Submitted:
29 April 2026
Posted:
30 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Reagent
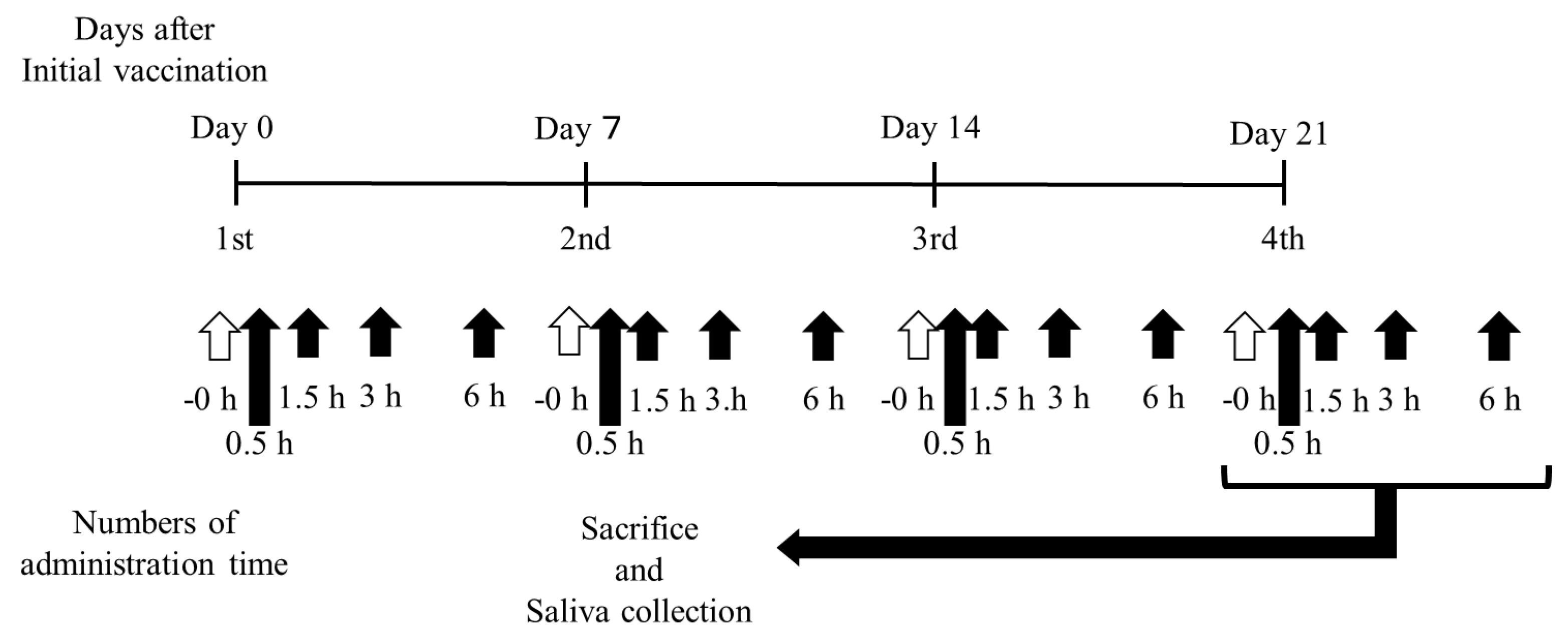
2.3. Nasal Immunization and Sample Collection Schedule
2.4. Measurement of SIgA and IgA Antibody-Forming Cells (AFCs)
2.5. Relative Metabolic Activity of IgA AFCs
2.6. Flow Cytometric Analysis for B220+CD38+ Memory B-Cell Populations
2.7. Statistical Analysis
3. Results
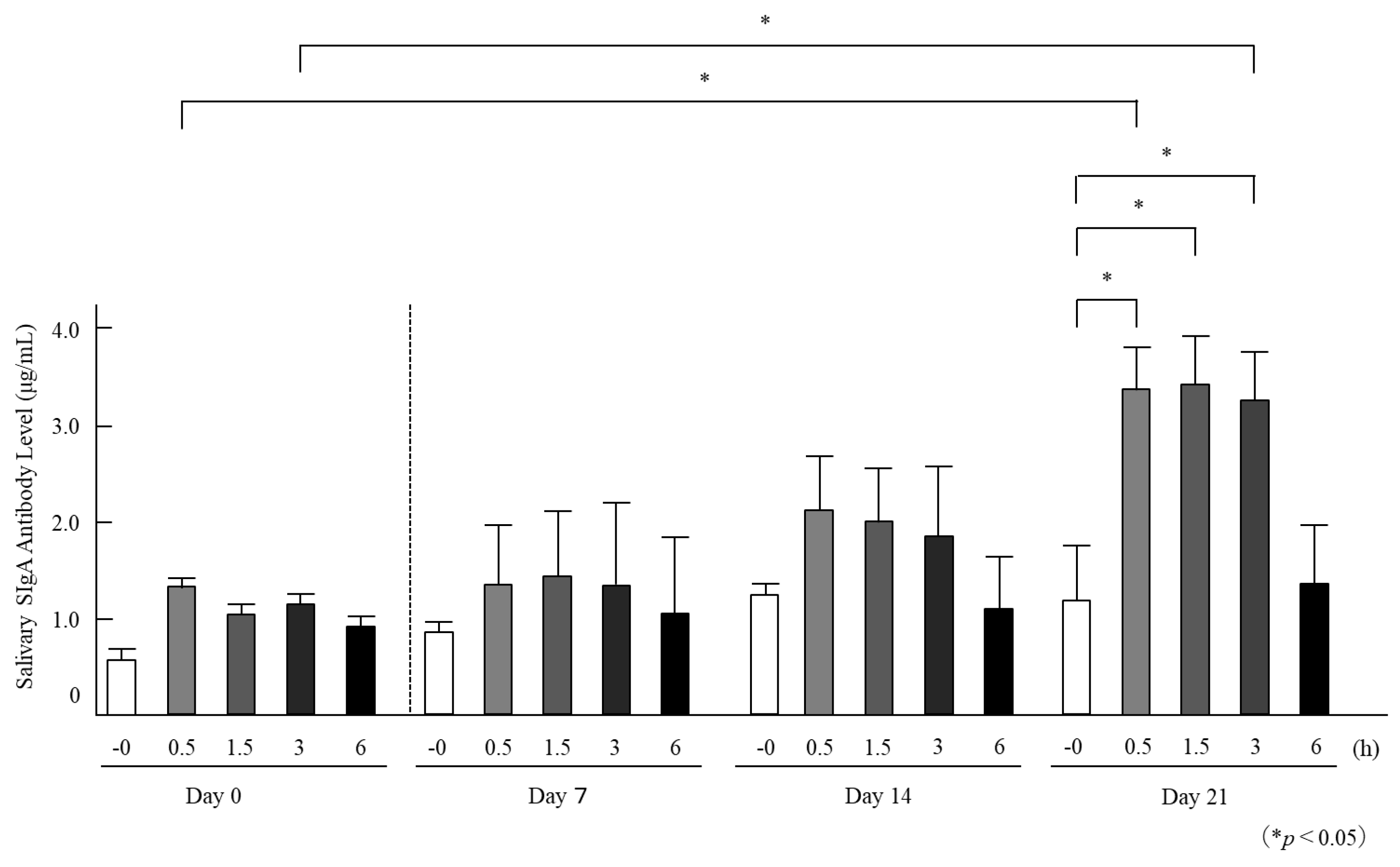
3.1. Temporal Changes in Salivary SIgA Antibody Levels Following HNK Administration
3.2. Rapid Kinetics of Salivary SIgA Antibody Secretion Following HNK Administration
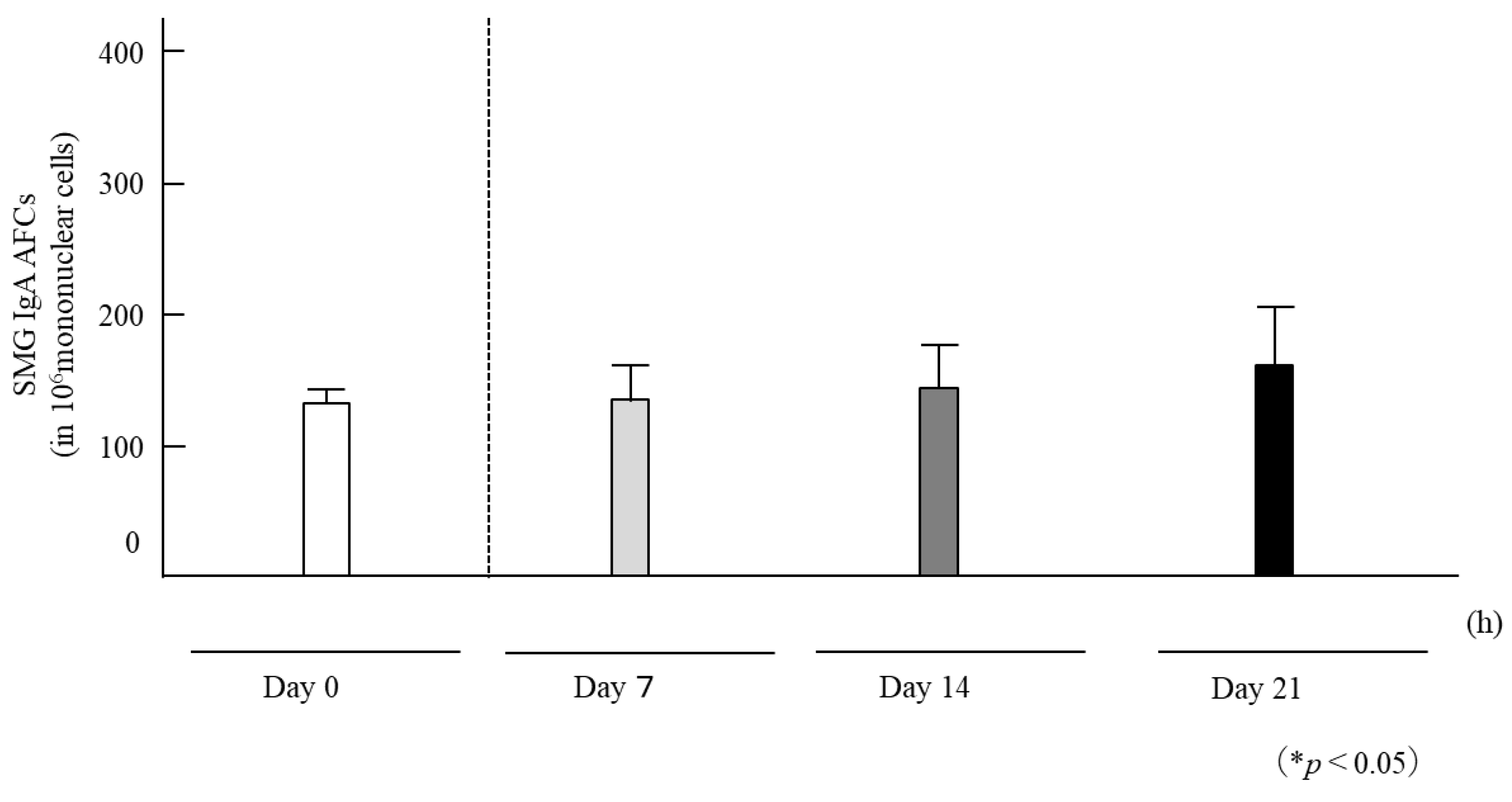
3.3. Quantification of IgA AFCs in SMGs on the Final Day of HNK Administration
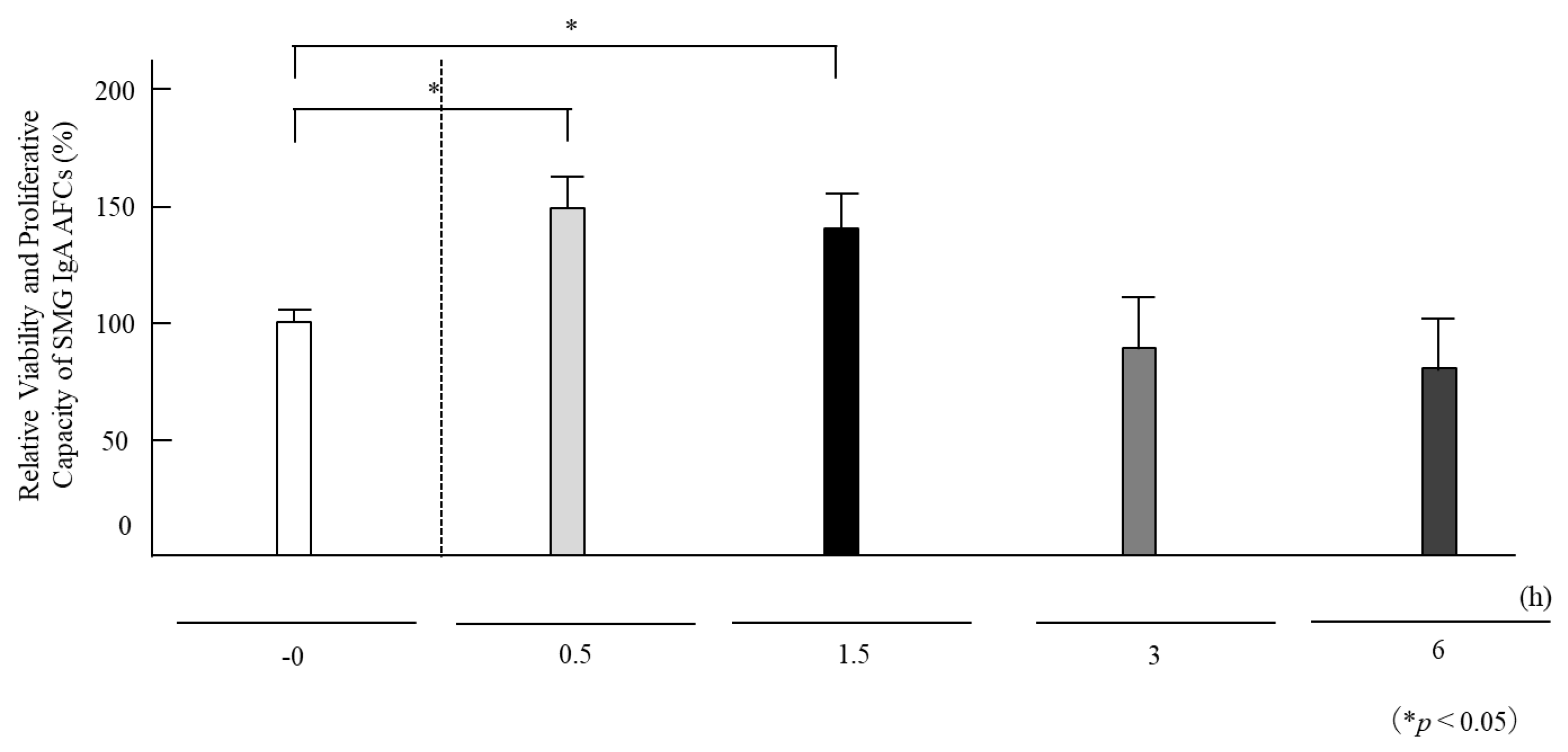
