Submitted:
02 May 2026
Posted:
05 May 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. CD40 Expression Landscape in PDAC
2.1. CD40 Expression on Non-Immune PDAC Compartments
2.2. CD40 Expression on Immune Cells Within the PDAC Tumor Microenvironment
3. Biological Functions and Signaling Pathways of CD40 in PDAC
3.1. Core CD40 Signaling Pathways Shaping Immune Activation
3.2. The CD40-CD40L Axis In Vivo: Ligand Sources and Signaling Context
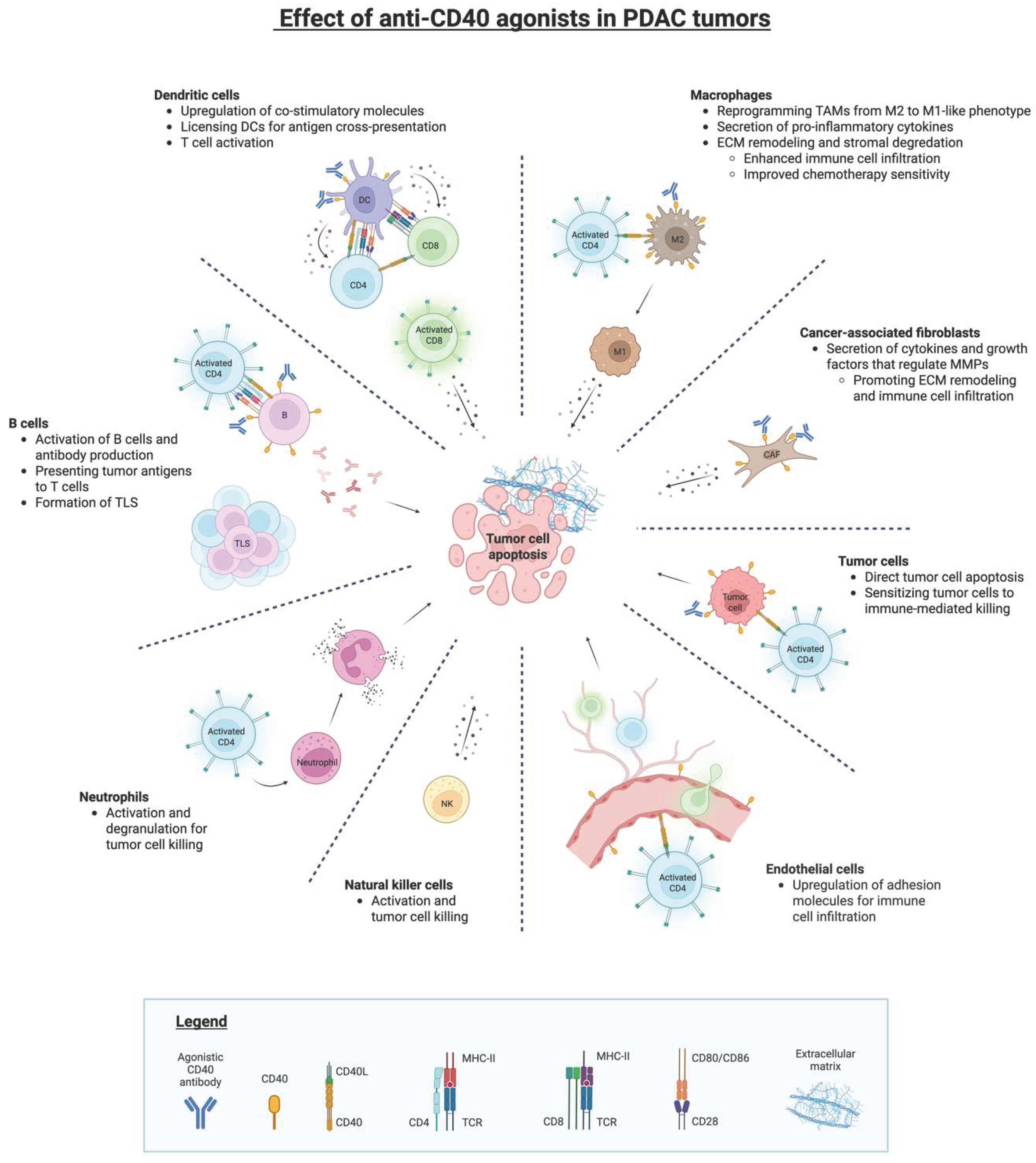
3.3. CD40-Driven Activation of Antigen-Presenting Cells
3.4. Reprogramming of Tumor-Associated Macrophages
3.5. Stromal and Vascular Remodeling Downstream of CD40 Activation
3.5. Tumor-Intrinsic CD40 Signaling in PDAC
4. Therapeutic Targeting of CD40 in PDAC
4.1. CD40 Agonistic Modalities and Design Principles
4.2. Combination Approaches with Chemotherapy
4.3. CD40 Agonism Combined with Immune Checkpoint Inhibition
4.4. Integration with Cancer Vaccines and Neoantigen-Directed Therapies
4.5. Neoadjuvant and Window-of-Opportunity Experience
4.6. Biomarkers: Prognostic Context and Predictors of Benefit
4.7. Safety Considerations and Toxicity Management
5. Challenges and Future Directions
5.1. Limitation to Consistent Clinical Benefit
5.1. Biomarkers and Patient Selection: Beyond Baseline CD40 Abundance
5.2. Optimization of Dosing, Scheduling, and Delivery
5.3. Context-Dependent Tumor-Intrinsic CD40 Signaling
5.4. Next-Generation CD40 Agonists and Rational Combinations
5.5. Standardization of CD40 Measurement and Reporting
6. Conclusion
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Bray, F.; Laversanne, M.; Sung, H.; et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2024, 74(3), 229–263. [Google Scholar] [CrossRef]
- Rahib, L.; Smith, B.D.; Aizenberg, R.; Rosenzweig, A.B.; Fleshman, J.M.; Matrisian, L.M. Projecting cancer incidence and deaths to 2030: the unexpected burden of thyroid, liver, and pancreas cancers in the United States. Cancer Res. 2014, 74(11), 2913–21. [Google Scholar] [CrossRef] [PubMed]
- 10.1158/0008-5472.CAN-14-0155.
- Stoop, T.F.; Javed, A.A.; Oba, A.; et al. Pancreatic cancer. Lancet 2025, 405(10485), 1182–1202. [Google Scholar] [CrossRef]
- Park, W.; Chawla, A.; O'Reilly, E.M. Pancreatic Cancer: A Review. Jama 2021, 326(9), 851–862. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.L.; Kratzer, T.B.; Wagle, N.S.; Sung, H.; Jemal, A. Cancer statistics, 2026. CA Cancer J. Clin. 2026, 76(1), e70043. [Google Scholar] [CrossRef]
- André, T.; Elez, E.; Lenz, H.J.; et al. Nivolumab plus ipilimumab versus nivolumab in microsatellite instability-high metastatic colorectal cancer (CheckMate 8HW): a randomised, open-label, phase 3 trial. Lancet 2025, 405(10476), 383–395. [Google Scholar] [CrossRef]
- Chalabi, M.; Fanchi, L.F.; Dijkstra, K.K.; et al. Neoadjuvant immunotherapy leads to pathological responses in MMR-proficient and MMR-deficient early-stage colon cancers. Nat. Med. 2020, 26(4), 566–576. [Google Scholar] [CrossRef] [PubMed]
- Mok, T.S.K.; Wu, Y.L.; Kudaba, I.; et al. Pembrolizumab versus chemotherapy for previously untreated, PD-L1-expressing, locally advanced or metastatic non-small-cell lung cancer (KEYNOTE-042): a randomised, open-label, controlled, phase 3 trial. Lancet 2019, 393(10183), 1819–1830. [Google Scholar] [CrossRef]
- Spicer, J.D.; Garassino, M.C.; Wakelee, H.; et al. Neoadjuvant pembrolizumab plus chemotherapy followed by adjuvant pembrolizumab compared with neoadjuvant chemotherapy alone in patients with early-stage non-small-cell lung cancer (KEYNOTE-671): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet 2024, 404(10459), 1240–1252. [Google Scholar] [CrossRef]
- Wolchok, J.D.; Chiarion-Sileni, V.; Rutkowski, P.; et al. Final, 10-Year Outcomes with Nivolumab plus Ipilimumab in Advanced Melanoma. N Engl. J. Med. 2025, 392(1), 11–22. [Google Scholar] [CrossRef]
- Janjigian, Y.Y.; Shitara, K.; Moehler, M.; et al. First-line nivolumab plus chemotherapy versus chemotherapy alone for advanced gastric, gastro-oesophageal junction, and oesophageal adenocarcinoma (CheckMate 649): a randomised, open-label, phase 3 trial. Lancet 2021, 398(10294), 27–40. [Google Scholar] [CrossRef]
- Tannir, N.M.; Albigès, L.; McDermott, D.F.; et al. Nivolumab plus ipilimumab versus sunitinib for first-line treatment of advanced renal cell carcinoma: extended 8-year follow-up results of efficacy and safety from the phase III CheckMate 214 trial. Ann. Oncol. 2024, 35(11), 1026–1038. [Google Scholar] [CrossRef]
- Abou-Alfa, G.K.; Lau, G.; Kudo, M.; et al. Tremelimumab plus Durvalumab in Unresectable Hepatocellular Carcinoma. NEJM Evid. 2022, 1(8), EVIDoa2100070. [Google Scholar] [CrossRef] [PubMed]
- Powles, T.; Valderrama, B.P.; Gupta, S.; et al. Enfortumab Vedotin and Pembrolizumab in Untreated Advanced Urothelial Cancer. N Engl. J. Med. 2024, 390(10), 875–888. [Google Scholar] [CrossRef] [PubMed]
- Brahmer, J.R.; Tykodi, S.S.; Chow, L.Q.; et al. Safety and activity of anti-PD-L1 antibody in patients with advanced cancer. N Engl. J. Med. 2012, 366(26), 2455–65. [Google Scholar] [CrossRef]
- Renouf, D.J.; Loree, J.M.; Knox, J.J.; et al. The CCTG PA.7 phase II trial of gemcitabine and nab-paclitaxel with or without durvalumab and tremelimumab as initial therapy in metastatic pancreatic ductal adenocarcinoma. Nat. Commun. 2022, 13(1), 5020. [Google Scholar] [CrossRef] [PubMed]
- O'Reilly, E.M.; Oh, D.Y.; Dhani, N.; et al. Durvalumab With or Without Tremelimumab for Patients With Metastatic Pancreatic Ductal Adenocarcinoma: A Phase 2 Randomized Clinical Trial. JAMA Oncol. 2019, 5(10), 1431–1438. [Google Scholar] [CrossRef]
