Submitted:
29 April 2026
Posted:
29 April 2026
You are already at the latest version
Abstract
Keywords:
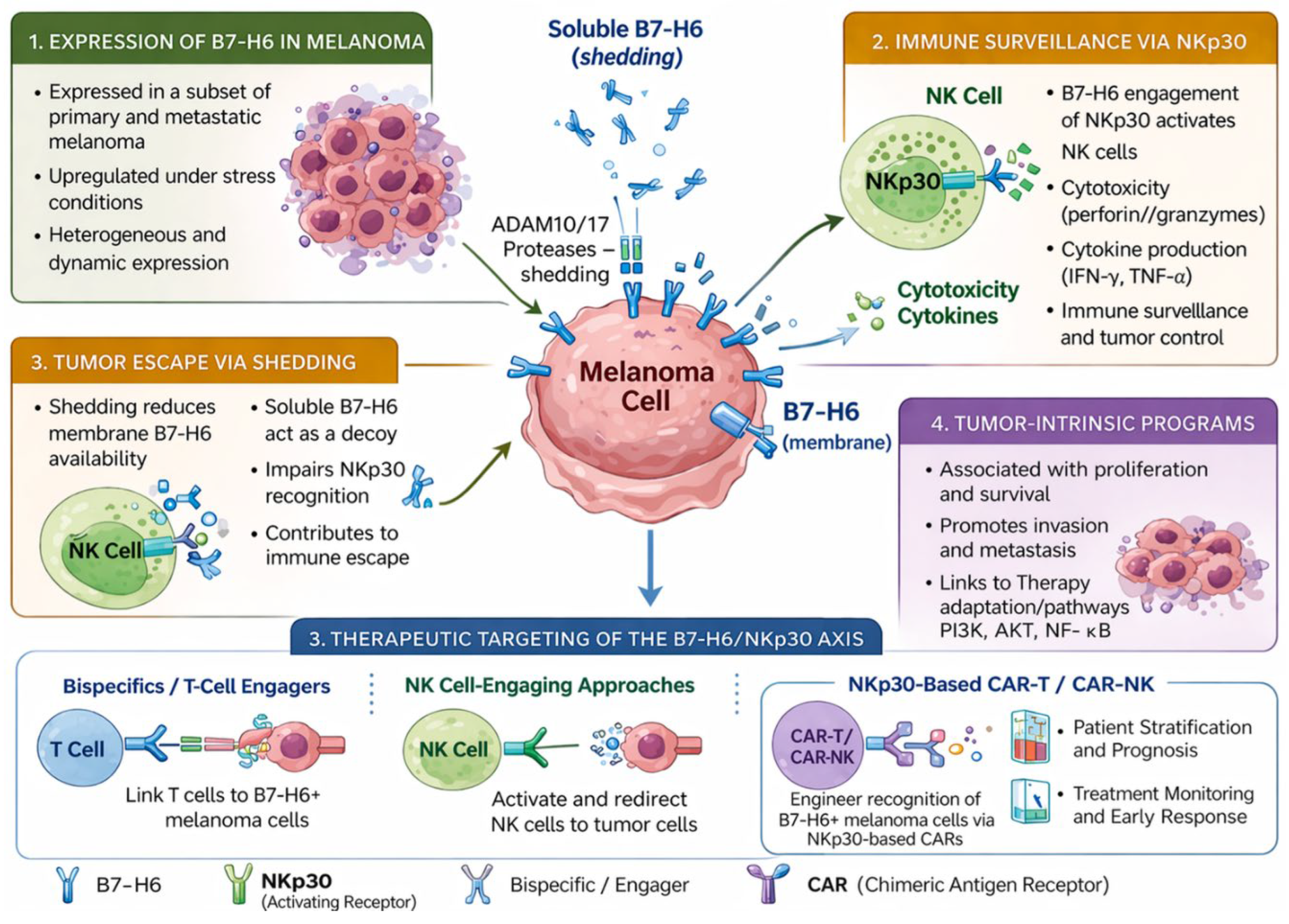
1. Introduction
2. B7-H6/NKp30 Axis in Immune Surveillance in Melanoma
3. Tumor Escape Mechanisms: Shedding, Soluble B7-H6, and Target Loss
4. Regulation and Tumor-Intrinsic Functions
5. Expression Landscape and Biomarker Opportunities
6. Therapeutic Targeting of the B7-H6/NKp30 Axis
6.1. T-cell Redirection and Bispecific Engagers
6.2. NK Cell–Engaging Approaches
6.3. NKp30-Based CAR Therapies
7. Combination Strategies and Clinical Positioning
8. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| B7-H6 | B7 homolog 6 |
| NKp30 | Natural cytotoxicity receptor 3 (NCR3) |
| ADAM10 | A disintegrin and metalloproteinase domain-containing protein 10 |
| ADAM17 | A disintegrin and metalloproteinase domain-containing protein 17 |
| CAR-T | Chimeric antigenic receptor T-cell therapy |
| c-Myc | Cellular homolog of v-Myc oncogene |
| PERK | Protein kinase R (PKR)-like endoplasmic reticulum kinase |
| CRISPR/Cas9 | Clustered regularly interspaced short palindromic repeat/CRISPR-associated protein 9 |
| HDAC2/3 | Histone deacetylase 2/3 |
References
- Haugh, A.M.; Salama, A.K.S.; Johnson, D.B. Advanced Melanoma: Resistance Mechanisms to Current Therapies. Hematol Oncol Clin North Am 2021, 35, 111-128. [CrossRef]
- Fenton, S.E.; Sosman, J.A.; Chandra, S. Resistance mechanisms in melanoma to immuneoncologic therapy with checkpoint inhibitors. Cancer Drug Resist 2019, 2, 744-761. [CrossRef]
- Brandt, C.S.; Baratin, M.; Yi, E.C.; Kennedy, J.; Gao, Z.; Fox, B.; Haldeman, B.; Ostrander, C.D.; Kaifu, T.; Chabannon, C.; et al. The B7 family member B7-H6 is a tumor cell ligand for the activating natural killer cell receptor NKp30 in humans. J Exp Med 2009, 206, 1495-1503. [CrossRef]
- Li, Y.; Wang, Q.; Mariuzza, R.A. Structure of the human activating natural cytotoxicity receptor NKp30 bound to its tumor cell ligand B7-H6. J Exp Med 2011, 208, 703-714. [CrossRef]
- Pinheiro, P.F.; Justino, G.C.; Marques, M.M. NKp30 - A prospective target for new cancer immunotherapy strategies. Br J Pharmacol 2020, 177, 4563-4580. [CrossRef]
- Kaifu, T.; Escalière, B.; Gastinel, L.N.; Vivier, E.; Baratin, M. B7-H6/NKp30 interaction: a mechanism of alerting NK cells against tumors. Cell Mol Life Sci 2011, 68, 3531-3539. [CrossRef]
- Schlecker, E.; Fiegler, N.; Arnold, A.; Altevogt, P.; Rose-John, S.; Moldenhauer, G.; Sucker, A.; Paschen, A.; von Strandmann, E.P.; Textor, S.; et al. Metalloprotease-mediated tumor cell shedding of B7-H6, the ligand of the natural killer cell-activating receptor NKp30. Cancer Res 2014, 74, 3429-3440. [CrossRef]
