Submitted:
02 May 2026
Posted:
04 May 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
| Feature | Brucellosis | Tuberculosis | Impact of Co-Infection | References |
|---|---|---|---|---|
| Causative Agent | Brucella melitensis, B. abortus, B. suis (Gram-negative coccobacillus) | Mycobacterium tuberculosis (acid-fast bacillus) | Dual intracellular infection: potential competition for macrophage niches | [16] |
| Primary Reservoir | Domestic animals (goats, cattle, swine); zoonotic | Humans (airborne transmission) | Overlap in endemic rural/agricultural regions | [17] |
| Intracellular Niche | Brucella-containing vacuole (BCV) that acquires ER markers | M. tuberculosis-containing phagosome that inhibits lysosomal fusion | Both evade phagolysosome fusion; may compete for host resources | [18] |
| Immune Response | Th1-mediated (IFN-γ, TNF-α) but with anti-inflammatory IL-10 modulation | Th1-dominated (IFN-γ, IL-12, TNF-α); granuloma formation | Potential immune exhaustion and dysregulation | [19] |
| Oxidative Stress Markers | ↑ MDA, ↓ SOD, ↓ GPx; limited human studies | ↑ MDA, ↑ 8-oxo-dG, ↓ GSH, ↓ total antioxidant capacity | Likely amplified oxidative stress; more severe DNA damage | [20] |
| Telomere Biology | Telomere shortening | Shorter telomere length in PBMCs; altered telomerase activity | Expected accelerated telomere attrition and immune senescence | [21] |
