Submitted:
28 April 2026
Posted:
30 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials and Sourcing
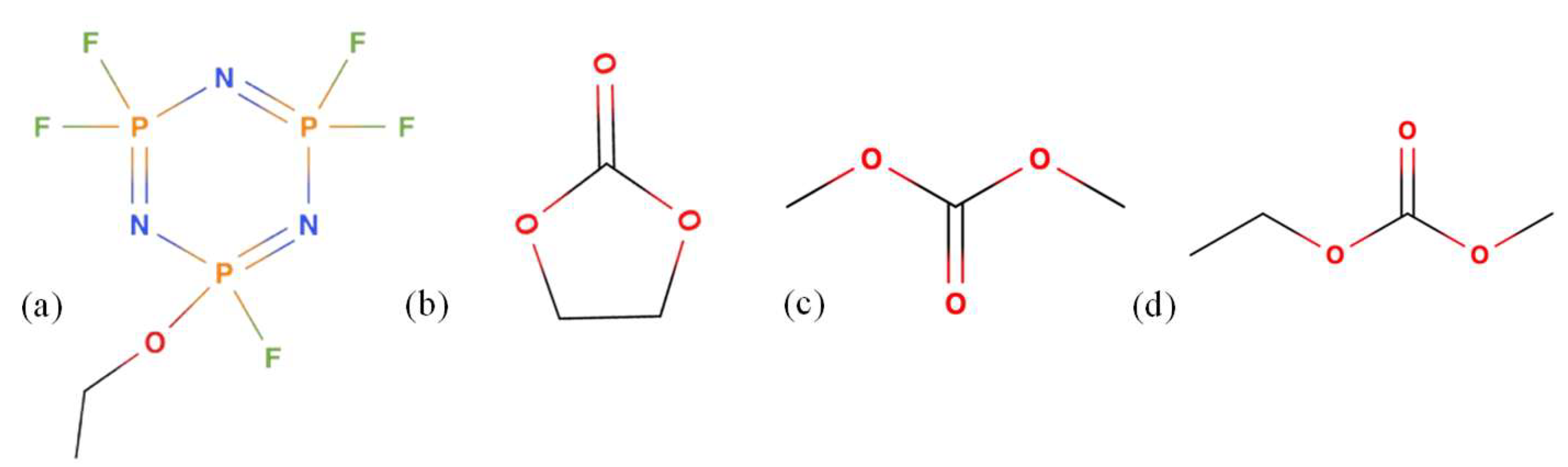
2.2. Self-Extinguishing Time Test
2.3. Differential Scanning Calorimetry
2.4. Ionic Conductivity Measurement
2.5. Electrochemical Performance Tests
3. Results and Discussion
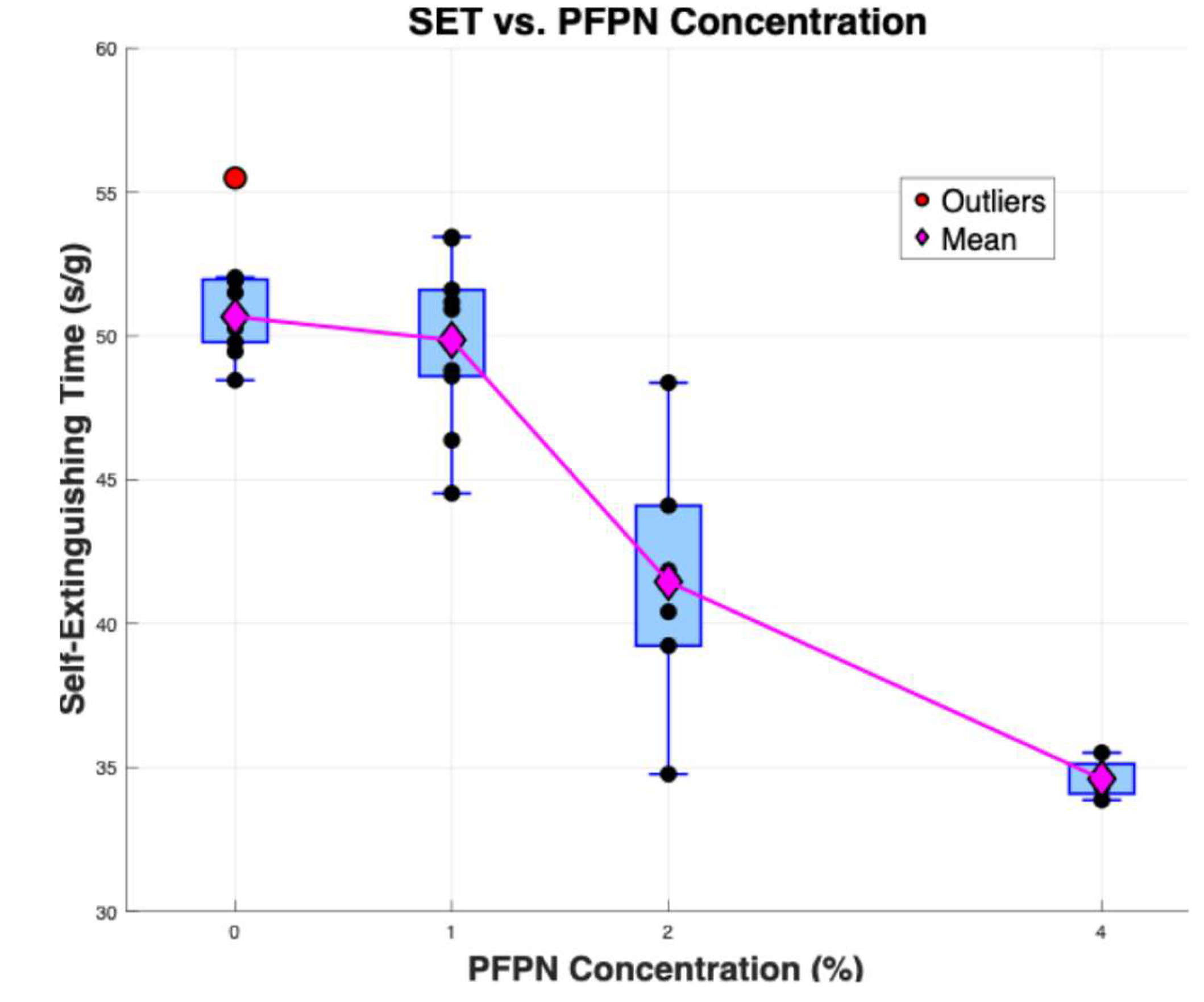
3.1. Self-Extinguishing Time
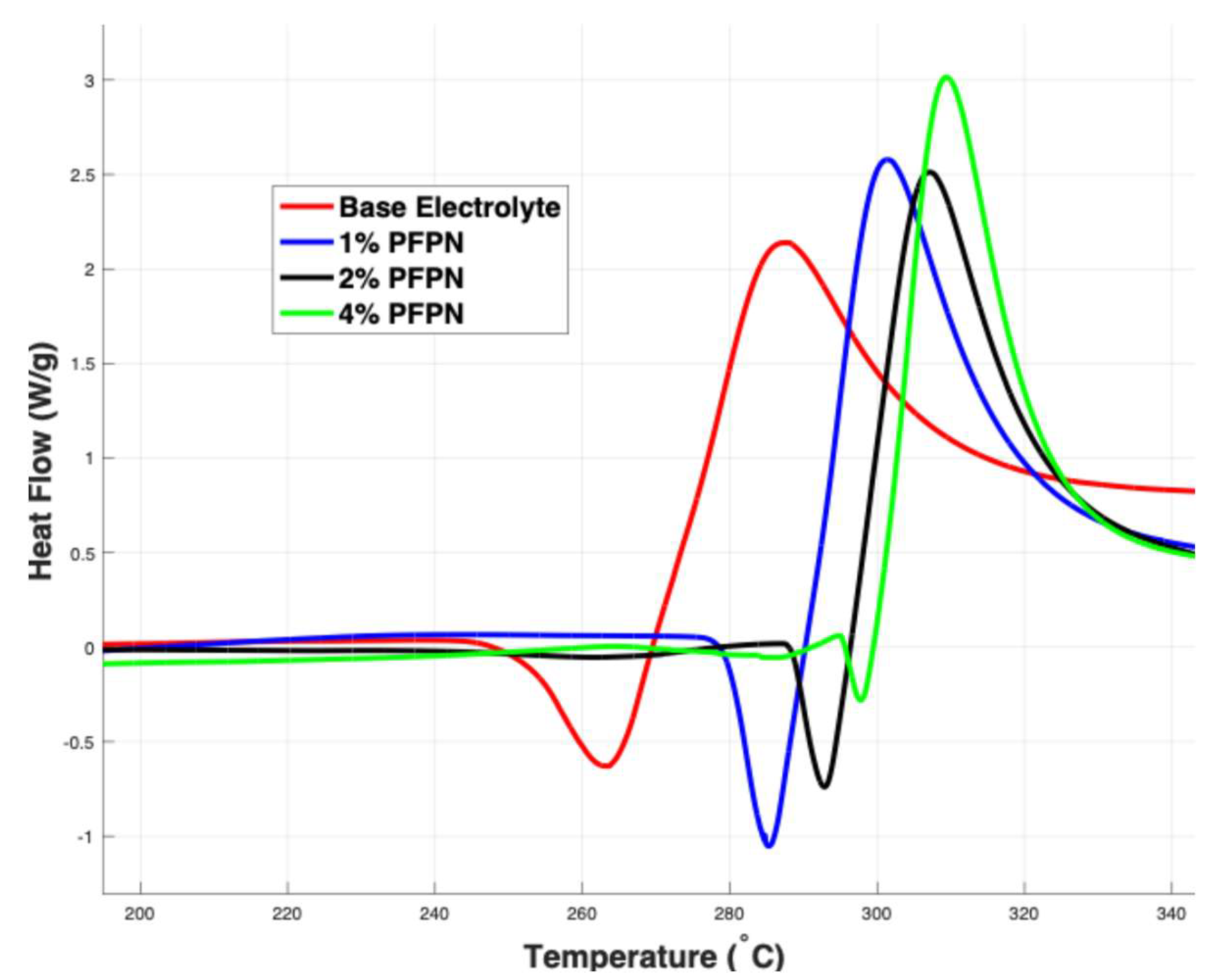
3.2. Differential Scanning Calorimetry
3.3. Ionic Conductivity
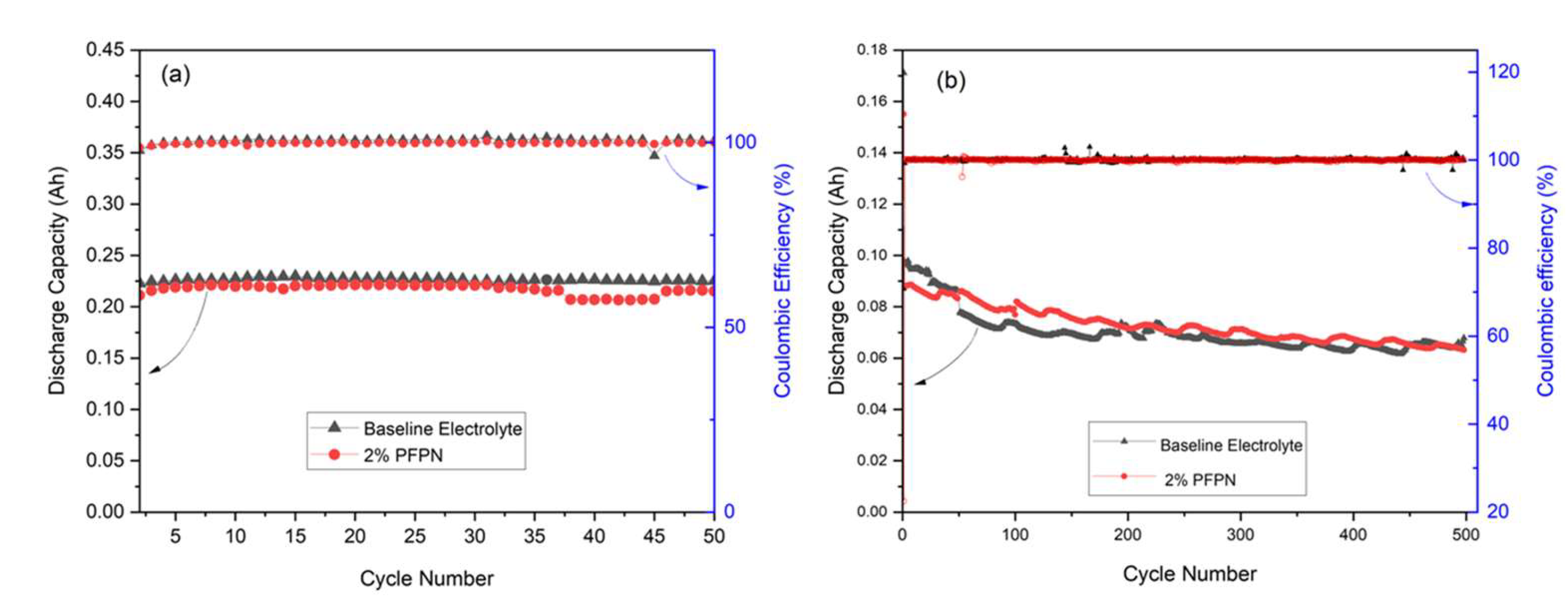
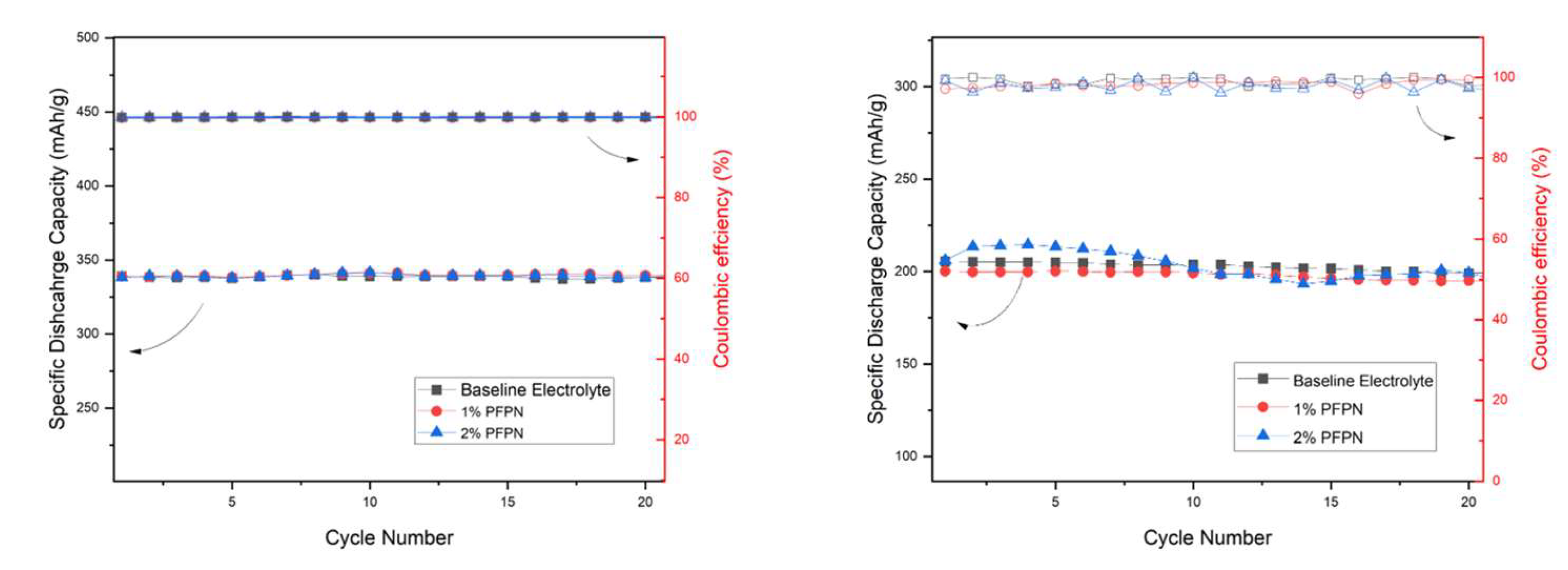
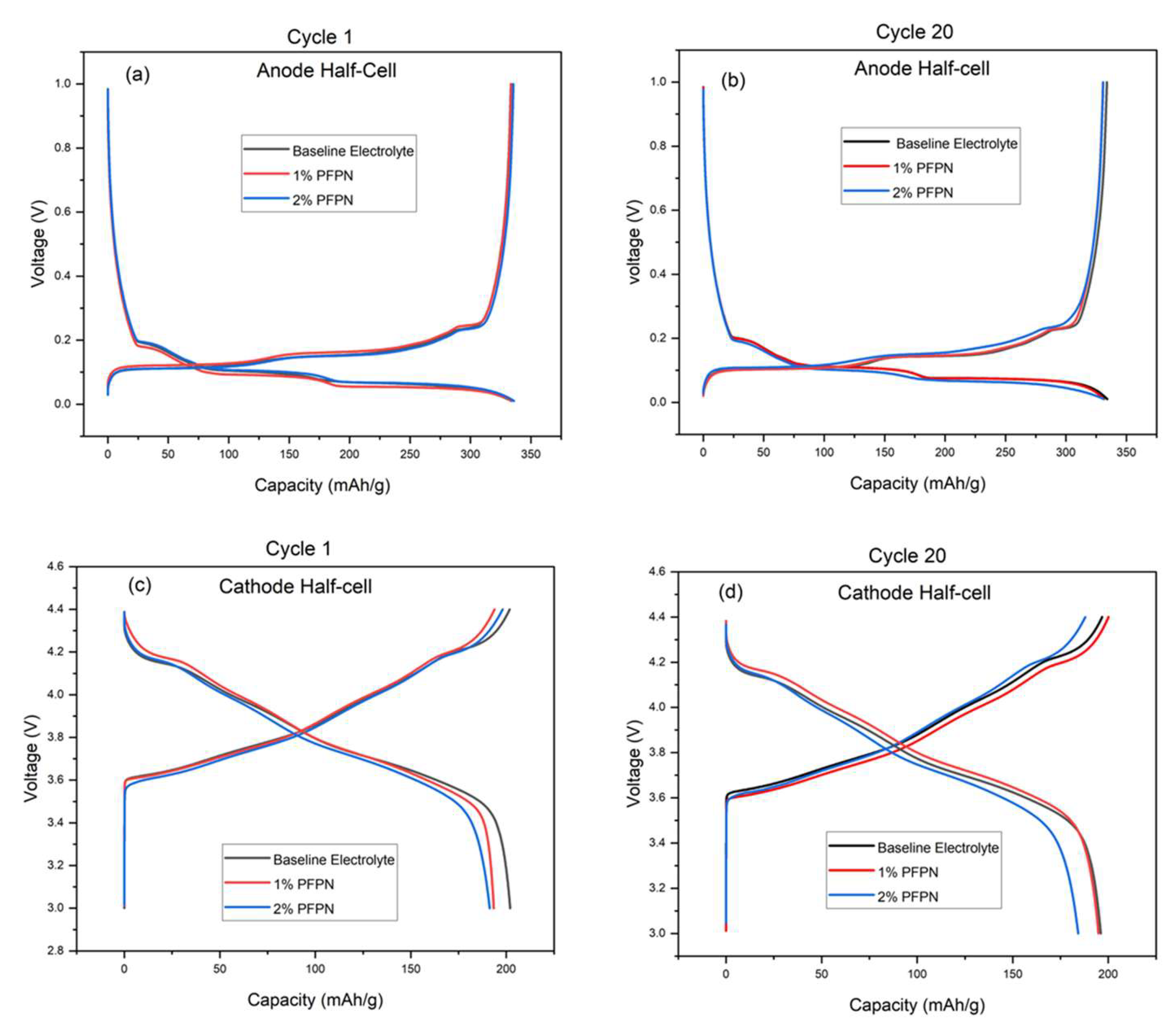
3.4. Coin cell Cycling
3.5. Pouch cell Cycling
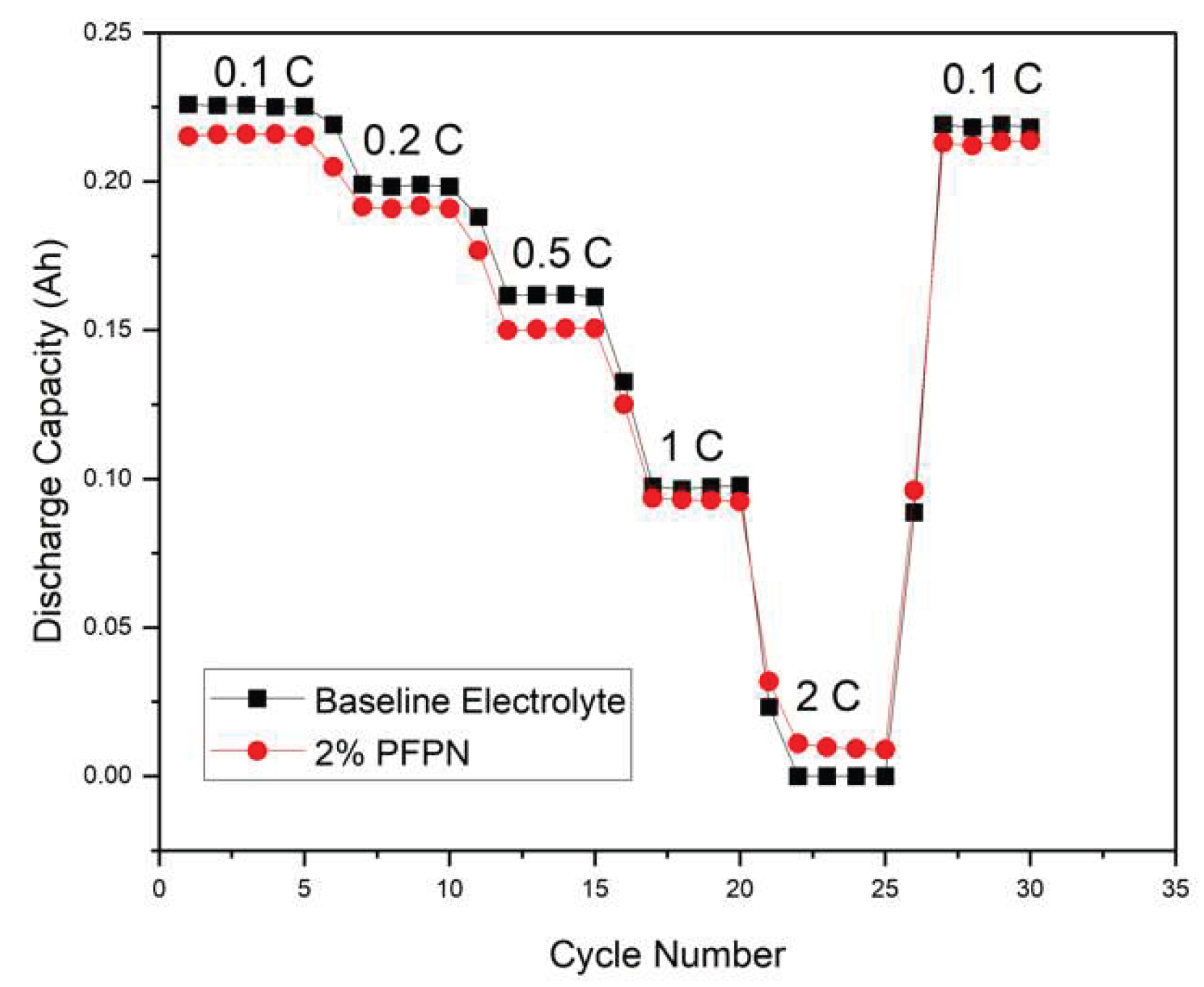
3.6. Rate Capability
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Luo, W.; Zhang, S.; Gao, Y.; Shen, C. Review of Mechanisms and Detection Methods of Internal Short Circuits in Lithium-Ion Batteries. Ionics Vol. 31(No. 5, 2025), 3945–3964. [CrossRef]
