Discussion
In this retrospective analysis of 248 intraoperative patients, arterial waveform–derived Pulse Energy Ratio (PER) demonstrated strong correlation with reference cardiac output. These findings support the hypothesis that PER, as a waveform-derived surrogate index, may reflect aspects of cardiovascular performance and vascular tone. Correlation strength improved with rolling averaging, suggesting that temporal smoothing reduces transient beat-to-beat variability while preserving the observed associations
These findings suggest that PER as a physiologically grounded, waveform-derived metric that may provide continuous insight into hemodynamic state using signals already available in routine clinical care. Unlike many existing cardiac output monitoring systems, PER does not require additional hardware, calibration, or invasive procedures beyond standard arterial pressure monitoring.
Understanding the Generation of Diastolic Pressure
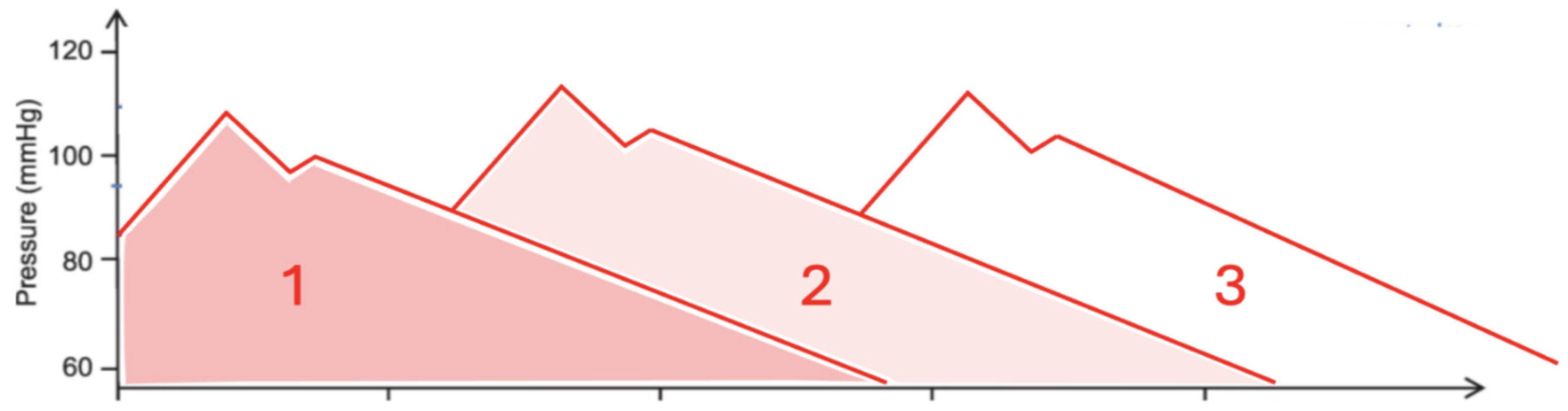
Diastolic pressure is not a static reservoir but the cumulative result of residual elastic energy from prior systolic ejections interacting with continuous distal runoff. Each cardiac cycle contributes to a temporally overlapping decay curve, such that the observed diastolic pressure represents a dynamic equilibrium between arterial recoil and peripheral flow. (
Figure 2)
Physiological Interpretation: Not All MAP Is Created Equal
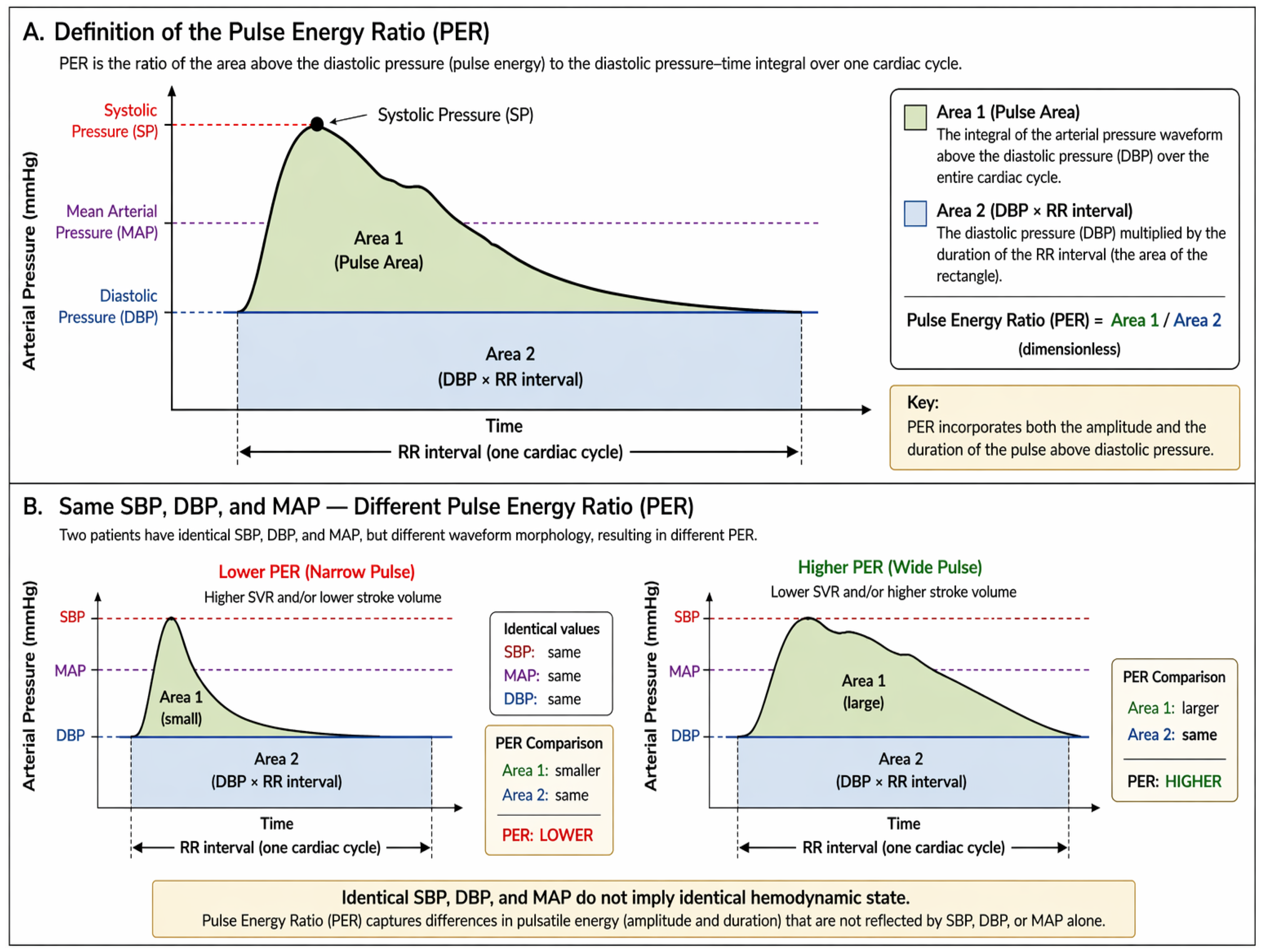
Mean arterial pressure remains the most commonly used endpoint for resuscitation and hemodynamic management. However, MAP alone provides limited insight into the underlying physiological determinants of circulation. Because MAP is governed by both cardiac output and systemic vascular resistance, identical MAP values may reflect markedly different hemodynamic states. One patient may maintain MAP through robust stroke volume and forward flow, while another may maintain the same pressure through vasoconstriction with a concomitantly reduced cardiac output.