| Primary Drug Resistance Mechanisms | rpoB mutations (rifampicin); Efflux pumps (TetA/B for doxycycline); Intracellular persistence/relapse | rpoB mutations (rifampicin); katG / inhA mutations (isoniazid); gyrA/B (fluoroquinolones); Efflux pumps | Overlapping rifampicin use; risk of cross-resistance; drug drug interactions (e.g., rifampicin reduces doxycycline levels) | [22] |
| Standard Treatment | Doxycycline + rifampicin OR doxycycline + gentamicin/streptomycin | RHZE (rifampicin, isoniazid, pyrazinamide, ethambutol) | Prolonged rifampicin exposure; complex regimens; higher relapse risk | [23] |
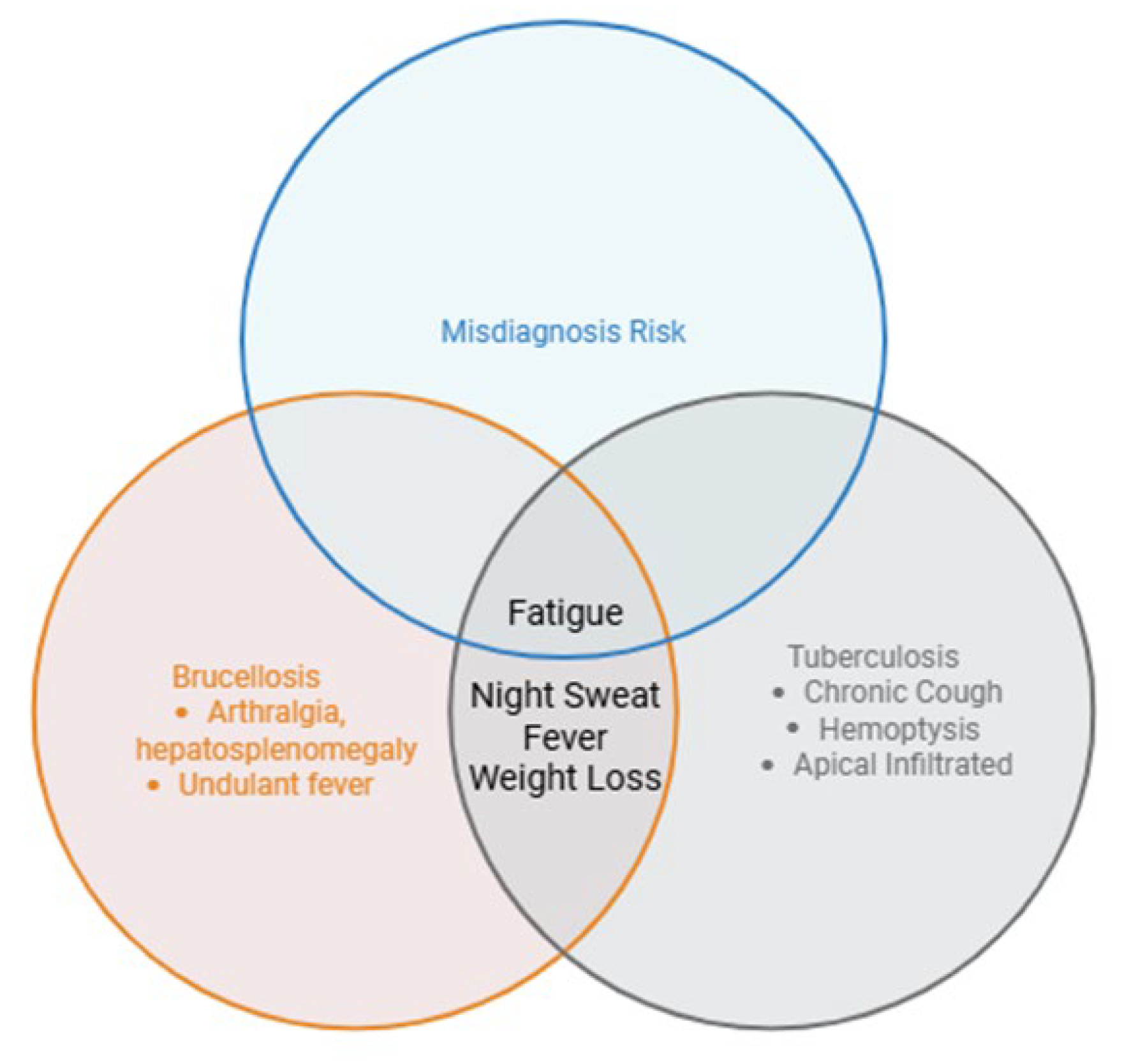
| Common Clinical Overlap | Fever, night sweats, weight loss, fatigue, arthralgia, hepatosplenomegaly | Fever, night sweats, weight loss, fatigue, chronic cough, hemoptysis | High risk of misdiagnosis or delayed treatment | [24] |
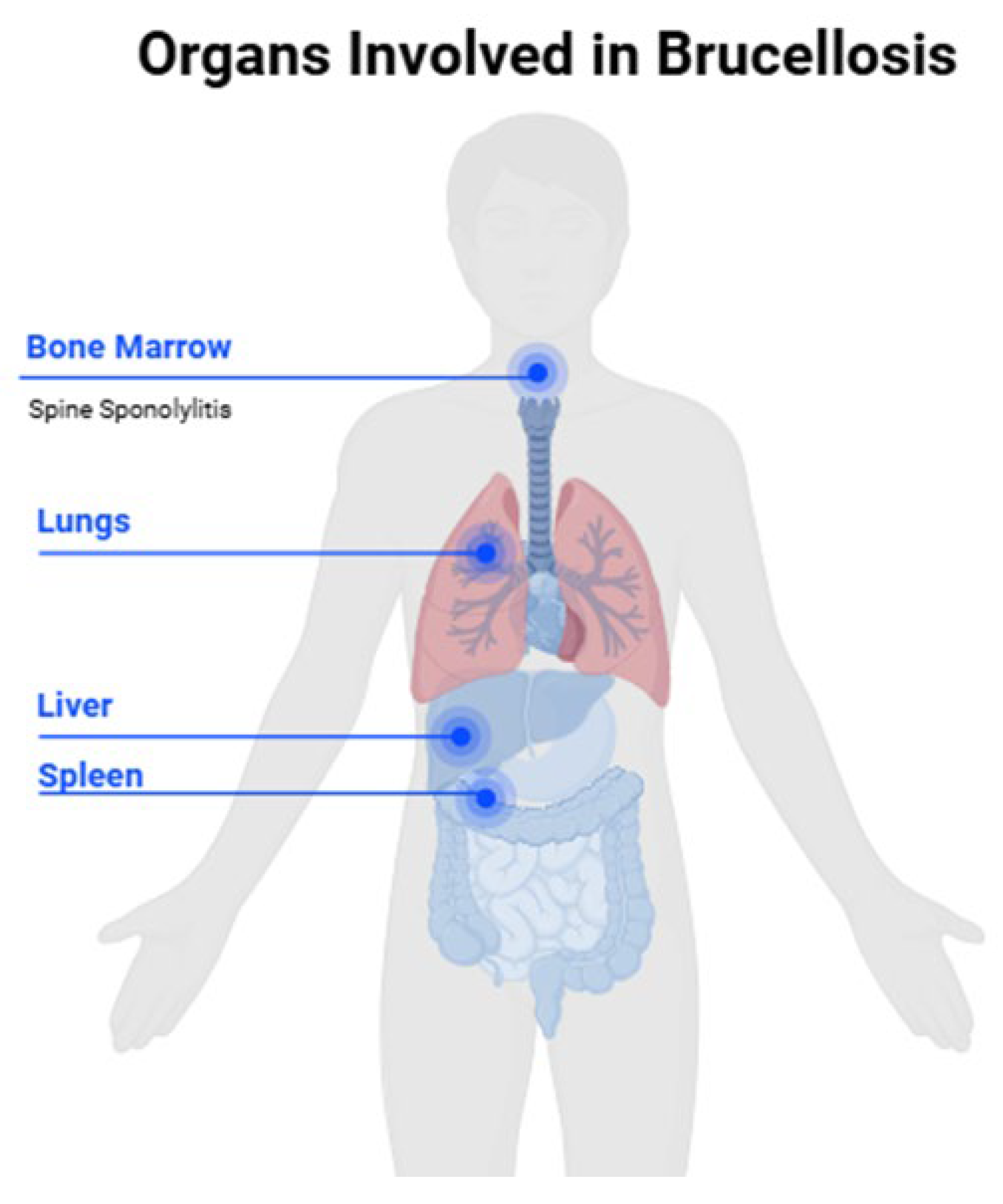
| Pathognomonic Finding | Non-caseating granulomas (liver, spleen, bone marrow) | Caseating granulomas with Langhans giant cells (lungs, lymph nodes) | Histology alone cannot distinguish; requires culture or molecular diagnostics | [25] |
2. Pathogenesis of Brucellosis and Tuberculosis
2.1. Brucellosis Pathogenesis
2.2. Tuberculosis Pathogenesis
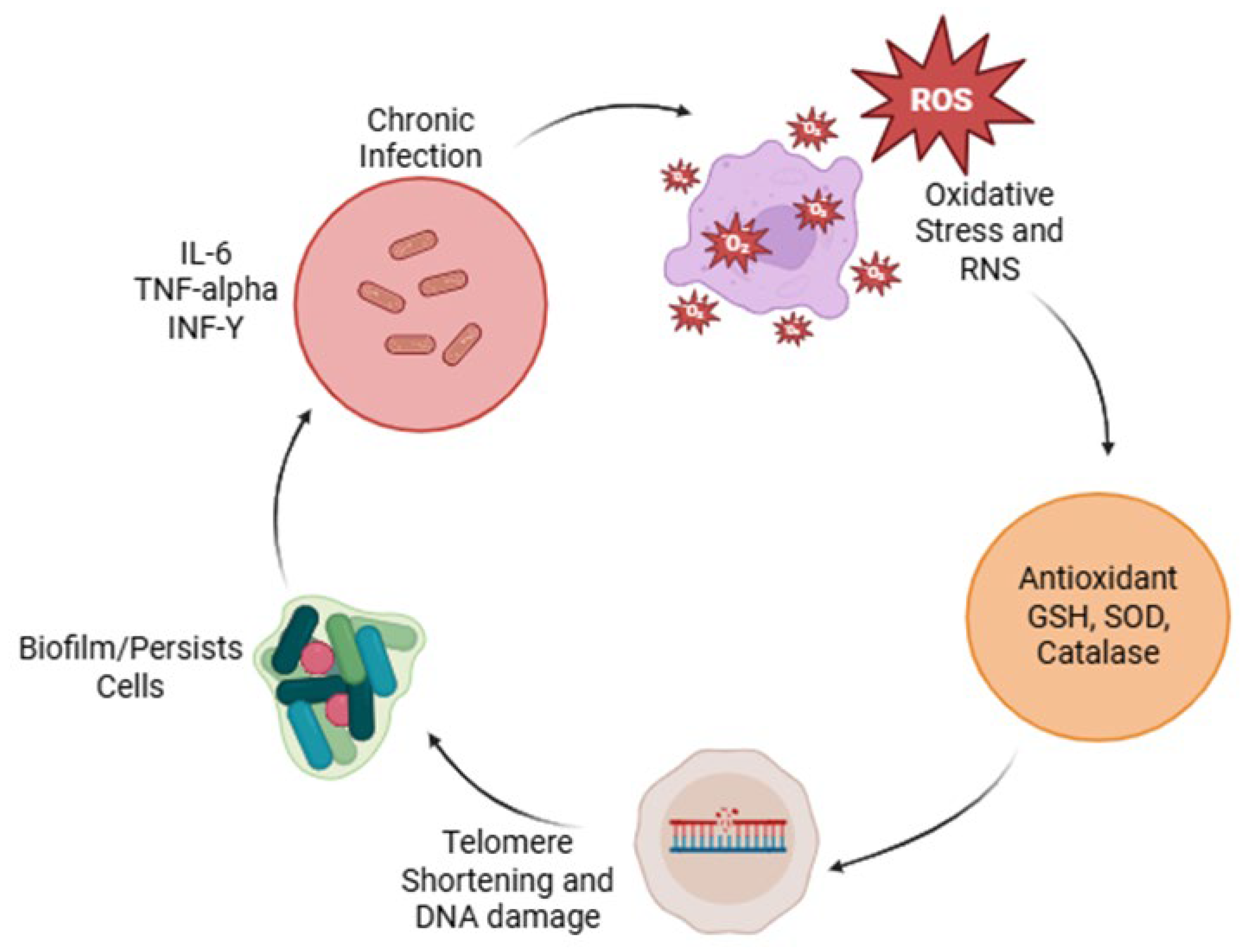
2.3. Co-Infection Dynamics
2.4. Oxidative Stress in Brucellosis and TB
2.5. Telomere Biology and Chronic Infection
3. Drug Resistance Mechanisms
3.1. Drug Resistance in Tuberculosis
3.2. Drug Resistance in Brucellosis
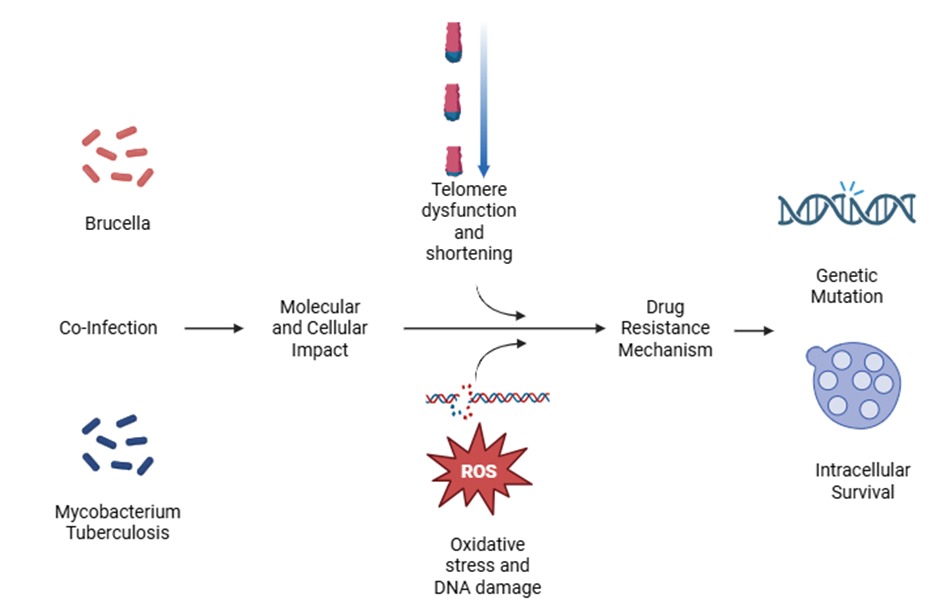
3.3. Impact of Co-Infection on Drug Resistance
3.4. Interconnection Between Telomere Biology, Oxidative Stress, and Drug Resistance
