Submitted:
01 May 2026
Posted:
05 May 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Optogenetics: From Evolution to Current Status
3. Opto-Biotechnology: Core Principle
3.1. Photoreceptor Network and Light Responsiveness Modules
3.2. Light and Metabolic Regulation
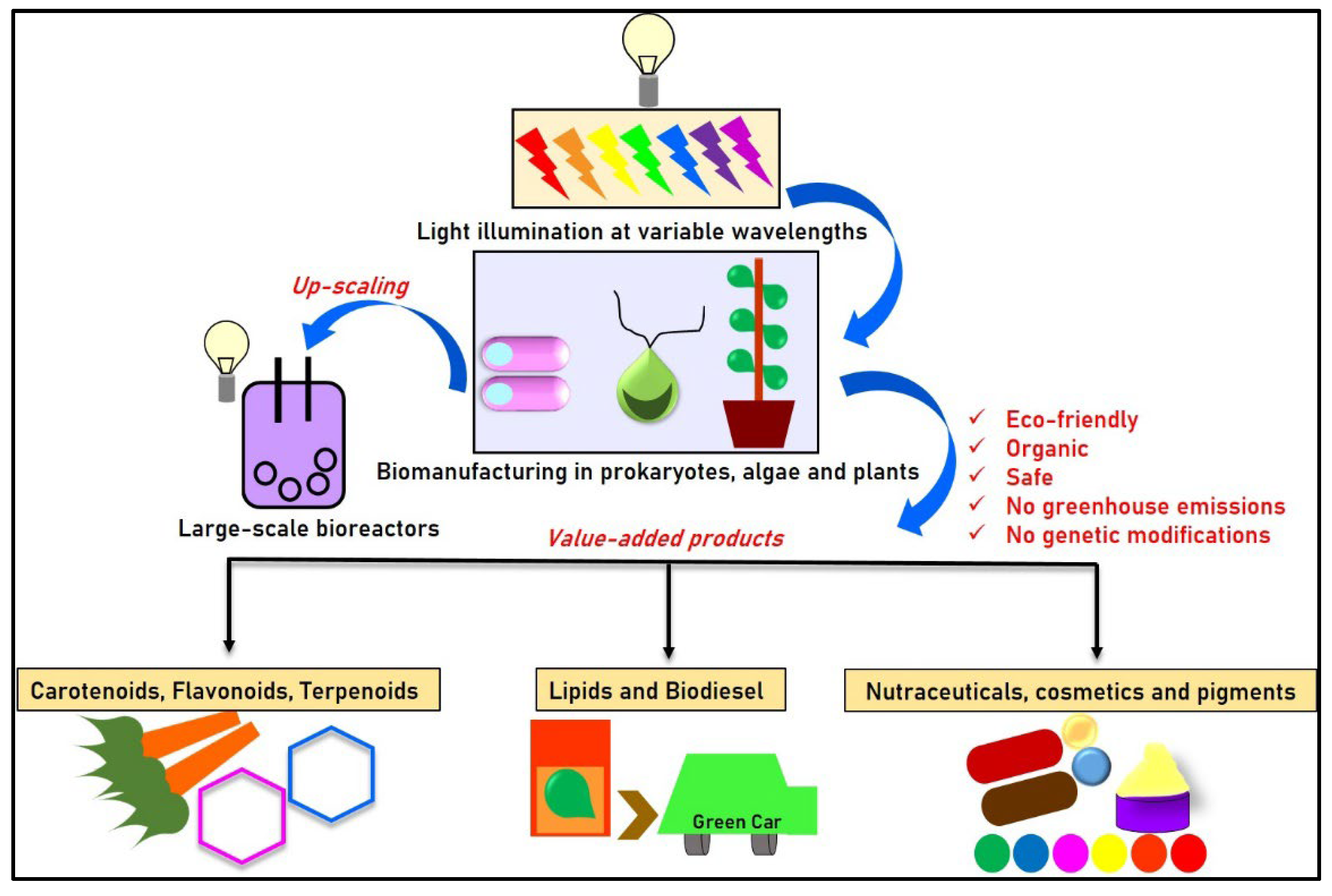
3.3. Mechanisms for Opto-Modulated Circuits and Biomanufacturing
4. Opto tools and Technologies in Opto-Bimanufacturing
4.1. Optogenetic and Light-Controlled Biomanufacturing
4.2. Optobioreactor Technologies
4.2.1. An Introduction to Light-Based Bioreactors
4.2.2. Effect of Artificial Light on Algal Cultivation
4.2.3. Conventional Methods of Artificial Illumination
4.2.3. Light-Emitting Diodes (LEDs)
4.3. Systems Biology, Artificial Intelligence and Machine Learning Approaches in Opto-Biomanufacturing
4.4. Biosynthetic Gene Clusters in Opto Biomanufacturing
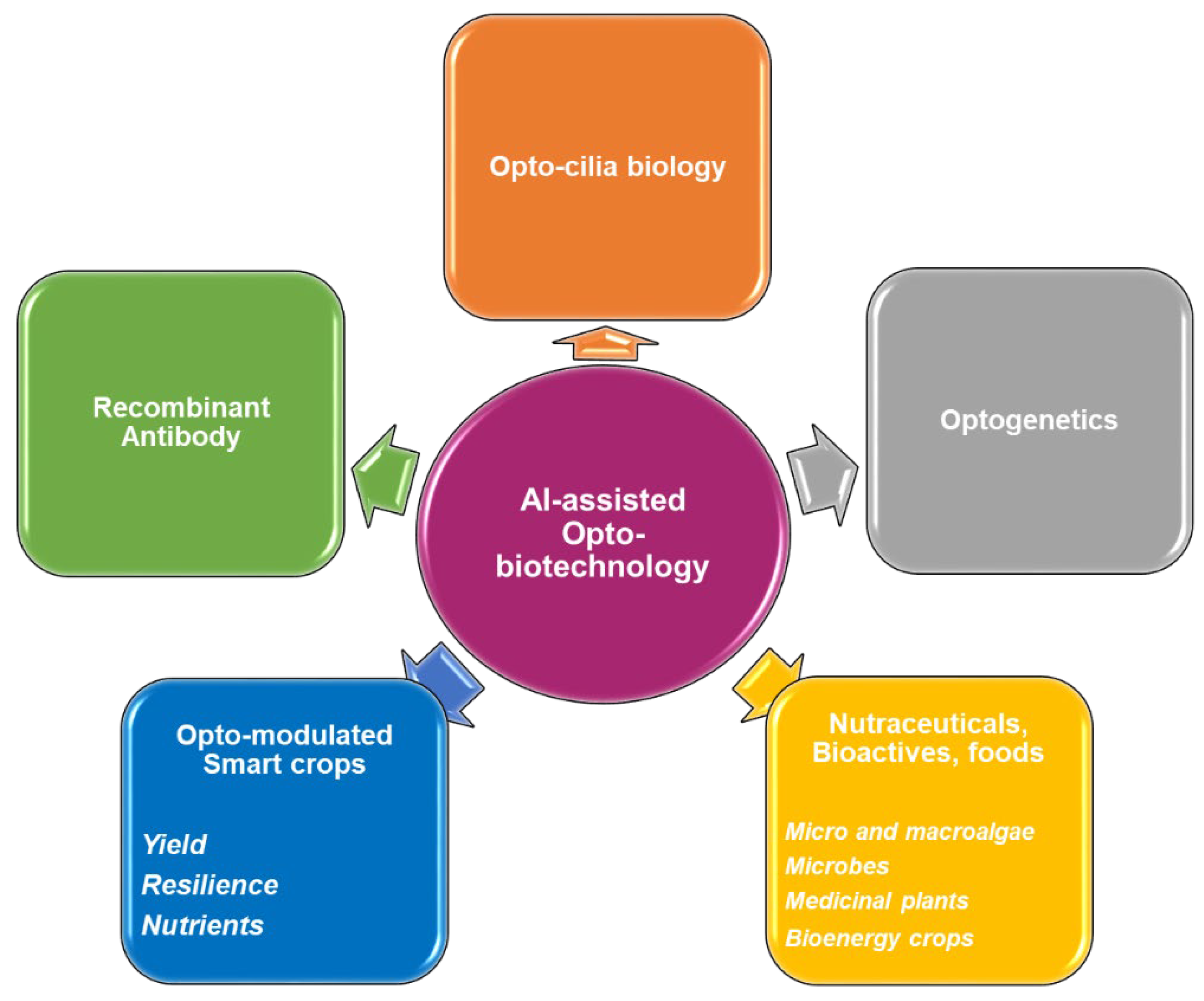
5. Scope in Different Sectors
6. Challenges and Future Directions
7. Conclusions
Author Contributions
Author’s approval
Declaration of interest
Acknowledgements
References
- Al-Qasmi, M.; Raut, N.; Talebi, S.; Al-Rajhi, S.; Al-Barwani, T. A review of effect of light on microalgae growth. Proc. World Congr. Eng. 2012, Vol. 1(No. 2), 1–7. [Google Scholar]
- Arnlund, D.; Johansson, L.C.; Wickstrand, C.; Barty, A.; Williams, G.J.; Malmerberg, E.; Davidsson, J.; Milathianaki, D.; DePonte, D.P.; Shoeman, R.L.; Wang, D. Visualizing a protein quake with time-resolved X-ray scattering at a free-electron laser. Nat. Methods 2014, 11(9), 923–926. [Google Scholar] [CrossRef]
- Baidya, A.; Akter, T.; Islam, M. R.; Shah, A. A.; Hossain, M. A.; Salam, M. A.; Paul, S. I. Effect of different wavelengths of LED light on the growth, chlorophyll, β-carotene content and proximate composition of Chlorella ellipsoidea. Heliyon 2021, 7(12). [Google Scholar] [CrossRef]
- Beel, B.; Prager, K.; Spexard, M.; Sasso, S.; Weiss, D.; Müller, N.; Heinnickel, M.; Dewez, D.; Ikoma, D.; Grossman, A.R.; Kottke, T. A flavin binding cryptochrome photoreceptor responds to both blue and red light in Chlamydomonas reinhardtii. Plant Cell 2012, 24(7), 2992–3008. [Google Scholar] [CrossRef]
- Blanken, W.; Cuaresma, M.; Wijffels, R. H.; Janssen, M. Cultivation of microalgae on artificial light comes at a cost. Algal Res. 2013, 2(4), 333–340. [Google Scholar] [CrossRef]
- Borella, L.; Sforza, E.; Bertucco, A. Effect of residence time in continuous photobioreactor on mass and energy balance of microalgal protein production. N Biotechnol. 2021, 64, 46–53. [Google Scholar] [CrossRef] [PubMed]
