Submitted:
29 April 2026
Posted:
30 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
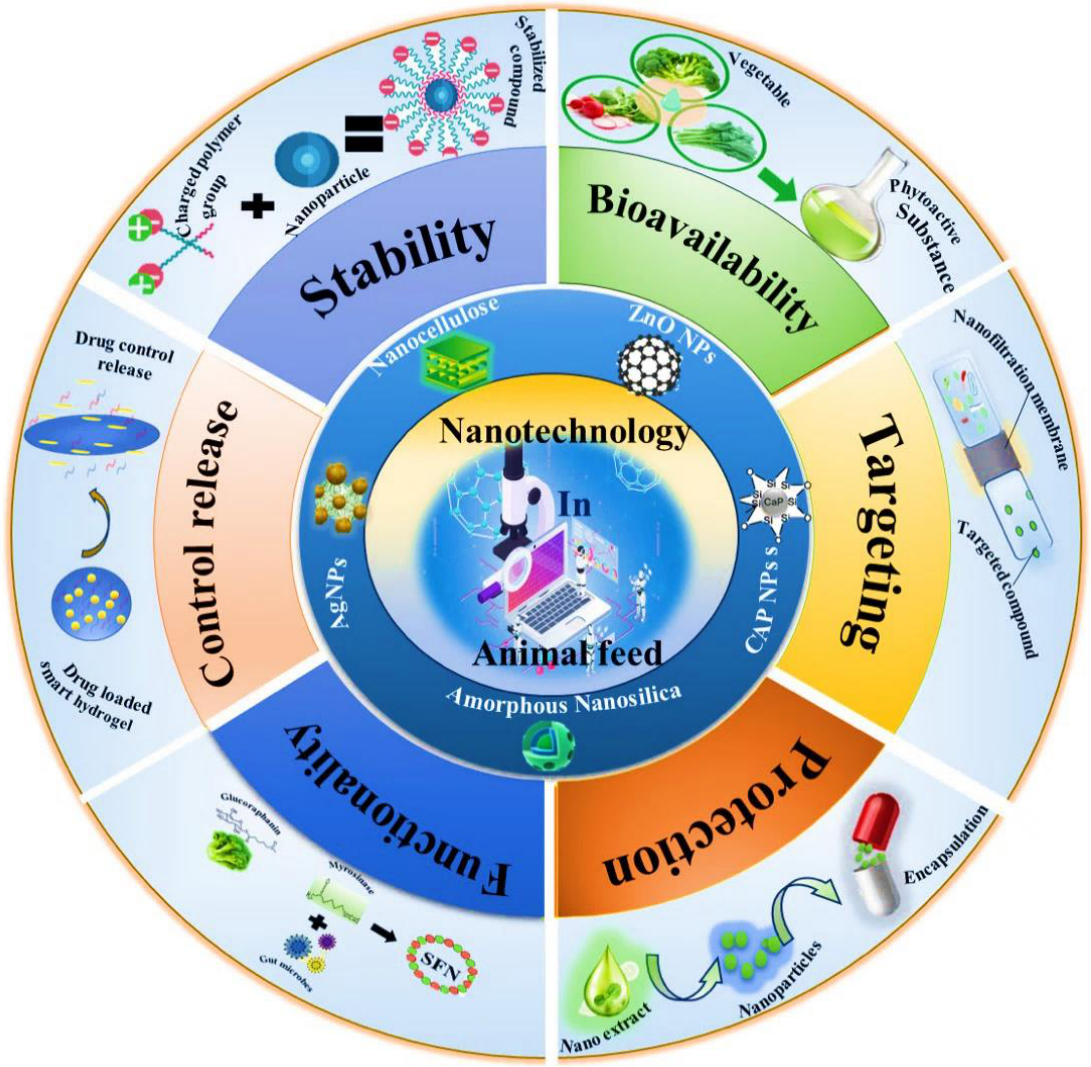
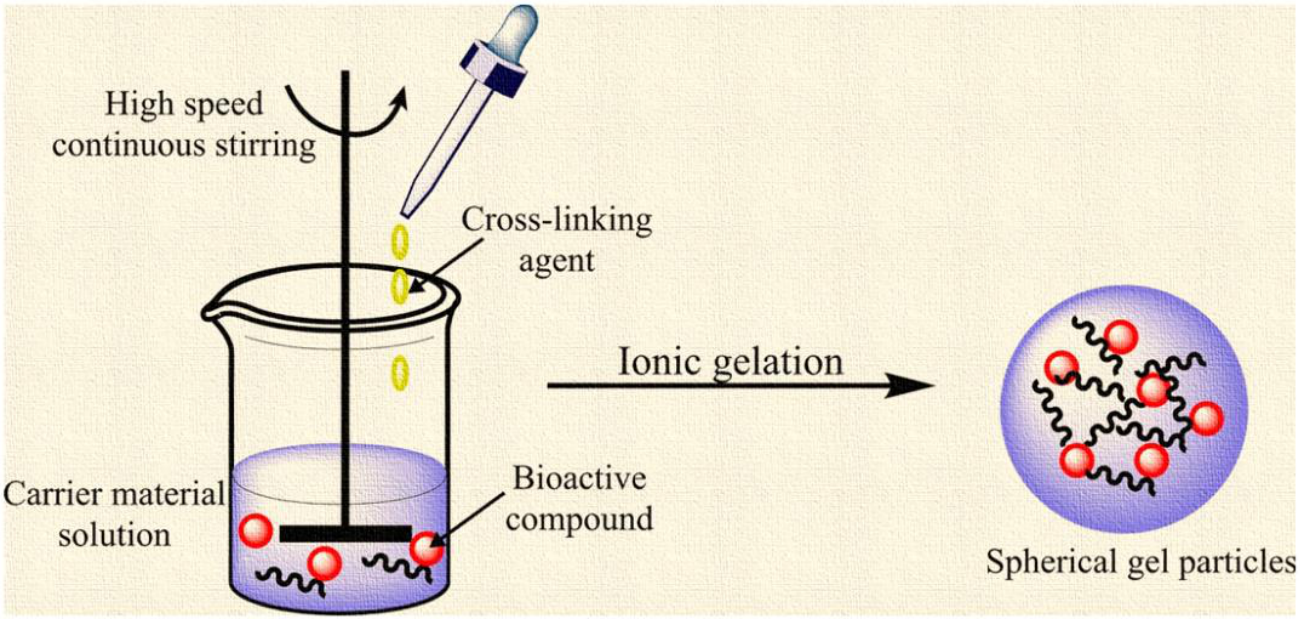
2. Ionic Gelation as an Engineering Framework for Nano-Delivery
2.1. Principle of Ionic Gelation
2.2. Application for Sulforaphane Encapsulation
2.3. Advantages of Ionic Gelation
3. Engineering Stability: Encapsulation Design and Protective Performance
3.1. Nano Extraction of Sulforaphane
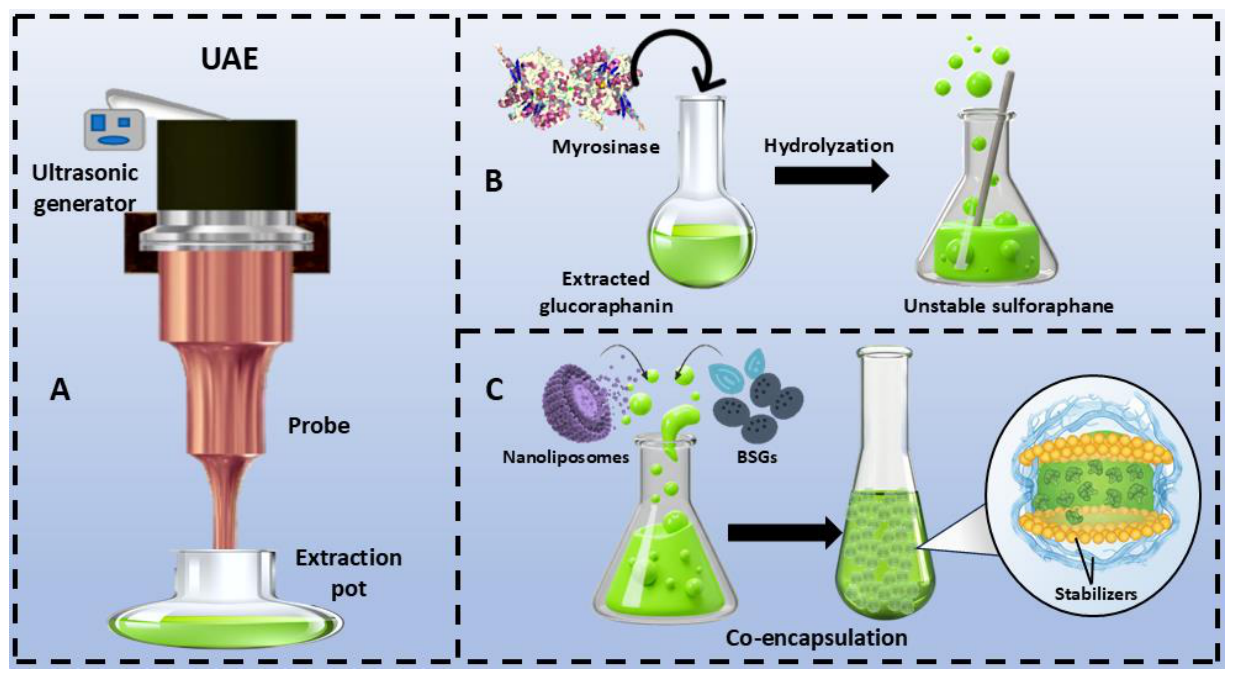
3.2. Enzymatic Hydrolysis of Glucoraphanin
3.3. Enrichment of Sulforaphane
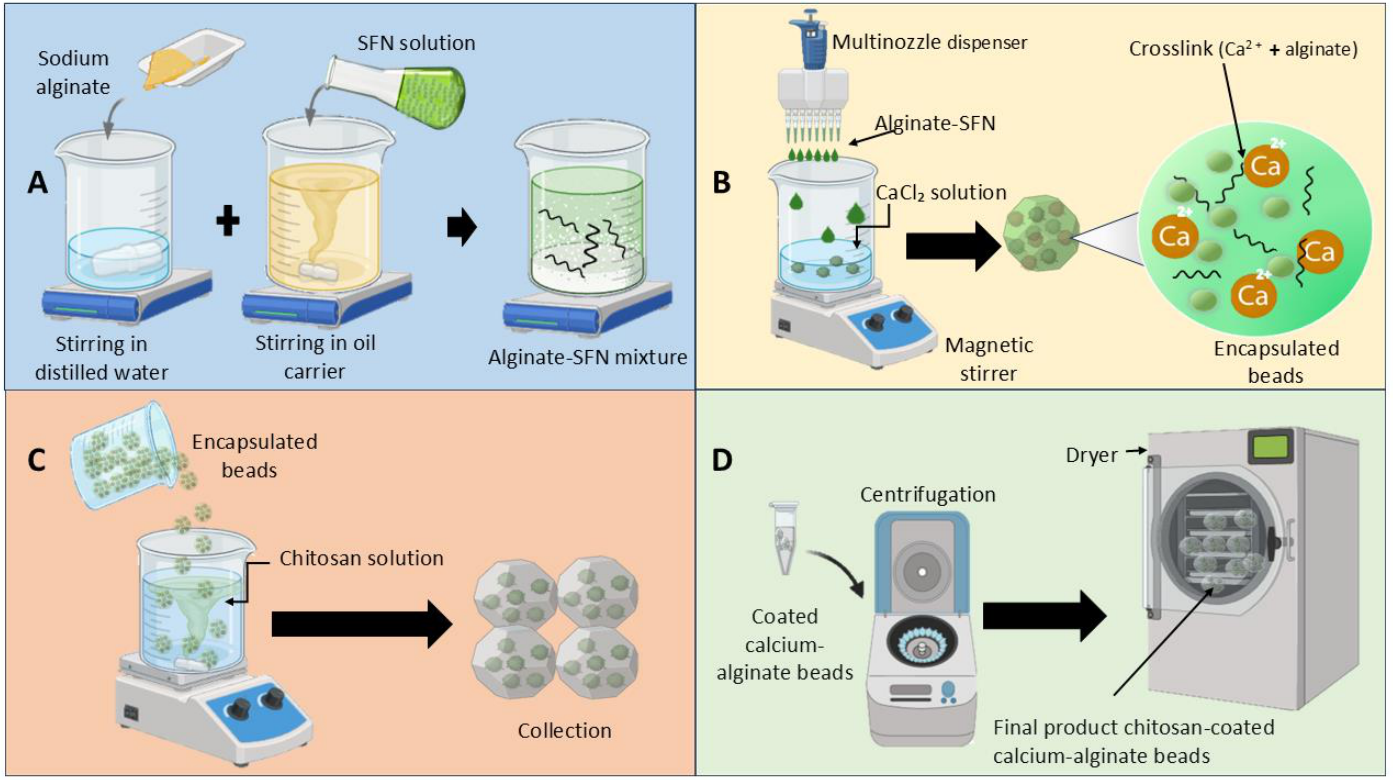
3.4. Encapsulation of Sulforaphane via Ionic Gelation
4. Measurable Efficacy in Animal Models
4.1. Quantifying Enhanced Bioavailability and Systemic Delivery
4.2. Modulation of Health and Resilience Markers
4.3. Species-Specific Delivery Challenges and Outcomes
5. Considerations and Safety Regulatory
5.1. Regulations on Nanotechnology in Feed Additives
5.2. Major Challenges for Approval of Nanotechnology in Animal Feed
5.3. Toxicity and Safety
| Parameter | Why It Matters | Regulatory Enforcement / Guidance | Reference |
|---|---|---|---|
| Particle size distribution | Determines whether material qualifies as a nanomaterial (<100 nm for 50%+ particles) | EU definition: ≥50% of particles by number must be 1–100 nm to qualify as a nanomaterial | [108,109] |
| Surface area (BET method) | High surface area influences reactivity and bioavailability | Required for nanomaterial identification in EFSA and OECD guidelines | [110] |
| Agglomeration and aggregation state | Affects particle behavior in biological systems and toxicity | EFSA requires evaluation in relevant media (e.g., feed matrix or digestive fluids) | [111] |
| Solubility and dissolution rate | Determines whether particles persist at nanoscale in the GI tract | Essential to decide whether nanospecific risk assessment is needed | [112] |
| Shape and aspect ratio | Rods, tubes, and fibers may behave differently than spheres | Required in OECD testing guidance and EFSA assessment | [113] |
| Surface charge (zeta potential) | Affects interaction with cells and proteins | Recommended by EFSA for risk and biointeraction studies | [114] |
| Impurities and chemical composition | Trace contaminants can influence toxicity and regulatory acceptance | EFSA and OECD require full elemental/chemical profiling | [113] |
| Bioavailability | Critical for feed effectiveness and systemic exposure | Often evaluated through in vitro or in vivo digestion models | [97] |
| Stability in biological media | Affects reliability of exposure and toxicity predictions | Testing in feed and GI-like conditions is recommended | [111] |
| Genotoxicity | Required for safety clearance of nanomaterials | EFSA: in vitro genotoxicity, 90-day rodent toxicity, toxicokinetic | [97,112] |
6. Future Direction and Opportunities
7. Conclusion
Declaration of competing interest
Data availability
Acknowledgments
References
- Bouranis, J.A.; Wong, C.P.; Beaver, L.M.; Uesugi, S.L.; Papenhausen, E.M.; Choi, J.; Davis, E.W., 2nd; Da Silva, A.N.; Kalengamaliro, N.; Chaudhary, R.; et al. Sulforaphane Bioavailability in Healthy Subjects Fed a Single Serving of Fresh Broccoli Microgreens. Foods 2023, 12. [Google Scholar] [CrossRef]
- Egner, P.; Chen, J.G.; Wang, J.B.; Wu, Y.; Sun, Y.; Lu, J.; Zhu, J.; Zhang, Y.H.; Chen, Y.-S.; Friesen, M.; et al. Bioavailability of Sulforaphane from Two Broccoli Sprout Beverages: Results of a Short-term, Cross-over Clinical Trial in Qidong, China. Cancer Prev. Res. 2011, 4, 384–395. [Google Scholar] [CrossRef]
- Clarke, J.; Hsu, A.; Riedl, K.; Bella, D.; Schwartz, S.; Stevens, J.; Ho, E. Bioavailability and inter-conversion of sulforaphane and erucin in human subjects consuming broccoli sprouts or broccoli supplement in a cross-over study design. Pharmacol. Res. 2011, 64 5, 456–463. [Google Scholar] [CrossRef]
- Abukhabta, S.; Ghawi, S.K.; Karatzas, K.; Charalampopoulos, D.; McDougall, G.; Allwood, W.; Verrall, S.; Lavery, S.; Latimer, C.; Pourshahidi, L.; et al. Sulforaphane-enriched extracts from glucoraphanin-rich broccoli exert antimicrobial activity against gut pathogens in vitro and innovative cooking methods increase in vivo intestinal delivery of sulforaphane. Eur. J. Nutr. 2020, 60, 1263–1276. [Google Scholar] [CrossRef] [PubMed]
