsic signalling factors associated with cancer cell-cell fusion. Cell Commun Signal 2023; 21: 68. [CrossRef]
- Lu X, Kang Y. Cell fusion as a hidden force in tumor progression. Cancer Res 2009; 69: 8536-8539. [CrossRef]
- Augimeri G, Gonzalez ME, Paoli A, Eido A, Choi Y, Burman B, Djomehri S, Karthikeyan SK, Varambally S, Buschhaus JM, Chen YC, Mauro L, Bonofiglio D, Nesvizhskii AI, Luker GD, Ando S, Yoon E, Kleer CG. A hybrid breast cancer/mesenchymal stem cell population enhances chemoresistance and metastasis. JCI Insight 2023; 8. [CrossRef]
- Chen YS, Chen ZP. Vasculogenic mimicry: a novel target for glioma therapy. Chin J Cancer 2014; 33: 74-79. [CrossRef]
- Mao JM, Liu J, Guo G, Mao XG, Li CX. Glioblastoma vasculogenic mimicry: signaling pathways progression and potential anti-angiogenesis targets. Biomark Res 2015; 3: 8. [CrossRef]
- Sun C, Dai X, Zhao D, Wang H, Rong X, Huang Q, Lan Q. Mesenchymal stem cells promote glioma neovascularization in vivo by fusing with cancer stem cells. BMC Cancer 2019; 19: 1240. [CrossRef]
- Mohammadalipour A, Dumbali SP, Wenzel PL. Mitochondrial Transfer and Regulators of Mesenchymal Stromal Cell Function and Therapeutic Efficacy. Front Cell Dev Biol 2020; 8: 603292. [CrossRef]
- Wang Y, Krivtsov AV, Sinha AU, North TE, Goessling W, Feng Z, Zon LI, Armstrong SA. The Wnt/beta-catenin pathway is required for the development of leukemia stem cells in AML. Science 2010; 327: 1650-1653. [CrossRef]
- Pavlova NN, Thompson CB. The Emerging Hallmarks of Cancer Metabolism. Cell Metab 2016; 23: 27-47. [CrossRef]
- Hoover G, Gilbert S, Curley O, Obellianne C, Lin MT, Hixson W, Pierce TW, Andrews JF, Alexeyev MF, Ding Y, Bu P, Behbod F, Medina D, Chang JT, Ayala G, Grelet S. Nerve-to-cancer transfer of mitochondria during cancer metastasis. Nature 2025; 644: 252-262. [CrossRef]
- Viale A, Pettazzoni P, Lyssiotis CA, Ying H, Sanchez N, Marchesini M, Carugo A, Green T, Seth S, Giuliani V, Kost-Alimova M, Muller F, Colla S, Nezi L, Genovese G, Deem AK, Kapoor A, Yao W, Brunetto E, Kang Y, Yuan M, Asara JM, Wang YA, Heffernan TP, Kimmelman AC, Wang H, Fleming JB, Cantley LC, DePinho RA, Draetta GF. Oncogene ablation-resistant pancreatic cancer cells depend on mitochondrial function. Nature 2014; 514: 628-632. [CrossRef]
- Ishikawa K, Takenaga K, Akimoto M, Koshikawa N, Yamaguchi A, Imanishi H, Nakada K, Honma Y, Hayashi J. ROS-generating mitochondrial DNA mutations can regulate tumor cell metastasis. Science 2008; 320: 661-664. [CrossRef]
- Shibata T, Hoshida Y. Mitochondrial genome diversity drives heterogeneity in HCC. Hepatology 2025; 82: 12-13. [CrossRef]
- Welch DR, Foster C, Rigoutsos I. Roles of mitochondrial genetics in cancer metastasis. Trends Cancer 2022; 8: 1002-1018. [CrossRef]
- Weinberg SE, Chandel NS. Targeting mitochondria metabolism for cancer therapy. Nat Chem Biol 2015; 11: 9-15. [CrossRef]