- Boyden, E.S.; Zhang, F.; Bamberg, E.; Nagel, G.; Deisseroth, K. Millisecond-timescale, genetically targeted optical control of neural activity. Nat. Neurosci. 2005, 8(9), 1263–1268. [Google Scholar] [CrossRef]
- Boyden, E.S. A history of optogenetics: the development of tools for controlling brain circuits with light. F1000 Biol. Rep. 2011, 3, 11. [Google Scholar] [CrossRef]
- Brzychczyk, B.; Hebda, T.; Pedryc, N. The influence of artificial lighting systems on the cultivation of algae: The example of chlorella vulgaris. Energies 2020, 13(22), 5994. [Google Scholar] [CrossRef]
- Carvalho, A. P.; Silva, S. O.; Baptista, J. M.; Malcata, F. X. Light requirements in microalgal photobioreactors: an overview of biophotonic aspects. Appl. Microbiol. Biotechnol. 2011, 89(5), 1275–1288. [Google Scholar] [CrossRef]
- Chen, I.W.; Papagiakoumou, E.; Emiliani, V. Towards circuit optogenetics. Curr. Opin. Neurobiol. 2018, 50, 179–189. [Google Scholar] [CrossRef]
- Deisseroth, K.; Feng, G.; Majewska, A.K.; Miesenböck, G.; Ting, A.; Schnitzer, M.J. Next-generation optical technologies for illuminating genetically targeted brain circuits. J. Neurosci. 2006, 26(41), 10380–10386. [Google Scholar] [CrossRef]
- Deisseroth, K. Optogenetics. Nat. Methods 2011, 8(1), 26–29. [Google Scholar] [CrossRef]
- Duan, X.; Zhu, M.; Gao, S. Two decades of optogenetic tools: a retrospective and a look ahead. Adv. Genet. 2025, 6(3), e00021. [Google Scholar] [CrossRef] [PubMed]
- Dutta Gupta, S.; Jatothu, B. Fundamentals and applications of light-emitting diodes (LEDs) in in vitro plant growth and morphogenesis. Plant Biotechnol. Rep. 2013, 7(3), 211–220. [Google Scholar] [CrossRef]
- Emiliani, V.; Entcheva, E.; Hedrich, R.; Hegemann, P.; Konrad, K. R.; Lüscher, C.; Yizhar, O. Optogenetics for light control of biological systems. Nat. Rev. Methods Prim. 2022, 2(1), 55. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, B. D.; Mota, A.; Teixeira, J. A.; Vicente, A. A. Continuous cultivation of photosynthetic microorganisms: approaches, applications and future trends. Biotechnol. Adv. 2015, 33(6), 1228–1245. [Google Scholar] [CrossRef]
- Fu, R.; Chen, Z.; Tian, H.; Hu, J.; Bu, F.; Zheng, P.; Chi, L.; Xue, L.; Jiang, Q.; Li, L.; Zhu, L. A review on the applications of machine learning in biomaterials, biomechanics, and biomanufacturing for tissue engineering. Smart Mater. Med. 2025, 6(2), 171–204. [Google Scholar] [CrossRef]