- Bouranis, J.; Beaver, L.; Wong, C.; Choi, J.; Hamer, S.; Davis, E.; Brown, K.; Jiang, D.; Sharpton, T.; Stevens, J.; et al. Sulforaphane and Sulforaphane-Nitrile Metabolism in Humans Following Broccoli Sprout Consumption: Inter-individual Variation, Association with Gut Microbiome Composition, and Differential Bioactivity. Mol. Nutr. Food Res. 2023. [Google Scholar] [CrossRef] [PubMed]
- Fahey, J.; Fahey, J.; Holtzclaw, W.; Wehage, S.; Wade, K.; Stephenson, K.; Talalay, P.; Talalay, P. Sulforaphane Bioavailability from Glucoraphanin-Rich Broccoli: Control by Active Endogenous Myrosinase. PLoS ONE 2015, 10. [Google Scholar] [CrossRef] [PubMed]
- Yepes-Molina, L.; Carvajal, M. Nanoencapsulation of sulforaphane in broccoli membrane vesicles and their in vitro antiproliferative activity. Pharm. Biol. 2021, 59, 1490–1504. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhao, X.; Liu, Y.; Yang, X. Sulforaphane and ophthalmic diseases. Food Sci. Nutr. 2024, 12, 5296–5311. [Google Scholar] [CrossRef]
- Van Steenwijk, H.; Winter, E.; Knaven, E.; Brouwers, J.; Van Baardwijk, M.; Van Dalum, J.; Luijendijk, T.; Van Osch, F.; Troost, F.; Bast, A.; et al. The beneficial effect of sulforaphane on platelet responsiveness during caloric load: a single-intake, double-blind, placebo-controlled, crossover trial in healthy participants. Front. Nutr. 2023, 10. [Google Scholar] [CrossRef]
- Somers, D.; Kushner, D.; McKinnis, A.; Mehmedovic, D.; Flame, R.; Arnold, T. Epigenetic weapons in plant-herbivore interactions: Sulforaphane disrupts histone deacetylases, gene expression, and larval development in Spodoptera exigua while the specialist feeder Trichoplusia ni is largely resistant to these effects. PLoS ONE 2023, 18. [Google Scholar] [CrossRef]
- Detsi, A.; Kavetsou, E.; Kostopoulou, I.; Pitterou, I.; Pontillo, A.R.N.; Tzani, A.; Christodoulou, P.; Siliachli, A.; Zoumpoulakis, P. Nanosystems for the Encapsulation of Natural Products: The Case of Chitosan Biopolymer as a Matrix. Pharmaceutics 2020, 12, 669. [Google Scholar] [CrossRef]
- Zambrano, V.; Bustos, R.; Mahn, A. Insights about stabilization of sulforaphane through microencapsulation. Heliyon 2019, 5. [Google Scholar] [CrossRef] [PubMed]
- Šantić, A.; Brinkkötter, M.; Portada, T.; Frkanec, L.; Cremer, C.; Schönhoff, M.; Moguš-Milanković, A. Supramolecular ionogels prepared with bis(amino alcohol)oxamides as gelators: ionic transport and mechanical properties. RSC Adv. 2020, 10, 17070–17078. [Google Scholar] [CrossRef] [PubMed]
- De Azevedo, E.S.; Noreña, C.Z. External ionic gelation as a tool for the encapsulation and stability of betacyanins from Bougainvillea glabra bracts extract in a food model. J. Food Process. Preserv. 2021. [Google Scholar] [CrossRef]
- Elzayat, A.; Adam-Cervera, I.; Albus, M.; Cháfer, A.; Badia, J.; Pérez-Pla, F.; Muñoz-Espí, R. Polysaccharide/Silica Microcapsules Prepared via Ionic Gelation Combined with Spray Drying: Application in the Release of Hydrophilic Substances and Catalysis. Polymers 2023, 15. [Google Scholar] [CrossRef]
- Farahmand, A.; Ghorani, B.; Emadzadeh, B.; Sarabi-Jamab, M.; Emadzadeh, M.; Modiri, A.; Tucker, N. Millifluidic-assisted ionic gelation technique for encapsulation of probiotics in double-layered polysaccharide structure. Food Res. Int. 2022, 160, 111699. [Google Scholar] [CrossRef] [PubMed]
- Paoletti, S.; Donati, I. Comparative Insights into the Fundamental Steps Underlying Gelation of Plant and Algal Ionic Polysaccharides: Pectate and Alginate. Gels 2022, 8, 784. [Google Scholar] [CrossRef]
- Yusefi, M.; Shameli, K.; Kia, P.; Hamrayev, H. Facile Fabrication of Polysaccharide Nanocomposites Using Ionic Gelation Method. J. Res. Nanosci. Nanotechnol. 2021. [Google Scholar] [CrossRef]
- Shard, P.; Sharma, D.; Bhatia, A. Optimization and effects of physico-chemical parameters on synthesis of Chitosan Nanoparticles by Ionic Gelation Technique. Int. J. Drug Deliv. 2014, 6, 58–63. [Google Scholar]
- Van Bavel, N.; Issler, T.; Pang, L.; Anikovskiy, M.; Prenner, E. A Simple Method for Synthesis of Chitosan Nanoparticles with Ionic Gelation and Homogenization. Molecules 2023, 28. [Google Scholar] [CrossRef]
- Sharma, R. Formulation and Characterization of Atenolol-loaded Gellan Gum Nanoparticles. Indian J. Pharm. Sci. 2021. [Google Scholar] [CrossRef]
- Yudhowijoyo, A.; Rafati, R.; Haddad, A.S.; Pokrajac, D.; Manzari, M. Developing Nanocomposite Gels from Biopolymers for Leakage Control in Oil and Gas Wells. Day 3 Thu, September 05, 2019 2019. [Google Scholar] [CrossRef]
- Vila-Sanjurjo, C.; Hembach, L.; Netzer, J.; Remuñán-López, C.; Vila-Sanjurjo, A.; Goycoolea, F. Covalently and ionically, dually crosslinked chitosan nanoparticles block quorum sensing and affect bacterial cell growth on a cell-density dependent manner. J. Colloid Interface Sci. 2020, 578, 171–183. [Google Scholar] [CrossRef]
