Submitted:
02 May 2026
Posted:
05 May 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
1.1. Scope and System Boundaries
2. Materials and Methods
2.1. Feedstock Description
2.2. Mlm-R™ Technology Description
2.3. Material Flow Analysis (Mfa) Tool and Reconciliation Approach
- (i)
- Goods Layer: Total Mass Flows (Kg Per Functional Basis);
- (ii)
- Carbon layer: elemental carbon flows (kg C per functional basis).
- (iii)
- A chemical-energy partitioning (HHV basis) was derived from reconciled mass flows and assigned heating values for the streams. Thermal energy flows (sensible heat, heat exchange across boundaries, and heat losses) were not modeled as STAN “energy flows” because the objective is to report chemical-energy partitioning consistent with the defined process boundary.
2.4. Framework for Petrochemical Feed Compatibility Discussion
3. Results
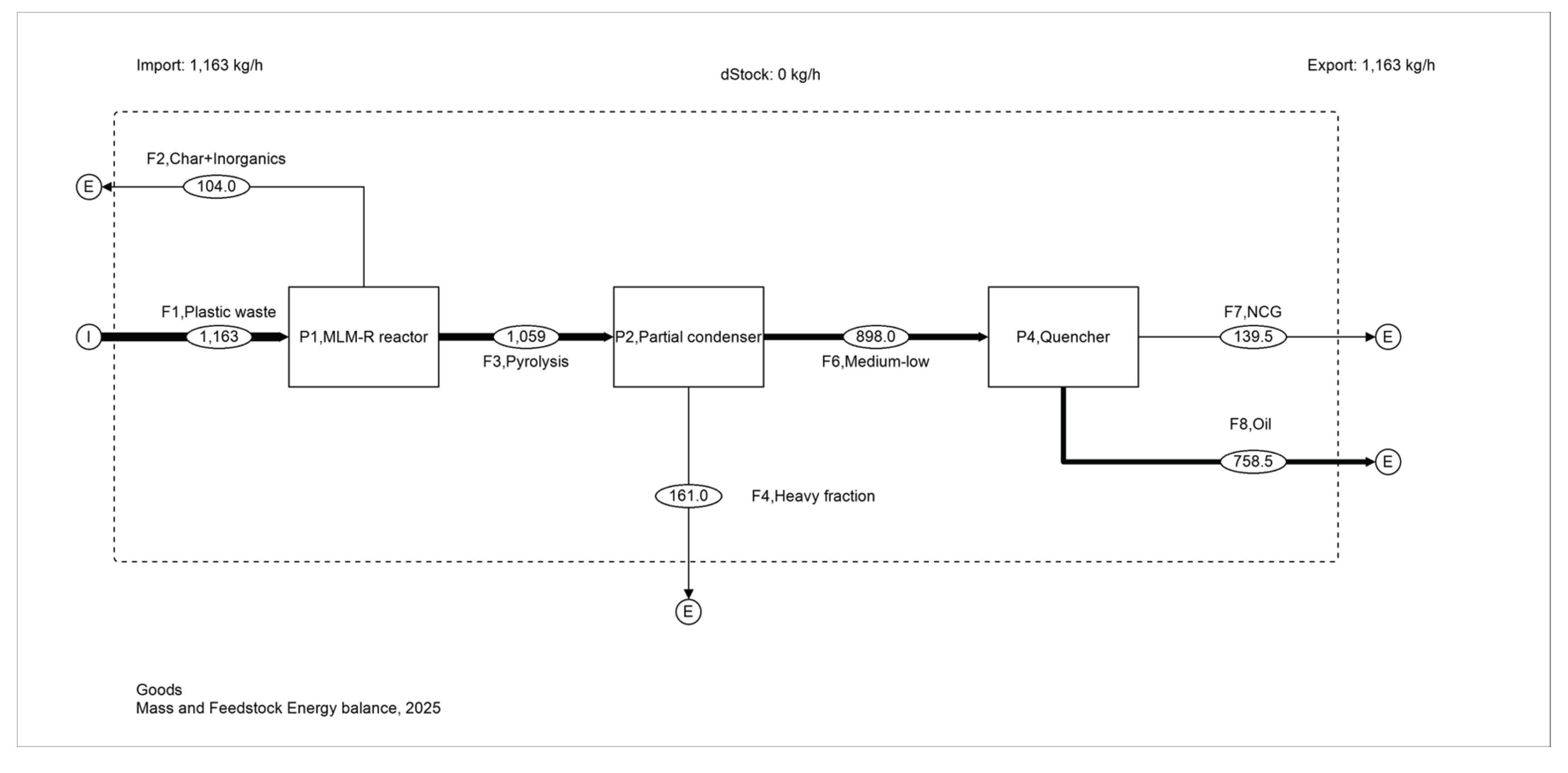
3.1. Reconciled Mass Balance
3.2. Chemical-Energy Partitioning
3.3. Robustness Considerations
4. Physicochemical Considerations for Mlm-Oil Integration
4.1. Linking Mfa Outcomes to Oil-Quality Targets
4.2. Impurity Control
5. Discussion
5.1. Industrial Relevance and Outlook
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Vollmer, I.; Jenks, M. J. F.; Roelands, M. C. P.; White, R. J.; van Harmelen, T.; de Wild, P.; van der Laan, G. P.; Meirer, F.; Keurentjes, J. T. F.; Weckhuysen, B. M. Beyond Mechanical Recycling: Giving New Life to Plastic Waste. Angew. Chem. Int. Ed. 2020, 59, 15402–15423. [Google Scholar] [CrossRef] [PubMed]
- Aguado, J.; Serrano, D. P.; Escola, J. M. Fuels from Waste Plastics by Thermal and Catalytic Processes: A Review. Ind. Eng. Chem. Res. 2008, 47, 7982–7992. [Google Scholar] [CrossRef]
- Aguado, J.; Serrano, D. P. Feedstock Recycling of Plastic Wastes; Royal Society of Chemistry: Cambridge, U.K., 2007. [Google Scholar] [CrossRef]
- Maqsood, T.; Dai, J.; Zhang, Y.; Guang, M.; Li, B. Pyrolysis of Plastic Species: A Review of Resources and Products. J. Anal. Appl. Pyrolysis 2021, 159, 105295. [Google Scholar] [CrossRef]
- Al-Salem, S. M.; Antelava, A.; Constantinou, A.; Manos, G.; Dutta, A. A Review on Thermal and Catalytic Pyrolysis of Plastic Solid Waste (PSW). J. Environ. Manag. 2017, 197, 177–198. [Google Scholar] [CrossRef] [PubMed]
- Di Blasi, C. Modeling Chemical and Physical Processes of Wood and Biomass Pyrolysis. Prog. Energy Combust. Sci. 2008, 34, 47–90. [Google Scholar] [CrossRef]