- Maher, K.; Boumaiza, A.; Amin, R. Understanding the Heat Generation Mechanisms and the Interplay between Joule Heat and Entropy Effects as a Function of State of Charge in Lithium-Ion Batteries. J. Power Sources 2024, Vol. 623, 235504. [Google Scholar] [CrossRef]
- Hou, J.; Feng, X.; Wang, L.; Liu, X.; Ohma, A.; Lu, L.; Ren, D.; Huang, W.; Li, Y.; Yi, M.; Wang, Y.; Ren, J.; Meng, Z.; Chu, Z.; Xu, G. L.; Amine, K.; He, X.; Wang, H.; Nitta, Y.; Ouyang, M. Unlocking the Self-Supported Thermal Runaway of High-Energy Lithium-Ion Batteries. Energy Storage Mater. 2021, Vol. 39, 395–402. [Google Scholar] [CrossRef]
- Tran, Y. H. T.; An, K.; Lim, G.; Kim, D.; Lee, Y. J.; Doh, C.; Song, S. W. Preventing Thermal Runaway of High-Nickel Li-Ion Battery through Nonflammable Carbonates-Based Electrolyte Formulation. Mater. Sci. Eng. R. Rep. Vol. 164(2025), 100980. [CrossRef]
- Sorensen, A.; Belt, J. Analyzing Thermal Runaway Propagation in Lithium-Ion Battery Modules with Reduced Flammability Electrolyte Cells. J. Electrochem. Soc. 2024, Vol. 171(No. 8), 080540. [Google Scholar] [CrossRef]
- Zhang, W.; Feng, X.; Huang, W.; Lu, L.; Wang, H.; Wang, L.; He, X.; Wei, M.; Ouyang, M.; Zhang, W.; Feng, X.; Huang, W.; Lu, L.; Wang, H.; Ouyang, M.; Wang, L.; He, X.; Wei, M. Thermal Runaway Inhibition of Lithium-Ion Batteries Employing Thermal-Driven Phosphazene Based Electrolytes. Adv. Funct. Mater. 2025, Vol. 35(No. 48), 2508688. [Google Scholar] [CrossRef]
- Dong, L.; Deng, L.; Wang, Z.; Liu, Y.; Zhan, J.; Wang, S.; Fang, Z.; Guo, F.; Liu, C.; Liu, H.; Chen, H. A Self Fire-Extinguishing and High Rate Lithium-Fluorinated Carbon Battery Realized by Ethoxy (Pentafluoro) Cyclotriphosphazene Electrolyte Additive Design. Nano Energy 2024, Vol. 131, 110309. [Google Scholar] [CrossRef]