- Ho, W.J.; Jaffee, E.M.; Zheng, L. The tumour microenvironment in pancreatic cancer - clinical challenges and opportunities. Nat. Rev. Clin. Oncol. 2020, 17(9), 527–540. [Google Scholar] [CrossRef]
- Murakami, T.; Hiroshima, Y.; Matsuyama, R.; Homma, Y.; Hoffman, R.M.; Endo, I. Role of the tumor microenvironment in pancreatic cancer. Ann. Gastroenterol. Surg. 2019, 3(2), 130–137. [Google Scholar] [CrossRef]
- Dallal, R.M.; Christakos, P.; Lee, K.; Egawa, S.; Son, Y.I.; Lotze, M.T. Paucity of dendritic cells in pancreatic cancer. Surgery 2002, 131(2), 135–8. [Google Scholar] [CrossRef]
- Hegde, S.; Krisnawan, V.E.; Herzog, B.H.; et al. Dendritic Cell Paucity Leads to Dysfunctional Immune Surveillance in Pancreatic Cancer. Cancer Cell 2020, 37(3), 289–307 e9. [Google Scholar] [CrossRef]
- Orhan, A.; Vogelsang, R.P.; Andersen, M.B.; et al. The prognostic value of tumour-infiltrating lymphocytes in pancreatic cancer: a systematic review and meta-analysis. Eur. J. Cancer 2020, 132, 71–84. [Google Scholar] [CrossRef]
- Schoenberger, S.P.; Toes, R.E.; van der Voort, E.I.; Offringa, R.; Melief, C.J. T-cell help for cytotoxic T lymphocytes is mediated by CD40-CD40L interactions. Nature 1998, 393(6684), 480–3. [Google Scholar] [CrossRef] [PubMed]
- Vonderheide, R.H. CD40 Agonist Antibodies in Cancer Immunotherapy. Annu Rev. Med. 2020, 71, 47–58. [Google Scholar] [CrossRef] [PubMed]
- Grewal, I.S.; Flavell, R.A. CD40 and CD154 in cell-mediated immunity. Annu Rev. Immunol. 1998, 16, 111–35. [Google Scholar] [CrossRef]
- van Kooten, C.; Banchereau, J. CD40-CD40 ligand. J. Leukoc. Biol. 2000, 67(1), 2–17. [Google Scholar] [CrossRef] [PubMed]
- Vonderheide, R.H. Prospect of targeting the CD40 pathway for cancer therapy. Clin. Cancer Res. 2007, 13(4), 1083–8. [Google Scholar] [CrossRef]
- Caux, C.; Massacrier, C.; Vanbervliet, B.; et al. Activation of human dendritic cells through CD40 cross-linking. J. Exp. Med. 1994, 180(4), 1263–72. [Google Scholar] [CrossRef]
- He, S.; Zhao, H.; Fei, M.; et al. Expression of the co-signaling molecules CD40-CD40L and their growth inhibitory effect on pancreatic cancer in vitro. Oncol. Rep. 2012, 28(1), 262–8. [Google Scholar]
- Bates, K.M.; Vathiotis, I.; MacNeil, T.; et al. Spatial characterization and quantification of CD40 expression across cancer types. BMC Cancer 2023, 23(1), 220. [Google Scholar] [CrossRef]
- Beatty, G.L.; Chiorean, E.G.; Fishman, M.P.; et al. CD40 agonists alter tumor stroma and show efficacy against pancreatic carcinoma in mice and humans. Science 2011, 331(6024), 1612–6. [Google Scholar] [CrossRef]
- Long, K.B.; Gladney, W.L.; Tooker, G.M.; Graham, K.; Fraietta, J.A.; Beatty, G.L. IFNγ and CCL2 Cooperate to Redirect Tumor-Infiltrating Monocytes to Degrade Fibrosis and Enhance Chemotherapy Efficacy in Pancreatic Carcinoma. Cancer Discov. 2016, 6(4), 400–413. [Google Scholar] [CrossRef]
- McVey, J.C.; Beatty, G.L. Facts and Hopes of CD40 Agonists in Cancer Immunotherapy. Clin. Cancer Res. 2025, 31(11), 2079–2087. [Google Scholar] [CrossRef]
- Mangsbo, S.M.; Broos, S.; Fletcher, E.; et al. The human agonistic CD40 antibody ADC-1013 eradicates bladder tumors and generates T-cell-dependent tumor immunity. Clin. Cancer Res. 2015, 21(5), 1115–26. [Google Scholar] [CrossRef] [PubMed]
- Irenaeus, S.M.M.; Nielsen, D.; Ellmark, P.; et al. First-in-human study with intratumoral administration of a CD40 agonistic antibody, ADC-1013, in advanced solid malignancies. Int. J. Cancer 2019, 145(5), 1189–1199. [Google Scholar] [CrossRef] [PubMed]
- Moreno, V.; Perets, R.; Peretz-Yablonski, T.; et al. A phase 1 study of intravenous mitazalimab, a CD40 agonistic monoclonal antibody, in patients with advanced solid tumors. Invest New Drugs 2023, 41(1), 93–104. [Google Scholar] [CrossRef]
- Baxendale, A.J.; Dawson, C.W.; Stewart, S.E.; et al. Constitutive activation of the CD40 pathway promotes cell transformation and neoplastic growth. Oncogene 2005, 24(53), 7913–23. [Google Scholar] [CrossRef] [PubMed]
- Murugaiyan, G.; Martin, S.; Saha, B. CD40-induced countercurrent conduits for tumor escape or elimination? Trends Immunol. 2007, 28(11), 467–73. [Google Scholar] [CrossRef]
- Korniluk, A.; Kemona, H.; Dymicka-Piekarska, V. Multifunctional CD40L: pro- and anti-neoplastic activity. Tumour Biol. 2014, 35(10), 9447–57. [Google Scholar] [CrossRef]
- Batrla, R.; Linnebacher, M.; Rudy, W.; Stumm, S.; Wallwiener, D.; Gückel, B. CD40-expressing carcinoma cells induce down-regulation of CD40 ligand (CD154) and impair T-cell functions. Cancer Res. 2002, 62(7), 2052–7. [Google Scholar]
- Khaliq, A.M.; Rajamohan, M.; Saeed, O.; et al. Spatial transcriptomic analysis of primary and metastatic pancreatic cancers highlights tumor microenvironmental heterogeneity. Nat. Genet. 2024, 56(11), 2455–2465. [Google Scholar] [CrossRef]
- Gong, R.; Chen, Y.; Li, C.; Zhang, H.; Liu, Z.; Yu, Q. Targeting myeloid cells in pancreatic ductal adenocarcinoma: from primary tumors to liver metastasis. Front Immunol. 2025, 16, 1555036. [Google Scholar] [CrossRef] [PubMed]
- Byrne, K.T.; Betts, C.B.; Mick, R.; et al. Neoadjuvant Selicrelumab, an Agonist CD40 Antibody, Induces Changes in the Tumor Microenvironment in Patients with Resectable Pancreatic Cancer. Clin. Cancer Res. 2021, 27(16), 4574–4586. [Google Scholar] [CrossRef] [PubMed]
- Ohlund, D.; Handly-Santana, A.; Biffi, G.; et al. Distinct populations of inflammatory fibroblasts and myofibroblasts in pancreatic cancer. J. Exp. Med. 2017, 214(3), 579–596. [Google Scholar] [CrossRef]
- 20162024 [pii].