- Jerby-Arnon, L.; Shah, P.; Cuoco, M.S.; Rodman, C.; Su, M.J.; Melms, J.C.; Leeson, R.; Kanodia, A.; Mei, S.; Lin, J.R.; et al. A Cancer Cell Program Promotes T Cell Exclusion and Resistance to Checkpoint Blockade. Cell 2018, 175, 984-997.e924. [CrossRef]
- Landsberg, J.; Kohlmeyer, J.; Renn, M.; Bald, T.; Rogava, M.; Cron, M.; Fatho, M.; Lennerz, V.; Wölfel, T.; Hölzel, M.; et al. Melanomas resist T-cell therapy through inflammation-induced reversible dedifferentiation. Nature 2012, 490, 412-416. [CrossRef]
- Sade-Feldman, M.; Jiao, Y.J.; Chen, J.H.; Rooney, M.S.; Barzily-Rokni, M.; Eliane, J.-P.; Bjorgaard, S.L.; Hammond, M.R.; Vitzthum, H.; Blackmon, S.M.; et al. Resistance to checkpoint blockade therapy through inactivation of antigen presentation. Nature Communications 2017, 8, 1136. [CrossRef]
- Sottile, R.; Pangigadde, P.N.; Tan, T.; Anichini, A.; Sabbatino, F.; Trecroci, F.; Favoino, E.; Orgiano, L.; Roberts, J.; Ferrone, S.; et al. HLA class I downregulation is associated with enhanced NK-cell killing of melanoma cells with acquired drug resistance to BRAF inhibitors. Eur J Immunol 2016, 46, 409-419. [CrossRef]
- Lehmann, J.; Caduff, N.; Krzywińska, E.; Stierli, S.; Salas-Bastos, A.; Loos, B.; Levesque, M.P.; Dummer, R.; Stockmann, C.; Münz, C.; et al. Escape from NK cell tumor surveillance by NGFR-induced lipid remodeling in melanoma. Sci Adv 2023, 9, eadc8825. [CrossRef]
- Porgador, A.; Mandelboim, O.; Restifo, N.P.; Strominger, J.L. Natural killer cell lines kill autologous beta2-microglobulin-deficient melanoma cells: implications for cancer immunotherapy. Proc Natl Acad Sci U S A 1997, 94, 13140-13145. [CrossRef]
- Mohammadi, A.; Najafi, S.; Amini, M.; Baradaran, B.; Firouzamandi, M. B7H6 silencing increases chemosensitivity to dacarbazine and suppresses cell survival and migration in cutaneous melanoma. Melanoma Res 2023, 33, 173-183. [CrossRef]
- Zhang, W.; Auguste, A.; Liao, X.; Walterskirchen, C.; Bauer, K.; Lin, Y.H.; Yang, L.; Sayedian, F.; Fabits, M.; Bergmann, M.; et al. A Novel B7-H6-Targeted IgG-Like T Cell-Engaging Antibody for the Treatment of Gastrointestinal Tumors. Clin Cancer Res 2022, 28, 5190-5201. [CrossRef]
- Obiedat, A.; Charpak-Amikam, Y.; Tai-Schmiedel, J.; Seidel, E.; Mahameed, M.; Avril, T.; Stern-Ginossar, N.; Springuel, L.; Bolsée, J.; Gilham, D.E.; et al. The integrated stress response promotes B7H6 expression. J Mol Med (Berl) 2020, 98, 135-148. [CrossRef]
- Laskowski, T.J.; Biederstädt, A.; Rezvani, K. Natural killer cells in antitumour adoptive cell immunotherapy. Nature Reviews Cancer 2022, 22, 557-575. [CrossRef]
- Lanier, L.L. NK cell recognition. Annu Rev Immunol 2005, 23, 225-274. [CrossRef]
- Kärre, K. Natural killer cell recognition of missing self. Nat Immunol 2008, 9, 477-480. [CrossRef]
- Kageshita, T.; Hirai, S.; Ono, T.; Hicklin, D.J.; Ferrone, S. Down-regulation of HLA class I antigen-processing molecules in malignant melanoma: association with disease progression. Am J Pathol 1999, 154, 745-754. [CrossRef]