4. Clinical Symptoms and Implications with Future Perspectives
5. Conclusions
Conflicts of Interest
References
- Haileamlak. Tuberculosis Continued as Global Challenge Though the Burden Remained High in Low-Income and High-Income Countries. In NLM (Medline); 01 Sep 2018. [Google Scholar] [CrossRef]
- Khairullah, R.; et al. Brucellosis: Unveiling the complexities of a pervasive zoonotic disease and its global impacts. In Faculty of Veterinary Medicine, University of Tripoli; 2024. [Google Scholar] [CrossRef]
- Rubach, M.P.; Halliday, J.E.B.; Cleaveland, S.; Crump, J.A. Brucellosis in low-income and middle-income countries. 2013. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, N.R.; Szostek, J.H. 52-year-old man with arthralgias, fever, and fatigue. Mayo Clin. Proc. 2010, vol. 85(no. 6), 568–571. [Google Scholar] [CrossRef] [PubMed]
- Zou, D.; Zhou, J.; Jiang, X. Diagnosis and management of spinal tuberculosis combined with brucellosis: A case report and literature review. In Spandidos Publications; 01 Apr 2018. [Google Scholar] [CrossRef]
- Assudani, P.J.; et al. Artificial intelligence and machine learning in infectious disease diagnostics: a comprehensive review of applications, challenges, and future directions; Elsevier Inc, 01 Nov 2025. [Google Scholar] [CrossRef]
- Yu, H.; Gu, X.; Wang, D.; Wang, Z. Brucella infection and Toll-like receptors. In Frontiers Media SA; 2024. [Google Scholar] [CrossRef]
- Moens, U.; Kostenko, S.; Sveinbjørnsson, B. The role of mitogen-activated protein kinase-activated protein kinases (MAPKAPKs) in inflammation. 2013. [Google Scholar] [CrossRef]