- Gao, L.; et al. Nanophotonic Approaches for Enhancing Light Utilization in Bioprocesses. Adv. Mater. 2023, 35(12), 2301045. [Google Scholar]
- García-Plazaola, J.I.; Esteban, R.; Fernández-Marín, B.; Kranner, I.; Porcar-Castell, A. Thermal energy dissipation and xanthophyll cycles beyond the Arabidopsis model. Photosynth. Res. 2012, 113(1), 89–103. [Google Scholar] [CrossRef]
- George, B.; Pancha, I.; Desai, C.; Chokshi, K.; Paliwal, C.; Ghosh, T.; Mishra, S. Effects of different media composition, light intensity and photoperiod on morphology and physiology of freshwater microalgae Ankistrodesmus falcatus–A potential strain for bio-fuel production. Bioresour. Technol. 2014, 171, 367–374. [Google Scholar] [CrossRef]
- Ghedifa, A. B.; Vega, J.; Korbee, N.; Mensi, F.; Figueroa, F. L.; Sadok, S. Effects of light quality on the photosynthetic activity and biochemical composition of Gracilaria gracilis (Rhodophyta). J. Appl. Phycol. 2021, 33, 3413–3425. [Google Scholar] [CrossRef]
- Glemser, M.; Heining, M.; Schmidt, J.; Becker, A.; Garbe, D.; Buchholz, R.; Brück, T. Application of light-emitting diodes (LEDs) in cultivation of phototrophic microalgae: current state and perspectives. Appl. Microbiol. Biotechnol. 2016, 100(3), 1077–1088. [Google Scholar] [CrossRef]
- Hifney, A.F.; Issa, A.A.; Fawzy, M.A. Abiotic stress induced production of ß-carotene, allophycocya-nin and total lipids in Spirulina sp. J. Biol. Earth Sci. 2013, 3, B54–B64. [Google Scholar]
- Im, C. S.; Eberhard, S.; Huang, K.; Beck, C. F.; Grossman, A. R. Phototropin involvement in the expression of genes encoding chlorophyll and carotenoid biosynthesis enzymes and LHC apoproteins in Chlamydomonas reinhardtii. Plant J. 2006, 48(1), 1–16. [Google Scholar] [CrossRef]
- Iasimone, F.; Panico, A.; De Felice, V.; Fantasma, F.; Iorizzi, M.; Pirozzi, F. Effect of light intensity and nutrients supply on microalgae cultivated in urban wastewater: Biomass production, lipids accumulation and settleability characteristics. J. Environ. Manag. 2018, 223, 1078–1085. [Google Scholar] [CrossRef]
- Konrad, K.R.; Gao, S.; Zurbriggen, M.D.; Nagel, G. Optogenetic methods in plant biology. Annu. Rev. Plant Biol. 2023, 74(1), 313–339. [Google Scholar] [CrossRef]
- Kumari, A.; Kumar, A.; Sharma, K.; Pati, S.R.; Mohanty, S.; Kateriya, S. Molecular characterization of unique multi-domain harbouring fungal rhodopsin for establishing their novel opto-synthetic biological usages. bioRxiv 2025, 2025–06. [Google Scholar]