- 10.1084/jem.20162024.
- Rebelo, R.; Xavier, C.P.R.; Giovannetti, E.; Vasconcelos, M.H. Fibroblasts in pancreatic cancer: molecular and clinical perspectives. Trends Mol. Med. 2023, 29(6), 439–453. [Google Scholar] [CrossRef] [PubMed]
- Karmann, K.; Hughes, C.C.; Schechner, J.; Fanslow, W.C.; Pober, J.S. CD40 on human endothelial cells: inducibility by cytokines and functional regulation of adhesion molecule expression. Proc. Natl. Acad. Sci. U S A 1995, 92(10), 4342–6. [Google Scholar] [CrossRef]
- Urban, D.; Thanabalasingam, U.; Stibenz, D.; et al. CD40/CD40L interaction induces E-selectin dependent leukocyte adhesion to human endothelial cells and inhibits endothelial cell migration. Biochem Biophys. Res. Commun. 2011, 404(1), 448–52. [Google Scholar] [CrossRef]
- Shintani, Y.; Fujiwara, A.; Kimura, T.; et al. IL-6 Secreted from Cancer-Associated Fibroblasts Mediates Chemoresistance in NSCLC by Increasing Epithelial-Mesenchymal Transition Signaling. J. Thorac. Oncol. 2016, 11(9), 1482–92. [Google Scholar] [CrossRef]
- Richards, K.E.; Zeleniak, A.E.; Fishel, M.L.; Wu, J.; Littlepage, L.E.; Hill, R. Cancer-associated fibroblast exosomes regulate survival and proliferation of pancreatic cancer cells. Oncogene 2017, 36(13), 1770–1778. [Google Scholar] [CrossRef]
- Chakravarthy, A.; Khan, L.; Bensler, N.P.; Bose, P.; De Carvalho, D.D. TGF-β-associated extracellular matrix genes link cancer-associated fibroblasts to immune evasion and immunotherapy failure. Nat. Commun. 2018, 9(1), 4692. [Google Scholar] [CrossRef]
- Grout, J.A.; Sirven, P.; Leader, A.M.; et al. Spatial Positioning and Matrix Programs of Cancer-Associated Fibroblasts Promote T-cell Exclusion in Human Lung Tumors. Cancer Discov. 2022, 12(11), 2606–2625. [Google Scholar] [CrossRef]
- Andersson, H.; Nyesiga, B.; Hermodsson, T.; et al. Next-generation CD40 agonists for cancer immunotherapy. Expert Opin. Biol. Ther. 2024, 24(5), 351–363. [Google Scholar] [CrossRef]
- Bellone, G.; Carbone, A.; Smirne, C.; et al. Cooperative induction of a tolerogenic dendritic cell phenotype by cytokines secreted by pancreatic carcinoma cells. J. Immunol. 2006, 177(5), 3448–60. [Google Scholar] [CrossRef]
- Xie, Z.; Fang, Y.; Zhang, X.; et al. The emerging role of dendritic cells in the tumor microenvironment: from antigen presentation to targeted immunotherapy. Cell Death Dis. 2025, 16(1), 900. [Google Scholar] [CrossRef]
- Zuo, C.; Baer, J.M.; Knolhoff, B.L.; et al. Stromal and therapy-induced macrophage proliferation promotes PDAC progression and susceptibility to innate immunotherapy. J. Exp. Med. 2023, 220(6). [Google Scholar] [CrossRef]
- jem.20212062 [pii].
- 10.1084/jem.20212062.
- Lehmann, J.; Thelen, M.; Kreer, C.; et al. Tertiary Lymphoid Structures in Pancreatic Cancer are Structurally Homologous, Share Gene Expression Patterns and B-cell Clones with Secondary Lymphoid Organs, but Show Increased T-cell Activation. Cancer Immunol. Res. 2025, 13(3), 323–336. [Google Scholar] [CrossRef]
- Pan, P.Y.; Ma, G.; Weber, K.J.; et al. Immune stimulatory receptor CD40 is required for T-cell suppression and T regulatory cell activation mediated by myeloid-derived suppressor cells in cancer. Cancer Res. 2010, 70(1), 99–108. [Google Scholar] [CrossRef] [PubMed]
- Minaei, E.; Wade, S.J.; Penney, C.; et al. Localized Delivery of Anti-CD40/PD-1 Immunotherapy via a Polymeric Implant is Efficacious, Remodels the Tumor Immune Microenvironment and Improves Safety in a PDAC Mouse Model. Adv. Healthc. Mater. 2026, 15(6), e03780. [Google Scholar] [CrossRef] [PubMed]
- Baumann, D.; Hägele, T.; Mochayedi, J.; et al. Proimmunogenic impact of MEK inhibition synergizes with agonist anti-CD40 immunostimulatory antibodies in tumor therapy. Nat. Commun. 2020, 11(1), 2176. [Google Scholar] [CrossRef]
- Carbone, E.; Ruggiero, G.; Terrazzano, G.; et al. A new mechanism of NK cell cytotoxicity activation: the CD40-CD40 ligand interaction. J. Exp. Med. 1997, 185(12), 2053–60. [Google Scholar] [CrossRef] [PubMed]
- Terrazzano, G.; Pisanti, S.; Grimaldi, S.; et al. Interaction between natural killer and dendritic cells: the role of CD40, CD80 and major histocompatibility complex class i molecules in cytotoxicity induction and interferon-gamma production. Scand. J. Immunol. 2004, 59(4), 356–62. [Google Scholar] [CrossRef] [PubMed]
- Jyothi, M.D.; Khar, A. Regulation of CD40L expression on natural killer cells by interleukin-12 and interferon gamma: its role in the elicitation of an effective antitumor immune response. Cancer Immunol. Immunother. 2000, 49(10), 563–72. [Google Scholar] [CrossRef]
- Turner, J.G.; Rakhmilevich, A.L.; Burdelya, L.; et al. Anti-CD40 antibody induces antitumor and antimetastatic effects: the role of NK cells. J. Immunol. 2001, 166(1), 89–94. [Google Scholar] [CrossRef] [PubMed]
- Tsukamoto, N.; Kobayashi, N.; Azuma, S.; Yamamoto, T.; Inoue, J. Two differently regulated nuclear factor kappaB activation pathways triggered by the cytoplasmic tail of CD40. Proc. Natl. Acad. Sci. U S A 1999, 96(4), 1234–9. [Google Scholar] [CrossRef] [PubMed]
- Foight, G.W.; Keating, A.E. Comparison of the peptide binding preferences of three closely related TRAF paralogs: TRAF2, TRAF3, and TRAF5. Protein Sci. 2016, 25(7), 1273–89. [Google Scholar] [CrossRef]
- Ma, Y.; Ding, J.; Chen, Y. The effect of CD40 agonist antibody therapy on the pancreatic cancer microenvironment. Naunyn Schmiedebergs Arch. Pharmacol. 2026, 399(2), 1897–1915. [Google Scholar] [CrossRef]
- Sun, S.C. Non-canonical NF-κB signaling pathway. Cell Res. 2011, 21(1), 71–85. [Google Scholar] [CrossRef]
- Clatza, A.; Bonifaz, L.C.; Vignali, D.A.; Moreno, J. CD40-induced aggregation of MHC class II and CD80 on the cell surface leads to an early enhancement in antigen presentation. J. Immunol. 2003, 171(12), 6478–87. [Google Scholar] [CrossRef]
- Cella, M.; Scheidegger, D.; Palmer-Lehmann, K.; Lane, P.; Lanzavecchia, A.; Alber, G. Ligation of CD40 on dendritic cells triggers production of high levels of interleukin-12 and enhances T cell stimulatory capacity: T-T help via APC activation. J. Exp. Med. 1996, 184(2), 747–52. [Google Scholar] [CrossRef]
- Gallagher, E.; Enzler, T.; Matsuzawa, A.; et al. Kinase MEKK1 is required for CD40-dependent activation of the kinases Jnk and p38, germinal center formation, B cell proliferation and antibody production. Nat. Immunol. 2007, 8(1), 57–63. [Google Scholar] [CrossRef]
- Tang, T.; Cheng, X.; Truong, B.; Sun, L.; Yang, X.; Wang, H. Molecular basis and therapeutic implications of CD40/CD40L immune checkpoint. Pharmacol. Ther. 2021, 219, 107709. [Google Scholar] [CrossRef]
- Yamato, I.; Sho, M.; Nomi, T.; et al. Clinical importance of B7-H3 expression in human pancreatic cancer. Br. J. Cancer 2009, 101(10), 1709–16. [Google Scholar] [CrossRef]
- Zhao, X.; Li, D.C.; Zhu, X.G.; et al. B7-H3 overexpression in pancreatic cancer promotes tumor progression. Int. J. Mol. Med. 2013, 31(2), 283–91. [Google Scholar] [CrossRef]
- Inamura, K.; Takazawa, Y.; Inoue, Y.; et al. Tumor B7-H3 (CD276) Expression and Survival in Pancreatic Cancer. J. Clin. Med. 2018, 7(7). [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Yang, Y.; Huang, Y.; et al. A B7H3-targeting antibody-drug conjugate in advanced solid tumors: a phase 1/1b trial. Nat. Med. 2025, 31(6), 1949–1957. [Google Scholar] [CrossRef] [PubMed]