- Ushio-Fukai, M.; Ash, D.; Nagarkoti, S.; De Chantemèle, E.J.B.; Fulton, D.J.R.; Fukai, T. Interplay between Reactive Oxygen/Reactive Nitrogen Species and Metabolism in Vascular Biology and Disease; Mary Ann Liebert Inc, 01 Jun 2021. [Google Scholar] [CrossRef]
- Manful, F.; Fordjour, E.; Subramaniam, D.; Sey, A.A.; Abbey, L.; Thomas, R. “Antioxidants and Reactive Oxygen Species: Shaping Human Health and Disease Outcomes,” Aug. 01. In Multidisciplinary Digital Publishing Institute (MDPI); 2025. [Google Scholar] [CrossRef]
- Barnes, R.P.; Fouquerel, E.; Opresko, P.L. The impact of oxidative DNA damage and stress on telomere homeostasis. In Elsevier Ireland Ltd; 01 Jan 2019. [Google Scholar] [CrossRef]
- Kam, M.L.W.; Nguyen, T.T.T.; Ngeow, J.Y.Y. Telomere biology disorders. In Nature Research; 01 Dec 2021. [Google Scholar] [CrossRef]
- Gavia-García, G.; Rosado-Pérez, J.; Arista-Ugalde, T.L.; Aguiñiga-Sánchez, I.; Santiago-Osorio, E.; Mendoza-Núñez, V.M. Telomere length and oxidative stress and its relation with metabolic syndrome components in the aging. In MDPI AG; 01 Apr 2021. [Google Scholar] [CrossRef]
- Katoto, P.D.M.C.; et al. Telomere length and outcome of treatment for pulmonary tuberculosis in a gold mining community. Sci. Rep. 2021, vol. 11(no. 1). [Google Scholar] [CrossRef]
- Seung, K.J.; Keshavjee, S.; Rich, M.L. “Multidrug-resistant tuberculosis and extensively drug-resistant tuberculosis,” Cold Spring Harb. Perspect. Med. 2015, vol. 5(no. 9). [Google Scholar] [CrossRef]
