Submitted:
28 April 2026
Posted:
28 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. The Evaluation of Oculomotor Dysfunction in Parkinson’s Disease
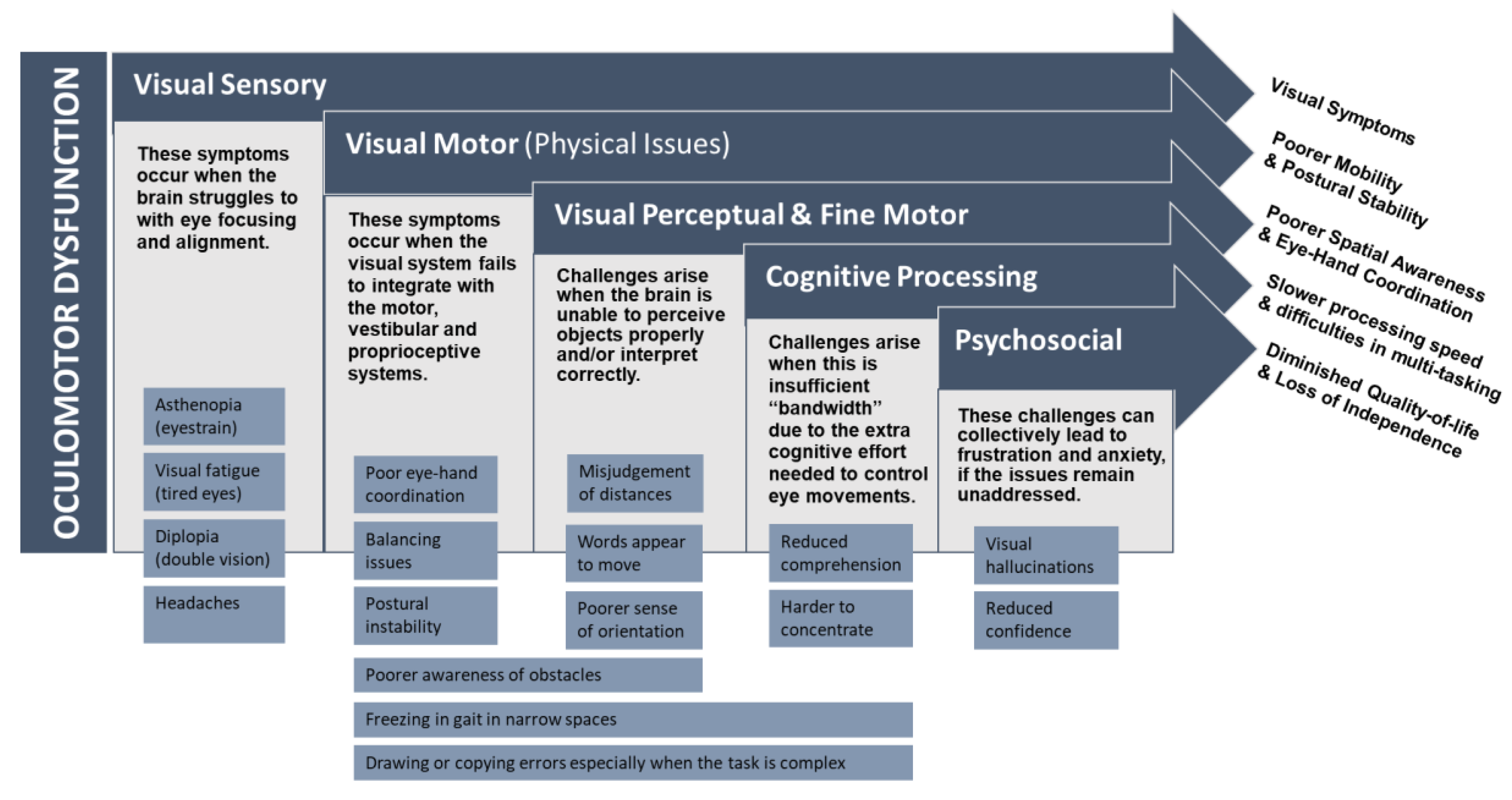
3. Implications of Oculomotor Dysfunction in Parkinson’s Disease
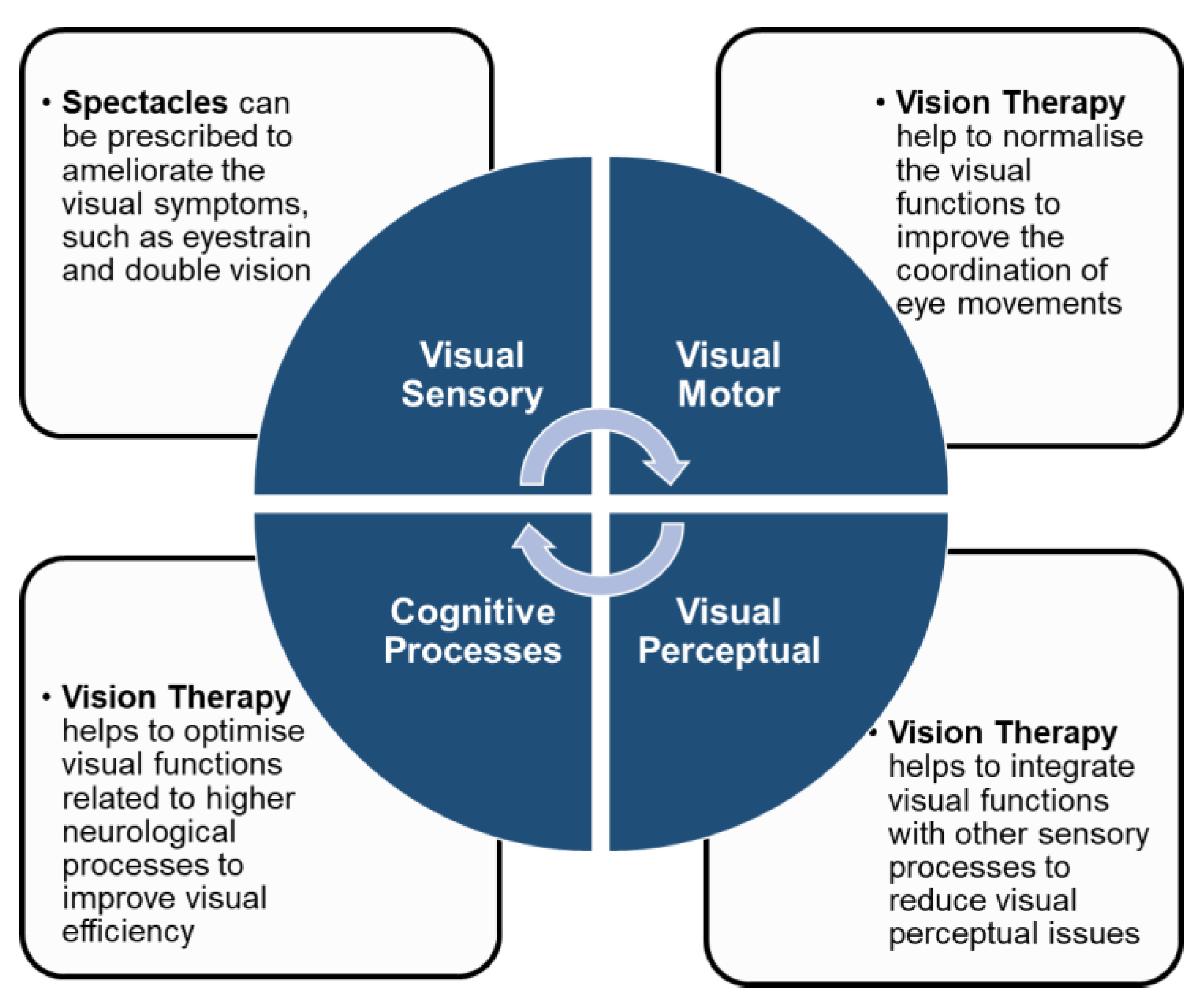
4. Clinical Management and Rehabilitation
5. Conclusion
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Weil, R.S.; Schrag, A.E.; Warren, J.D.; Crutch, S.J.; Lees, A.J.; Morris, H.R. Visual dysfunction in Parkinson’s disease. Brain A J. Neurol. 2016, 139(11), 2827–2843. [Google Scholar] [CrossRef]
- Mleczek, J.; Forjindam, A.; Shaikh, A.; Ghasia, F. Color Vision Deficits and Binocular Vision Dysfunction in Parkinson’s Disease. Brain Sci. 2026, 16(2). [Google Scholar] [CrossRef] [PubMed]
- Nowalk, N.; Matthews, J.; Walley, M.; Salmasinia, D.; Maitland, C. Investigational Study on the Degree of Contrast Sensitivity Visual Acuity Defects in Early Stages of Parkinsonism (P06.001). 2013, 80((7_) supplement, P06.001–P006.001. [Google Scholar] [CrossRef]
- Yap, T.P.; Stern, C. Advances in the Management of Oculomotor Dysfunction in Adults and Children with Concussion. In Current Advances in Optometry.; Shu, D., Singh, R.B., Ichhpujani, P., Eds.; Springer Nature Singapore: Singapore, 2024; pp. 59–75. [Google Scholar]
- Obeso, J.A.; Rodríguez-Oroz, M.C.; Rodríguez, M.; et al. Pathophysiology of the basal ganglia in Parkinson’s disease. Trends Neurosci. 2000, 23((10) Suppl, S8–19. [Google Scholar] [CrossRef] [PubMed]
- Hikosaka, O.; Takikawa, Y.; Kawagoe, R. Role of the basal ganglia in the control of purposive saccadic eye movements. Physiol. Rev. 2000, 80(3), 953–978. [Google Scholar] [CrossRef]
- Urwyler, P.; Nef, T.; Killen, A.; et al. Visual complaints and visual hallucinations in Parkinson’s disease. Park. Relat. Disord. 2014, 20(3), 318–322. [Google Scholar] [CrossRef]