- Lee, H. H.; Xing, Q.; Park, J. S.; Lee, H.; Yarish, C.; Kim, J. K. Effects of different artificial photosynthetically active radiation (PAR) sources and intensity on the growth and nutrient uptake in Ulva prolifera and Neopyropia yezoensis. Algal Res. 2023, 72, 103151. [Google Scholar] [CrossRef]
- Liu, J.; Yuan, C.; Hu, G.; Li, F. Effects of light intensity on the growth and lipid accumulation of microalga Scenedesmus sp. 11-1 under nitrogen limitation. Appl. Biochem. Biotechnol. 2012, 166(8), 2127–2137. [Google Scholar] [CrossRef]
- Long, B.; Fischer, B.; Zeng, Y.; Amerigian, Z.; Li, Q.; Bryant, H.; Li, M.; Dai, S.Y.; Yuan, J.S. Machine learning-informed and synthetic biology-enabled semi-continuous algal cultivation to unleash renewable fuel productivity. Nat. Commun. 2022, 13(1), 541. [Google Scholar] [CrossRef]
- Möglich, A.; Moffat, K. Structural basis for light-dependent signaling in the dimeric LOV domain of the photosensor YtvA. J. Mol. Biol. 2007, 373(1), 112–126. [Google Scholar] [CrossRef]
- Mc Gee, D.; Archer, L.; Smyth, T. J.; Fleming, G. T.; Touzet, N. Bioprospecting and LED-based spectral enhancement of antimicrobial activity of microalgae isolated from the west of Ireland. Algal Res. 2020, 45, 101704. [Google Scholar] [CrossRef]
- Oesterhelt, D.; Stoeckenius, W. Functions of a new photoreceptor membrane. Proc. Natl. Acad. Sci. 1973, 70(10), 2853–2857. [Google Scholar] [CrossRef]
- Park, S.; et al. Light-Induced Oxidative Stress and Its Role in High-Value Bioproduct Formation. Biochem. Eng. J. 2020, 156, 107634. [Google Scholar]
- Petroutsos, D.; Tokutsu, R.; Maruyama, S.; Flori, S.; Greiner, A.; Magneschi, L.; Cusant, L.; Kottke, T.; Mittag, M.; Hegemann, P.; Finazzi, G. A blue-light photoreceptor mediates the feedback regulation of photosynthesis. Nature 2016, 537(7621), 563–566. [Google Scholar] [CrossRef]
- Polstein, L.R.; Gersbach, C.A. Light-inducible spatiotemporal control of gene activation by customizable zinc finger transcription factors. J. Am. Chem. Soc. 2012, 134(40), 16480–16483. [Google Scholar] [CrossRef]
- Metsoviti, M. N.; Papapolymerou, G.; Karapanagiotidis, I. T.; Katsoulas, N. Effect of light intensity and quality on growth rate and composition of Chlorella vulgaris. Plants 2019, 9(1), 31. [Google Scholar] [CrossRef]
- Nwoba, E. G.; Parlevliet, D. A.; Laird, D. W.; Alameh, K.; Moheimani, N. R. Light management technologies for increasing algal photobioreactor efficiency. Algal Res. 2019, 39, 101433. [Google Scholar] [CrossRef]