- Batt, J.; Khan, K. Responsible use of rifampin for the treatment of latent tuberculosis infection. In Canadian Medical Association; 2019. [Google Scholar] [CrossRef]
- Zhang, T.; et al. The impacts of animal agriculture on One Health—Bacterial zoonosis, antimicrobial resistance, and beyond; Elsevier B.V., 01 Jun 2024. [Google Scholar] [CrossRef]
- Olowu, I.; Zakariya, M.E.; Abdulkareem, A.A.; Okewale, O.T.; Idris, M.H.; Olayiwola, H.O. Drug Design and Delivery for Intracellular Bacteria: Emerging Paradigms. In John Wiley and Sons Inc; 01 Dec 2025. [Google Scholar] [CrossRef]
- Cavalcanti, Y.V.N.; Brelaz, M.C.A.; Neves, J.K.D.A.L.; Ferraz, J.C.; Pereira, V.R.A. Role of TNF-alpha, IFN-gamma, and IL-10 in the development of pulmonary tuberculosis. 2012. [Google Scholar] [CrossRef] [PubMed]
- Borrego, S.; et al. Oxidative stress and DNA damage in human gastric carcinoma: 8-Oxo-7’8-dihydro-2’-deoxyguanosine (8-oxo-dG) as a possible tumor marker. Int. J. Mol. Sci. 2013, vol. 14(no. 2), 3467–3486. [Google Scholar] [CrossRef]
- Lin, J.; et al. Systematic and Cell Type-Specific Telomere Length Changes in Subsets of Lymphocytes. J. Immunol. Res. 2016, vol. 2016. [Google Scholar] [CrossRef]
- Meng, F.; et al. Efflux pumps positively contribute to rifampin resistance in rpoB mutant Mycobacterium tuberculosis. Ann. Clin. Microbiol. Antimicrob. 2025, vol. 24(no. 1). [Google Scholar] [CrossRef]
- Alavi, S.M.; Alavi, L. Systematic Review Treatment of brucellosis: a systematic review of studies in recent twenty years.