3.4. Evaluation of the Viability and Proliferative Capacity of IgA AFCs in SMGs on the Final Day of HNK Administration
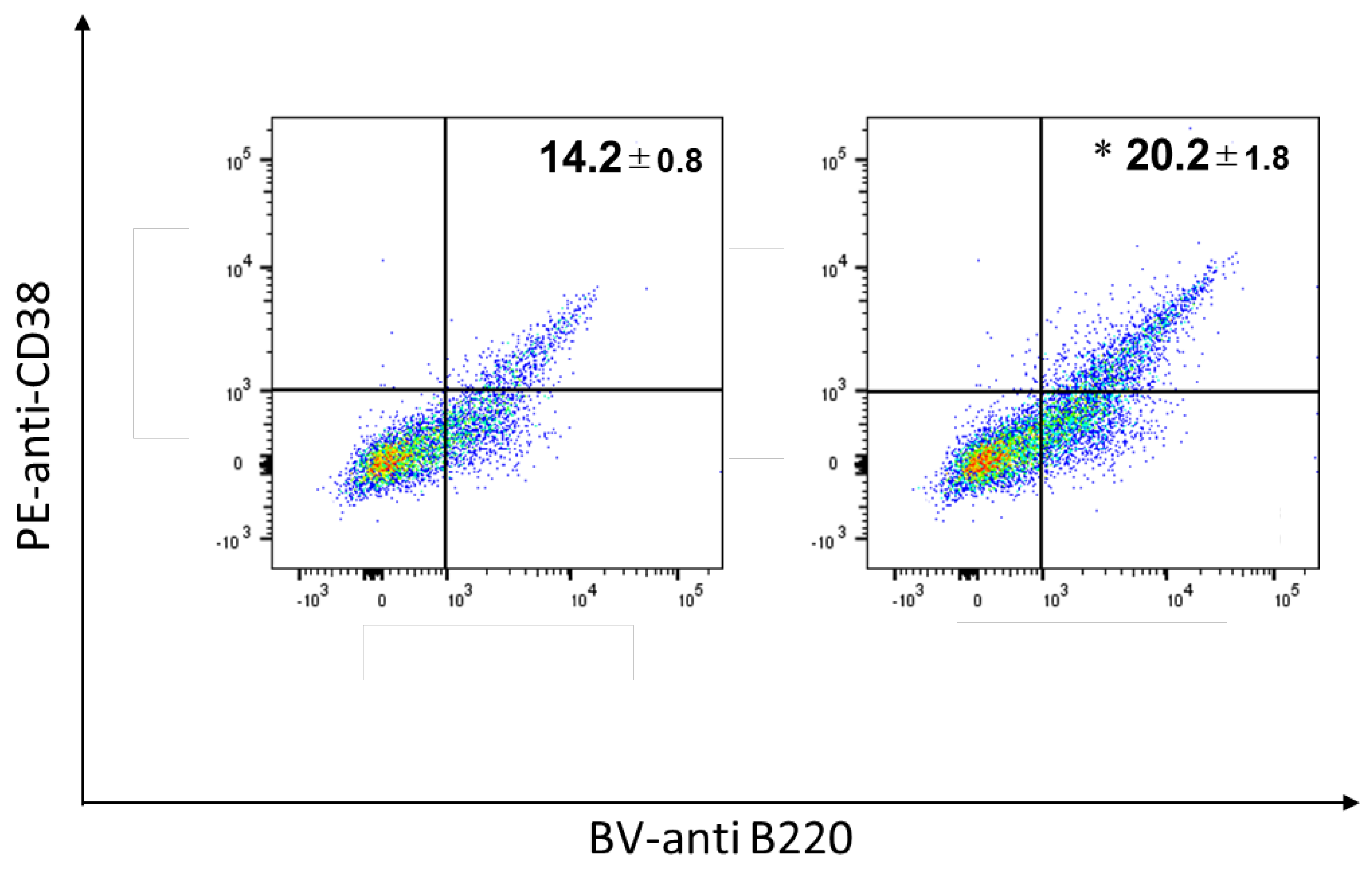
3.5. Induction of B220+CD38+ Memory B-Cells in SMGs
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| HNK | Hinokitiol |
| SIgA | Secretory immunoglobulin A |
| AFC | Antibody-forming cell |
| DC | Dendritic cell |
| SMG | Submandibular gland |
| DMSO | Dimethyl sulfoxide |
| PBS | Phosphate-buffered saline |
| ELISA | Enzyme-linked immunosorbent assay |
| ELISPOT | Enzyme-linked immunospot |
| NALT | Nasal-associated lymphoid tissue |
References
- Goto, Y.; Kurashima, Y.; Kiyono, H. The gut microbiota and inflammatory bowel disease. Curr Opin Rheumatol 2015, 27, 388–96. [CrossRef]
- Kurashima, Y.; Goto, Y.; Kiyono, H. Mucosal innate immune cells regulate both gut homeostasis and intestinal inflammation. Eur J Immunol 2013, 43, 3108–3115. [CrossRef]