- Kim, J.-H.; Hyun, J.-H.; Kim, S.; Park, H.; Yu, S.-H.; Kim, J.-H.; Hyun, J.-H.; Park, W. H.; Yu, S.-H.; Aiiso, S. K.; Li, Y. Phosphorus-Based Flame-Retardant Electrolytes for Lithium Batteries. Adv. Energy Mater. Vol. 15(No. 23, 2025), 2500587. [CrossRef]
- Dagger, T.; Rad, B. R.; Schappacher, F. M.; Winter, M. Comparative Performance Evaluation of Flame Retardant Additives for Lithium Ion Batteries – I. Safety, Chemical and Electrochemical Stabilities. Energy Technol. 2018, Vol. 6(No. 10), 2011–2022. [Google Scholar] [CrossRef]
- Li, X.; Li, W.; Chen, L.; Lu, Y.; Su, Y.; Bao, L.; Wang, J.; Chen, R.; Chen, S.; Wu, F. Ethoxy (Pentafluoro) Cyclotriphosphazene (PFPN) as a Multi-Functional Flame Retardant Electrolyte Additive for Lithium-Ion Batteries. J. Power Sources 2018, Vol. 378, 707–716. [Google Scholar] [CrossRef]
- Chen, L.; Nian, Q.; Ruan, D.; Fan, J.; Li, Y.; Chen, S.; Tan, L.; Luo, X.; Cui, Z.; Cheng, Y.; Li, C.; Ren, X. High-Safety and High-Efficiency Electrolyte Design for 4.6 V-Class Lithium-Ion Batteries with a Non-Solvating Flame-Retardant. Chem. Sci. 2023, Vol. 14(No. 5), 1184–1193. [Google Scholar] [CrossRef] [PubMed]
- Hess, S.; Wohlfahrt-Mehrens, M.; Wachtler, M. Flammability of Li-Ion Battery Electrolytes: Flash Point and Self-Extinguishing Time Measurements. J. Electrochem. Soc. 2015, Vol. 162(No. 2), A3084–A3097. [Google Scholar] [CrossRef]
- Arbizzani, C.; Gabrielli, G.; Mastragostino, M. Thermal Stability and Flammability of Electrolytes for Lithium-Ion Batteries. J. Power Sources 2011, Vol. 196(No. 10), 4801–4805. [Google Scholar] [CrossRef]