- Levy, L.; Nasereddin, A.; Rav-Acha, M.; Kedmi, M.; Rund, D.; Gatt, M.E. Prolonged fever, hepatosplenomegaly, and pancytopenia in a 46-year-old woman. 2009. [Google Scholar] [CrossRef]
- Kumar, S.N.; Prasad, T.S.; Narayan, P.A.; Muruganandhan, J. Granuloma with langhans giant cells: An overview. J. Oral. Maxillofac. Pathol. 2013, vol. 17(no. 3), 420–423. [Google Scholar] [CrossRef]
- Guimarães, S.; Gomes, M.T.R.; de Araujo, A.C.V.S.C.; Ramos, K.K.S.; Oliveira, S.C. Pathogenicity and virulence of Brucella: Strategies for metabolic adaptation and immune evasion; Taylor and Francis Ltd, 2025. [Google Scholar] [CrossRef]
- Huy, T.X.N.; Nguyen, T.T.; Kim, H.; Reyes, A.W.B.; Kim, S. Brucella Phagocytosis Mediated by Pathogen-Host Interactions and Their Intracellular Survival. In MDPI; 01 Oct 2022. [Google Scholar] [CrossRef]
- Ke, Y.; Wang, Y.; Li, W.; Chen, Z. Type IV secretion system of Brucella spp. and its effectors; Frontiers Media S.A, 2015. [Google Scholar] [CrossRef]
- Pellegrini, J.M.; Gorvel, J.P.; Mémet, S. Immunosuppressive mechanisms in brucellosis in light of chronic bacterial diseases. Microorganisms 2022, vol. 10(no. 7). [Google Scholar] [CrossRef] [PubMed]
- Su, Y.; Zhang, L.; Liu, R.; Sa, R.; Zheng, L. Spatiotemporal dynamics of Brucella immune evasion across infection stages. Front. Immunol. 2026, vol. 16. [Google Scholar] [CrossRef]
- Podinovskaia, M.; Lee, W.; Caldwell, S.; Russell, D.G. Infection of macrophages with Mycobacterium tuberculosis induces global modifications to phagosomal function. Cell. Microbiol. 2013, vol. 15(no. 6), 843–859. [Google Scholar] [CrossRef]
- Queval, J. Mycobacterium tuberculosis Controls Phagosomal Acidification by Targeting CISH-Mediated Signaling. Cell Rep. 2017, vol. 20(no. 13), 3188–3198. [Google Scholar] [CrossRef] [PubMed]
- Nisa, A.; Kipper, F.C.; Panigrahy, D.; Tiwari, S.; Kupz, A.; Subbian, S. Different modalities of host cell death and their impact on Mycobacterium tuberculosis infection. In American Physiological Society; 01 Nov 2022. [Google Scholar] [CrossRef]
- Salina, G.; Makarov, V. Mycobacterium tuberculosis Dormancy: How to Fight a Hidden Danger. In MDPI; 01 Dec 2022. [Google Scholar] [CrossRef]
- Masoudian, M.; Derakhshandeh; Seno, G. Brucella melitensis and Mycobacterium tuberculosis depict overlapping gene expression patterns induced in infected THP-1 macrophages. 2015. Available online: http://rest.gene-.
