Submitted:
29 April 2026
Posted:
29 April 2026
You are already at the latest version
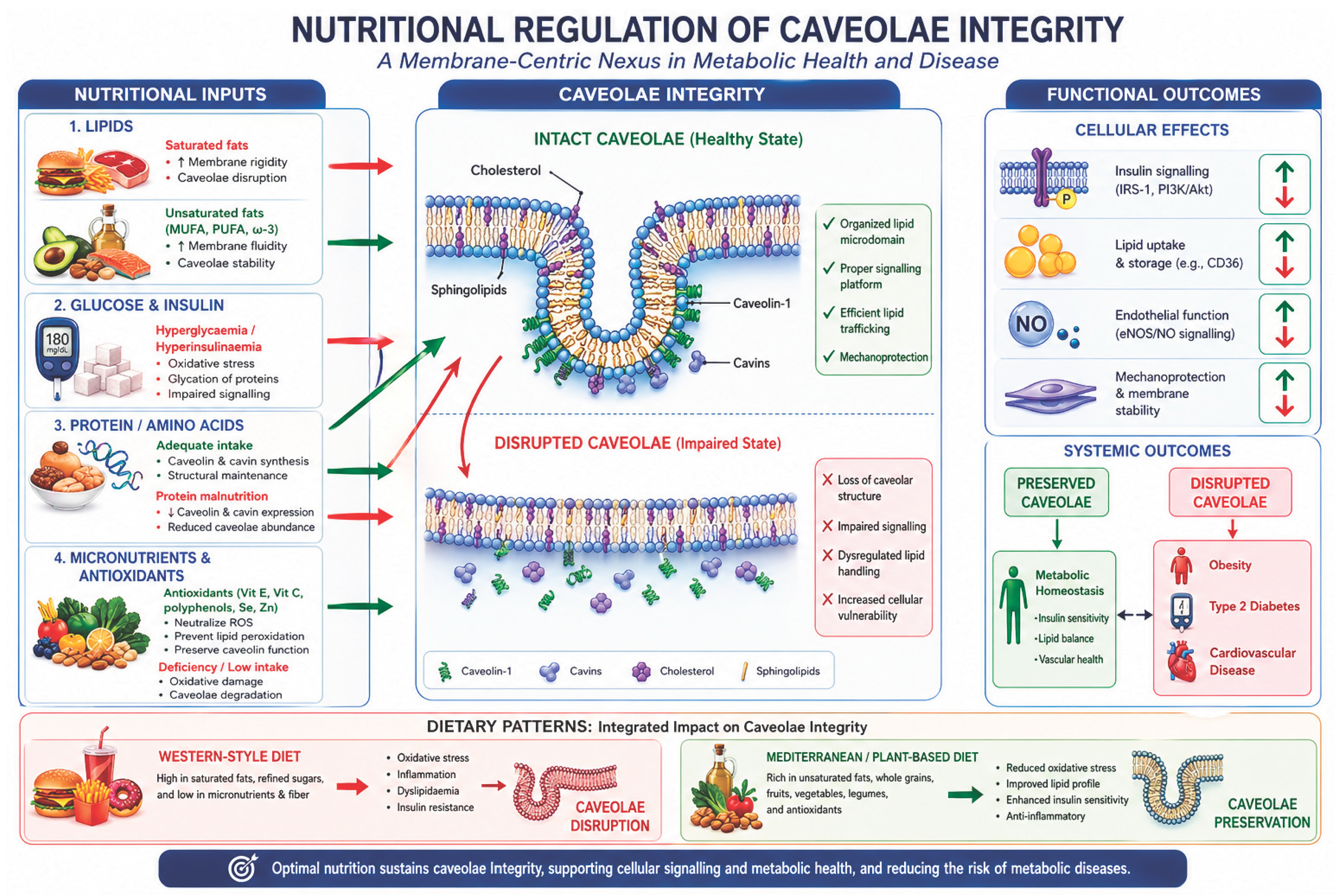
Abstract
Keywords:
Introduction
1. Lipid Composition as a Primary Determinant of Caveolae Stability
2. Glucose and Insulin Signaling: Metabolic Inputs to Caveolae Function
3. Protein and Amino Acid Availability: Structural and Regulatory Roles
4. Micronutrients and Antioxidant defense: Protecting Caveolar Integrity
5. Dietary Patterns and Systemic Effects on Caveolae
6. Caveolae as Nutrient Sensors and Mediators
7. Pathophysiological Implications
8. Future Directions and Research Gaps
Conclusions
Finding
Conflicts of interest
References
- Stea, D.M.; D’Alessio, A. Caveolae: Metabolic Platforms at the Crossroads of Health and Disease. Int. J. Mol. Sci. 2025, Vol. 26. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Lim, Y.W.; McMahon, K.A.; Martel, N.; Rae, J.; Lo, H.P.; et al. Pro-ferroptotic lipids as key control points for caveola formation and disassembly. Cell. Rep. [Internet] 2025, 44(6), 115789. Available online: https://www.sciencedirect.com/science/article/pii/S2211124725005601. [CrossRef] [PubMed]
- Zeng, W.; Cai, N.; Liu, J.; Liu, K.; Lin, S.; Yang, X.; et al. Caveolin-1 deficiency Improved Glucose Metabolism via Modulation of Beta-cell Autophagy on High-Fat-Diet Mice. J. Biol. Chem. [Internet] Available from. 2026. [Google Scholar] [CrossRef] [PubMed]
- Carpentier, M.; Omrane, M.; Shaaban, R.; Träger, J.; El Khallouki, N.; Le Lay, S.; et al. Seipin Governs caveolin-1 trafficking through modulating sphingolipid-glycerolipid balance. Cell. Rep. [Internet] Available from. 2025, 44(10). [Google Scholar] [CrossRef] [PubMed]