- Morgado, S.; Sanchez-Correa, B.; Casado, J.G.; Duran, E.; Gayoso, I.; Labella, F.; Solana, R.; Tarazona, R. NK cell recognition and killing of melanoma cells is controlled by multiple activating receptor-ligand interactions. J Innate Immun 2011, 3, 365-373. [CrossRef]
- Textor, S.; Bossler, F.; Henrich, K.O.; Gartlgruber, M.; Pollmann, J.; Fiegler, N.; Arnold, A.; Westermann, F.; Waldburger, N.; Breuhahn, K.; et al. The proto-oncogene Myc drives expression of the NK cell-activating NKp30 ligand B7-H6 in tumor cells. Oncoimmunology 2016, 5, e1116674. [CrossRef]
- Balsamo, M.; Scordamaglia, F.; Pietra, G.; Manzini, C.; Cantoni, C.; Boitano, M.; Queirolo, P.; Vermi, W.; Facchetti, F.; Moretta, A.; et al. Melanoma-associated fibroblasts modulate NK cell phenotype and antitumor cytotoxicity. Proc Natl Acad Sci U S A 2009, 106, 20847-20852. [CrossRef]
- Jilani, S.; Saco, J.D.; Mugarza, E.; Pujol-Morcillo, A.; Chokry, J.; Ng, C.; Abril-Rodriguez, G.; Berger-Manerio, D.; Pant, A.; Hu, J.; et al. CAR-T cell therapy targeting surface expression of TYRP1 to treat cutaneous and rare melanoma subtypes. Nat Commun 2024, 15, 1244. [CrossRef]
- Anikeeva, N.; Panteleev, S.; Mazzanti, N.W.; Terai, M.; Sato, T.; Sykulev, Y. Efficient killing of tumor cells by CAR-T cells requires greater number of engaged CARs than TCRs. J Biol Chem 2021, 297, 101033. [CrossRef]
- Tang, J.; Gong, Y.; Ma, X. Bispecific Antibodies Progression in Malignant Melanoma. Front Pharmacol 2022, 13, 837889. [CrossRef]
- Ponath, V.; Hoffmann, N.; Bergmann, L.; Mäder, C.; Alashkar Alhamwe, B.; Preußer, C.; Pogge von Strandmann, E. Secreted Ligands of the NK Cell Receptor NKp30: B7-H6 Is in Contrast to BAG6 Only Marginally Released via Extracellular Vesicles. Int J Mol Sci 2021, 22. [CrossRef]
- Gutierrez-Franco, J.; Hernandez-Gutierrez, R.; Bueno-Topete, M.R.; Haramati, J.; Navarro-Hernandez, R.E.; Escarra-Senmarti, M.; Vega-Magaña, N.; Del Toro-Arreola, A.; Pereira-Suarez, A.L.; Del Toro-Arreola, S. Characterization of B7H6, an endogenous ligand for the NK cell activating receptor NKp30, reveals the identity of two different soluble isoforms during normal human pregnancy. Immunobiology 2018, 223, 57-63. [CrossRef]
- Lee, S.; Kim, J.H.; Jang, I.H.; Jo, S.; Lee, S.Y.; Oh, S.C.; Kim, S.M.; Kong, L.; Ko, J.; Kim, T.D. Harnessing B7-H6 for Anticancer Immunotherapy: Expression, Pathways, and Therapeutic Strategies. Int J Mol Sci 2024, 25. [CrossRef]
- Reuben, A.; Spencer, C.N.; Prieto, P.A.; Gopalakrishnan, V.; Reddy, S.M.; Miller, J.P.; Mao, X.; De Macedo, M.P.; Chen, J.; Song, X.; et al. Genomic and immune heterogeneity are associated with differential responses to therapy in melanoma. npj Genomic Medicine 2017, 2, 10. [CrossRef]