- Zhang, T.; Hashizume, T.; Kurita-Ochiai, T.; Yamamoto, M. Sublingual vaccination with outer membrane protein of Porphyromonas gingivalis and Flt3 ligand elicits protective immunity in the oral cavity. Biochem Biophys Res Commun 2009, 390, 937–941. [CrossRef]
- Mestecky, J.; McGhee, J.R. Immunoglobulin A (IgA): molecular and cellular interactions involved in IgA biosynthesis and immune response. Adv Immunol 1987, 40, 153–245. [CrossRef]
- Michalek, S.M.; Childers, N.K. Development and outlook for a caries vaccines. Crit Rev Oral Biol Med 1990, 1, 37–54. [CrossRef]
- Ogawa, T.; Shimauchi, H.; Hamada, S. Mucosal and systemic immune response in BALB/c mice to Bacteroides gingivalis fimbriae administered orally. Infect Immun 1989, 57, 3466–3471. [CrossRef]
- Czerkinsky, C.; Russell, M.W.; Lycke, N.; Lindblad, M.; Holmgren, J. Oral administration of a streptococcal antigen coupled to cholera toxin B submit evokes strong antibody responses in salivary glands and extramucosal tissues. Infect Immun 1989, 57, 1072–1077. [CrossRef]
- Fotopoulou, T.; Ćirić, A.; Kritsi, E.; Calhelha, R.C.; Ferreira, I.C.F.R.; Soković, M., Zoumpoulakis, P.; Koufaki, M. Antimicrobial/antibiofilm activity and cytotoxic studies of β-thujaplicin derivatives. Arch Pharm Chem Life Sci 2016, 349, 698–709. [CrossRef]
- Kim, D.J.; Lee, M.W.; Choi, J.S.; Lee, S.G.; Park, J.Y.; Kim, S.W. Inhibitory activity of hinokitiol against biofilm formation in fluconazole-resistant Candida species. PLoS One 2017, 12, e0171244. [CrossRef]