- Dong, Y.; Ng, W.; Shen, S.; Kim, S.; Tan, R. Scalable ionic gelation synthesis of chitosan nanoparticles for drug delivery in static mixers. Carbohydr. Polym. 2013, 94 2, 940–945. [Google Scholar] [CrossRef]
- Yammine, J.; Chihib, N.-E.; Gharsallaoui, A.; Ismail, A.; Karam, L. Advances in essential oils encapsulation: development, characterization and release mechanisms. Polym. Bull. 2024, 81, 3837–3882. [Google Scholar] [CrossRef]
- da Silva, B.D.; Bernardes, P.C.; Pinheiro, P.F.; Fantuzzi, E.; Roberto, C.D. Chemical composition, extraction sources and action mechanisms of essential oils: Natural preservative and limitations of use in meat products. Meat Sci. 2021, 176, 108463. [Google Scholar] [CrossRef]
- Zambrano, V.; Bustos, R.; Mahn, A. Insights about stabilization of sulforaphane through microencapsulation. Heliyon 2019, 5, e02951. [Google Scholar] [CrossRef]
- García-Saldaña, J.; Campas-Baypoli, O.; López-Cervantes, J.; Sánchez-Machado, D.; Cantú-Soto, E.; Rodríguez-Ramirez, R. Microencapsulation of sulforaphane from broccoli seed extracts by gelatin/gum arabic and gelatin/pectin complexes. Food Chem. 2016, 201, 94–100. [Google Scholar] [CrossRef]
- Wu, Y.; Zou, L.; Mao, J.-W.; Huang, J.; Liu, S. Stability and encapsulation efficiency of sulforaphane microencapsulated by spray drying. Carbohydr. Polym. 2014, 102, 497–503. [Google Scholar] [CrossRef] [PubMed]
- Azarashkan, Z.; Motamedzadegan, A.; Ghorbani-Hasansaraei, A.; Rahaiee, S.; Biparva, P. Improvement of the Stability and Release of Sulforaphane-enriched Broccoli Sprout Extract Nanoliposomes by Co-encapsulation into Basil Seed Gum. Food Bioprocess Technol. 2022, 15, 1573–1587. [Google Scholar] [CrossRef]
- Pai, S.; Rizvi, S.; D'Souza, M. Development of sulforaphane-encapsulated microspheres for cancer epigenetic therapy. Int. J. Pharm. 2010, 386 1-2, 114–121. [Google Scholar] [CrossRef] [PubMed]
- Yepes Molina, L.; Carvajal, M. Nanoencapsulation of sulforaphane in broccoli membrane vesicles and their in vitro antiproliferative activity. Pharm. Biol. 2021, 59, 1490–1504. [Google Scholar] [CrossRef]
- Liu, Y.; Zhang, D.; Li, X.; Xiao, J.; Guo, L. Enhancement of ultrasound-assisted extraction of sulforaphane from broccoli seeds via the application of microwave pretreatment. Ultrason. Sonochemistry 2022, 87. [Google Scholar] [CrossRef]
- Choi, A.R.; Lee, G.-S.; Chae, H. Effects of ultrasonication on the analysis of sulforaphane content in vegetables. Korea Acad.-Ind. Coop. Soc. 2008, 9, 794–799. [Google Scholar] [CrossRef]
- Liang, H.; Li, C.; Yuan, Q.; Vriesekoop, F. Separation and Purification of Sulforaphane from Broccoli Seeds by Solid Phase Extraction and Preparative High-Performance Liquid Chromatography. J. Agric. Food Chem. 2007, 55, 8047–8053. [Google Scholar] [CrossRef] [PubMed]
- Hafezian, S.M.; Azizi, S.N.; Biparva, P.; Bekhradnia, A. High-efficiency purification of sulforaphane from the broccoli extract by nanostructured SBA-15 silica using solid-phase extraction method. J. Chromatogr. B Anal. Technol. BioMed Life Sci. 2019, 1108, 1–10. [Google Scholar] [CrossRef]
- Wang, X.; Song, F.; Yang, Y. Orthogonal experimental design optimizes the compound extraction process of sulforaphane from broccoli seeds. 2019. [Google Scholar]
- Wang, L.; Jiang, H.; Qiu, Y.; Dong, Y.; Hamouda, H.; Balah, M.; Mao, X. Biochemical Characterization of a Novel Myrosinase Rmyr from Rahnella inusitata for High-Level Preparation of Sulforaphene and Sulforaphane. J. Agric. Food Chem. 2022. [Google Scholar] [CrossRef]
- Wang, L.; Hamouda, H.; Dong, Y.; Jiang, H.; Quan, Y.; Chen, Y.; Liu, Y.; Wang, J.; Balah, M.; Mao, X. High-level and reusable preparation of sulforaphane by yeast cells expressing myrosinase. Food Chem. X 2023, 18, 100668. [Google Scholar] [CrossRef]
- Sangkret, S.; Pongmalai, P.; Devahastin, S.; Chiewchan, N. Enhanced production of sulforaphane by exogenous glucoraphanin hydrolysis catalyzed by myrosinase extracted from Chinese flowering cabbage (Brassica rapa var. parachinensis). Sci. Rep. 2019, 9. [Google Scholar] [CrossRef] [PubMed]
- Panda, D.; Manickam, S. Cavitation technology—The future of greener extraction method: A review on the extraction of natural products and process intensification mechanism and perspectives. Appl. Sci. 2019, 9, 766. [Google Scholar] [CrossRef]