- Cao, G.; Wang, J.; Zheng, X.; Wei, H.; Tian, Z.; Sun, R. Tumor Therapeutics Work as Stress Inducers to Enhance Tumor Sensitivity to Natural Killer (NK) Cell Cytolysis by Up-regulating NKp30 Ligand B7-H6. J Biol Chem 2015, 290, 29964-29973. [CrossRef]
- Chen, H.; Zhang, Y.; Shen, Y.; Jiang, L.; Zhang, G.; Zhang, X.; Xu, Y.; Fu, F. Deficiency of N-linked glycosylation impairs immune function of B7-H6. Front Immunol 2023, 14, 1255667. [CrossRef]
- Rambow, F.; Marine, J.C.; Goding, C.R. Melanoma plasticity and phenotypic diversity: therapeutic barriers and opportunities. Genes Dev 2019, 33, 1295-1318. [CrossRef]
- Vendramin, R.; Katopodi, V.; Cinque, S.; Konnova, A.; Knezevic, Z.; Adnane, S.; Verheyden, Y.; Karras, P.; Demesmaeker, E.; Bosisio, F.M.; et al. Activation of the integrated stress response confers vulnerability to mitoribosome-targeting antibiotics in melanoma. J Exp Med 2021, 218. [CrossRef]
- Čaval, T.; Alisson-Silva, F.; Schwarz, F. Roles of glycosylation at the cancer cell surface: opportunities for large scale glycoproteomics. Theranostics 2023, 13, 2605-2615. [CrossRef]
- Rodriguez, E. Tumor Glycosylation: A Main Player in the Modulation of Immune Responses. Eur J Immunol 2025, 55, e202451318. [CrossRef]
- Givi, S.; Lohnes, B.J.; Ebrahimi, S.; Riedel, S.; Khokhali, S.; Khan, S.A.; Keller, M.; Wölfel, C.; Echchannaoui, H.; Bockamp, E.; et al. CRISPR/Cas9 TCR-Edited NKp30 CAR T Cells Exhibit Superior Anti-Tumor Immunity to B7H6-Expressing Leukemia and Melanoma. Int J Mol Sci 2025, 26. [CrossRef]
- Rusakiewicz, S.; Perier, A.; Semeraro, M.; Pitt, J.M.; Pogge von Strandmann, E.; Reiners, K.S.; Aspeslagh, S.; Pipéroglou, C.; Vély, F.; Ivagnes, A.; et al. NKp30 isoforms and NKp30 ligands are predictive biomarkers of response to imatinib mesylate in metastatic GIST patients. Oncoimmunology 2017, 6, e1137418. [CrossRef]
- Messaoudene, M.; Fregni, G.; Enot, D.; Jacquelot, N.; Neves, E.; Germaud, N.; Garchon, H.J.; Boukouaci, W.; Tamouza, R.; Chanal, J.; et al. NKp30 isoforms and NKp46 transcripts in metastatic melanoma patients: Unique NKp30 pattern in rare melanoma patients with favorable evolution. Oncoimmunology 2016, 5, e1154251. [CrossRef]
- Zhang, T.; Wu, M.R.; Sentman, C.L. An NKp30-based chimeric antigen receptor promotes T cell effector functions and antitumor efficacy in vivo. J Immunol 2012, 189, 2290-2299. [CrossRef]
- Butler, S.E.; Brog, R.A.; Chang, C.H.; Sentman, C.L.; Huang, Y.H.; Ackerman, M.E. Engineering a natural ligand-based CAR: directed evolution of the stress-receptor NKp30. Cancer Immunol Immunother 2022, 71, 165-176. [CrossRef]
- Wu, M.R.; Zhang, T.; Gacerez, A.T.; Coupet, T.A.; DeMars, L.R.; Sentman, C.L. B7H6-Specific Bispecific T Cell Engagers Lead to Tumor Elimination and Host Antitumor Immunity. J Immunol 2015, 194, 5305-5311. [CrossRef]