- Guan B, Liu Y, Xie B, Zhao S, Yalikun A, Chen W, Zhou M, Gu Q, Yan D. Mitochondrial genome transfer drives metabolic reprogramming in adjacent colonic epithelial cells promoting TGFbeta1-mediated tumor progression. Nat Commun 2024; 15: 3653. [CrossRef]
- Park JH, Vithayathil S, Kumar S, Sung PL, Dobrolecki LE, Putluri V, Bhat VB, Bhowmik SK, Gupta V, Arora K, Wu D, Tsouko E, Zhang Y, Maity S, Donti TR, Graham BH, Frigo DE, Coarfa C, Yotnda P, Putluri N, Sreekumar A, Lewis MT, Creighton CJ, Wong LC, Kaipparettu BA. Fatty Acid Oxidation-Driven Src Links Mitochondrial Energy Reprogramming and Oncogenic Properties in Triple-Negative Breast Cancer. Cell Rep 2016; 14: 2154-2165. [CrossRef]
- Woytash JA, Lefebvre A, Zhang Z, Xu B, Harchenko SA, Le HT, McColloch AR, Shi X, Digman MA, Razorenova OV. CDCP1/mitochondrial Src axis increases electron transport chain function to promote metastasis in triple-negative breast cancer. Br J Cancer 2025; 133: 1265-1277. [CrossRef]
- Liu X, Si W, He L, Yang J, Peng Y, Ren J, Liu X, Jin T, Yu H, Zhang Z, Cheng X, Zhang W, Xia L, Huang Y, Wang Y, Liu S, Shan L, Zhang Y, Yang X, Li H, Liang J, Sun L, Shang Y. The existence of a nonclassical TCA cycle in the nucleus that wires the metabolic-epigenetic circuitry. Signal Transduct Target Ther 2021; 6: 375. [CrossRef]
- English J, Son JM, Cardamone MD, Lee C, Perissi V. Decoding the rosetta stone of mitonuclear communication. Pharmacol Res 2020; 161: 105161. [CrossRef]
- Jo C, Park S, Oh S, Choi J, Kim EK, Youn HD, Cho EJ. Histone acylation marks respond to metabolic perturbations and enable cellular adaptation. Exp Mol Med 2020; 52: 2005-2019. [CrossRef]
- Ambrosini G, Cordani M, Zarrabi A, Alcon-Rodriguez S, Sainz RM, Velasco G, Gonzalez-Menendez P, Dando I. Transcending frontiers in prostate cancer: the role of oncometabolites on epigenetic regulation, CSCs, and tumor microenvironment to identify new therapeutic strategies. Cell Commun Signal 2024; 22: 36. [CrossRef]
- Sun L, Zhang H, Gao P. Metabolic reprogramming and epigenetic modifications on the path to cancer. Protein Cell 2022; 13: 877-919. [CrossRef]
- Haws SA, Leech CM, Denu JM. Metabolism and the Epigenome: A Dynamic Relationship. Trends Biochem Sci 2020; 45: 731-747. [CrossRef]
- Bernasocchi T, Mostoslavsky R. Subcellular one carbon metabolism in cancer, aging and epigenetics. Front Epigenet Epigenom 2024; 2. [CrossRef]
- Russo M, Pileri F, Ghisletti S. Novel insights into the role of acetyl-CoA producing enzymes in epigenetic regulation. Front Endocrinol (Lausanne) 2023; 14: 1272646. [CrossRef]
- Ottens F, Franz A, Hoppe T. Build-UPS and break-downs: metabolism impacts on proteostasis and aging. Cell Death Differ 2021; 28: 505-521. [CrossRef]
- Leung J, Gaudin V. Who Rules the Cell? An Epi-Tale of Histone, DNA, RNA, and the Metabolic Deep State. Front Plant Sci 2020; 11: 181. [CrossRef]
- Anselme M, He H, Lai C, Luo W, Zhong S. Targeting mitochondrial transporters and metabolic reprogramming for disease treatment. J Transl Med 2025; 23: 1111. [CrossRef]