- Fahey, J.W.; Holtzclaw, W.D.; Wehage, S.L.; Wade, K.L.; Stephenson, K.K.; Talalay, P. Sulforaphane Bioavailability from Glucoraphanin-Rich Broccoli: Control by Active Endogenous Myrosinase. PLoS ONE 2015, 10, e0140963. [Google Scholar] [CrossRef] [PubMed]
- Azarashkan, Z.; Farahani, S.; Abedinia, A.; Akbarmivehie, M.; Motamedzadegan, A.; Heidarbeigi, J.; Hayaloğlu, A.A. Co-encapsulation of broccoli sprout extract nanoliposomes into basil seed gum: effects on in vitro antioxidant, antibacterial and anti-Listeria activities in ricotta cheese. Int. J. Food Microbiol. 2022, 376, 109761. [Google Scholar] [CrossRef]
- Zambrano, V.; Bustos, R.; Arozarena, Y.; Mahn, A. Optimization of a Microencapsulation Process Using Oil-in-Water (O/W) Emulsion to Increase Thermal Stability of Sulforaphane. Foods 2023, 12. [Google Scholar] [CrossRef] [PubMed]
- Di Santo, M.; Antoni, C.L.D.; Rubio, A.D.; Alaimo, A.; Pérez, O. Chitosan-tripolyphosphate nanoparticles designed to encapsulate polyphenolic compounds for biomedical and pharmaceutical applications - A review. Biomed. Pharmacother. Biomed. Pharmacother. 2021, 142, 111970. [Google Scholar] [CrossRef]
- Malagutti, E.; Pinho, S.G.B.; Thomazini, M.; Tapia-Blácido, D.; Tosi, M.M. Optimizing Encapsulation of Active Compounds of Carrot By-Product in TPP-Chitosomes. Foods 2024, 13. [Google Scholar] [CrossRef]
- Kim, E.; Baek, Y.; Yoo, H.; Lee, J.-S.; Lee, H. Chitosan-Tripolyphosphate Nanoparticles Prepared by Ionic Gelation Improve the Antioxidant Activities of Astaxanthin in the In Vitro and In Vivo Model. Antioxidants 2022, 11. [Google Scholar] [CrossRef]
- Gadziński, P.; Froelich, A.; Jadach, B.; Wojtyłko, M.; Tatarek, A.; Białek, A.; Krysztofiak, J.; Gackowski, M.; Otto, F.; Osmałek, T. Ionotropic Gelation and Chemical Crosslinking as Methods for Fabrication of Modified-Release Gellan Gum-Based Drug Delivery Systems. Pharmaceutics 2022, 15, 108. [Google Scholar] [CrossRef]
- Guerra-Valle, M.; Petzold, G.; Orellana-Palma, P. Optimization of Encapsulation by Ionic Gelation Technique of Cryoconcentrated Solution: A Response Surface Methodology and Evaluation of Physicochemical Characteristics Study. Polymers 2022, 14, 1031. [Google Scholar] [CrossRef] [PubMed]
- Meyer-Déru, L.; David, G.; Auvergne, R. Chitosan chemistry review for living organisms encapsulation. Carbohydr. Polym. 2022, 295, 119877. [Google Scholar] [CrossRef]
- Öner, B.; Köprüalan, Ö.; Sumer Okkali, G.; Sarıkahya, N.; Sahin-Nadeem, H.; KaymakErtekin, F. Isolation, Purification, Structural Characterization, and Encapsulation of Sulforaphane from Cabbage Seeds Using the Ionic Gelation Method. Food Bioprocess Technol. 2025, 18, 9891–9911. [Google Scholar] [CrossRef]
- Alizadeh, H.; Ahmadi, M.; Heydari Shayesteh, O. On chip synthesis of a pH sensitive gefitinib anticancer drug nanocarrier based on chitosan/alginate natural polymers. Sci. Rep. 2024, 14, 772. [Google Scholar] [CrossRef]
- Zhao, L.; Yang, G.; Shi, Y.; Su, C.; Chang, J. Co-delivery of Gefitinib and chloroquine by chitosan nanoparticles for overcoming the drug acquired resistance. J. Nanobiotechnology 2015, 13, 57. [Google Scholar] [CrossRef] [PubMed]
- Lai, K.-C.; Chueh, F.-S.; Hsiao, Y.-T.; Cheng, Z.-Y.; Lien, J.-C.; Liu, K.-C.; Peng, S.-F.; Chung, J.-G. Gefitinib and curcumin-loaded nanoparticles enhance cell apoptosis in human oral cancer SAS cells in vitro and inhibit SAS cell xenografted tumor in vivo. Toxicol. Appl. Pharmacol. 2019, 382, 114734. [Google Scholar] [CrossRef]
- Jin, M.; Li, S.; Wu, Y.; Li, D.; Han, Y. Construction of chitosan/alginate nano-drug delivery system for improving dextran sodium sulfate-induced colitis in mice. Nanomaterials 2021, 11, 1884. [Google Scholar] [CrossRef]
- Li, P.; Dai, Y.-N.; Zhang, J.-P.; Wang, A.-Q.; Wei, Q. Chitosan-alginate nanoparticles as a novel drug delivery system for nifedipine. Int. J. Biomed. Sci. IJBS 2008, 4, 221. [Google Scholar] [CrossRef]
- Zhang, T.; Bao, J.; Zhang, M.; Ge, Y.; Wei, J.; Li, Y.; Wang, W.; Li, M.; Jin, Y. Chemo-photodynamic therapy by pulmonary delivery of gefitinib nanoparticles and 5-aminolevulinic acid for treatment of primary lung cancer of rats. Photodiagnosis Photodyn. Ther. 2020, 31, 101807. [Google Scholar] [CrossRef] [PubMed]
- Van Bavel, N.; Issler, T.; Pang, L.; Anikovskiy, M.; Prenner, E.J. A Simple Method for Synthesis of Chitosan Nanoparticles with Ionic Gelation and Homogenization. Molecules 2023, 28, 4328. [Google Scholar] [CrossRef]
- Chen, S.; Osaka, A.; Hayakawa, S.; Tsuru, K.; Fujii, E.; Kawabata, K. Microstructure evolution in Stöber-type silica nanoparticles and their in vitro apatite deposition. J. Sol.-Gel Sci. Technol. 2008, 48, 322–335. [Google Scholar] [CrossRef]