- Duan, J.; Sun, Y.; Wang, Q.; et al. HS-20093, a B7-H3-targeted antibody-drug conjugate in lung cancer: Results from the ARTEMIS-001 phase 1a/b trial. Cancer Cell. 2026, 44(4), 846–857 e3. [Google Scholar] [CrossRef] [PubMed]
- Hostager, B.S.; Bishop, G.A. CD40-Mediated Activation of the NF-κB2 Pathway. Front Immunol. 2013, 4, 376. [Google Scholar] [CrossRef]
- Zhou, Y.; Richmond, A.; Yan, C. Harnessing the potential of CD40 agonism in cancer therapy. Cytokine Growth Factor Rev. 2024, 75, 40–56. [Google Scholar] [CrossRef]
- Hanissian, S.H.; Geha, R.S. Jak3 is associated with CD40 and is critical for CD40 induction of gene expression in B cells. Immunity 1997, 6(4), 379–87. [Google Scholar] [CrossRef]
- Revy, P.; Hivroz, C.; Andreu, G.; et al. Activation of the Janus kinase 3-STAT5a pathway after CD40 triggering of human monocytes but not of resting B cells. J. Immunol. 1999, 163(2), 787–93. [Google Scholar] [CrossRef]
- Säemann, M.D.; Diakos, C.; Kelemen, P.; et al. Prevention of CD40-triggered dendritic cell maturation and induction of T-cell hyporeactivity by targeting of Janus kinase 3. Am. J. Transplant. 2003, 3(11), 1341–9. [Google Scholar] [CrossRef]
- Wattenberg, M.M.; Herrera, V.M.; Giannone, M.A.; Gladney, W.L.; Carpenter, E.L.; Beatty, G.L. Systemic inflammation is a determinant of outcomes of CD40 agonist-based therapy in pancreatic cancer patients. JCI Insight 2021, 6(5). [Google Scholar] [CrossRef] [PubMed]
- Yu, H.; Pardoll, D.; Jove, R. STATs in cancer inflammation and immunity: a leading role for STAT3. Nat. Rev. Cancer 2009, 9(11), 798–809. [Google Scholar] [CrossRef] [PubMed]
- Lesina, M.; Kurkowski, M.U.; Ludes, K.; et al. Stat3/Socs3 activation by IL-6 transsignaling promotes progression of pancreatic intraepithelial neoplasia and development of pancreatic cancer. Cancer Cell. 2011, 19(4), 456–69. [Google Scholar] [CrossRef] [PubMed]
- Corcoran, R.B.; Contino, G.; Deshpande, V.; et al. STAT3 plays a critical role in KRAS-induced pancreatic tumorigenesis. Cancer Res. 2011, 71(14), 5020–9. [Google Scholar] [CrossRef]
- Fukuda, A.; Wang, S.C.; Morris JPt et, a.l. Stat3 and MMP7 contribute to pancreatic ductal adenocarcinoma initiation and progression. Cancer Cell. 2011, 19(4), 441–55. [Google Scholar] [CrossRef]
- Li, H.; Huang, C.; Huang, K.; et al. STAT3 knockdown reduces pancreatic cancer cell invasiveness and matrix metalloproteinase-7 expression in nude mice. PLoS ONE 2011, 6(10), e25941. [Google Scholar] [CrossRef]
- Chang, Q.; Bournazou, E.; Sansone, P.; et al. The IL-6/JAK/Stat3 feed-forward loop drives tumorigenesis and metastasis. Neoplasia 2013, 15(7), 848–62. [Google Scholar] [CrossRef]
- Scholz, A.; Heinze, S.; Detjen, K.M.; et al. Activated signal transducer and activator of transcription 3 (STAT3) supports the malignant phenotype of human pancreatic cancer. Gastroenterology 2003, 125(3), 891–905. [Google Scholar] [CrossRef]
- Wei, D.; Le, X.; Zheng, L.; et al. Stat3 activation regulates the expression of vascular endothelial growth factor and human pancreatic cancer angiogenesis and metastasis. Oncogene 2003, 22(3), 319–29. [Google Scholar] [CrossRef]
- Hurwitz, H.I.; Uppal, N.; Wagner, S.A.; et al. Randomized, Double-Blind, Phase II Study of Ruxolitinib or Placebo in Combination With Capecitabine in Patients With Metastatic Pancreatic Cancer for Whom Therapy With Gemcitabine Has Failed. J. Clin. Oncol. 2015, 33(34), 4039–47. [Google Scholar] [CrossRef]
- Hurwitz, H.; Van Cutsem, E.; Bendell, J.; et al. Ruxolitinib + capecitabine in advanced/metastatic pancreatic cancer after disease progression/intolerance to first-line therapy: JANUS 1 and 2 randomized phase III studies. Invest New Drugs 2018, 36(4), 683–695. [Google Scholar] [CrossRef]
- Beatty, G.L.; Shahda, S.; Beck, T.; et al. A Phase Ib/II Study of the JAK1 Inhibitor, Itacitinib, plus nab-Paclitaxel and Gemcitabine in Advanced Solid Tumors. Oncologist 2019, 24(1), 14–e10. [Google Scholar] [CrossRef]
- Parampalli Yajnanarayana, S.; Stübig, T.; Cornez, I.; et al. JAK1/2 inhibition impairs T cell function in vitro and in patients with myeloproliferative neoplasms. Br. J. Haematol. 2015, 169(6), 824–33. [Google Scholar] [CrossRef]
- Klein, K.; Stoiber, D.; Sexl, V.; Witalisz-Siepracka, A. Untwining Anti-Tumor and Immunosuppressive Effects of JAK Inhibitors-A Strategy for Hematological Malignancies? Cancers 2021, 13(11). [Google Scholar] [CrossRef]
- Kubo, S.; Yamaoka, K.; Kondo, M.; et al. The JAK inhibitor, tofacitinib, reduces the T cell stimulatory capacity of human monocyte-derived dendritic cells. Ann. Rheum. Dis. 2014, 73(12), 2192–8. [Google Scholar] [CrossRef]
- Chand Dakal, T.; Dhabhai, B.; Agarwal, D.; et al. Mechanistic basis of co-stimulatory CD40-CD40L ligation mediated regulation of immune responses in cancer and autoimmune disorders. Immunobiology 2020, 225(2), 151899. [Google Scholar] [CrossRef]
- Johnson, P.; Challis, R.; Chowdhury, F.; et al. Clinical and biological effects of an agonist anti-CD40 antibody: a Cancer Research UK phase I study. Clin. Cancer Res. 2015, 21(6), 1321–8. [Google Scholar] [CrossRef]
- Ino, Y.; Yamazaki-Itoh, R.; Shimada, K.; et al. Immune cell infiltration as an indicator of the immune microenvironment of pancreatic cancer. Br. J. Cancer 2013, 108(4), 914–23. [Google Scholar] [CrossRef]
- Hou, Y.C.; Chao, Y.J.; Hsieh, M.H.; Tung, H.L.; Wang, H.C.; Shan, Y.S. Low CD8⁺ T Cell Infiltration and High PD-L1 Expression Are Associated with Level of CD44⁺/CD133⁺ Cancer Stem Cells and Predict an Unfavorable Prognosis in Pancreatic Cancer. Cancers 2019, 11(4). [Google Scholar] [CrossRef]
- Fukunaga, A.; Miyamoto, M.; Cho, Y.; et al. CD8+ tumor-infiltrating lymphocytes together with CD4+ tumor-infiltrating lymphocytes and dendritic cells improve the prognosis of patients with pancreatic adenocarcinoma. Pancreas 2004, 28(1), e26-31. [Google Scholar]
- Garcia Garcia, C.J.; Huang, Y.; Fuentes, N.R.; et al. Stromal HIF2 Regulates Immune Suppression in the Pancreatic Cancer Microenvironment. Gastroenterology 2022, 162(7), 2018–2031. [Google Scholar] [CrossRef]
- Caronni, N.; La Terza, F.; Vittoria, F.M.; et al. IL-1β(+) macrophages fuel pathogenic inflammation in pancreatic cancer. Nature 2023, 623(7986), 415–422. [Google Scholar] [CrossRef]
- Shakiba, M.; Tuveson, D.A. Macrophages and fibroblasts as regulators of the immune response in pancreatic cancer. Nat. Immunol. 2025, 26(5), 678–691. [Google Scholar] [CrossRef]
- Ichikawa, J.; Okuda, H.; Kawano, K.; et al. A Combinatorial Effect of Immune Checkpoint Inhibitors and CD40 Agonistic Antibody in Murine Pancreatic Cancer Model. Cancer Sci. 2026, 117(1), 8–18. [Google Scholar]
- Byrne, K.T.; Vonderheide, R.H. CD40 Stimulation Obviates Innate Sensors and Drives T Cell Immunity in Cancer. Cell Rep. 2016, 15(12), 2719–32. [Google Scholar] [CrossRef]
- Lau, S.P.; van Montfoort, N.; Kinderman, P.; et al. Dendritic cell vaccination and CD40-agonist combination therapy licenses T cell-dependent antitumor immunity in a pancreatic carcinoma murine model. J. Immunother. Cancer 2020, 8(2). [Google Scholar] [CrossRef]
- 10.1136/jitc-2020-000772.
