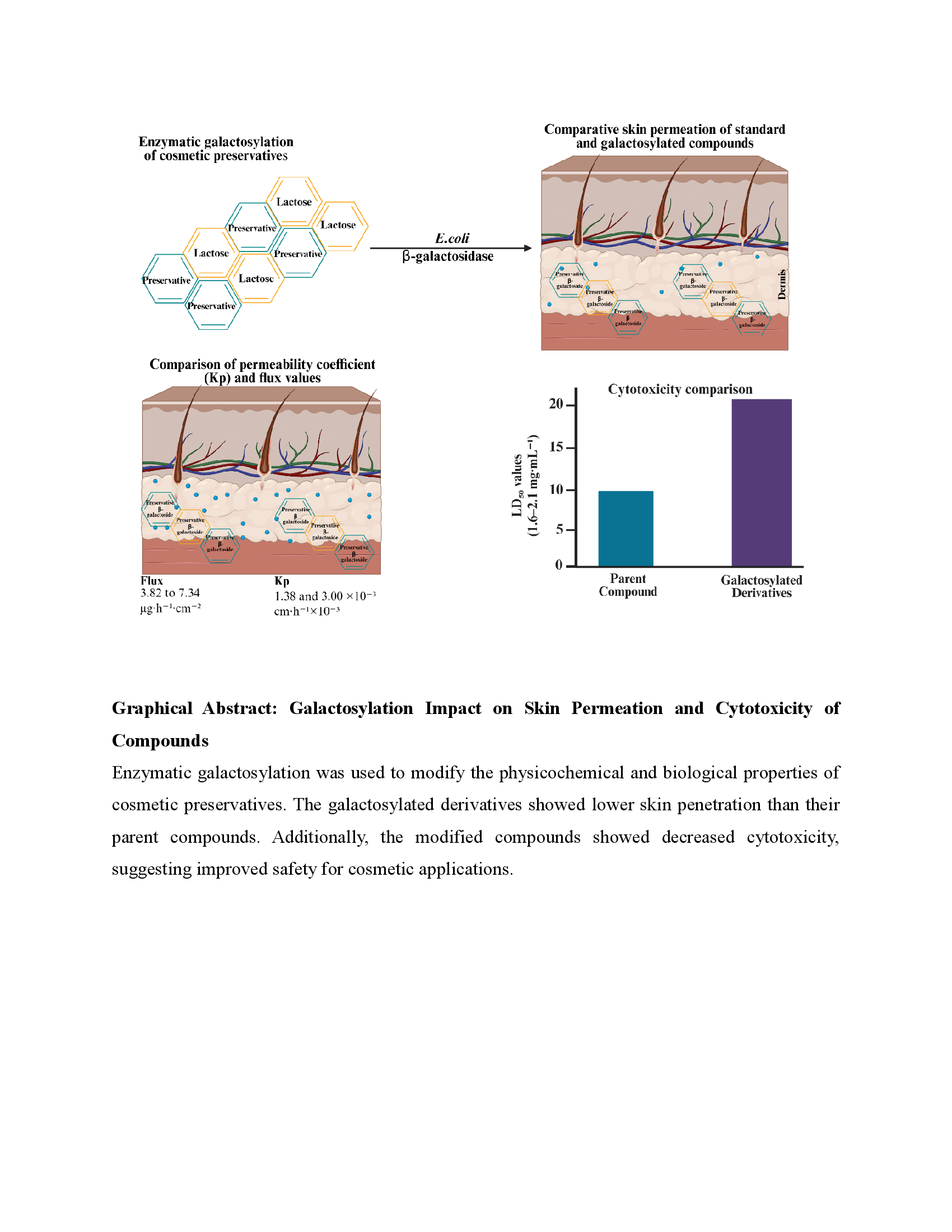
Cosmetic preservatives should have reduced percutaneous absorption to lower the risk of systemic exposure and skin irritation. In this work, Escherichia coli β-galactosidase was used to enzymatically modify several of the commonly used cosmetic preservatives to produce their corresponding galactosylated derivatives: benzyl alcohol β-D-galactopyranoside 7, 2-phenoxyethanol β-D-galactopyranoside 8, chlorphenesin β-D-galactopyranoside 9, 1,2-hexanediol β-D-galactopyranoside 10, 1,2-octanediol β-D-galactopyranoside 11, and 2-phenylethyl β-D-galactopyranoside 12.
HPLC and NMR spectroscopy were used to analyze the synthesized derivatives. The Franz diffusion cell assay was used to evaluate skin penetration. 2-phenoxyethanol (PE), chlorphenesin (CPN), and 2-phenylethanol (PhE), exhibited measurable skin penetration with flux values ranging from 3.82 to 7.34 µg·h⁻¹·cm⁻² and permeability coefficients (Kp) between 1.38 and 3.00 ×10⁻³ cm·h⁻¹. In contrast, their galactosylated derivatives showed markedly reduced permeation under the same experimental conditions.
Moreover, brine shrimp lethality assays indicated that galactosylated derivatives had significantly higher LD₅₀ values (1.6–2.1 mg/mL) than their parent compounds (0.1–0.79 mg/mL), suggesting lower cytotoxicity. These findings suggest that enzymatic galactosylation can significantly decrease skin permeability and the toxicity of cosmetic preservatives, highlighting its potential as a strategy to improve the safety of cosmetic ingredients.