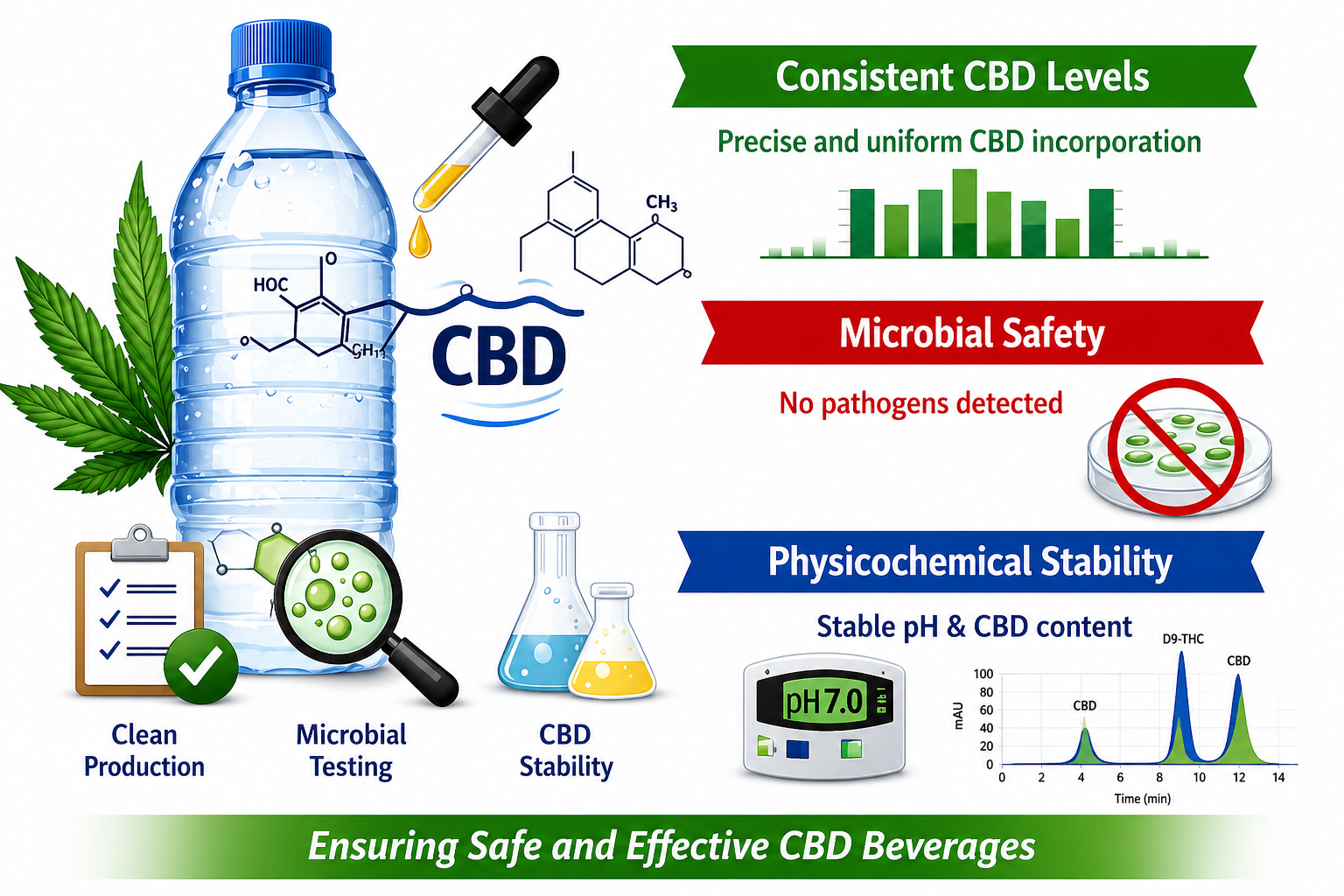
This paper was about creating and testing quality, microbial safety, chemical stability, and shelf life of CBD infused bottled water. Regular water does not mix well with lipophilic cannabidiol, which results in dose inconsistency, degradation, microbial contamination, and limited stability. To counteract these problems, a controlled CBD incorporation method was combined with clean, room bottling and systematic quality control protocols. The bottled water was subjected to various tests after being stored for 28 days, including cannabidiol concentration, degradation products, physicochemical parameters (pH, total solids, water activity) and microbial safety, total plate counts, yeast, mold, and pathogenic bacteria. CBD concentration was maintained with negligible degradation and microbial analyses revealed that total counts were low and no pathogens were detected. This proves that aseptic processing is very effective. Physicochemical parameters did not change, which means that the beverage matrix was not affected by either the addition of CBD or the storage. These results guarantee consistent potency, chemical integrity, microbial safety and product stability effectively solving the problem of producing CBD beverages. The paper demonstrates a reliable method of making safe and high, quality CBD functional beverages with a good shelf life. The results are relevant for manufacturing operations of different scales and supply insight on standardized production, quality monitoring, and storage practices. This research is in line with regulatory compliance and consumer safety and consistent product performance, providing a foundation for the safe commercialization of CBD-infused bottled water.