- Ra, C. H.; Sirisuk, P.; Jung, J. H.; Jeong, G. T.; Kim, S. K. Effects of light-emitting diode (LED) with a mixture of wavelengths on the growth and lipid content of microalgae. Bioprocess Biosyst. Eng. 2018, 41(4), 457–465. [Google Scholar] [CrossRef]
- Reshetnikov, V.V.; Smolskaya, S.V.; Feoktistova, S.G.; Verkhusha, V.V. Optogenetic approaches in biotechnology and biomaterials. Trends Biotechnol. 2022, 40(7), 858–874. [Google Scholar] [CrossRef]
- Sandgruber, F.; Gielsdorf, A.; Baur, A.C.; Schenz, B.; Müller, S.M.; Schwerdtle, T.; Stang, G.I.; Grieh, C.; Lorkowski, S.; Dawczynski, C. Variability in Macro- and Micronutrients of 15 Commercially Available Microalgae Powders. Mar. Drugs. 2021, 19, 310. [Google Scholar] [CrossRef] [PubMed]
- Schulze, P.S.; Guerra, R.; Pereira, H.; Schüler, L.M.; Varela, J.C. Flashing LEDs for microalgal production. Trends Biotechnol. 2017, 35(11), 1088–1101. [Google Scholar] [CrossRef]
- Seong, J.; Lin, M. Z. Optobiochemistry: Genetically encoded control of protein activity by light. Annu. Rev. Biochem. 2021, 90(1), 475–501. [Google Scholar] [CrossRef] [PubMed]
- Sforza, E.; Simionato, D.; Giacometti, G.M.; Bertucco, A.; Morosinotto, T. Adjusted light and dark cycles can optimize photosynthetic efficiency in algae growing in photobioreactors. PLoS ONE 2012, 7(6), e38975. [Google Scholar] [CrossRef]
- Singh, V.; Mishra, V. A review on the current application of light-emitting diodes for microalgae cultivation and its fiscal analysis. Crit. Rev. Biotechnol. 2023, 43(5), 665–679. [Google Scholar] [CrossRef] [PubMed]
- Silva-Stenico, M.E.; Silva, C.S.P.; Lorenzi, A.S.; Shishido, T.K.; Etchegaray, A.; Lira, S.P.; Moraes, L.A.B.; Fiore, M.F. Non-ribosomal peptides produced by Brazilian cyanobacterial isolates with antimicrobial activity. Microbiol. Res. 2011, 166(3), 161–175. [Google Scholar] [CrossRef]
- Süssmuth, R.; Müller, J.; von Döhren, H.; Molnár, I. Fungal cyclooligomer depsipeptides: from classical biochemistry to combinatorial biosynthesis. Nat. Prod. Rep. 2011, 28, 99–124. [Google Scholar] [CrossRef]
- Sumanasekara, H.; Jayasingha, H.; Amarasooriya, G.; Dayarathne, N.; Mainali, B.; Senevirathna, L.; Gamage, A.; Merah, O. Hybrid machine learning models for predicting the impact of light wavelengths on algal growth in freshwater ecosystems. Phycology 2025, 5(2), 23. [Google Scholar] [CrossRef]
- Takala, H.; Björling, A.; Berntsson, O.; Lehtivuori, H.; Niebling, S.; Hoernke, M.; Kosheleva, I.; Henning, R.; Menzel, A.; Ihalainen, J.A.; Westenhoff, S. Signal amplification and transduction in phytochrome photosensors. Nature 2014, 509(7499), 245–248. [Google Scholar] [CrossRef]