- Pekar, L.; Klausz, K.; Busch, M.; Valldorf, B.; Kolmar, H.; Wesch, D.; Oberg, H.H.; Krohn, S.; Boje, A.S.; Gehlert, C.L.; et al. Affinity Maturation of B7-H6 Translates into Enhanced NK Cell-Mediated Tumor Cell Lysis and Improved Proinflammatory Cytokine Release of Bispecific Immunoligands via NKp30 Engagement. J Immunol 2021, 206, 225-236. [CrossRef]
- Lee, J.H.; Shklovskaya, E.; Lim, S.Y.; Carlino, M.S.; Menzies, A.M.; Stewart, A.; Pedersen, B.; Irvine, M.; Alavi, S.; Yang, J.Y.H.; et al. Transcriptional downregulation of MHC class I and melanoma de- differentiation in resistance to PD-1 inhibition. Nature Communications 2020, 11, 1897. [CrossRef]
- Ma, X.; He, H.; Zhu, Y.; Zuo, D.; Wang, F.; Feng, M.; Ji, K.; Chen, X. Dual T/NK cell engagement via B7-H6-targeted bispecific antibodies and IL-15 eradicates chemo-resistant solid tumors. Front Immunol 2025, 16, 1625813. [CrossRef]
- Fiegler, N.; Textor, S.; Arnold, A.; Rölle, A.; Oehme, I.; Breuhahn, K.; Moldenhauer, G.; Witzens-Harig, M.; Cerwenka, A. Downregulation of the activating NKp30 ligand B7-H6 by HDAC inhibitors impairs tumor cell recognition by NK cells. Blood 2013, 122, 684-693. [CrossRef]
| Study | Key melanoma-linked observation | Targeting implication | Main limitation |
|---|---|---|---|
| Brandt et al. (2009) [3] | Identifies B7-H6 as the human tumor-cell ligand for NKp30; melanoma cell lines were among the first reported to express the target. | Establishes melanoma as a plausible cell-surface targeting context for the NKp30/B7-H6 axis. | Primarily discovery-phase; limited melanoma-specific clinical annotation. |
| Schlecker et al. (2014) [7] | Shows ADAM10/17-mediated shedding of B7-H6, elevated soluble B7-H6 in melanoma serum, and increased tissue expression in melanoma specimens in situ. | Directly links melanoma biology to target loss, soluble biomarker potential, and a defined resistance mechanism. | Subset-based serum analysis; no therapeutic intervention study. |
| Messaoudene et al. (2016) [39] | Defines unusual NKp30 transcript patterns in metastatic melanoma and rare mucosal melanoma. | Supports the importance of the receptor side in clinically distinct melanoma states. | Does not directly measure tumor-cell B7-H6. |
| Obiedat et al. (2020) [16] | Integrates stress response and PERK signaling increased B7-H6 expression; ISR inhibitor reversed this effect. | Provides a rationale for pharmacologic target induction and combination design. | Preclinical and not melanoma-specific, but mechanistically informative. |
| Mohammadi et al. (2023) [14] | B7-H6 silencing in A375 melanoma cells reduces survival, migration, clonogenicity, and increased dacarbazine sensitivity. | Suggests that B7-H6-positive melanoma states may have therapeutic importance beyond immune recognition. | Single-cell-line study; no in vivo melanoma validation. |