- Zhang, M.; Xiao, J.; Tang, W.; He, Y.; Tan, P.; Haranczyk, M.; Wang, D. Y. A Novel Benchmarking Approach to Assess Fire Safety of Liquid Electrolytes in Lithium-Ion Batteries. Adv. Energy Mater. 2024, Vol. 14(No. 31). [Google Scholar] [CrossRef]
- Fernandes, Y.; Bry, A.; de Persis, S. Thermal Degradation Analyses of Carbonate Solvents Used in Li-Ion Batteries. J. Power Sources 2019, Vol. 414, 250–261. [Google Scholar] [CrossRef]
- Farris, S.; Pozzoli, S.; Biagioni, P.; Duó, L.; Mancinelli, S.; Piergiovanni, L. The Fundamentals of Flame Treatment for the Surface Activation of Polyolefin Polymers – A Review. Polymer 2010, Vol. 51(No. 16), 3591–3605. [Google Scholar] [CrossRef]
- Dong, L.; Deng, L.; Wang, Z.; Liu, Y.; Zhan, J.; Wang, S.; Fang, Z.; Guo, F.; Liu, C.; Liu, H.; Chen, H. A Self Fire-Extinguishing and High Rate Lithium-Fluorinated Carbon Battery Realized by Ethoxy (Pentafluoro) Cyclotriphosphazene Electrolyte Additive Design. 2024. [Google Scholar] [CrossRef]