- Apol, Á.D.; Winckelmann, A.A.; Duus, R.B.; Bukh, J.; Weis, N. The Role of CTLA-4 in T Cell Exhaustion in Chronic Hepatitis B Virus Infection. In MDPI; 01 May 2023. [Google Scholar] [CrossRef]
- Chaudhary, P.; et al. Oxidative stress, free radicals and antioxidants: potential crosstalk in the pathophysiology of human diseases; Frontiers Media S.A, 2023. [Google Scholar] [CrossRef]
- Kulkarni, R.; Deshpande, A.; Saxena, R.; Saxena, K. A study of serum malondialdehyde and cytokine in tuberculosis patients. J. Clin. Diagn. Res. 2013, vol. 7(no. 10), 2140–2142. [Google Scholar] [CrossRef]
- Graille, M.; Wild, P.; Sauvain, J.J.; Hemmendinger, M.; Canu, I.G.; Hopf, N.B. Urinary 8-OHDG as a biomarker for oxidative stress: A systematic literature review and meta-analysis. In MDPI AG; 01 Jun 2020. [Google Scholar] [CrossRef]
- Tyagi, P.; Dharmaraja, A.T.; Bhaskar, A.; Chakrapani, H.; Singh, A. Mycobacterium tuberculosis has diminished capacity to counteract redox stress induced by elevated levels of endogenous superoxide. Free Radic. Biol. Med. 2015, vol. 84, 344–354. [Google Scholar] [CrossRef]
- Gholami, M.; Roushan, M.H.; Mahjoub, S.; Bijani, A. How is total antioxidant status in plasma of Patients with brucellosis?
- Gusti, A.M.T.; Qusti, S.Y.; Alshammari, E.M.; Toraih, E.A.; Fawzy, M.S. Antioxidants-related superoxide dismutase (Sod), catalase (cat), glutathione peroxidase (gpx), glutathione-s-transferase (gst), and nitric oxide synthase (nos) gene variants analysis in an obese population: A preliminary case-control study. Antioxidants 2021, vol. 10(no. 4). [Google Scholar] [CrossRef] [PubMed]
- Kell, L.; Simon, A.K.; Alsaleh, G.; Cox, L.S. The central role of DNA damage in immunosenescence. In Frontiers Media SA; 2023. [Google Scholar] [CrossRef]
- Galati, A.; et al. TRF1 and TRF2 binding to telomeres is modulated by nucleosomal organization. Nucleic Acids Res. 2015, vol. 43(no. 12), 5824–5837. [Google Scholar] [CrossRef]
- Assalve; et al. Exploring the Link Between Telomeres and Mitochondria: Mechanisms and Implications in Different Cell Types. In Multidisciplinary Digital Publishing Institute (MDPI); 01 Feb 2025. [Google Scholar] [CrossRef]
- Kordinas, V.; Ioannidis, A.; Chatzipanagiotou, S. The telomere/telomerase system in chronic inflammatory diseases. Cause or effect? In MDPI AG; 03 Sep 2016. [Google Scholar] [CrossRef]
- Choi, J.; Fauce, S.R.; Effros, R.B. Reduced telomerase activity in human T lymphocytes exposed to cortisol. Brain Behav. Immun. 2008, vol. 22(no. 4), 600–605. [Google Scholar] [CrossRef]
- Johnson, A.; Rought, T.; Aronov, J.; Pokharel, P.; Chiu, A.; Nasuhidehnavi, A. The impacts of chronic infections on shaping cellular senescence. In BioMed Central Ltd; 01 Dec 2025. [Google Scholar] [CrossRef]
- Karlseder, J.; et al. The telomeric protein TRF2 binds the ATM Kinase and Can Inhibit the ATM-dependent DNA damage response. PLoS Biol. 2004, vol. 2(no. 8). [Google Scholar] [CrossRef] [PubMed]
- Aramburu, T.; Plucinsky, S.; Skordalakes, E. POT1-TPP1 telomere length regulation and disease; Elsevier B.V, 01 Jan 2020. [Google Scholar] [CrossRef]
- Maleki, M.R. Genetic Analysis of Molecular Mechanisms of Drug Resistance in Mycobacterium tuberculosis Against Four Major First-Line Anti-Tuberculosis Drugs (Isoniazid, Rifampin, Ethambutol, and Pyrazinamide). In Dove Medical Press Ltd; 2025. [Google Scholar] [CrossRef]
- Mboowa; Namaganda, C.; Ssengooba, W. “Rifampicin resistance mutations in the 81 bp RRDR of rpoB gene in Mycobacterium tuberculosis clinical isolates using Xpert® MTB/RIF in Kampala, Uganda: a retrospective study,” 2014. Available online: http://www.biomedcentral.com/1471-2334/14/481.