- Lee-Okada, H.C.; Xue, C.; Yokomizo, T. Recent advances on the physiological and pathophysiological roles of polyunsaturated fatty acids and their biosynthetic pathway. Biochim Biophys. Acta-Mol. Cell. Biol. Lipids [Internet] 2025, 1870(1), 159564. Available online: https://www.sciencedirect.com/science/article/pii/S1388198124001148. [CrossRef] [PubMed]
- Warda, M.; Tekin, S.; Gamal, M.; Khafaga, N.; Çelebi, F.; Tarantino, G. Lipid rafts: novel therapeutic targets for metabolic, neurodegenerative, oncological, and cardiovascular diseases. Lipids Health Dis. [Internet] Available from. 2025, 24(1), 147. [Google Scholar] [CrossRef] [PubMed]
- zheng, Zhang W. An association of metabolic syndrome constellation with cellular membrane caveolae. Pathobiol. Aging Age-Relat. Dis. 2014, 4(1). [Google Scholar] [CrossRef] [PubMed]
- Russo, P.; Sirangelo, I.; Siani, A. Dietary Advanced Glycation End Products (dAGEs): Pathogenesis and nutritional strategies for health longevity-A critical view. Nutr. Metab. Cardiovasc Dis. [Internet] Available from. 2026, 36(5). [Google Scholar] [CrossRef] [PubMed]
- Parton, R.G.; Kozlov, M.M.; Lundmark, R. A lipid-centric view of endocytosis by caveolae. Nat Cell Biol [Internet]. Available from. 2026. [CrossRef]
- Russo, L.; Babboni, S.; Andreassi, M.G.; Daher, J.; Canale, P.; Del Turco, S.; et al. Treating Metabolic Dysregulation and Senescence by Caloric Restriction: Killing Two Birds with One Stone? Antioxidants 2025, Vol. 14, 99. [Google Scholar] [CrossRef] [PubMed]
- Mei, J.; Yang, F.; yuan; Gong, Q. Branched-chain amino acids and insulin resistance in type 2 diabetes: from metabolic dysregulation to therapeutic targets. Front Endocrinol (Lausanne) [Internet]. 2026, Volume 16. Available online: https://www.frontiersin.org/journals/endocrinology/articles/10.3389/fendo.2025.1643231.