The PER provides a continuous morphological assessment of the arterial waveform that reflects this distinction. Higher PER values are associated with broader, higher-amplitude arterial waveforms consistent with greater forward flow, while lower PER values are associated with narrower waveforms consistent with increased vascular tone or reduced stroke volume. In this way, PER may provide insight into the physiological "quality" of arterial pressure, helping to distinguish pressure supported by flow from pressure supported by vasoconstriction.
This distinction may be particularly important during resuscitation, where therapeutic interventions such as fluid administration, vasopressors, and inotropes may produce similar changes in MAP but have different effects on cardiac output and tissue perfusion. Continuous waveform-derived monitoring may therefore provide additional context for interpreting changes in arterial pressure and guiding therapeutic decisions.
Importance of Trending and Continuous Monitoring
From a clinical perspective, the most valuable feature of waveform-derived indices such as PER is their ability to provide continuous trending information. In hemodynamic monitoring, trends are often more informative than isolated values, as they reflect the physiologic response to interventions and allow early detection of evolving instability. (6)
Because PER can be derived continuously from the arterial waveform, directional changes may provide real-time insight into whether therapeutic interventions are improving or worsening cardiovascular performance. For example, an increase in PER following fluid administration may reflect improved stroke volume and forward flow, whereas a decrease in PER despite stable MAP may suggest worsening cardiac performance or increasing vascular tone. Similarly, an increase in MAP following vasopressor initiation accompanied by a decrease in PER may indicate ventriculo-arterial uncoupling, prompting reassessment of perfusion and cardiac function, while concordant increases in both MAP and PER would support true hemodynamic improvement.