- Irving, E.L.; Chriqui, E.; Law, C.; et al. Prevalence of Convergence Insufficiency in Parkinson’s Disease. Mov. Disord. Clin. Pract. 2017, 4(3), 424–429. [Google Scholar] [CrossRef]
- Repka, M.X.; Claro, M.C.; Loupe, D.N.; Reich, S.G. Ocular motility in Parkinson’s disease. J. Pediatr. Ophthalmol. Strabismus 1996, 33(3), 144–147. [Google Scholar] [CrossRef]
- Lepore, F.E., Md. Parkinson’s Disease and Diplopia. Neuro-Ophthalmology 2006, 30(2-3), 37–40. [Google Scholar] [CrossRef]
- Diotaiuti, P.; Marotta, G.; Di Siena, F.; et al. Eye Tracking in Parkinson’s Disease: A Review of Oculomotor Markers and Clinical Applications. 2025, 15(4), 362. [Google Scholar] [CrossRef]
- Dæhlen, A.; Heldal, I.; Ali, Q. Technologies Supporting Screening Oculomotor Problems: Challenges for Virtual Reality. 2023, 12(7), 134. [Google Scholar] [CrossRef]
- Health Policy Institute; American Optometric Association. Care Coordination between Optometry (OD), Occupational Therapy (OT), Physical Therapy (PT) and Other Rehabilitation Team Members for PatientCentric Care; 2023. [Google Scholar]
- Yaramothu, C.; Morris, C.J.; d’Antonio-Bertagnolli, J.V.; Alvarez, T.L. OculoMotor Assessment Tool Test Procedure and Normative Data. Optom. Vis. Sci. 2021, 98(6), 636–643. [Google Scholar] [CrossRef]
- González-Vides, L.; Hernández-Verdejo, J.L.; Cañadas-Suárez, P. Eye Tracking in Optometry: A Systematic Review. J. Eye Mov. Res. 2023, 16(3). [Google Scholar] [CrossRef]
- Shaikh, A.G.; Ghasia, F.F. Chapter 5—Saccades in Parkinson’s disease: Hypometric, slow, and maladaptive. In Progress in Brain Research.; Ramat, S., Shaikh, A.G., Eds.; Elsevier, 2019; Volume 249, pp. 81–94. [Google Scholar]
- Fooken, J.; Patel, P.; Jones, C.B.; McKeown, M.J.; Spering, M. Preservation of Eye Movements in Parkinson’s Disease Is Stimulus- and Task-Specific. J. Neurosci. 2022, 42(3), 487–499. [Google Scholar] [CrossRef]