- Wu, Y.; Lim, Y.W.; Stroud, D.A.; Martel, N.; Hall, T.E.; Lo, H.P.; et al. Caveolae sense oxidative stress through membrane lipid peroxidation and cytosolic release of CAVIN1 to regulate NRF2. Dev. Cell. 2023, 58(5). [Google Scholar] [CrossRef] [PubMed]
- Canner, S.W.; Zhu, F.; Feller, S.E.; Wassall, S.R. A Role for Lipid-Lipid Interactions in Vitamin E’s Function as a Membrane Antioxidant. Biophys. J. [Internet] Available from. 2019, 116(3), 227a. [Google Scholar] [CrossRef]
- Kalogerakou, T.; Antoniadou, M. The Role of Dietary Antioxidants, Food Supplements and Functional Foods for Energy Enhancement in Healthcare Professionals; Antioxidants, 2024; Vol. 13. [Google Scholar]
- Terao, J. Caveolae and caveolin-1 as targets of dietary polyphenols for protection against vascular endothelial dysfunction. J. Clin. Biochem Nutr. 2024, 75(1), 7–16. [Google Scholar] [CrossRef] [PubMed]
- de Oliveira, A.R.S.; Cruz, K.J.C.; Morais, J.B.S.; dos Santos, L.R.; de Sousa Melo, S.R.; Fontenelle, L.C.; et al. Magnesium, selenium and zinc deficiency compromises antioxidant defense in women with obesity. BioMetals [Internet] Available from. 2024, 37(6), 1551–63. [Google Scholar] [CrossRef] [PubMed]
- Liu, I.F.; Lin, T.C.; Wang, S.C.; Yen, C.H.; Li, C.Y.; Kuo, H.F.; et al. Long-term administration of Western diet induced metabolic syndrome in mice and causes cardiac microvascular dysfunction, cardiomyocyte mitochondrial damage, and cardiac remodeling involving caveolae and caveolin-1 expression. Biol. Direct 2023, 18(1). [Google Scholar] [CrossRef] [PubMed]
- Detopoulou, P.; Antonopoulou, S.; Douvogianni, P.; Demopoulos, C.A. The Mediterranean Diet and Cardiovascular Protection: Biochemical Mechanisms with Emphasis on Platelet-Activating Factor; Nutrients, 2026; Vol. 18. [Google Scholar]
- López-Moreno, M.; Fresán, U. Do the Health Benefits of the Mediterranean Diet Increase with a Higher Proportion of Whole Plant-Based Foods? Curr. Nutr. Rep. [Internet] Available from. 2025, 14(1), 52. [Google Scholar] [CrossRef] [PubMed]
- Vergara Nieto, Á.A.; Diaz, A.H.; Hernández, M.; Sagredo, D. A Narrative Review about Metabolic Pathways, Molecular Mechanisms and Clinical Implications of Intermittent Fasting as Autophagy Promotor. Curr. Nutr. Rep. [Internet] Available from. 2025, 14(1), 78. [Google Scholar] [CrossRef] [PubMed]
- Briand, N.; Prado, C.; Mabilleau, G.; Lasnier, F.; Le Lièpvre, X.; Covington, J.D.; et al. Caveolin-1 Expression and Cavin Stability Regulate Caveolae Dynamics in Adipocyte Lipid Store Fluctuation. Diabetes [Internet] Available from. 2014, 63(12), 4032–44. [Google Scholar] [CrossRef] [PubMed]
- Hwej, A.S.; Alsharif, M.; Al-Ferjani, A.; Kennedy, S. Regulation of NO Synthesis by Caveolin-1: A Review of Its Importance in Blood Vessels, Perivascular Adipose Tissue and in Atherosclerosis; Applied Biosciences, 2026; Vol. 5, p. 11. [Google Scholar]
- Zhang, W.Z. Chapter 7 - Pharmacologically targeting caveolae in metabolic diseases; de Oliveira MRBTPT in, M.D., Ed.; Academic Press, 2026; pp. 127–39. Available online: https://www.sciencedirect.com/science/article/pii/B9780443273704000123.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.