- Yoshimatsu, H.; Kanda, R.; Kano, K. In vitro synergistic effects of hinokitiol and fluconazole against Candida albicans. Appl. Sci 2026, 16, 2126. [CrossRef]
- Kanda, R.; Yoshimatsu, H.; Lyu, X. Investigation of antibacterial effects of hinokitiol on Lactobacillus casei. J Osaka Dent Univ 2025, 59, 7–11. [CrossRef]
- Tsuji, N.; Yoshimatsu, H.; Kanda, R.; Hashimoto, N.; Maeda, H. Investigation of antibacterial effects of hinokitiol on Fusobacterium nucleatum. World J Adv Res Rev 2025, 25, 103–108. [CrossRef]
- Yoshimatsu, H.; Kanda, R.; Xinghui, L.; Kano, K. Hinokitiol exhibits potent anticancer effects in human oral squamous cell carcinoma by inducing apoptosis and inhibition of cell migration. J Oral Maxillofac Surg Med Pathol 2025, 37, 1038–1043. [CrossRef]
- Koyanagi, K.; Kataoka, K.; Yoshimatsu, H.; Fujihashi, K.; Miyake, T. Human salivary protein-derived peptides specific-salivary SIgA antibodies enhanced by nasal double DNA adjuvant in mice play an essential role in preventing Porphyromonas gingivalis colonization: an in-vitro study. BMC Oral Health 2023, 23, 123. [CrossRef]
- Sekine, S.; Kataoka, K.; Fukuyama, Y.; Adachi, Y.; Davydova, J.; Yamamoto, M.; Kobayashi, R.; Fujihashi, K.; Suzuki, H.; Curiel, D.T.; Shizukuishi, S.; McGhee, J.R.; Fujihashi, K. A novel adenovirus expressing Flt3 ligand enhances mucosal immunity by inducing mature nasopharyngeal-associated lymphoreticular tissue dendritic cell migration. J Immunol 2008, 180, 8126–8134. [CrossRef]
- Osawa, K.; Matsumoto, T.; Maruyama, T.; Takiguchi, T.; Takazoe, I. Studies the antibacterial activity of plant extracts and their constituents against periodontopathic bacteria. Bull Tokyo Dent Coll 1990, 31, 17–21.