- Herrero-Gracia, A.; Hernández-Andrés, R.; Merino, C.V.; Muedra, C.P.; Ciuffreda, K.J.; Díez-Ajenjo, M.A. Parkinson’s disease and reading performance. Ophthalmic Physiol. Opt. 2025, 45(7), 1653–1661. [Google Scholar] [CrossRef] [PubMed]
- Koh, S.B.; Suh, S.I.; Kim, S.H.; Kim, J.H. Stereopsis and extrastriate cortical atrophy in Parkinson’s disease: a voxel-based morphometric study. Neuroreport 2013, 24(5), 229–232. [Google Scholar] [CrossRef]
- Liu, L.D.; Pack, C.C. The Contribution of Area MT to Visual Motion Perception Depends on Training. Neuron 2017, 95(2), 436–446.e433. [Google Scholar] [CrossRef] [PubMed]
- Lawton, T.; Shelley-Tremblay, J. Training on Movement Figure-Ground Discrimination Remediates Low-Level Visual Timing Deficits in the Dorsal Stream, Improving High-Level Cognitive Functioning, Including Attention, Reading Fluency, and Working Memory. Front. Hum. Neurosci. 2017, 11, 236. [Google Scholar] [CrossRef]
- Karnath, H.O. Spatial orientation and the representation of space with parietal lobe lesions. Philos. Trans. R. Soc. Lond. Ser. B Biol. Sci. 1997, 352(1360), 1411–1419. [Google Scholar] [CrossRef]
- Salehi, M.A.; Rezagholi, F.; Mohammadi, S.; et al. Optical coherence tomography angiography measurements in Parkinson’s disease: A systematic review and meta-analysis. Eye 2023, 37(15), 3145–3156. [Google Scholar] [CrossRef] [PubMed]
- Gupta, P.; Beylergil, S.; Murray, J.; et al. Effects of Parkinson Disease on Blur-Driven and Disparity-Driven Vergence Eye Movements. J. Neuro-Ophthalmol. Off. J. North Am. Neuro-Ophthalmol. Soc. 2021, 41(4), 442–451. [Google Scholar] [CrossRef]
- Padula, W.V.; Subramanian, P.; Spurling, A.; Jenness, J. Risk of fall (RoF) intervention by affecting visual egocenter through gait analysis and yoked prisms. NeuroRehabilitation 2015, 37(2), 305–314. [Google Scholar] [CrossRef]
- P. D.culomotor Dysfunction in Parkinson’s Disease. Eur. J. Geriatr. Gerontol. 2020, 2(3), 87–89. [CrossRef]