- Foster, A. J.; Jae, J.; Cheng, Y.-T.; Huber, G. W.; Lobo, R. F. Optimizing the Aromatic Yield and Distribution from Catalytic Fast Pyrolysis of Biomass over ZSM-5. Appl. Catal. 2012, A 423-424, 154–161. [Google Scholar] [CrossRef]
- Westerhout, R. W. J.; Waanders, J.; Kuipers, J. A. M.; van Swaaij, W. P. M. Kinetics of the Low-Temperature Pyrolysis of Polyethene, Polypropene, and Polystyrene: Modeling, Experimental Determination, and Comparison with Literature Models and Data. Ind. Eng. Chem. Res. 1997, 36, 1955–1964. [Google Scholar] [CrossRef]
- Gracida-Alvarez, U. R.; Mitchell, M. K.; Sacramento-Rivero, J. C.; Shonnard, D. R. Effect of Temperature and Vapor Residence Time on the Micropyrolysis Products of Waste High Density Polyethylene. Ind. Eng. Chem. Res. 2018, 57, 1911–1924. [Google Scholar] [CrossRef]
- Miskolczi, N.; Bartha, L.; Deak, G.; Jover, B.; Kallo, D. Thermal and Thermo-Catalytic Degradation of High-Density Polyethylene Waste. J. Anal. Appl. Pyrolysis 2004, 72, 235–242. [Google Scholar] [CrossRef]
- Glicksman, L. R. Fluidized Bed Scaleup. In Handbook of Fluidization and Fluid-Particle Systems; Yang, W.-C., Ed.; Marcel Dekker: New York, 2003; pp. 343–378. [Google Scholar]
- Werther, J.; Hartge, E.-U. chapter in. In Handbook of Fluidization and Fluid-Particle Systems; Yang, W.-C., Ed.; Marcel Dekker: New York, 2003. [Google Scholar]
- Handbook of Fluidization and Fluid-Particle Systems; Yang, W.-C., Ed.; Marcel Dekker: New York, 2003. [Google Scholar]
- Arena, U. Process and Technological Aspects of Municipal Solid Waste Gasification. A Review. Waste Manag. 2012, 32, 625–639. [Google Scholar] [CrossRef] [PubMed]
- Kunii, D.; Levenspiel, O. Fluidization Engineering, 2nd ed.; Butterworth-Heinemann: Boston, 1991. [Google Scholar]
- Basu, P. Combustion and Gasification in Fluidized Beds; CRC Press/Taylor & Francis: Boca Raton, FL, 2006. [Google Scholar]
- Grace, J. R.; Bi, H. T.; Golriz, M. Circulating Fluidized Beds. Adv. Chem. Eng. 2008, 35, 1–52. [Google Scholar]
- Knowlton, T. M. Standpipes and Nonmechanical Valves. In Handbook of Fluidization and Fluid-Particle Systems; Yang, W.-C., Ed.; Marcel Dekker: New York, 2003; pp. 571–598. [Google Scholar]
- Bi, H. T.; Grace, J. R. Flow Regime Diagrams for Gas-Solid Fluidization and Upward Transport. Int. J. Multiph. Flow 1995, 21, 1229–1236. [Google Scholar] [CrossRef]
- Mastellone, M. L. Plastic-to-Oil Plant for Converting Plastic Waste into Petrochemical Products. Eur. Patent EP 3 894 516 B1, 29 Nov 2023. [Google Scholar]