- Chen, S.; Zhang, H.; Chinnathambi, S.; Hanagata, N. Synthesis of novel chitosan–silica/CpG oligodeoxynucleotide nanohybrids with enhanced delivery efficiency. Mater. Sci. Eng. C 2013, 33, 3382–3388. [Google Scholar] [CrossRef] [PubMed]
- Sacco, P.; Brun, F.; Donati, I.; Porrelli, D.; Paoletti, S.; Turco, G. On the Correlation between the Microscopic Structure and Properties of Phosphate-Cross-Linked Chitosan Gels. ACS Appl. Mater. Interfaces 2018, 10, 10761–10770. [Google Scholar] [CrossRef]
- Iswanti, F.C.; Nurulita, I.; Djauzi, S.; Sadikin, M.; Witarto, A.B.; Yamazaki, T. Preparation, characterization, and evaluation of chitosan-based nanoparticles as CpG ODN carriers. Biotechnol. Biotechnol. Equip. 2019, 33, 390–396. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, Y.; Li, X.; Luo, L.; Ning, F.; Liu, T.; Li, J. Lactobacillus plantarum increase the sulforaphane formation efficiency via microbial-targeted delivery system in vivo. Food Biosci. 2024, 62, 105544. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, W.; Zhao, Y.; Peng, R.; Zhang, Z.; Xu, Z.; Simal-Gandara, J.; Yang, H.; Deng, J. Bioactive sulforaphane from cruciferous vegetables: advances in biosynthesis, metabolism, bioavailability, delivery, health benefits, and applications. Crit. Rev. Food Sci. Nutr. 2025, 65, 3027–3047. [Google Scholar] [CrossRef]
- Mangla, B.; Javed, S.; Sultan, M.H.; Kumar, P.; Kohli, K.; Najmi, A.; Alhazmi, H.A.; Al Bratty, M.; Ahsan, W. Sulforaphane: A review of its therapeutic potentials, advances in its nanodelivery, recent patents, and clinical trials. Phyther. Res. 2021, 35, 5440–5458. [Google Scholar] [CrossRef] [PubMed]
- Cascajosa-Lira, A.; Prieto, A.I.; Pichardo, S.; Jos, A.; Cameán, A.M. Protective effects of sulforaphane against toxic substances and contaminants: A systematic review. Phytomedicine 2024, 130, 155731. [Google Scholar] [CrossRef] [PubMed]
- Houghton, C.A. Sulforaphane: Its "Coming of Age" as a Clinically Relevant Nutraceutical in the Prevention and Treatment of Chronic Disease. Oxid. Med. Cell Longev. 2019, 2019, 2716870. [Google Scholar] [CrossRef]
- Shao, D.; Gao, Z.; Zhao, Y.; Fan, M.; Zhao, X.; Wei, Q.; Pan, M.; Ma, B. Sulforaphane Suppresses H(2)O(2)-Induced Oxidative Stress and Apoptosis via the Activation of AMPK/NFE2L2 Signaling Pathway in Goat Mammary Epithelial Cells. Int. J. Mol. Sci. 2023, 24. [Google Scholar] [CrossRef]
- Taguchi, K.; Yamamoto, M. The KEAP1–NRF2 System in Cancer. Front. Oncol. 2017, 7–2017. [Google Scholar] [CrossRef]
- Ahmed, S.M.U.; Luo, L.; Namani, A.; Wang, X.J.; Tang, X. Nrf2 signaling pathway: Pivotal roles in inflammation. Biochim. Et. Biophys. Acta (BBA) -Mol. Basis Dis. 2017, 1863, 585–597. [Google Scholar] [CrossRef]
- Ruhee, R.T.; Suzuki, K. The integrative role of sulforaphane in preventing inflammation, oxidative stress and fatigue: A review of a potential protective phytochemical. Antioxidants 2020, 9, 521. [Google Scholar] [CrossRef]
- Wei, L.-Y.; Zhang, J.-K.; Zheng, L.; Chen, Y. The functional role of sulforaphane in intestinal inflammation: a review. Food Funct. 2022, 13, 514–529. [Google Scholar] [CrossRef]
- Shao, D.; Gao, Z.; Zhao, Y.; Fan, M.; Zhao, X.; Wei, Q.; Pan, M.; Ma, B. Sulforaphane Suppresses H2O2-Induced Oxidative Stress and Apoptosis via the Activation of AMPK/NFE2L2 Signaling Pathway in Goat Mammary Epithelial Cells. Int. J. Mol. Sci. 2023, 24, 1070. [Google Scholar] [CrossRef]
- Liu, Y.; Li, T.; Liao, Y.; Chen, J.; Sheng, Y.; Zhao, Y.; He, Z.; Tang, H.; Wang, Y.; Bian, Z.; et al. Novel therapeutic approach in periodontitis: Sulforaphane attenuates disease progression via Nrf2-mediated antioxidant defense. Int. Immunopharmacol. 2025, 161, 115002. [Google Scholar] [CrossRef] [PubMed]
- Danish Rizvi, S.M.; Abu Lila, A.S.; Moin, A.; Khafagy, E.S.; Rajab, A.A.H.; Hegazy, W.A.H.; Bendary, M.M. Sulforaphane Is Not Only a Food Supplement: It Diminishes the Intracellular Survival and Colonization of Salmonella enterica. ACS Omega 2025, 10, 2969–2977. [Google Scholar] [CrossRef]
- Zhang, R.; Neuhoff, C.; Yang, Q.; Cinar, M.U.; Uddin, M.J.; Tholen, E.; Schellander, K.; Tesfaye, D. Sulforaphane Enhanced Proliferation of Porcine Satellite Cells via Epigenetic Augmentation of SMAD7. Animals 2022, 12, 1365. [Google Scholar] [CrossRef]
- Zhang, L.; Wang, S.; Zhang, Y.; Li, F.; Yu, C. Sulforaphane alleviates lung ischemia-reperfusion injury through activating Nrf-2/HO-1 signaling. Exp. Ther. Med. 2023, 25, 265. [Google Scholar] [CrossRef]