- Wagner, A.H.; Gebauer, M.; Pollok-Kopp, B.; Hecker, M. Cytokine-inducible CD40 expression in human endothelial cells is mediated by interferon regulatory factor-1. Blood 2002, 99(2), 520–5. [Google Scholar] [CrossRef]
- Antoniades, C.; Bakogiannis, C.; Tousoulis, D.; Antonopoulos, A.S.; Stefanadis, C. The CD40/CD40 ligand system: linking inflammation with atherothrombosis. J. Am. Coll. Cardiol. 2009, 54(8), 669–77. [Google Scholar] [CrossRef] [PubMed]
- Vuong, J.T.; Stein-Merlob, A.F.; Nayeri, A.; Sallam, T.; Neilan, T.G.; Yang, E.H. Immune Checkpoint Therapies and Atherosclerosis: Mechanisms and Clinical Implications: JACC State-of-the-Art Review. J. Am. Coll. Cardiol. 2022, 79(6), 577–593. [Google Scholar] [CrossRef] [PubMed]
- Prontera, C.; Martelli, N.; Evangelista, V.; et al. Homocysteine modulates the CD40/CD40L system. J. Am. Coll. Cardiol. 2007, 49(22), 2182–90. [Google Scholar] [CrossRef] [PubMed]
- Mach, F.; Schönbeck, U.; Fabunmi, R.P.; et al. T lymphocytes induce endothelial cell matrix metalloproteinase expression by a CD40L-dependent mechanism: implications for tubule formation. Am. J. Pathol. 1999, 154(1), 229–38. [Google Scholar] [CrossRef] [PubMed]
- Eriksson, E.; Moreno, R.; Milenova, I.; et al. Activation of myeloid and endothelial cells by CD40L gene therapy supports T-cell expansion and migration into the tumor microenvironment. Gene Ther. 2017, 24(2), 92–103. [Google Scholar] [CrossRef]
- Luheshi, N.M.; Coates-Ulrichsen, J.; Harper, J.; et al. Transformation of the tumour microenvironment by a CD40 agonist antibody correlates with improved responses to PD-L1 blockade in a mouse orthotopic pancreatic tumour model. Oncotarget 2016, 7(14), 18508–20. [Google Scholar] [CrossRef] [PubMed]
- Melter, M.; Reinders, M.E.; Sho, M.; et al. Ligation of CD40 induces the expression of vascular endothelial growth factor by endothelial cells and monocytes and promotes angiogenesis in vivo. Blood 2000, 96(12), 3801–8. [Google Scholar] [CrossRef]
- Reinders, M.E.; Sho, M.; Robertson, S.W.; Geehan, C.S.; Briscoe, D.M. Proangiogenic function of CD40 ligand-CD40 interactions. J. Immunol. 2003, 171(3), 1534–41. [Google Scholar] [CrossRef]
- Chung, H.W.; Lim, J.B. Clinical significance of elevated serum soluble CD40 ligand levels as a diagnostic and prognostic tumor marker for pancreatic ductal adenocarcinoma. J. Transl. Med. 2014, 12, 102. [Google Scholar] [CrossRef] [PubMed]
- Flaxenburg, J.A.; Melter, M.; Lapchak, P.H.; Briscoe, D.M.; Pal, S. The CD40-induced signaling pathway in endothelial cells resulting in the overexpression of vascular endothelial growth factor involves Ras and phosphatidylinositol 3-kinase. J. Immunol. 2004, 172(12), 7503–9. [Google Scholar] [CrossRef] [PubMed]
- Selvaraj, S.; Raundhal, M.; Patidar, A.; Saha, B. Anti-VEGF antibody enhances the antitumor effect of CD40. Int. J. Cancer 2014, 135(8), 1983–8. [Google Scholar] [CrossRef]
- Kashyap, A.S.; Schmittnaegel, M.; Rigamonti, N.; et al. Optimized antiangiogenic reprogramming of the tumor microenvironment potentiates CD40 immunotherapy. Proc. Natl. Acad. Sci. U S A 2020, 117(1), 541–551. [Google Scholar] [CrossRef]
- Shoji, Y.; Miyamoto, M.; Ishikawa, K.; et al. The CD40-CD154 interaction would correlate with proliferation and immune escape in pancreatic ductal adenocarcinoma. J. Surg. Oncol. 2011, 103(3), 230–8. [Google Scholar] [CrossRef] [PubMed]
- Alexandroff, A.B.; Jackson, A.M.; Paterson, T.; et al. Role for CD40-CD40 ligand interactions in the immune response to solid tumours. Mol. Immunol. 2000, 37(9), 515–26. [Google Scholar] [CrossRef] [PubMed]
- Schiele, P.; Japp, A.S.; Stark, R.; et al. CD8(+) T cell-derived CD40L mediates noncanonical cytotoxicity in CD40-expressing cancer cells. Sci. Adv. 2025, 11(21), eadr9331. [Google Scholar] [CrossRef]
- Davies, C.C.; Mason, J.; Wakelam, M.J.; Young, L.S.; Eliopoulos, A.G. Inhibition of phosphatidylinositol 3-kinase- and ERK MAPK-regulated protein synthesis reveals the pro-apoptotic properties of CD40 ligation in carcinoma cells. J. Biol. Chem. 2004, 279(2), 1010–9. [Google Scholar] [CrossRef]
- Elmetwali, T.; Young, L.S.; Palmer, D.H. CD40 ligand-induced carcinoma cell death: a balance between activation of TNFR-associated factor (TRAF) 3-dependent death signals and suppression of TRAF6-dependent survival signals. J. Immunol. 2010, 184(2), 1111–20. [Google Scholar] [CrossRef]
- Li, D.; Zhong, Y.; Zhou, Y.; et al. Autocrine TNF-α-mediated NF-κB activation is a determinant for evasion of CD40-induced cytotoxicity in cancer cells. Biochem Biophys. Res. Commun. 2013, 436(3), 467–72. [Google Scholar] [CrossRef]
- Ahmed-Choudhury, J.; Russell, C.L.; Randhawa, S.; Young, L.S.; Adams, D.H.; Afford, S.C. Differential induction of nuclear factor-kappaB and activator protein-1 activity after CD40 ligation is associated with primary human hepatocyte apoptosis or intrahepatic endothelial cell proliferation. Mol. Biol. Cell. 2003, 14(4), 1334–45. [Google Scholar]
- Fonsatti, E.; Maio, M.; Altomonte, M.; Hersey, P. Biology and clinical applications of CD40 in cancer treatment. Semin Oncol. 2010, 37(5), 517–23. [Google Scholar]
- Murugaiyan, G.; Agrawal, R.; Mishra, G.C.; Mitra, D.; Saha, B. Differential CD40/CD40L expression results in counteracting antitumor immune responses. J. Immunol. 2007, 178(4), 2047–55. [Google Scholar]
- Murgaski, A.; Kiss, M.; Van Damme, H.; et al. Efficacy of CD40 Agonists Is Mediated by Distinct cDC Subsets and Subverted by Suppressive Macrophages. Cancer Res. 2022, 82(20), 3785–3801. [Google Scholar] [CrossRef]
- Dahan, R.; Barnhart, B.C.; Li, F.; Yamniuk, A.P.; Korman, A.J.; Ravetch, J.V. Therapeutic Activity of Agonistic, Human Anti-CD40 Monoclonal Antibodies Requires Selective FcγR Engagement. Cancer Cell 2016, 29(6), 820–831. [Google Scholar] [CrossRef] [PubMed]
- Beatty, G.L.; Winograd, R.; Evans, R.A.; et al. Exclusion of T Cells From Pancreatic Carcinomas in Mice Is Regulated by Ly6C(low) F4/80(+) Extratumoral Macrophages. Gastroenterology 2015, 149(1), 201–10. [Google Scholar]
- Troiano, G.; Caponio, V.C.A.; Adipietro, I.; et al. Prognostic significance of CD68(+) and CD163(+) tumor associated macrophages in head and neck squamous cell carcinoma: A systematic review and meta-analysis. Oral. Oncol. 2019, 93, 66–75. [Google Scholar] [PubMed]
- López-Janeiro, Á.; Padilla-Ansala, C.; de Andrea, C.E.; Hardisson, D.; Melero, I. Prognostic value of macrophage polarization markers in epithelial neoplasms and melanoma. A systematic review and meta-analysis. Mod. Pathol. 2020, 33(8), 1458–1465. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Lin, Y.; Zhu, H.; et al. The Prognostic and Clinical Value of Tumor-Associated Macrophages in Patients With Breast Cancer: A Systematic Review and Meta-Analysis. Front Oncol. 2022, 12, 905846. [Google Scholar]
- Van Laethem, J.L.; Borbath, I.; Prenen, H.; et al. Combining CD40 agonist mitazalimab with mFOLFIRINOX in previously untreated metastatic pancreatic ductal adenocarcinoma (OPTIMIZE-1): a single-arm, multicentre phase 1b/2 study. Lancet Oncol. 2024, 25(7), 853–864. [Google Scholar]