- Takeshita, T.; Ota, S.; Yamazaki, T.; Hirata, A.; Zachleder, V.; Kawano, S. Starch and lipid accumulation in eight strains of six Chlorella species under comparatively high light intensity and aeration culture conditions. Bioresour. Technol. 2014, 158, 127–134. [Google Scholar] [CrossRef]
- Teo, C. L.; Atta, M.; Bukhari, A.; Taisir, M.; Yusuf, A. M.; Idris, A. Enhancing growth and lipid production of marine microalgae for biodiesel production via the use of different LED wavelengths. Bioresour. Technol. 2014, 162, 38–44. [Google Scholar] [CrossRef]
- Toledo-Ortiz, G.; Huq, E.; Rodríguez-Concepción, M. Direct regulation of phytoene synthase gene expression and carotenoid biosynthesis by phytochrome-interacting factors. Proc. Natl. Acad. Sci. 2010, 107(25), 11626–11631. [Google Scholar] [CrossRef]
- Trippens, J.; Greiner, A.; Schellwat, J.; Neukam, M.; Rottmann, T.; Lu, Y.; Kateriya, S.; Hegemann, P.; Kreimer, G. Phototropin influence on eyespot development and regulation of phototactic behavior in Chlamydomonas reinhardtii. Plant Cell 2012, 24(11), 4687–4702. [Google Scholar] [CrossRef]
- Wang, Y.; Jiang, X.; Hu, C.; Sun, T.; Zeng, Z.; Cai, X.; Li, H.; Hu, Z. Optogenetic regulation of artificial microRNA improves H. 2017. [Google Scholar]
- Yuan, Y.; Iannetta, A.A.; Kim, M.; Sadecki, W.; Arend, M.; Tsichla, A.; Águila Ruiz-Sola, M.; Kepesidis, G.; Falconet, D.; Thevenon, E.; Tardif, M. Phototropin connects blue light perception to starch metabolism in green algae. Nat. Commun. 2025, 16(1), 2545. [Google Scholar] [CrossRef] [PubMed]
- Ziegler, T.; Möglich, A. Photoreceptor engineering. Front. Mol. Biosci. 2015, 2, 30. [Google Scholar] [CrossRef]
- Zhao, E.M.; Zhang, Y.; Mehl, J.; Park, H.; Lalwani, M.A.; Toettcher, J.E.; Avalos, J.L. Optogenetic regulation of engineered cellular metabolism for microbial chemical production. Nature 2018, 555(7698), 683–687. [Google Scholar] [CrossRef] [PubMed]
- Zhao, E.M.; Lalwani, M.A.; Lovelett, R.J.; García-Echauri, S.A.; Hoffman, S.M.; Gonzalez, C.L.; Toettcher, J.E.; Kevrekidis, I.G.; Avalos, J.L. Design and Characterization of Rapid Optogenetic Circuits for Dynamic Control in Yeast Metabolic Engineering. ACS Synth. Biol. 2020, 9(12), 3254–3266. [Google Scholar] [CrossRef] [PubMed Central]
- Zhao, E.M.; Lalwani, M.A.; Chen, J.M.; Orillac, P.; Toettcher, J.E.; Avalos, J.L. Optogenetic Amplification Circuits for Light-Induced Metabolic Control. ACS Synth. Biol. 2021, 10(5), 1143–1154. [Google Scholar] [CrossRef] [PubMed]