- Thiruvengadam, M.; Venkidasamy, B.; Subramanian, U.; Samynathan, R.; Shariati, M.A.; Rebezov, M.; Girish, S.; Thangavel, S.; Dhanapal, A.R.; Fedoseeva, N.; et al. Bioactive Compounds in Oxidative Stress-Mediated Diseases: Targeting the NRF2/ARE Signaling Pathway and Epigenetic Regulation. Antioxidants 2021, 10. [Google Scholar] [CrossRef]
- Liu, L.; Chen, D.; Yu, B.; Luo, Y.; Huang, Z.; Zheng, P.; Mao, X.; Yu, J.; Luo, J.; Yan, H.; et al. Influences of Selenium-Enriched Yeast on Growth Performance, Immune Function, and Antioxidant Capacity in Weaned Pigs Exposure to Oxidative Stress. BioMed Res. Int. 2021, 2021. [Google Scholar] [CrossRef]
- Merz, T.; Denoix, N.; Wigger, D.; Waller, C.; Wepler, M.; Vettorazzi, S.; Tuckermann, J.; Radermacher, P.; McCook, O. The Role of Glucocorticoid Receptor and Oxytocin Receptor in the Septic Heart in a Clinically Relevant, Resuscitated Porcine Model With Underlying Atherosclerosis. Front. Endocrinol. 2020, 11. [Google Scholar] [CrossRef] [PubMed]
- Wohlert, A.; Pézsa, N.P.; Móritz, A.; Jerzsele, Á.; Farkas, O.; Pászti-Gere, E. Luteolin and Chrysin Could Prevent E. coli Lipopolysaccharide-Ochratoxin A Combination-Caused Inflammation and Oxidative Stress in In Vitro Porcine Intestinal Model. Anim. An. Open Access J. From MDPI 2022, 12. [Google Scholar] [CrossRef] [PubMed]
- Hu, R.; He, Z.; Liu, M.; Tan, J.; Zhang, H.; Hou, D.; He, J.; Wu, S. Dietary protocatechuic acid ameliorates inflammation and up-regulates intestinal tight junction proteins by modulating gut microbiota in LPS-challenged piglets. J. Anim. Sci. Biotechnol. 2020, 11. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.; Kim, H.-D.; Park, S.-Y.; Pan, J.G.; Kim, J.H.; Yum, D. Dietary Supplementation With a Bacillus Superoxide Dismutase Protects Against &ggr;-Radiation-induced Oxidative Stress and Ameliorates Dextran Sulphate Sodium-induced Ulcerative Colitis in Mice. J. Crohn's Colitis 2018, 12, 860. [Google Scholar] [CrossRef]
- Zhu, W.; Cremonini, E.; Mastaloudis, A.F.; Mitchell, A.E.; Bornhorst, G.M.; Oteiza, P.I. Optimization of sulforaphane bioavailability from a glucoraphanin-rich broccoli seed extract in a model of dynamic gastric digestion and absorption by Caco-2 cell monolayers. Food Funct. 2025, 16, 314–328. [Google Scholar] [CrossRef]
- Wu YuanFeng, W.Y.; Mao JianWei, M.J.; You YuRu, Y.Y.; Liu ShiWang, L.S. Study on degradation kinetics of sulforaphane in broccoli extract. 2014. [Google Scholar]
- Wang, Z.-J.; Jiang, Q.; Li, P.; Shi, P.; Liu, C.; Wang, W.; Huang, K.; Yin, Y.; Huang, P. The Water Extract of Ampelopsis grossedentata Alleviates Oxidative Stress and Intestinal Inflammation. Antioxidants 2023, 12. [Google Scholar] [CrossRef]
- Yuan, Z.; Huang, L.; Hu, Z.; Deng, J.; Duan, Y.; Jiang, Q.; Tan, B.e.; Ma, X.; Zhang, C.; Tang, X. Lactobacillus fermentum ZC529 Protects Intestinal Epithelial Barrier Integrity by Activating the Keap1-Nrf2 Signaling Pathway and Inhibiting the NF-κB Signaling Pathway. Antioxidants 2025, 14, 732. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Guo, K. Tannic acid alleviates ETEC K88-induced intestinal damage through regulating the p62-keap1-Nrf2 and TLR4-NF-κB-NLRP3 pathway in IPEC-J2 cells. J. Sci. Food Agric. 2024, 104, 5186–5196. [Google Scholar] [CrossRef]
- Kim, M.J.; Zhang, T.; Kim, K.-N.; Bae, G.-W.; Yoon, S.; Yue, Y.; Wu, X.; Park, S. Alleviation of Cognitive and Physical Fatigue with Enzymatic Porcine Placenta Hydrolysate Intake through Reducing Oxidative Stress and Inflammation in Intensely Exercised Rats. Biology 2022, 11. [Google Scholar] [CrossRef]
- Liu, J.; Chandaka, G.; Zhang, R.; Parfenova, H. Acute antioxidant and cytoprotective effects of sulforaphane in brain endothelial cells and astrocytes during inflammation and excitotoxicity. Pharmacol. Res. Perspect. 2020, 8. [Google Scholar] [CrossRef]
- Amenta, V.; Aschberger, K.; Arena, M.; Bouwmeester, H.; Moniz, F.B.; Brandhoff, P.; Gottardo, S.; Marvin, H.; Mech, A.; Pesudo, L.Q.; et al. Regulatory aspects of nanotechnology in the agri/feed/food sector in EU and non-EU countries. Regul. Toxicol. Pharmacol. RTP 2015, 73 1, 463–476. [Google Scholar] [CrossRef]
- Gupta, N. 2 Nanotechnology in agrifood sector: Ethical, regulatory, and governance landscape in EU. In Ethics in Nanotechnology; 2021. [Google Scholar] [CrossRef]
- Aschberger, K.; Gottardo, S.; Amenta, V.; Arena, M.; Moniz, F.; Bouwmeester, H.; Brandhoff, P.; Mech, A.; Pesudo, L.; Rauscher, H.; et al. Nanomaterials in Food - Current and Future Applications and Regulatory Aspects. J. Phys. Conf. Ser. 2015, 617. [Google Scholar] [CrossRef]