- O'Hara, M.H.; O'Reilly, E.M.; Varadhachary, G.; et al. CD40 agonistic monoclonal antibody APX005M (sotigalimab) and chemotherapy, with or without nivolumab, for the treatment of metastatic pancreatic adenocarcinoma: an open-label, multicentre, phase 1b study. Lancet Oncol. 2021, 22(1), 118–131. [Google Scholar] [CrossRef]
- Padrón, L.J.; Maurer, D.M.; O'Hara, M.H.; et al. Sotigalimab and/or nivolumab with chemotherapy in first-line metastatic pancreatic cancer: clinical and immunologic analyses from the randomized phase 2 PRINCE trial. Nat. Med. 2022, 28(6), 1167–1177. [Google Scholar] [CrossRef]
- Van Laethem, J.L.; Geboes, K.; Borbath, I.; et al. CD40 agonist mitazalimab with mFOLFIRINOX in untreated metastatic pancreatic cancer: Biomarkers associated with outcomes from OPTIMIZE-1. Cell Rep. Med. 2025, 6(10), 102407. [Google Scholar] [CrossRef] [PubMed]
- Beatty, G.L.; Torigian, D.A.; Chiorean, E.G.; et al. A phase I study of an agonist CD40 monoclonal antibody (CP-870,893) in combination with gemcitabine in patients with advanced pancreatic ductal adenocarcinoma. Clin. Cancer Res. 2013, 19(22), 6286–95. [Google Scholar] [CrossRef]
- Winograd, R.; Byrne, K.T.; Evans, R.A.; et al. Induction of T-cell Immunity Overcomes Complete Resistance to PD-1 and CTLA-4 Blockade and Improves Survival in Pancreatic Carcinoma. Cancer Immunol. Res. 2015, 3(4), 399–411. [Google Scholar] [CrossRef] [PubMed]
- Morrison, A.H.; Diamond, M.S.; Hay, C.A.; Byrne, K.T.; Vonderheide, R.H. Sufficiency of CD40 activation and immune checkpoint blockade for T cell priming and tumor immunity. Proc. Natl. Acad. Sci. U S A 2020, 117(14), 8022–8031. [Google Scholar] [CrossRef] [PubMed]
- Burrack, A.L.; Rollins, M.R.; Spartz, E.J.; et al. CD40 Agonist Overcomes T Cell Exhaustion Induced by Chronic Myeloid Cell IL-27 Production in a Pancreatic Cancer Preclinical Model. J. Immunol. 2021, 206(6), 1372–1384. [Google Scholar] [CrossRef]
- Sanborn, R.; Gabrail, N.; Carneiro, B.; et al. Abstract 596: Results from a phase 1 study of CDX-1140, a fully human anti-CD40 agonist monoclonal antibody (mAb), in combination with pembrolizumab. J. Immunother. Cancer 2022, 10 (Suppl 2). [Google Scholar] [CrossRef]
- Sanborn, R.E.; Gabrail, N.Y.; Bhardwaj, N.; et al. Abstract LB-194: First-in-human Phase I study of the CD40 agonist mAb CDX-1140 and in combination with CDX-301 (rhFLT3L) in patients with advanced cancers: Interim results. Cancer Res. 2019, 79 13_Supplement, LB-194-LB-194. [Google Scholar] [CrossRef]
- Hogg, G.D.; Weinstein, A.G.; Kingston, N.L.; et al. Combined Flt3L and CD40 agonism restores dendritic cell-driven T cell immunity in pancreatic cancer. Sci. Immunol. 2025, 10(110), eadp3978. [Google Scholar] [CrossRef]
- Fu, S. Abstract e14501: Early safety and efficacy from a phase I open-label clinical study of LVGN7409 (CD40 agonist antibody) in patients with advanced or metastatic malignancies. presented at: ASCO Annual Meeting; 2022; Session Developmental Therapeutics—Immunotherapy. Accessed 4/28/2026.
- Sanchez-Paulete, A.R.; Labiano, S.; Rodriguez-Ruiz, M.E.; et al. Deciphering CD137 (4-1BB) signaling in T-cell costimulation for translation into successful cancer immunotherapy. Eur. J. Immunol. 2016, 46(3), 513–22. [Google Scholar] [CrossRef] [PubMed]
- Schmiechen, Z.C.; Nanda, H.A.; Burrack, A.L.; et al. IL-15 Complex Enhances Agonistic Anti-CD40 + Anti-PDL1 by Correcting the T-bet to Tox Ratio in CD8+ T cells Infiltrating Pancreatic Ductal Adenocarcinoma. Cancer Immunol. Res. 2025, 13(6), 847–866. [Google Scholar] [CrossRef]
- Rojas, L.A.; Sethna, Z.; Soares, K.C.; et al. Personalized RNA neoantigen vaccines stimulate T cells in pancreatic cancer. Nature 2023, 618(7963), 144–150. [Google Scholar] [CrossRef] [PubMed]
- Pant, S.; Wainberg, Z.A.; Weekes, C.D.; et al. Lymph-node-targeted, mKRAS-specific amphiphile vaccine in pancreatic and colorectal cancer: the phase 1 AMPLIFY-201 trial. Nat. Med. 2024, 30(2), 531–542. [Google Scholar] [CrossRef]
- Sethna, Z.; Guasp, P.; Reiche, C.; et al. RNA neoantigen vaccines prime long-lived CD8(+) T cells in pancreatic cancer. Nature 2025, 639(8056), 1042–1051. [Google Scholar] [CrossRef]
- Vonderheide, R.H. The Immune Revolution: A Case for Priming, Not Checkpoint. Cancer Cell 2018, 33(4), 563–569. [Google Scholar] [CrossRef]
- Yamauchi, T.; Hoki, T.; Oba, T.; et al. CD40 and CD80/86 signaling in cDC1s mediate effective neoantigen vaccination and generation of antigen-specific CX3CR1(+) CD8(+) T cells. Cancer Immunol. Immunother. 2022, 71(1), 137–151. [Google Scholar] [CrossRef]
- Kucukcelebi, S.; van 't Land, F.R.; van der Burg, S.H.; et al. REACtiVe-2: phase I evaluation of dendritic cell vaccination and agonistic CD40 therapy following (m)FOLFIRINOX in metastatic pancreatic cancer. Nat. Commun. 2025, 16(1), 10609. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Wen, X.; Tian, L.; et al. Irreversible electroporation reverses resistance to immune checkpoint blockade in pancreatic cancer. Nat. Commun. 2019, 10(1), 899. [Google Scholar] [CrossRef] [PubMed]
- Sabel, M.S.; Yamada, M.; Kawaguchi, Y.; Chen, F.A.; Takita, H.; Bankert, R.B. CD40 expression on human lung cancer correlates with metastatic spread. Cancer Immunol. Immunother. 2000, 49(2), 101–8. [Google Scholar] [CrossRef]
- Ishikawa, K.; Miyamoto, M.; Yoshioka, T.; et al. Up-regulation of CD40 with juxtacrine activity in human nonsmall lung cancer cells correlates with poor prognosis. Cancer 2008, 113(3), 530–41. [Google Scholar] [CrossRef]
- Guo, J.; Xiao, J.J.; Zhang, X.; Fan, K.X. CD40 expression and its prognostic significance in human gastric carcinoma. Med. Oncol. 2015, 32(3), 63. [Google Scholar] [CrossRef] [PubMed]
- Werner, J.M.; Kuhl, S.; Ulrich, K.; et al. Expression of CD40 Correlates Negatively with Overall and Progression-Free Survival of Low- and High-Grade Gliomas. World Neurosurg. 2019, 130, e17–e25. [Google Scholar] [CrossRef]
- Grazia, G.A.; Bastos, D.R.; Villa, L.L. CD40/CD40L expression and its prognostic value in cervical cancer. Braz. J. Med. Biol. Res. 2023, 56, e13047. [Google Scholar] [CrossRef]
- Uehara, Y.; Nishizaki, D.; Fujiwara, Y.; et al. CD40 transcriptomic expression patterns across malignancies: implications for clinical trials of CD40 agonists. Cancer Immunol. Immunother. 2025, 74(12), 359. [Google Scholar] [CrossRef]
- Digomann, D.; Heiduk, M.; Reiche, C.; et al. Serum immune checkpoint profiling identifies soluble CD40 as a biomarker for pancreatic cancer. npj Precis Oncol. 2023, 7(1), 104. [Google Scholar] [CrossRef] [PubMed]
- Vonderheide, R.H.; Flaherty, K.T.; Khalil, M.; et al. Clinical activity and immune modulation in cancer patients treated with CP-870,893, a novel CD40 agonist monoclonal antibody. J. Clin. Oncol. 2007, 25(7), 876–83. [Google Scholar] [CrossRef]
- Rüter, J.; Antonia, S.J.; Burris, H.A.; Huhn, R.D.; Vonderheide, R.H. Immune modulation with weekly dosing of an agonist CD40 antibody in a phase I study of patients with advanced solid tumors. Cancer Biol. Ther. 2010, 10(10), 983–93. [Google Scholar] [CrossRef]
- Richman, L.P.; Vonderheide, R.H. Role of crosslinking for agonistic CD40 monoclonal antibodies as immune therapy of cancer. Cancer Immunol. Res. 2014, 2(1), 19–26. [Google Scholar] [CrossRef]