| Givi et al. (2025) [37] | Primary melanoma samples and melanoma cell lines express B7-H6; CRISPR/Cas9-edited NKp30 CAR-T cells showed in vitro cytotoxicity and in vivo control in an A375 xenograft model. | Strengthens the translational case for B7-H6-directed cellular therapy and highlights protein-level heterogeneity. | Small melanoma sample set; broader clinical heterogeneity remains untested. |
| Platform | Mechanistic concept | Development strength | Key caveat |
|---|---|---|---|
| Native NKp30-based CAR-T | Uses the natural NKp30 extracellular domain to recognize tumor-cell B7-H6 | In vivo proof-of-concept: receptor biology is directly linked to targeting | Native receptor affinity may be modest; potential off-tumor concerns remain |
| Affinity-matured NKp30 CARs | Directs evolution generates higher-affinity NKp30 variants for CAR recognition | Expands binding range and may improve performance in lower-density target states | Still preclinical; performance in melanoma-specific models remains limited |
| B7-H6/CD3 T-cell engager BI 765049 | Bispecific binder links B7-H6-positive tumor cells to CD3-positive T cells | Demonstrates the feasibility of industrial and clinical development for the target | Most public efficacy data are outside the melanoma setting |
| CRISPR/Cas9 TCR-edited NKp30 CAR-T | Engineered T cells combine NKp30-based recognition with TCR deletion | Direct melanoma activity has been shown in vitro and in vivo; the platform allows precise product engineering | Manufacturing, persistence, antigen heterogeneity, and safety questions remain |
| Dual T/NK + cytokine-anchored strategies | Combine B7-H6-targeted immune redirection with tumor-localized IL-15/IL-15Rα signaling to amplify effector-cell activation at B7-H6-positive tumor sites | Extends the platform beyond target binding alone; supports both NK- and T-cell activity, with particularly strong preclinical activity in NK-engaging formats in resistant solid-tumor models | Not melanoma-specific; cytokine dosing, safety, and generalizability remain to be defined |
| Antigen-priming approaches | Stress-inducing therapies or ISR activation may raise B7-H6 density before cell-based or engager-based treatment | Creates a route to rational combinations and biomarker-guided scheduling | Timing, dosing, and simultaneous induction of shedding require resolution |
| Shedding-aware strategies | Reduce ADAM10/17-mediated B7-H6 shedding and consider soluble B7-H6 as a complementary pharmacodynamic biomarker | Strong mechanistic rationale based on melanoma serum data and established shedding biology | No melanoma-specific intervention study has yet shown clinical benefit, and direct metalloprotease inhibition remains difficult to implement therapeutically |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