- Sodano, V. Nanotechnology and Food System: Assessing the European Union Regulatory System. Eur. J. Dev. Stud. 2023. [Google Scholar] [CrossRef]
- Allan, J.; Belz, S.; Hoeveler, A.; Hugas, M.; Okuda, H.; Patri, A.; Rauscher, H.; Silva, P.; Slikker, W.; Sokull-Kluettgen, B.; et al. Regulatory landscape of nanotechnology and nanoplastics from a global perspective. Regul. Toxicol. Pharmacol. 2021, 122. [Google Scholar] [CrossRef]
- Murashov, V.; Schulte, P.; Geraci, C.; Howard, J. Regulatory approaches to worker protection in nanotechnology industry in the USA and European union. Ind. Health 2011, 49 3, 280–296. [Google Scholar] [CrossRef] [PubMed]
- Sauer, U. Eating Nanomaterials: Cruelty-free and Safe? The EFSA Guidance on Risk Assessment of Nanomaterials in Food and Feed. Altern. To Lab. Anim. 2011, 39, 567–575. [Google Scholar] [CrossRef]
- Xavier, M.; Parente, I.; Rodrigues, P.; Cerqueira, M.; Pastrana, L.; Gonçalves, C. Safety and fate of nanomaterials in food: The role of in vitro tests. Trends Food Sci. Technol. 2021, 109, 593–607. [Google Scholar] [CrossRef]
- Peters, R.; Bouwmeester, H.; Gottardo, S.; Amenta, V.; Arena, M.; Brandhoff, P.; Marvin, H.; Mech, A.; Moniz, F.; Pesudo, L.; et al. Nanomaterials for products and application in agriculture, feed and food. Trends Food Sci. Technol. 2016, 54, 155–164. [Google Scholar] [CrossRef]
- Kumari, R.; Suman, K.; Karmakar, S.; Mishra, V.; Lakra, S.G.; Saurav, G.; Mahto, B. Regulation and safety measures for nanotechnology-based agri-products. Front. Genome Ed. 2023, 5. [Google Scholar] [CrossRef] [PubMed]
- Shah, S.; Nene, S.; Rangaraj, N.; Raghuvanshi, R.; Singh, S.; Srivastava, S. Bridging the gap: academia, industry and FDA convergence for nanomaterials. Drug Dev. Ind. Pharm. 2020, 46, 1735–1746. [Google Scholar] [CrossRef]
- Elmanakhly, M.E.; Rizk, M.; Shaaban, M. Nanotechnology risk, toxicity and its effect on safety and human health. 2021, 33–37. [Google Scholar] [CrossRef]
- D, R.; Rao, P. Nanoparticles: Is Toxicity a Concern? EJIFCC 2011, 22, 92–101. [Google Scholar]
- Joshi, S.; Kaushik, U. Nanoparticles And Reproductive Toxicity: An Overview; 2013. [Google Scholar]
- Zibaei, A.; Jafari, S. Safety and toxicity aspects of food nanoparticles. In Safety and Regulatory Issues of Nanoencapsulated Food Ingredients; 2021. [Google Scholar] [CrossRef]
- Stern, S.; McNeil, S. Nanotechnology safety concerns revisited. Toxicol. Sci. An. Off. J. Soc. Toxicol. 2008, 101 1, 4–21. [Google Scholar] [CrossRef]
- Boyes, W. Safety Assessment of Nanotechnology Products. 2014, 9, 34–43. [Google Scholar] [CrossRef]
- Thomas, L.; Gert, R.; Douglas, G.; Luigi, C.; François, R.; Peter, G.; Christoph, K. Requirements on measurements for the implementation of the European Commission definition of the term 'nanomaterial'. 2012. [Google Scholar] [CrossRef]
- Rauscher, H.; Sokull-Kluettgen, B.; Stamm, H. The European Commission's recommendation on the definition of nanomaterial makes an impact. Nanotoxicology 2012, 7, 1195–1197. [Google Scholar] [CrossRef]
- Rasmussen, K.; Rauscher, H.; Mech, A.; Sintes, J.R.; Gilliland, D.; González, M.; Kearns, P.; Moss, K.; Visser, M.; Groenewold, M.; et al. Physico-chemical properties of manufactured nanomaterials - Characterisation and relevant methods. An outlook based on the OECD Testing Programme. Regul. Toxicol. Pharmacol. 2018, 92, 8–28. [Google Scholar] [CrossRef] [PubMed]
- Yuhui. 1. Characterization of Nanomaterials in Nanotoxicological Analyses. 2016, 1–21. [Google Scholar] [CrossRef]
- More, S.; Bampidis, V.; Benford, D.; Bragard, C.; Halldorsson, T.; Hernandez-Jerez, A.; Bennekou, S.; Koutsoumanis, K.; Lambré, C.; Machera, K.; et al. Guidance on technical requirements for regulated food and feed product applications to establish the presence of small particles including nanoparticles. EFSA J. 2021, 19. [Google Scholar] [CrossRef]
- Bushell, M.; Beauchemin, S.; Kunc, F.; Gardner, D.; Ovens, J.; Toll, F.; Kennedy, D.; Nguyen, K.; Vladisavljevic, D.; Rasmussen, P.; et al. Characterization of Commercial Metal Oxide Nanomaterials: Crystalline Phase, Particle Size and Specific Surface Area. Nanomaterials 2020, 10. [Google Scholar] [CrossRef] [PubMed]
- Catita, J. Challenges in Nanomaterial Characterization - From Definition to Analysis. Adv. Exp. Med. Biol. 2022, 1357, 3–17. [Google Scholar] [CrossRef]
- Speedy, A.; Battaglia, D. Safety of feed and animal products. 2019, 99–103. [Google Scholar]