- Sarkar S, Chang CI, Jean J, Wu MJ. TCA cycle-derived oncometabolites in cancer and the immune microenvironment. J Biomed Sci 2025; 32: 87. [CrossRef]
- Kim S, Ramalho TR, Haynes CM. Regulation of proteostasis and innate immunity via mitochondria-nuclear communication. J Cell Biol 2024; 223. [CrossRef]
- Xu S, Zhang X, Liu C, Liu Q, Chai H, Luo Y, Li S. Role of Mitochondria in Neurodegenerative Diseases: From an Epigenetic Perspective. Front Cell Dev Biol 2021; 9: 688789. [CrossRef]
- Kincaid JW, Berger NA. NAD metabolism in aging and cancer. Exp Biol Med (Maywood) 2020; 245: 1594-1614. [CrossRef]
- Meacham CE, Morrison SJ. Tumour heterogeneity and cancer cell plasticity. Nature 2013; 501: 328-337. [CrossRef]
- Zhang X, Powell K, Li L. Breast Cancer Stem Cells: Biomarkers, Identification and Isolation Methods, Regulating Mechanisms, Cellular Origin, and Beyond. Cancers (Basel) 2020; 12. [CrossRef]
- Fenu G, Grinan-Lison C, Etzi F, Gonzalez-Titos A, Pisano A, Toledo B, Farace C, Sabalic A, Carrillo E, Marchal JA, Madeddu R. Functional Characterization of miR-216a-5p and miR-125a-5p on Pancreatic Cancer Stem Cells. Int J Mol Sci 2025; 26. [CrossRef]
- Zhou Y, Xia L, Wang H, Oyang L, Su M, Liu Q, Lin J, Tan S, Tian Y, Liao Q, Cao D. Cancer stem cells in progression of colorectal cancer. Oncotarget 2018; 9: 33403-33415. [CrossRef]
- Prabavathy D, Swarnalatha Y, Ramadoss N. Lung cancer stem cells-origin, characteristics and therapy. Stem Cell Investig 2018; 5: 6. [CrossRef]
- Patel S, Shah K, Mirza S, Shah K, Rawal R. Circulating tumor stem like cells in oral squamous cell carcinoma: An unresolved paradox. Oral Oncol 2016; 62: 139-146. [CrossRef]
- Feitelson MA, Arzumanyan A, Kulathinal RJ, Blain SW, Holcombe RF, Mahajna J, Marino M, Martinez-Chantar ML, Nawroth R, Sanchez-Garcia I, Sharma D, Saxena NK, Singh N, Vlachostergios PJ, Guo S, Honoki K, Fujii H, Georgakilas AG, Bilsland A, Amedei A, Niccolai E, Amin A, Ashraf SS, Boosani CS, Guha G, Ciriolo MR, Aquilano K, Chen S, Mohammed SI, Azmi AS, Bhakta D, Halicka D, Keith WN, Nowsheen S. Sustained proliferation in cancer: Mechanisms and novel therapeutic targets. Semin Cancer Biol 2015; 35 Suppl: S25-S54. [CrossRef]
- Galassi C, Manic G, Esteller M, Galluzzi L, Vitale I. Epigenetic regulation of cancer stemness. Signal Transduct Target Ther 2025; 10: 243. [CrossRef]
- French R, Pauklin S. Epigenetic regulation of cancer stem cell formation and maintenance. Int J Cancer 2021; 148: 2884-2897. [CrossRef]
- Thiery JP, Acloque H, Huang RY, Nieto MA. Epithelial-mesenchymal transitions in development and disease. Cell 2009; 139: 871-890. [CrossRef]
- Nieto MA, Huang RY, Jackson RA, Thiery JP. Emt: 2016. Cell 2016; 166: 21-45. [CrossRef]
- Gaude E, Frezza C. Tissue-specific and convergent metabolic transformation of cancer correlates with metastatic potential and patient survival. Nat Commun 2016; 7: 13041. [CrossRef]