- Eriksson, E.; Milenova, I.; Wenthe, J.; Moreno, R.; Alemany, R.; Loskog, A. IL-6 Signaling Blockade during CD40-Mediated Immune Activation Favors Antitumor Factors by Reducing TGF-β, Collagen Type I, and PD-L1/PD-1. J. Immunol. 2019, 202(3), 787–798. [Google Scholar] [CrossRef]
- Plesca, I.; Benešová, I.; Beer, C.; et al. Clinical Significance of Tumor-Infiltrating Conventional and Plasmacytoid Dendritic Cells in Pancreatic Ductal Adenocarcinoma. Cancers 2022, 14(5). [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T.; Masuda, A.; Inoue, J.; et al. Integrated analysis of tertiary lymphoid structures in relation to tumor-infiltrating lymphocytes and patient survival in pancreatic ductal adenocarcinoma. J. Gastroenterol. 2023, 58(3), 277–291. [Google Scholar] [CrossRef]
- Kinker, G.S.; Vitiello, G.A.F.; Diniz, A.B.; et al. Mature tertiary lymphoid structures are key niches of tumour-specific immune responses in pancreatic ductal adenocarcinomas. Gut 2023, 72(10), 1927–1941. [Google Scholar] [CrossRef]
- Yousuf, S.; Qiu, M.; Voith von Voithenberg, L.; et al. Spatially Resolved Multi-Omics Single-Cell Analyses Inform Mechanisms of Immune Dysfunction in Pancreatic Cancer. Gastroenterology 2023, 165(4), 891–908 e14. [Google Scholar] [CrossRef]
- Hwang, W.L.; Jagadeesh, K.A.; Guo, J.A.; et al. Single-nucleus and spatial transcriptome profiling of pancreatic cancer identifies multicellular dynamics associated with neoadjuvant treatment. Nat. Genet. 2022, 54(8), 1178–1191. [Google Scholar] [CrossRef]
- Mi, H.; Sivagnanam, S.; Betts, C.B.; et al. Quantitative Spatial Profiling of Immune Populations in Pancreatic Ductal Adenocarcinoma Reveals Tumor Microenvironment Heterogeneity and Prognostic Biomarkers. Cancer Res. 2022, 82(23), 4359–4372. [Google Scholar] [CrossRef]
- Vonderheide, R.H.; Glennie, M.J. Agonistic CD40 antibodies and cancer therapy. Clin. Cancer Res. 2013, 19(5), 1035–43. [Google Scholar] [CrossRef]
- Liu, H.C.; Davila Gonzalez, D.; Viswanath, D.I.; et al. Sustained Intratumoral Administration of Agonist CD40 Antibody Overcomes Immunosuppressive Tumor Microenvironment in Pancreatic Cancer. Adv. Sci. (Weinh) 2023, 10(9), e2206873. [Google Scholar] [CrossRef]
- Chen, S.; Lin, Y.; Li, D.; et al. Development of a Tumor-Specific Multivalent CD40 Agonist Antibody FAPxCD40 for Cancer Therapy: Balancing Efficacy and Toxicity. Mol. Cancer Ther. 2025, 24(7), 1063–1074. [Google Scholar] [CrossRef]
- McIntyre, C.A.; Grimont, A.; Park, J.; et al. Distinct clinical outcomes and biological features of specific KRAS mutants in human pancreatic cancer. Cancer Cell 2024, 42(9), 1614–1629 e5. [Google Scholar] [CrossRef]
- Chen, Y.; Shen, X.; Tang, Y.; et al. The diverse pancreatic tumor cell-intrinsic response to IFNγ is determined by epigenetic heterogeneity. Cancer Lett. 2023, 562, 216153. [Google Scholar] [CrossRef]
- Osorio, J.C.; Knorr, D.A.; Weitzenfeld, P.; et al. Fc-optimized CD40 agonistic antibody elicits tertiary lymphoid structure formation and systemic antitumor immunity in metastatic cancer. Cancer Cell 2025, 43(10), 1902–1916 e9. [Google Scholar] [CrossRef]
- Wang, L.M.; Yang, C.; Sun, X.; et al. SHR-2005, an FcγR-Dependent CD40 Agonistic Antibody with Potent Antitumor Activities and Favorable Safety Profile. Mol. Cancer Ther. 2025, 24(11), 1671–1684. [Google Scholar] [CrossRef]
- Ceglia, V.; Zurawski, S.; Montes, M.; et al. Anti-CD40 Antibodies Fused to CD40 Ligand Have Superagonist Properties. J. Immunol. 2021, 207(8), 2060–2076. [Google Scholar] [CrossRef]
- Salomon, R.; Dahan, R. Next Generation CD40 Agonistic Antibodies for Cancer Immunotherapy. Front Immunol. 2022, 13, 940674. [Google Scholar] [CrossRef] [PubMed]
- Sum, E.; Rapp, M.; Fröbel, P.; et al. Fibroblast Activation Protein α-Targeted CD40 Agonism Abrogates Systemic Toxicity and Enables Administration of High Doses to Induce Effective Antitumor Immunity. Clin. Cancer Res. 2021, 27(14), 4036–4053. [Google Scholar] [CrossRef]
- Althobaiti, S.; Parajuli, P.; Luong, D.; et al. Enhanced safety and efficacy profile of CD40 antibody upon encapsulation in pHe-triggered membrane-adhesive nanoliposomes. Nanomedicine (Lond) 2025, 20(2), 155–166. [Google Scholar] [CrossRef]
- Rech, A.J.; Dada, H.; Kotzin, J.J.; et al. Radiotherapy and CD40 Activation Separately Augment Immunity to Checkpoint Blockade in Cancer. Cancer Res. 2018, 78(15), 4282–4291. [Google Scholar] [CrossRef]
- Maltez, V.I.; Arora, C.; Gribbin, K.P.; et al. Agonistic anti-CD40 antibody treatment converts resident regulatory T cells into activated type 1 effectors within the tumor microenvironment. Immunity 2026, 59(4), 1058–1074 e7. [Google Scholar] [CrossRef]
- Wartenberg, M.; Cibin, S.; Zlobec, I.; et al. Integrated Genomic and Immunophenotypic Classification of Pancreatic Cancer Reveals Three Distinct Subtypes with Prognostic/Predictive Significance. Clin. Cancer Res. 2018, 24(18), 4444–4454. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.A.; Hoffman, H.I.; Weekes, C.D.; Zheng, L.; Ting, D.T.; Hwang, W.L. Refining the Molecular Framework for Pancreatic Cancer with Single-cell and Spatial Technologies. Clin. Cancer Res. 2021, 27(14), 3825–3833. [Google Scholar] [CrossRef] [PubMed]
- Steele, N.G.; Carpenter, E.S.; Kemp, S.B.; et al. Multimodal Mapping of the Tumor and Peripheral Blood Immune Landscape in Human Pancreatic Cancer. Nat. Cancer 2020, 1(11), 1097–1112. [Google Scholar] [CrossRef]
- Karamitopoulou, E. Tumour microenvironment of pancreatic cancer: immune landscape is dictated by molecular and histopathological features. Br. J. Cancer 2019, 121(1), 5–14. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Choi, J.H.; Lim, Y.; et al. Artificial Intelligence-Powered Spatial Analysis of Immune Phenotypes in Resected Pancreatic Cancer. JAMA Surg. 2025, 160(8), 884–892. [Google Scholar] [CrossRef] [PubMed]
- Cao, L.; Huang, C.; Cui Zhou, D.; et al. Proteogenomic characterization of pancreatic ductal adenocarcinoma. Cell 2021, 184(19), 5031–5052 e26. [Google Scholar] [CrossRef]
- Coveler, A.L.; Smith, D.C.; Phillips, T.; et al. Phase 1 dose-escalation study of SEA-CD40: a non-fucosylated CD40 agonist, in advanced solid tumors and lymphomas. J. Immunother. Cancer 2023, 11(6). [Google Scholar] [CrossRef] [PubMed]
| Agent | Other names | Developer | Antibody class | Fc engineering |
|---|---|---|---|---|
| Sotigalimab | APX005M | Apexigen / Pyxis Oncology | IgG1 | Enhanced FcγRIIB binding |
| Mitazalimab | JNJ-64457107/ADC-1013 | Alligator Bioscience | IgG1 | FcγR crosslinking-dependent; no specific Fc engineering reported |
| Selicrelumab | RG7876/CP-870,893 | Pfizer / Genentech / Roche | IgG2 | None |
| CDX-1140 | - | Celldex Therapeutics | IgG2 | None |
| ChiLob 7/4 | - | University of Southampton | IgG1 | None |
| SEA-CD40 | - | Seagen | IgG1 | Non-fucosylated Fc; Enhanced FcγRIIIa binding |
| LVGN7409 | - | Lyvgen Biopharma | IgG1 | Selective FcγRIIB binding |
| Regimen | Design | Tumor type | Treatment scheme | Clinical outcomes | Immunological outcomes | Toxicity | Clinical trial/ reference |
|---|---|---|---|---|---|---|---|
