Submitted:
27 April 2026
Posted:
28 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction and Research Aims
2. Materials and Methods
2.1. Wood Samples
2.2. Surface Efflorescence
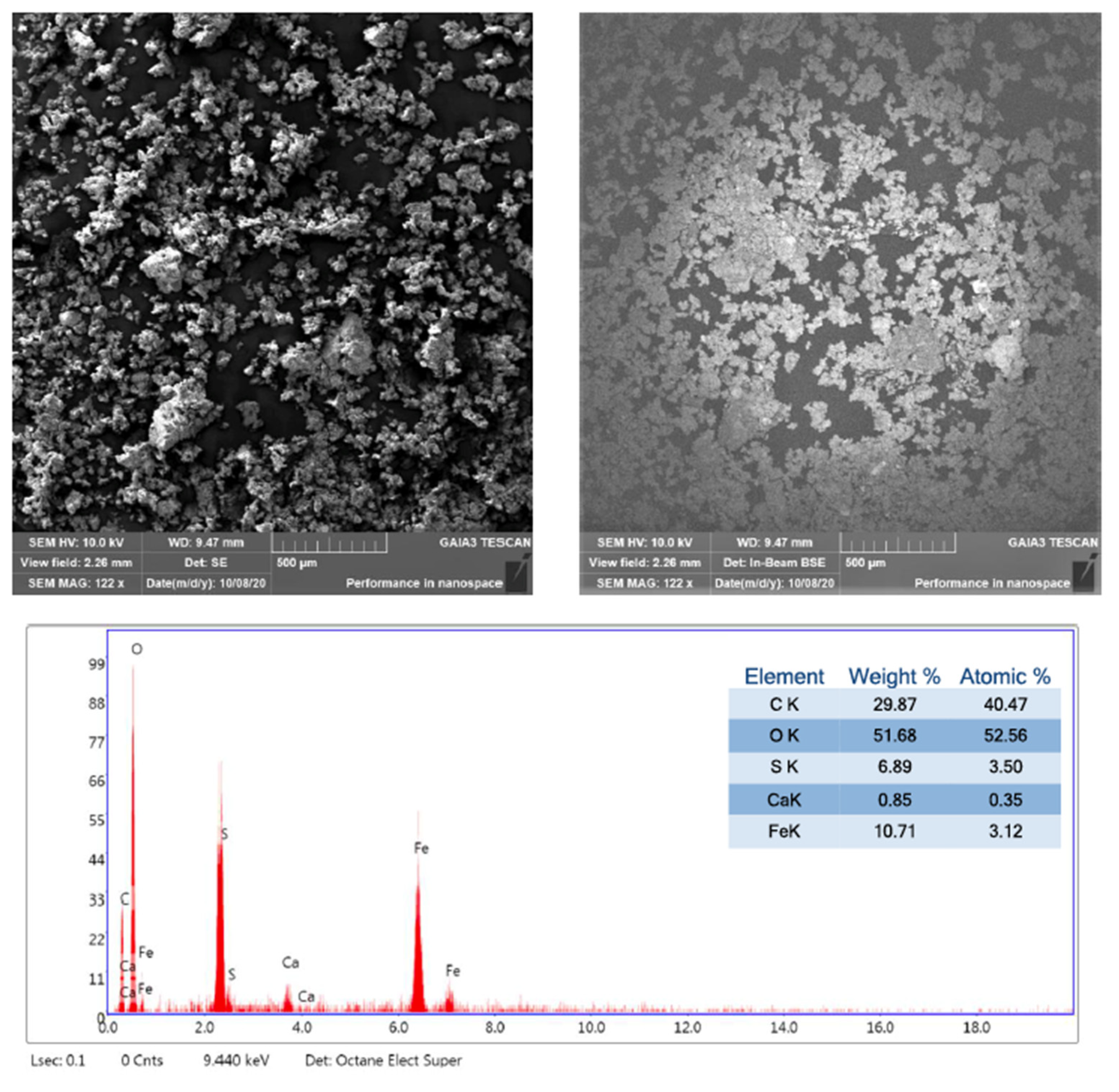
2.2.1. EDS Analysis
2.2.2. XRPD Analysis
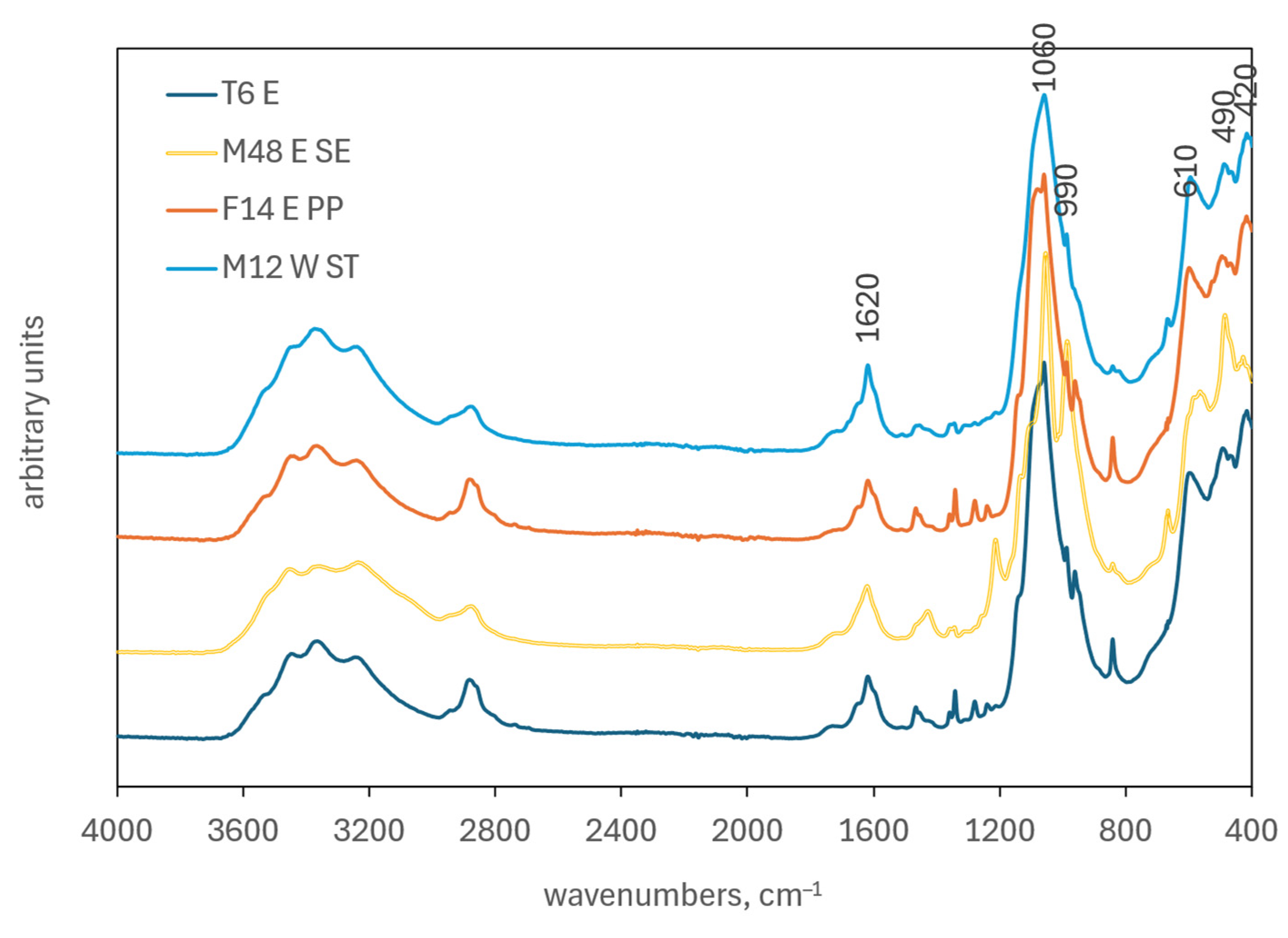
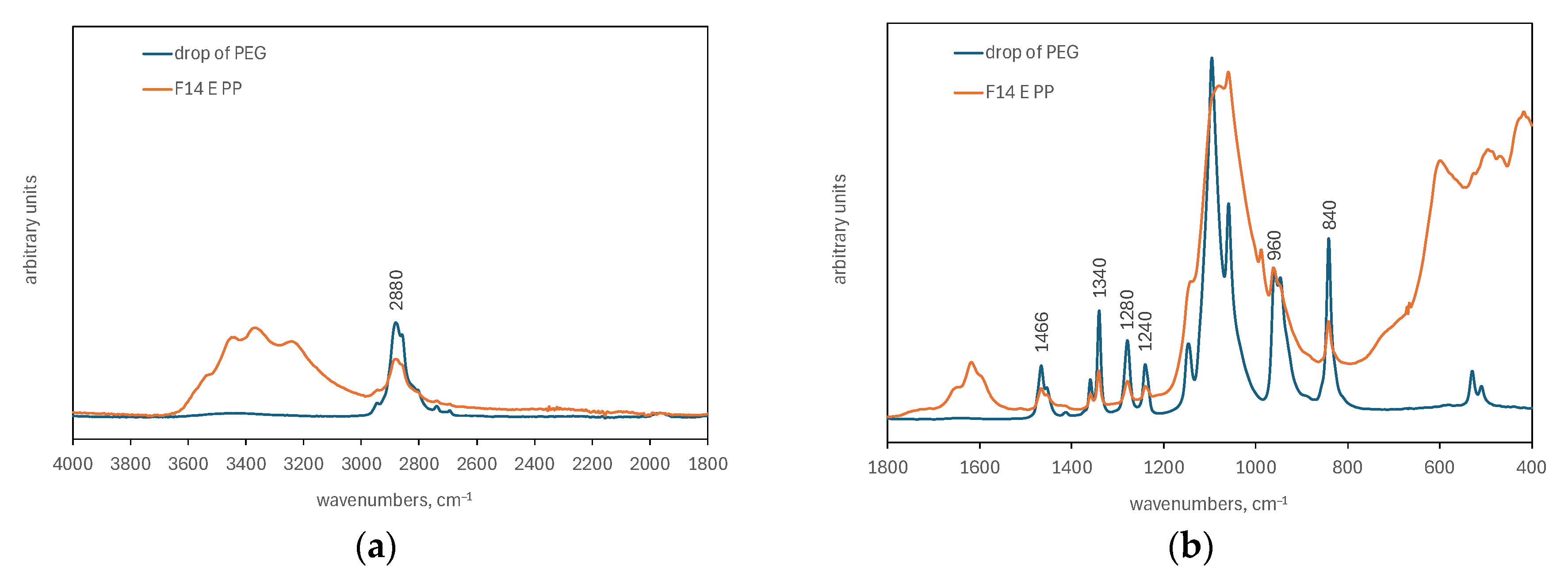
2.2.3. FTIR Analysis
2.3. Conditioning Tests
3. Results and Discussion
3.1. Identification of Surface Efflorescence
3.1.1. EDS and XRPD Analysis
3.1.2. ATR-FTIR Analysis
3.2. Conditioning Tests
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Dell’Amico, P. Il Relitto Di Grado: Considerazioni Preliminari. Archeol. Subacquea Studi Ric. E Doc. 1997, 2, 93–128. [Google Scholar]