- Pasquier J, Guerrouahen BS, Al Thawadi H, Ghiabi P, Maleki M, Abu-Kaoud N, Jacob A, Mirshahi M, Galas L, Rafii S, Le Foll F, Rafii A. Preferential transfer of mitochondria from endothelial to cancer cells through tunneling nanotubes modulates chemoresistance. J Transl Med 2013; 11: 94. [CrossRef]
- Desai SP, Bhatia SN, Toner M, Irimia D. Mitochondrial localization and the persistent migration of epithelial cancer cells. Biophys J 2013; 104: 2077-2088. [CrossRef]
- Kaplan RN, Riba RD, Zacharoulis S, Bramley AH, Vincent L, Costa C, MacDonald DD, Jin DK, Shido K, Kerns SA, Zhu Z, Hicklin D, Wu Y, Port JL, Altorki N, Port ER, Ruggero D, Shmelkov SV, Jensen KK, Rafii S, Lyden D. VEGFR1-positive haematopoietic bone marrow progenitors initiate the pre-metastatic niche. Nature 2005; 438: 820-827. [CrossRef]
- Sceneay J, Chow MT, Chen A, Halse HM, Wong CS, Andrews DM, Sloan EK, Parker BS, Bowtell DD, Smyth MJ, Moller A. Primary tumor hypoxia recruits CD11b+/Ly6Cmed/Ly6G+ immune suppressor cells and compromises NK cell cytotoxicity in the premetastatic niche. Cancer Res 2012; 72: 3906-3911. [CrossRef]
- Gerlinger M, Rowan AJ, Horswell S, Math M, Larkin J, Endesfelder D, Gronroos E, Martinez P, Matthews N, Stewart A, Tarpey P, Varela I, Phillimore B, Begum S, McDonald NQ, Butler A, Jones D, Raine K, Latimer C, Santos CR, Nohadani M, Eklund AC, Spencer-Dene B, Clark G, Pickering L, Stamp G, Gore M, Szallasi Z, Downward J, Futreal PA, Swanton C. Intratumor heterogeneity and branched evolution revealed by multiregion sequencing. N Engl J Med 2012; 366: 883-892. [CrossRef]
- Vitale I, Shema E, Loi S, Galluzzi L. Intratumoral heterogeneity in cancer progression and response to immunotherapy. Nat Med 2021; 27: 212-224. [CrossRef]
- Ju YS, Alexandrov LB, Gerstung M, Martincorena I, Nik-Zainal S, Ramakrishna M, Davies HR, Papaemmanuil E, Gundem G, Shlien A, Bolli N, Behjati S, Tarpey PS, Nangalia J, Massie CE, Butler AP, Teague JW, Vassiliou GS, Green AR, Du MQ, Unnikrishnan A, Pimanda JE, Teh BT, Munshi N, Greaves M, Vyas P, El-Naggar AK, Santarius T, Collins VP, Grundy R, Taylor JA, Hayes DN, Malkin D, Group IBC, Group ICMD, Group IPC, Foster CS, Warren AY, Whitaker HC, Brewer D, Eeles R, Cooper C, Neal D, Visakorpi T, Isaacs WB, Bova GS, Flanagan AM, Futreal PA, Lynch AG, Chinnery PF, McDermott U, Stratton MR, Campbell PJ. Origins and functional consequences of somatic mitochondrial DNA mutations in human cancer. Elife 2014; 3. [CrossRef]
- Yuan Y, Ju YS, Kim Y, Li J, Wang Y, Yoon CJ, Yang Y, Martincorena I, Creighton CJ, Weinstein JN, Xu Y, Han L, Kim HL, Nakagawa H, Park K, Campbell PJ, Liang H, Consortium P. Comprehensive molecular characterization of mitochondrial genomes in human cancers. Nat Genet 2020; 52: 342-352. [CrossRef]
- A FCL. Mitochondrial metabolism and DNA methylation: a review of the interaction between two genomes. Clin Epigenetics 2020; 12: 182. [CrossRef]
- Roehlecke C, Schmidt MHH. Tunneling Nanotubes and Tumor Microtubes in Cancer. Cancers (Basel) 2020; 12. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