| Light Quality | Algal Strain/Group | Key Metabolites/Proteins Affected | Mechanism/Notes | Reference | ||
| Low intensity + short residence (continuous batch) | Microalgae (general) | Increased total protein content | Optimizes photosynthesis, reduces photoinhibition | Borella et al., 2021 | ||
| Green (520-550 nm) | Red algae (Gracilaria gracilis) | R-phycoerythrin, soluble proteins | Phycobiliprotein biosynthesis | Ghedifa et al., 2021 | ||
| UV (280-400 nm) | Red algae (Gracilaria gracilis) | Mycosporine-like amino acids (MAAs) | UV protectants, antioxidants (synergistic with green) | Ghedifa et al., 2021 | ||
| Blue (400-500 nm) | Gracilaria gracilis | Antioxidants, phenolic compounds | Phototropin kinase signaling, phenylpropanoids | Ghedifa et al., 2021 | ||
| Blue shift | Prymnesium sp. DMGCW_41 | Polar fractions (antimicrobial) | Terpenoids/polyketides upregulated | McGee et al., 2020 | ||
| Blue shift | cf. Chlorococcum sp. DMGCW_43 | Polar fractions (antimicrobial) | Terpenoids/polyketides upregulated | McGee et al., 2020 | ||
| Blue (phototropin) | Green algae (C. reinhardtii) | Carotenoids (2-3x increase) | Kinase cascades, HPLC-detectable | Das et al., 2019 | ||
| White/Red (growth phase) + Blue/Green shift | General microalgae | Biomass → high-value (astaxanthin, MAAs) | Two-phase: growth then accumulation | Liu et al., 2012; Sforza et al., 2012; etc. |
||
| Pathway/Product | Host | OT Circuit/Module | Yield Improvement | Light Dose/Condition |
References |
|
| Lactic acid | S. cerevisiae | OptoAMP4/OptoINVRT7 | 12.8-fold vs dark | 1.3% pulses (blue) | ||
| Isobutanol | S. cerevisiae | OptoAMP/EXP | 8.49 g/L (2-fold) | 1.7% production phase |
Zhao et al., 2020; Zhao et al., 2021 Reshetnikov et al., 2022 |
|
| Naringenin | S. cerevisiae | OptoAMP4 | >20-fold vs plant | Variable two-phase | ||
| Lycopene | E. coli | PhyB-PIF3 | ~2-fold vs IPTG | Red light pulses | ||
| Hydrogen | C. reinhardtii |
CRY2-CIB1 (PSII repression) |
2.7-fold vs WT | Blue light (continuous) | ||
| Astaxanthin | Phaffia rhodozyma (yeast-like) | CRY2 clustering | 2.5-fold | Blue pulses | ||
| Isoprenoids/ sesquiterpenes |
Nicotiana benthamiana (higher plant) | EL222-VP64 (leaf agroinfiltration) | 10-50-fold transient | Blue light (450 nm) | ||
| Carotenoids | Arabidopsis | PULSE (UVR8-COP1 degron) | 3-fold flux redirection | UV-B pulses |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