- Armstrong, R.A. Visual symptoms in Parkinson’s disease. Park. Dis. 2011, 2011, 908306. [Google Scholar] [CrossRef] [PubMed]
- Friedman, Z.; Neumann, E. Benzhexol-induced blindness in Parkinson’s disease. Br. Med. J. 1972, 1(5800), 605. [Google Scholar] [CrossRef]
- Leigh, R.J.; Zee, D.S. The Neurology of Eye Movements.; Oxford University Press, 2015. [Google Scholar]
- Terao, Y.; Fukuda, H.; Ugawa, Y.; Hikosaka, O. New perspectives on the pathophysiology of Parkinson’s disease as assessed by saccade performance: a clinical review. Clin. Neurophysiol. Off. J. Int. Fed. Clin. Neurophysiol. 2013, 124(8), 1491–1506. [Google Scholar] [CrossRef] [PubMed]
- Angelopoulou, E.; Papadopoulos, A.N.; Spantideas, N.; Bougea, A. Migraine, Tension-Type Headache and Parkinson’s Disease: A Systematic Review and Meta-Analysis. Medicina 2022, 58(11). [Google Scholar] [CrossRef]
- Jeffery, K.; Guo, W.; Ball, D.; Rodriguez-Sanchez, J. Visual imagination and cognitive mapping of a virtual building. J. Navig. 2022, 75(1), 1–14. [Google Scholar] [CrossRef]
- Diotaiuti, P.; Marotta, G.; Di Siena, F.; et al. Eye Tracking in Parkinson’s Disease: A Review of Oculomotor Markers and Clinical Applications. Brain Sci. 2025, 15(4). [Google Scholar] [CrossRef]
- Rayner, K. Eye movements in reading and information processing: 20 years of research. Psychol. Bull. 1998, 124(3), 372–422. [Google Scholar] [CrossRef]
- Archibald, N.K.; Clarke, M.P.; Mosimann, U.P.; Burn, D.J. Visual symptoms in Parkinson’s disease and Parkinson’s disease dementia. Mov. Disord. Off. J. Mov. Disord. Soc. 2011, 26(13), 2387–2395. [Google Scholar] [CrossRef]
- Lewis, G.N.; Byblow, W.D.; Walt, S.E. Stride length regulation in Parkinson’s disease: the use of extrinsic, visual cues. Brain A J. Neurol. 2000, 123 Pt 10, 2077–2090. [Google Scholar] [CrossRef]