- Zinigrad, E.; Larush-Asraf, L.; Gnanaraj, J. S.; Sprecher, M.; Aurbach, D. On the Thermal Stability of LiPF6. Thermochim. Acta 2005, Vol. 438(Nos. 1–2), 184–191. [Google Scholar] [CrossRef]
- Ravdel, B.; Abraham, K. M.; Gitzendanner, R.; DiCarlo, J.; Lucht, B.; Campion, C. Thermal Stability of Lithium-Ion Battery Electrolytes. J. Power Sources 2003, Vols. 119–121, 805–810. [Google Scholar] [CrossRef]
- Wang, Q.; Sun, J.; Yao, X.; Chen, C. Thermal Stability of LiPF6/EC + DEC Electrolyte with Charged Electrodes for Lithium Ion Batteries. Thermochim. Acta 2005, Vol. 437(Nos. 1–2), 12–16. [Google Scholar] [CrossRef]
- Xia, L.; Xia, Y.; Liu, Z. A Novel Fluorocyclophosphazene as Bifunctional Additive for Safer Lithium-Ion Batteries. J. Power Sources 2015, Vol. 278, 190–196. [Google Scholar] [CrossRef]
- Liu, J.; Song, X.; Zhou, L.; Wang, S.; Song, W.; Liu, W.; Long, H.; Zhou, L.; Wu, H.; Feng, C.; Guo, Z. Fluorinated Phosphazene Derivative – A Promising Electrolyte Additive for High Voltage Lithium Ion Batteries: From Electrochemical Performance to Corrosion Mechanism. Nano Energy 2018, Vol. 46, 404–414. [Google Scholar] [CrossRef]
- Li, N.; Zhang, Y.; Zhang, S.; Shi, L.; Zhang, J. Y.; Song, K. M.; Li, J. C.; Zeng, F. L. Insight into the Probability of Ethoxy(Pentafluoro)Cyclotriphosphazene (PFPN) as the Functional Electrolyte Additive in Lithium–Sulfur Batteries. RSC Adv. 2024, Vol. 14(No. 18), 12754. [Google Scholar] [CrossRef] [PubMed]
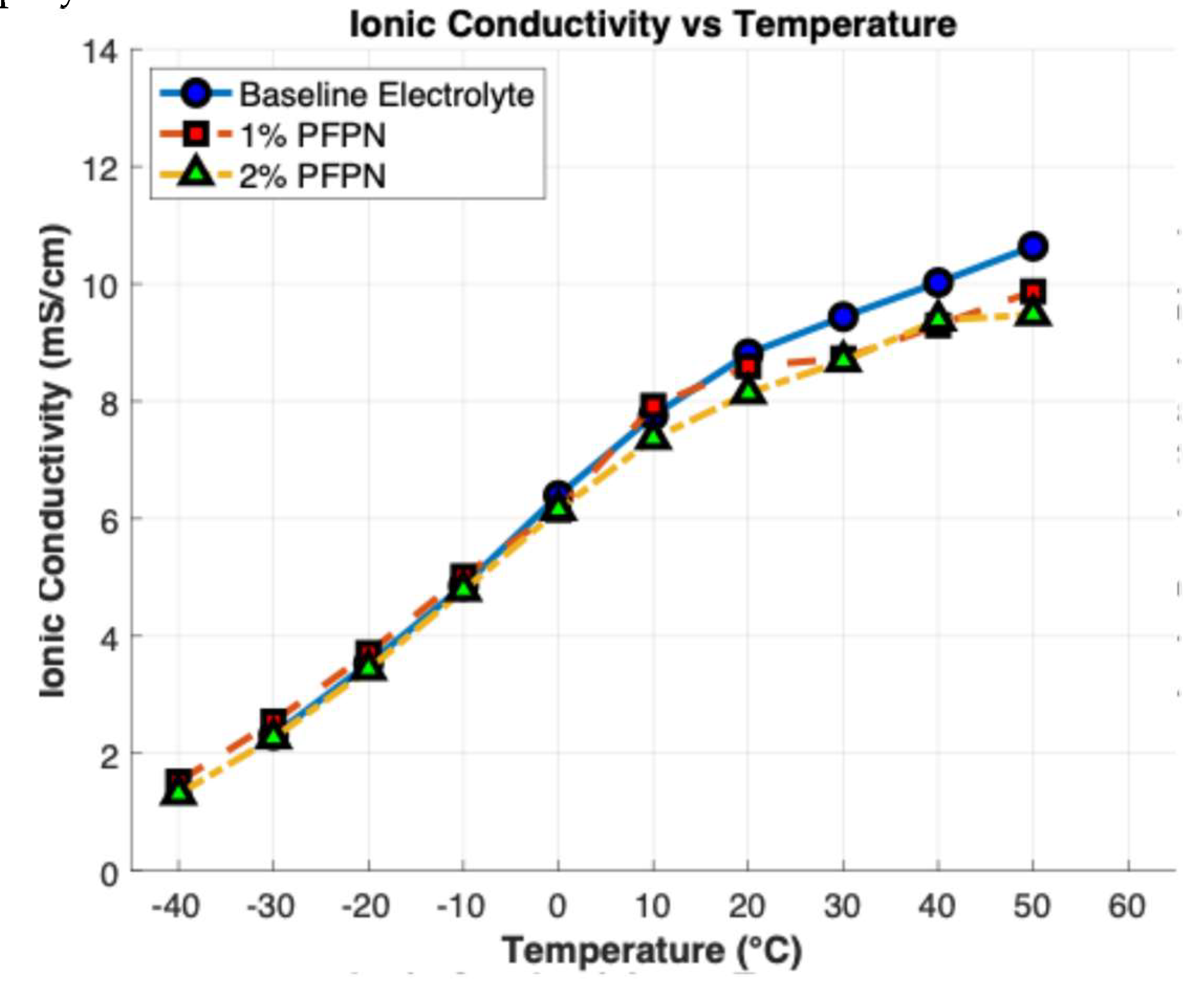
| PFPN Concertation (wt %) | 0% PFPN | 1% PFPN | 2% PFPN | 4% PFPN |
| Ionic Conductivity (mS/cm) | 10.26 | 10.02 | 9.88 | 9.12 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