- PRUVIA GmbH. MLM-R Technology—Integrated Pyrolysis Loop for Plastic Waste Conversion. Available online: https://www.pruvia.com/technology-in-detail/ (accessed on 2026-03-08).
- Brunner, P. H.; Rechberger, H. Practical Handbook of Material Flow Analysis; CRC Press: Boca Raton, FL, 2016. [Google Scholar]
- Cencic, O. Nonlinear Data Reconciliation in Material Flow Analysis with Software STAN. Sustain. Environ. Res. 2016, 26, 291–298. [Google Scholar] [CrossRef]
- Allesch, A.; Brunner, P. H. Material Flow Analysis as a Tool to Improve Waste Management Systems: The Case of Austria. Environ. Sci. Technol. 2017, 51, 540–551. [Google Scholar] [CrossRef] [PubMed]
- Zaccariello, L.; Cremiato, R.; Mastellone, M. L. Evaluation of Municipal Solid Waste Management Performance by Material Flow Analysis: Theoretical Approach and Case Study. Waste Manag. Res. 2015, 33(10), 871–885. [Google Scholar] [CrossRef] [PubMed]
- Mastellone, M. L.; Brunner, P. H.; Arena, U. Scenarios of Waste Management for a Waste Emergency Area: A Substance Flow Analysis. J. Ind. Ecol. 2009, 13(5), 735–757. [Google Scholar] [CrossRef]
- Arena, U.; Di Gregorio, F. A Waste Management Planning Based on Substance Flow Analysis. Resour. Conserv. Recycl. 2014, 85, 54–66. [Google Scholar] [CrossRef]
| Component, % | Carbon*, % | Feedstock Energy, MJ/kg | |
|---|---|---|---|
| Polyolefins fraction | 86.00% | 86.08% | 45.73 |
| Other polymers | 4.00% | 56.85% | 23.25 |
| Inorganics | 2.00% | 0.00% | 0 |
| Biogenic | 3.00% | 50.00% | 20 |
| Moisture | 5.00% | 0.00% | 0 |
| Stream | Mass | Notes |
| Plastic feed | 1163 | System input |
| Condensed liquid | 919.26 | Pyrolysis-side product |
| Non-condensable gas (NCG) | 139.53 | Pyrolysis-side product |
| Char to regenerator off-gas | 80.74 (103.9 incl. inorganics) | Combustion-side outlet |
| Stream | Carbon (kg C per basis) | Fraction of feed carbon (%) |
| Feed carbon | 904.9 | 100 |
| Carbon in Oil | 600.9 (Light) +137.6 (Heavy) | 81.0 |
| Carbon in NCG | 105.1 | 11.6 |
| Carbon internally combusted | 61.3 | 6.8 |
| Energy term | Energy (MJ per basis) | Share of feed energy (%) |
| Feedstock chemical energy (HHV basis) | 47512 | 100 |
| Recovered in Oil (chemical) | 39618 | 83.3 |
| Recovered in NCG (chemical) | 5093 | 10.7 |
| Converted to heat via internal combustion | 2801 | 5.89 |
| Impurity | In the Oil |
In the Flue Gas (from char oxidation) |
| Chlorine | 13% | 87% |
| Nitrogen | 44% | 56% |
| Sulphur | 5% | 95% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