- Gottardo, S.; Mech, A.; Drbohlavová, J.; Małyska, A.; Bøwadt, S.; Sintes, J.R.; Rauscher, H. Towards safe and sustainable innovation in nanotechnology: State-of-play for smart nanomaterials. Nanoimpact 2021, 21. [Google Scholar] [CrossRef] [PubMed]
- Reddy, P.R.K.; Yasaswini, D.; Reddy, P.; Zeineldin, M.; Adegbeye, M.; Hyder, I. Applications, challenges, and strategies in the use of nanoparticles as feed additives in equine nutrition. Vet. World 2020, 13, 1685–1696. [Google Scholar] [CrossRef] [PubMed]
- Danafar, H.; Sharafi, A.; Kheiri, S.; Manjili, H.K. Co -delivery of Sulforaphane and Curcumin with PEGylated Iron Oxide-Gold Core Shell Nanoparticles for Delivery to Breast Cancer Cell Line. Iran. J. Pharm. Res. IJPR 2018, 17, 480–494. [Google Scholar] [CrossRef]
- Kiadeh, S.G.H.; Rahaiee, S.; Azizi, H.; Govahi, M. The synthesis of broccoli sprout extract-loaded silk fibroin nanoparticles as efficient drug delivery vehicles: development and characterization. Pharm. Dev. Technol. 2024, 29, 359–370. [Google Scholar] [CrossRef]
- Essien, E.N.; Revi, N.; Khatri, V.; Liu, S.; Van Thiel, G.; Bijukumar, D. Methotrexate and Sulforaphane loaded PBA-G5-PAMAM dendrimers as a combination therapy for anti-inflammatory response in an intra-articular joint arthritic animal model. Int. J. Pharm. 2023, 123150. [Google Scholar] [CrossRef]
- Danafar, H.; Sharafi, A.; Manjili, H.K.; Andalib, S. Sulforaphane delivery using mPEG–PCL co-polymer nanoparticles to breast cancer cells. Pharm. Dev. Technol. 2017, 22, 642–651. [Google Scholar] [CrossRef]
- Thakkar, A.; Chenreddy, S.; Thio, A.; Khamas, W.; Wang, J.; Prabhu, S. Preclinical systemic toxicity evaluation of chitosan-solid lipid nanoparticle-encapsulated aspirin and curcumin in combination with free sulforaphane in BALB/c mice. Int. J. Nanomed. 2016, 11, 3265–3276. [Google Scholar] [CrossRef]
- Mangla, B.; Neupane, Y.; Singh, A.; Kumar, P.; Shafi, S.; Kohli, K. Lipid-nanopotentiated combinatorial delivery of tamoxifen and sulforaphane: ex vivo, in vivo and toxicity studies. Nanomedicine 2020. [Google Scholar] [CrossRef]
- Wang, L.; Rose, D.; Rao, P.; Zhang, Y. Development of prolamin-based composite nanoparticles for controlled-release of sulforaphane. J. Agric. Food Chem. 2019. [Google Scholar] [CrossRef]
- Hemati, H.; Haghiralsadat, F.; Hemati, M.; Sargazi, G.; Razi, N. Design and Evaluation of Liposomal Sulforaphane-Loaded Polyvinyl Alcohol/Polyethylene Glycol (PVA/PEG) Hydrogels as a Novel Drug Delivery System for Wound Healing. Gels 2023, 9. [Google Scholar] [CrossRef] [PubMed]
- Yepes-Molina, L.; Pérez-Jiménez, M.I.; Martínez-Esparza, M.; Teruel, J.; Ruiz-Alcaraz, A.; García-Peñarrubia, P.; Carvajal, M. Membrane Vesicles for Nanoencapsulated Sulforaphane Increased Their Anti-Inflammatory Role on an In Vitro Human Macrophage Model. Int. J. Mol. Sci. 2022, 23. [Google Scholar] [CrossRef] [PubMed]
- Abdelnour, S.; Alagawany, M.; Hashem, N.; Farag, M.; Alghamdi, E.; Hassan, F.; Bilal, R.; Elnesr, S.; Dawood, M.; Nagadi, S.; et al. Nanominerals: Fabrication Methods, Benefits and Hazards, and Their Applications in Ruminants with Special Reference to Selenium and Zinc Nanoparticles. Anim. An. Open Access J. From MDPI 2021, 11. [Google Scholar] [CrossRef]
- Rajendran, D.; Ezhuthupurakkal, P.B.; Lakshman, R.; Gowda, N.; Manimaran, A.; Rao, S. Application of encapsulated nano materials as feed additive in livestock and poultry: a review. Vet. Res. Commun. 2022, 46, 315–328. [Google Scholar] [CrossRef]
- Idowu, A.O.; Ojebiyi, O.; Lateef, A.; Odunsi, A.; Ola, A.; Akinola, A.O. APPLICATIONS OF NANOTECHNOLOGY IN ANIMAL NUTRITION: A REVIEW. Sci. Technol. Nanomater. 2024. [Google Scholar] [CrossRef]
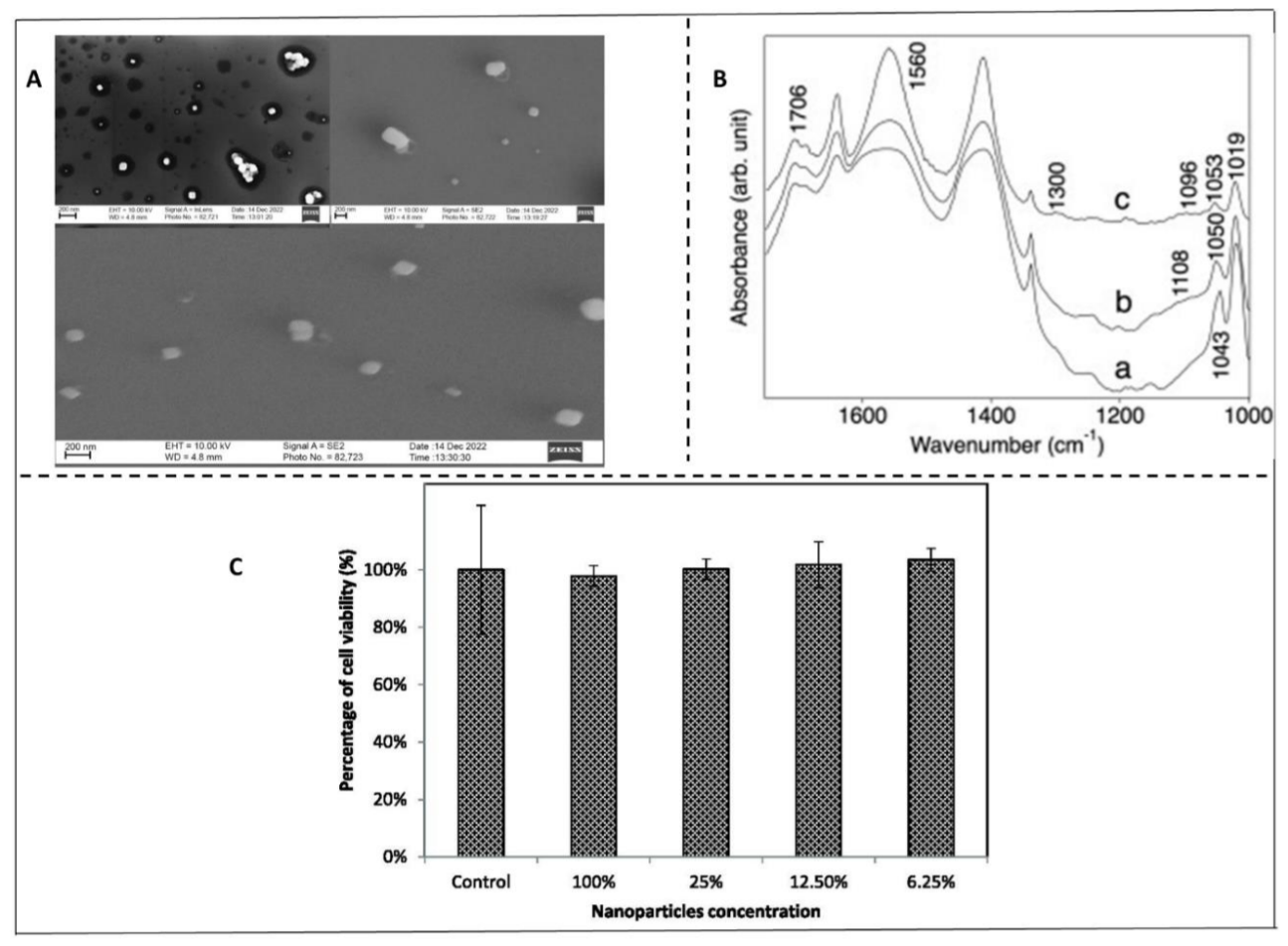
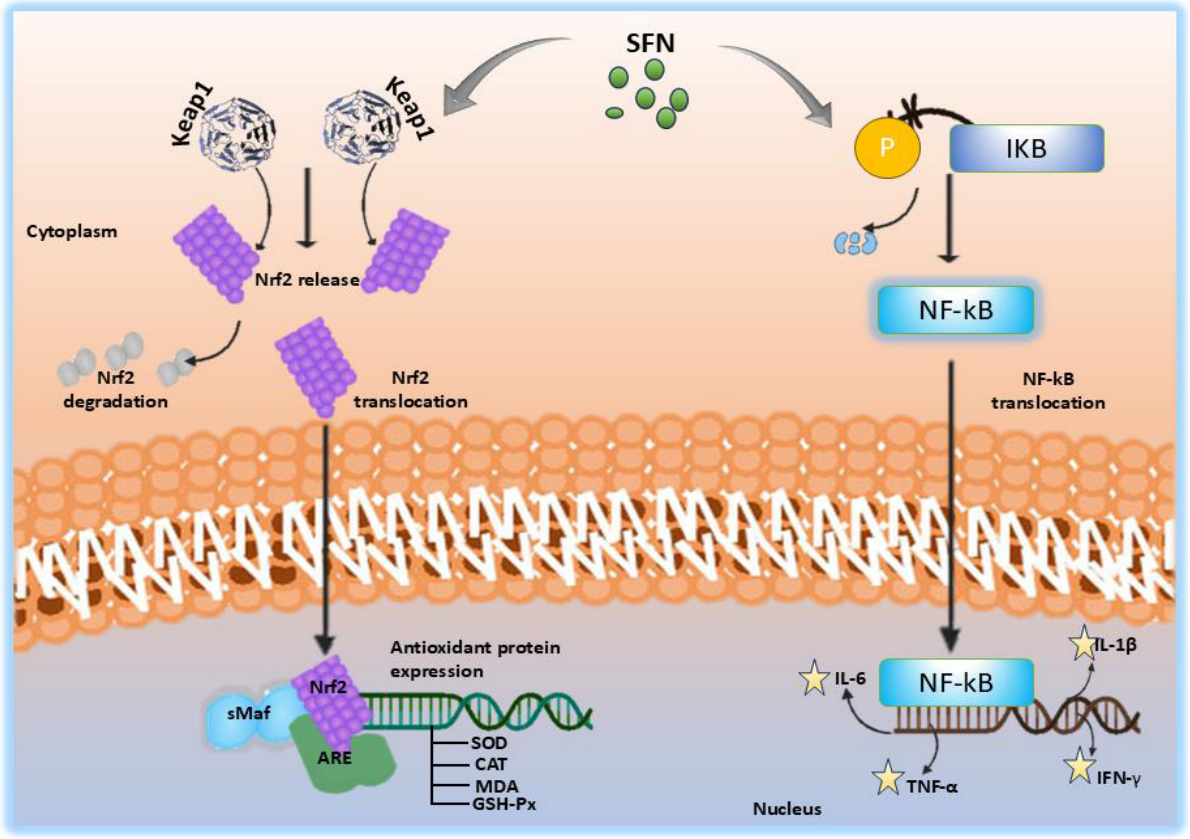
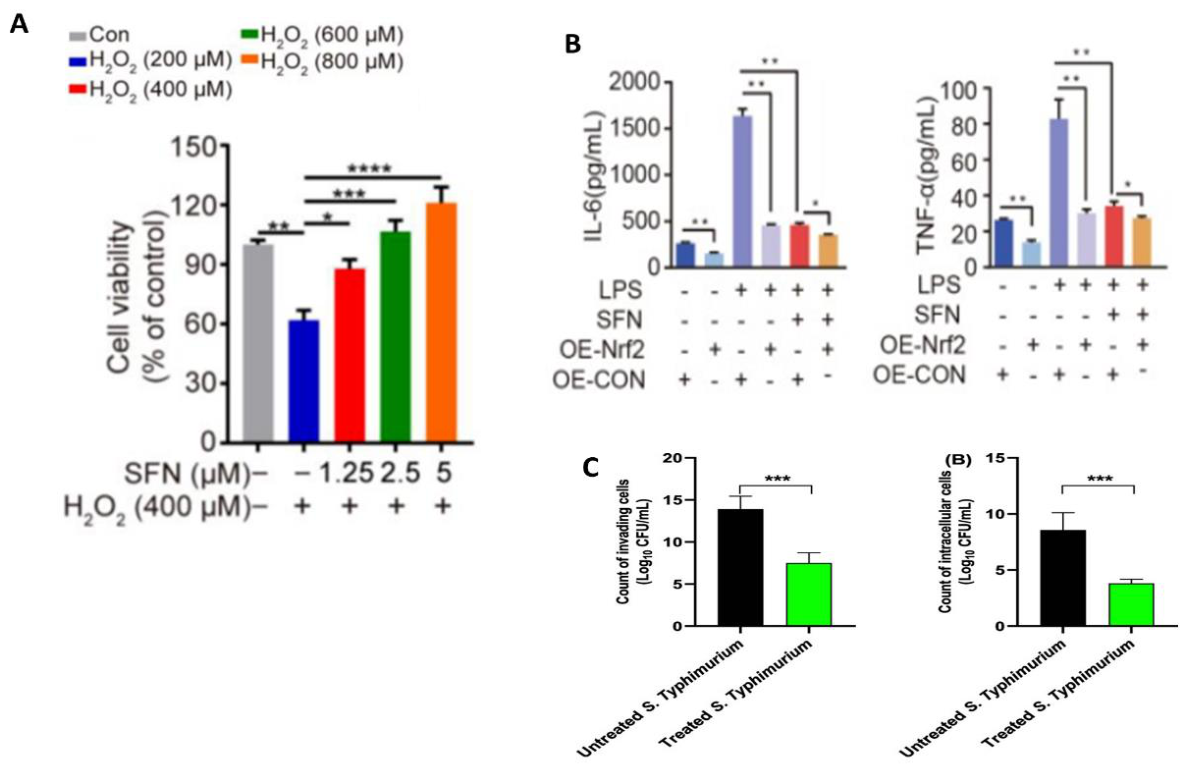
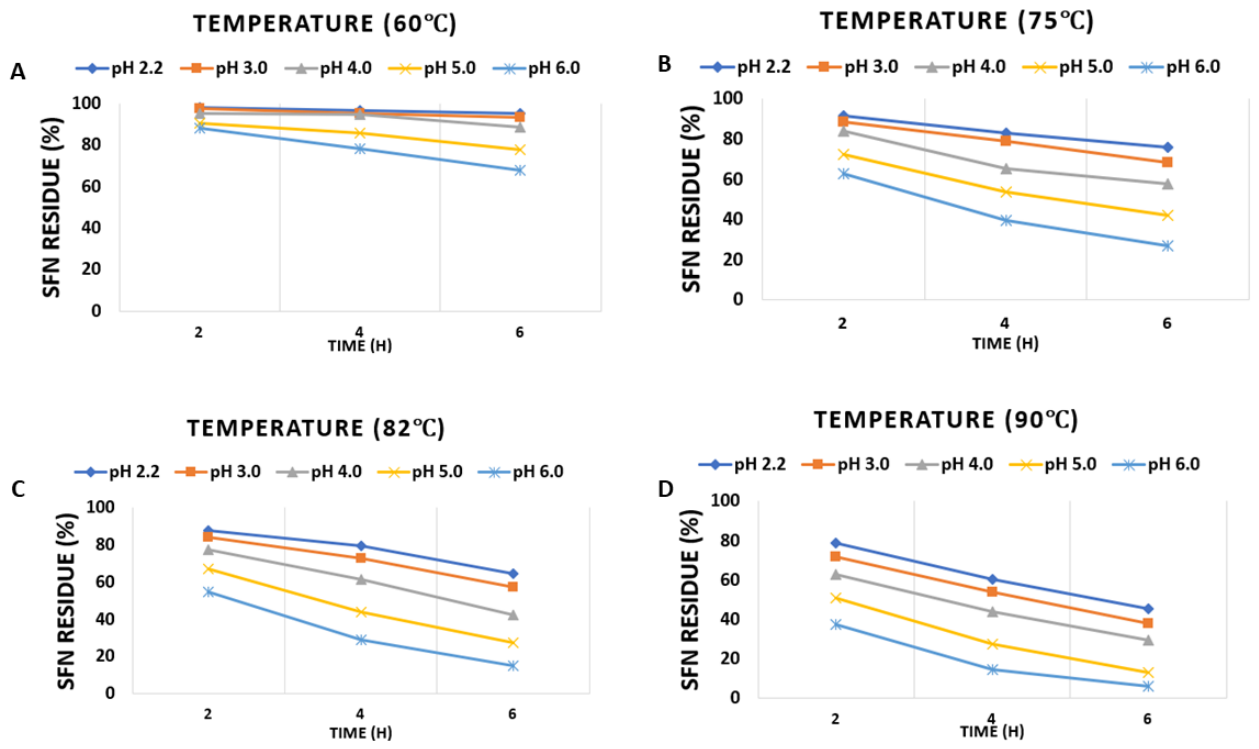
| Synthesis method | Nanocarrier main components | Synthesis time (min) | Loading capacity (mg g⁻¹) | Encapsulation efficiency (%) | Nanocarrier size (nm) | Ref. |
|---|---|---|---|---|---|---|
| Microfluidics-assisted self-assembly | Chitosan + alginate | 5 | 50.2 | 68.4 | 5.3 | [52] |
| Ionic gelation | Chitosan + TPP | 60 | – | 80.7 | 80.8 | [53] |
| Emulsion solvent evaporation | PLGA + γ-PGA | 240 | – | 89.5 | 548.5 | [54] |
| Emulsification solvent volatilization | PLGA + chitosan + alginate | 300 | 256.7 | 87.23 | 255 | [55] |
| Ionic pre-gelation + complexation | Chitosan + alginate | 90 | 6.8 | – | 20–50 | [56] |
| Emulsion solvent evaporation | PLGA + PVA | 300 | – | – | 222 | [57] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