- Yamano, H.; Yamazaki, T.; Sato, K.; Shiga, S.; Hagiwara, T.; Ouchi, K.; Kishimoto, T. In vitro inhibitory effects of hinokitiol on proliferation of Chlamydia trachomatis. Antimicrob Agents Chemother 2005, 49, 2519–2521. [CrossRef]
- Byeon, S.E.; Lee, Y.G.; Kim, J.-C.; Han, J.G.; Lee, H.Y.; Cho, J.Y. Hinokitiol, a natural tropolone derivative, inhibits TNF-alpha production in LPS-activated macrophages via suppression of NF-kB. Planta Med 2008, 74, 828–833. [CrossRef]
- Roberts-Thomson, I.C.; Whittingham, S.; Youngchaiyud, U.; Mackay, I.R. Aging, immune response, and mortality. Lancet 1974, 2, 368–370. [CrossRef]
- Kataoka, K.; McGhee, J.R.; Kobayashi, R.; Fujihashi, K.; Shizukuishi, S.; Fujihashi, K. Nasal Flt3 ligand cDNA elicits CD11c+ CD8+ dendritic cells for enhanced mucosal immunity. J Immunol 2004, 172, 3612–3619. [CrossRef]
- Miletic, I.D.; Schiffman, S.S.; Miletic, V.D.; Sattely-Miller, E.A. Salivary IgA rate in young and elderly persons. Physiol Behav 1996, 60, 243–248. [CrossRef]
- Evans, P.; Der, G.; Ford, G.; Hucklebridge, F.; Hunt, K.; Lambert, S. Social class, sex, and age differences in mucosal immunity in a large community sample. Brain Behav Immun 2000, 14, 41–48. [CrossRef]
- Kurosaki, T.; Kometani, K.; Ise, W. Memory B cells. Nat Rev Immunol 2015, 15, 149–159. [CrossRef]
- Kiyono, H.; Fukuyama, S. NALT-versus Peyer’s-patch-mediated mucosal immunity. Nat Rev Immunol 2004, 4, 699–710. [CrossRef]
- Suzuki, K.; Maruya, M.; Kawamoto, S.; Sitnik, K.; Kitamura, H.; Agace, W.W.; Fagarasan, S. The sensing of environmental stimuli by follicular dendritic cells promotes immunoglobulin A generation in gut. Immunity 2010, 33, 71–83. [CrossRef]
- Belkaid, Y.; Hand, T.W. Role of the microbiota in immunity and inflammation. Cell 2014, 157, 121–141. [CrossRef]
- Ramos, MJJ. Microinjection of NMDA-neurotoxin into the superior salivatory nucleus of the rat: Short-term secretory and long-term drinking behavior effects. Physiol Behav 2023, 269. [CrossRef]
- Proctor, GB.; Carpenter, GH. Regulation of salivary gland function by autonomic nerves. Auton Neurosci 2007, 133, 3-18. [CrossRef]
- Ueda, H.; Mitoh, Y.; Fujita, M.; Kobayashi, M.; Yamashiro, T.; Sugimoto, T.; Ichikawa, H.; Matsuo, R. Muscarinic receptor immunoreactivity in the superior salivatory nucleus neurons innervating the salivary glands of the rat. Neurosci Lett 2011, 15, 42-46. [CrossRef]
- Madsen, BK.; Hischer, M.; Zetner, D.; Rosenberg, J. Adverse reactions of dimethyl sulfoxide in humans: a systematic review. F1000Res 2018, 7, 1746. [CrossRef]
- Arranz, E.; Mahony, SO.; Barton, JR.; Ferguson, A. Immunosenescence and mucosal immunity: significant effects of old age on secretory IgA concentrations and intraepithelial lymphocyte counts. Gut 1992, 33, 882-886. [CrossRef]
- Zheng, H.; Zhang, C.; Wang, Q.; Feng, S.; Fang, Y.; Zhang, S. The impact of aging on intestinal mucosal immune function and clinical applications. Front Immunol 2022, 13. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