- Aarsland, D.; Marsh, L.; Schrag, A. Neuropsychiatric symptoms in Parkinson’s disease. Mov. Disord. Off. J. Mov. Disord. Soc. 2009, 24(15), 2175–2186. [Google Scholar] [CrossRef] [PubMed]
- Geroin, C.; Artusi, C.A.; Gandolfi, M.; et al. Does the Degree of Trunk Bending Predict Patient Disability, Motor Impairment, Falls, and Back Pain in Parkinson’s Disease? Front. Neurol. 2020, 11, 207. [Google Scholar] [CrossRef] [PubMed]
- Savitt, J.; Aouchiche, R. Management of Visual Dysfunction in Patients with Parkinson’s Disease. J. Park. Dis. 2020, 10(s1), S49–s56. [Google Scholar] [CrossRef]
- Raggi, A.; Serretti, A.; Ferri, R. Treatment options for depression in Parkinson’s disease: a mini-review. Int. Clin. Psychopharmacol. 2025, 40(6), 312–320. [Google Scholar] [CrossRef]
- Berry, A.J.; Costello, H.; Jesús, S.; Price, G.; Jha, A. Management of Anxiety in Parkinson’s Disease. Mov. Disord. Clin. Pract. 2025, 12(10), 1490–1501. [Google Scholar] [CrossRef]
- Amara, A.W.; Chahine, L.M.; Videnovic, A. Treatment of Sleep Dysfunction in Parkinson’s Disease. Curr. Treat. Options Neurol. 2017, 19(7), 26. [Google Scholar] [CrossRef] [PubMed]
- Nemanich, S.T.; Earhart, G.M. Prism adaptation in Parkinson disease: comparing reaching to walking and freezers to non-freezers. Exp. Brain Res. 2015, 233(8), 2301–2310. [Google Scholar] [CrossRef]
- Meglio, M.; Olivola, E.; Santilli, M.; et al. Effects of Prismatic Lenses on Lateral Axial Dystonia in Parkinson’s Disease: A Pilot Study. Innov. Clin. Neurosci. 2021, 18(1-3), 39–42. [Google Scholar]
- Borm, C.; Bloem, B.R.; Hoyng, C.; de Vries, N.M.; Theelen, T. The Many Faces of Blurry Vision in Parkinson’s Disease: An Illustrative Case Series. Case Rep. Neurol. 2022, 14(1), 173–178. [Google Scholar] [CrossRef]
- Shayler, G. The Use of Models to Help Our Understanding of Vision; 2015. [Google Scholar]
- Milner, A.D.; Goodale, M.A. Two visual systems re-viewed. Neuropsychologia 2008, 46(3), 774–785. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