- Hazbón, M.H.; et al. Population genetics study of isoniazid resistance mutations and evolution of multidrug-resistant Mycobacterium tuberculosis. Antimicrob. Agents Chemother. 2006, vol. 50(no. 8), 2640–2649. [Google Scholar] [CrossRef]
- Liu, J.; et al. Mutations in efflux pump Rv1258c (Tap) cause resistance to pyrazinamide, isoniazid, and streptomycin in M. tuberculosis. Front. Microbiol. 2019, vol. 10, no. FEB. [Google Scholar] [CrossRef] [PubMed]
- Alamian, S.; Dadar, M.; Etemadi, A.; Afshar, D.; Alamian, M.M. “Antimicrobial susceptibility of Brucella spp. isolated from Iranian patients during 2016 to 2018,” 2019. Available online: http://www.oie.int/en/animal-health-in-the-.
- Marianelli, C.; Ciuchini, F.; Tarantino, M.; Pasquali, P.; Adone, R. Genetic bases of the rifampin resistance phenotype in Brucella spp. J. Clin. Microbiol. 2004, vol. 42(no. 12), 5439–5443. [Google Scholar] [CrossRef] [PubMed]
- Wachino, J.I.; Doi, Y.; Arakawa, Y. Aminoglycoside Resistance: Updates with a Focus on Acquired 16S Ribosomal RNA Methyltransferases. In W.B. Saunders; 01 Dec 2020. [Google Scholar] [CrossRef]
- Mode, S.; Ketterer, M.; Québatte, M.; Dehio, C. Antibiotic persistence of intracellular Brucella abortus. PLoS Negl. Trop. Dis. 2022, vol. 16(no. 7). [Google Scholar] [CrossRef]
- Chen, J.; Raymond, K. Roles of rifampicin in drug-drug interactions: Underlying molecular mechanisms involving the nuclear pregnane X receptor; 15 Feb 2006. [Google Scholar] [CrossRef]
- Pattanaik, K.P.; Sengupta, S.; Jit, B.P.; Kotak, R.; Sonawane, A. Host-mycobacteria conflict: Immune responses of the host vs. the mycobacteria TLR2 and TLR4 ligands and concomitant host-directed therapy. In Elsevier GmbH; 01 Nov 2022. [Google Scholar] [CrossRef]
- Sienkiewicz, M.; Sroka, K.; Binienda, A.; Jurk, D.; Fichna, J. A new face of old cells: An overview about the role of senescence and telomeres in inflammatory bowel diseases. In Elsevier Ireland Ltd; 01 Nov 2023. [Google Scholar] [CrossRef]
- Liebenberg, D.; Gordhan, B.G.; Kana, B.D. Drug resistant tuberculosis: Implications for transmission, diagnosis, and disease management; Frontiers Media S.A, 23 Sep 2022. [Google Scholar] [CrossRef]
- Akter, R.; Bishakha, A.C.; Rajib, R.; Raj, A.; Roja, M.S.; Noor, F. Targeting telomere dynamics with plant-derived compounds: Molecular strategies against aging; Elsevier B.V., 01 Jan 2025. [Google Scholar] [CrossRef]
- Voghel, G.; et al. Chronic treatment with N-acetyl-cystein delays cellular senescence in endothelial cells isolated from a subgroup of atherosclerotic patients. Mech. Ageing Dev. 2008, vol. 129(no. 5), 261–270. [Google Scholar] [CrossRef]
- Lamba, A.S.; et al. Brucellosis Masquerading as Disseminated Tuberculosis: a Clinical Case report. 2023. [Google Scholar] [CrossRef]
- Xu, N.; et al. Evaluating the efficacy of serological testing of clinical specimens collected from patients with suspected brucellosis. PLoS Negl. Trop. Dis. 2023, vol. 17(no. 2). [Google Scholar] [CrossRef]
- Vecchio; Jamil, T.; Khamesipour, F.; Heidary, M. Prevalence, diagnosis, and manifestations of brucellosis: A systematic review and meta-analysis.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