| ChiLob 7/4 | Phase I | CD-40 expressing solid tumors and diffuse large B cell lymphoma (DLBCL), including 2 pancreatic cancer patients (total n=28) | ChiLob7/4 weekly for 4 doses | No objective responses. Disease stabilization in 15/29 treatments (52%), with median duration of 6 months. | Signs for immune activation and effector cell trafficking. | Well-tolerated. One DLT. Infusion reactions could be prevented with single-dose corticosteroid premedication. | NCT01561911 [98] |
| SEA-CD40 | Phase I | Advanced solid tumors (n=56) and lymphoma (n=11), including 3 pancreatic cancer patients (total n=67) |
SEA-CD40 monotherapy in 21-day cycle. | One CR and three SD in seven lymphoma patients. | Cytokine induction and activation of T cells and NK cells in the peripheral blood | Acceptable safety profile. Infusion/hypersensitivity reactions (IHRs) in 73% of the patients, primarily grade 1-2. | NCT02376699 [193] |
| CP-870,893 (=selicrelumab) + gemcitabine | Open-label, dose-escalation, phase I |
Chemotherapy-naive patients with advanced PDAC (n=22). | Gemcitabine once weekly for three weeks with CP-870893 48 hours after gemcitabine on day three of each 28-day cycle. | Median PFS 5.2 months (95% CI: 1.9-7.4). Median OS 8.4 months (95% CI: 5.3-11.8). 1-year OS 28.6%. ORR 19% (4/22 patients). |
Increase in inflammatory cytokines. Increase in B cell expression of costimulatory molecules. Transient depletion of B cells. | Well-tolerated. One DLT. Most common AE was CRS (grade 1 to 2). | NCT00711191 [140] |
| Selicrelumab +/- Gem/Nab | Open-label, phase I |
Resectable PDAC (n=16). |
Arm I (n=16): neoadjuvant selicrelumab two weeks prior to surgery. Arm II (n=11): neoadjuvant Gem/Nab followed by selicrelumab two days later, prior to surgery. Adjuvant Gem/Nab followed by selicrelumab two days later, up to four 28-day cycles, in both arms. |
Both arms (n=16): Median OS 23.4 months (95% CI: 18.0-28.8). Median DFS 13.9 months (95% CI: 2.9-24.8). Arm I: Median OS 23.4 months (95% CI: 9.1-37.6). Median DFS 9.8 months (95% CI: 0.4-19.2). 1-year DFS 49.9%. 1-year OS 81.8%. Arm II: Median OS and DFS are not reached. 1-year DFS 75.0%. 1-year OS 100%. |
82% of the treated tumors were T cell enriched. More active and proliferative T cells in both TME and circulation. Reduced tumor fibrosis. Less M2-like macrophages. More mature intratumoral DCs. Systemically increased inflammatory cytokines. | Acceptable toxicity profile. Selicrelumab related AEs were mostly mild; 5 patients with grade 3 AEs, and one patient with grade 4 AE. Three SAEs observed in two patients. | NCT02588443 [43] |
|
Mitazalimab + mFOLFIRINOX (OPTIMIZE-1) |
Single-arm, phase Ib/II |
Chemotherapy-naive patients with metastasized PDAC (n=70) |
During the first 21 day treatment cycle, mitazalimab on day 1 (priming dose) and on day 10, and mFOLFIRINOX on day 8. During subsequent 14-day treatment cycles, mFOLFIRINOX on day 1 and mitazalimab on day 3. | Median PFS 7.7 months (95% CI: 5.8-11.3). Median OS 14.3 months (95% CI: 10.0-21.6). 1-year PFS 34%. 1-year OS 59%. ORR 40% (23/57 patients). |
Mitazalimab-induced increases in activated circulating myeloid, B cell, and T cell frequencies correlate with better outcomes. Intratumoral myeloid and T cell activation in objective responders. |
Manageable safety. One DLT observed. Most common grade 3 or worse AEs: neutropenia 26%, hypokalaemia 16%, anaemia and thrombocytopenia 11%. SAEs in 41%, none considered related to mitazalimab. No treatment-related deaths. |
NCT04888312 [136,139] |
|
Sotigalimab (APX005M) + Gem/nab +/- nivolumab (PRINCE) |
Non-randomized, open-label, four cohort, phase Ib |
First line treatment for metastasized PDAC (n=30) |
1) Nivolumab + Gem/Nab, 2) Sotigalimab + Gem/Nab, 3) Sotigalimab + nivolumab + Gem/Nab. Nivolumab on days 1 and 15. Sotigalimab on day 3 (=2 days after chemotherapy), or on day 10 if patients received chemotherapy on day 8. |
Median PFS 11.7 months (95% CI: 7.1-17.8). Median OS 20.1 months (95% CI: 10.5-not estimable). ORR 58% (14/24 DLT-evaluable patients). |
Decrease of naïve B cells and increase of plasmablasts. Increased frequency of CD141-negative myeloid DCs and pDCs. Increased proportions of activated CD8+ and CD4+ T cells. Increased proportions of CD4+ naïve, central memory, and regulatory T cells. Decreased KRAS VAF (in 12/14 patients who had detectable KRAS mutations in plasma). |
Treatment is tolerable. Two DLTs (grade 3 and 4 febrile neutropenia), however deemed unrelated to either sotigalimab or nivolumab. 14 (47%) patients with treatment-related SAE. Most common were pyrexia, sepsis, haemolytic uraemic syndrome, and nausea. Overall, grade 3 or 4 treatment-related adverse events occurred in 28 (93%) of 30 patients and were clinically manageable. Most common grade 3-4 treatment-related AEs were haematological and generally transient (lymphocyte count decrease, neutrophil count decrease, and anaemia). No grade 3-4 CRS and infusion reactions. Two deaths due to AEs related to Gem/Nab (sepsis and septic shock in the setting of neutropenia). One death from an unknown cause occurring 4 months after the last study intervention. |
NCT03214250 [137] |
|
Sotigalimab (APX005M) + Gem/Nab +/- nivolumab (PRINCE) |
Randomized, open-label, phase II |
First line treatment for metastasized PDAC (n=105) |
1) Nivolumab + Gem/Nab, 2) Sotigalimab + Gem/Nab, 3) Sotigalimab + nivolumab + Gem/Nab. Nivolumab on days 1 and 15. Sotigalimab on day 3 (=2 days after chemotherapy), or on day 10 if patients received chemotherapy on day 8. |
1) Nivo/chemo (n=34): Median PFS 6.4 months (95% CI: 5.2-8.8). Median OS 16.7 months (95% CI: 9.8-18.4). 1-year OS 57.7% ORR 50% (95% CI: 32-68). 2) Sotiga/chemo (n=36): Median PFS 7.3 months (95% CI: 5.4-9.2). Median OS 11.4 months (95% CI: 7.2-20.1) 1-year OS 48.1% ORR 33% 3) Sotiga/nivo/chemo (n=35): Median PFS 6.7 months (95% CI: 4.2-9.8). Median OS 10.1 months (95% CI: 7.9-13.2). 1-year OS 41.3% ORR 31%. |
98% of the patients had at least one treatment related AE. Most common grade 3-4 treatment related AEs were hematologic and generally transient. Two patients died due to an AE. One from acute hepatic failure possibly related to sotiga/chemo. One from intracranial haemorrhage possibly related sotiga/nivo/chemo. |
NCT03214250 [138] |
|
|
Mitazalimab + autologous DC vaccine (REACtiVe-2) |
Open-label, dose-escalation, phase I |
Metastasized PDAC (n=16) |
Biweekly 25 × 10[6] DCs (1/3 i.d. and 2/3 i.v.) co-administered with mitazalimab for the first three administrations, followed by a fourth and fifth administration if no disease progression. | In patients without PD at baseline: Median PFS 2.76 months (IQR: 2.40-6.86), and median OS 12.1 months (IQR: 5.74-21.77). 1-year PFS rate 13% 1-year OS rate 50%. No objective radiological response. 8/16 patients (50%) with SD after 3x administrations. | Systemic increase in activated and vaccine-specific T cell responses. Increased T cell infiltration and decreased collagen deposition in post-treatment biopsies. | Safe and well-tolerated. One transient DLT (grade 3 fever). | NCT05650918 [155] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