Importantly, changes in PER may precede changes in conventional vital signs. Progressive reductions in PER may signal declining cardiac output or increasing vascular tone before a fall in MAP becomes apparent, offering an earlier warning of hemodynamic deterioration. This may be particularly valuable in perioperative and critical care settings, where rapid physiologic changes are common.
By enabling continuous, real-time assessment of physiologic response, PER provides an additional signal to guide fluid therapy, vasopressor titration, and inotropic support. Rather than relying solely on static pressure targets, clinicians may assess whether interventions are moving the patient toward improved hemodynamic conditions and adequate forward flow.
Increasing Capability of Basic Invasive Monitoring
A particularly important implication of this work is that PER requires only an adequate arterial pressure waveform. No additional sensors, calibration procedures, or invasive devices are necessary beyond standard arterial line monitoring, which is already widely used in perioperative and critical care settings.
Because PER is derived from waveform morphology, the analysis can be implemented in software and integrated directly into existing patient monitoring systems. This creates the potential for broad deployment without additional hardware costs or procedural risks. In principle, any monitor capable of displaying a continuous arterial waveform could incorporate PER analysis.
Furthermore, advances in computer vision and artificial intelligence may extend this concept even further. Waveform analysis could potentially be performed using camera-based systems capturing monitor displays, enabling retrospective or real-time analysis without direct integration into monitoring hardware. Such approaches could expand access to advanced hemodynamic interpretation in settings where proprietary monitoring platforms are unavailable.
These possibilities suggest a pathway toward democratizing hemodynamic monitoring. Rather than requiring specialized and often expensive cardiac output devices, physiologic insight could be derived from routinely available waveform data using software-based analysis. This approach may be particularly valuable in resource-limited environments, smaller hospitals, or settings where advanced hemodynamic monitoring is not routinely available.
Comparison with Existing Monitoring Approaches
Traditional cardiac output monitoring systems rely on thermodilution, pulse contour analysis, bioreactance, or Doppler-based approaches, often requiring additional hardware, calibration, or operator expertise. While these technologies provide valuable information, they may be limited by cost, invasiveness, or operational complexity.
In contrast, PER is derived directly from the arterial waveform without calibration or model-based assumptions regarding arterial compliance. This simplicity may offer advantages in robustness, ease of implementation, and interpretability. Additionally, because PER reflects waveform morphology rather than absolute volumetric measurements, it may be particularly useful for trending physiologic changes over time.
The correlation magnitudes observed in this study are comparable to those reported for uncalibrated pulse contour methods in perioperative populations. (7,8) While correlation does not imply interchangeability with cardiac output measurement, these findings support the potential clinical utility of waveform-derived morphology metrics as physiologic surrogates.
Limitations
This study has several limitations. First, the retrospective observational design limits causal inference and does not allow assessment of clinical outcomes associated with PER-guided management. Second, correlation analysis does not establish interchangeability with cardiac output measurement, and future studies using agreement and trending analyses are warranted. Thirdly, the reference cardiac output was derived from a semi-calibrated pulse contour system, which may introduce measurement variability, particularly between calibration intervals. Fourth, patient-level clinical variables such as vasopressor use, fluid administration, and surgical factors were not controlled for in this analysis. Fifth, this correlation was done using radial artery catheter data, while many resuscitationists have transitioned to using femoral arterial lines, hence this should be repeated with more central catheters.
Additionally, arterial compliance varies between patients and may influence waveform morphology independently of cardiac output or systemic vascular resistance. Future studies may explore whether patient-specific factors influence PER performance.
Finally, this dataset was derived from intraoperative patients at a single institution, which may limit generalizability to other clinical environments such as intensive care or emergency settings.
Future Directions
These findings support further investigation of waveform-derived Pulse Energy Ratio in prospective clinical studies. Future work may include:
Prospective validation studies
Trending and concordance analysis
Evaluation in critically ill populations
Assessment of therapeutic responsiveness
Integration into bedside monitoring systems
Additionally, integration of PER into machine learning–based waveform analysis platforms may enable multi-parameter hemodynamic interpretation and automated clinical decision support.