- Beltrame, C.; Gaddi, D. Documentazione e Analisi Degli Elementi Strutturali Dello Scafo Della Nave Romana Di Grado (GO). 2000.
- Beltrame, C.; Gaddi, D. Preliminary Analysis of the Hull of the Roman Ship from Grado, Gorizia, Italy. Int. J. Naut. Archaeol. 2007, 36, 138–147. [Google Scholar] [CrossRef]
- Giacobelli, M. I Vetri Del Relitto Di Grado. Atti Convegno Naz. Archeol. Subacquea Anzio 1996, 311–314. [Google Scholar]
- Auriemma, R. Le Anfore Del Relitto Di Grado e Il Loro Contenuto. MEFRA Mélanges LÉcole Fr. Rome Antiq. 112 1 2000 2000, 1000–1025. [Google Scholar] [CrossRef]
- Costa, E.; Beltrame, C. 3D Modelling from Archive and Legacy Data: Preliminary Data Processing on the Roman Shipwreck Grado I. Archeol. E Calcolatori 2021, 32, 156–166. [Google Scholar] [CrossRef]
- Contessa, A.; Novello, M.; Condò, F. Il Nuovo Museo Di Archeologia Subacquea Dell’Alto Adriatico Di Grado (GO). In Proceedings of the Allestire l’archeologia. Progetti in corso e nuove proposte per i musei e i parchi archeologici nazionali, Atti del convegno a cura di M. Osanna e M. Bressan; Franco Cosimo Panini, February 26 2024; pp. 207–215.
- Fors, Y. Sulfur-Related Conservation Concerns in Marine Archaeological Wood: The Origin, Speciation and Distribution of Accumulated Sulfur with Some Remedies for the Vasa. 2008.
- Fors, Y.; Jalilehvand, F.; Damian Risberg, E.; Björdal, C.; Phillips, E.; Sandström, M. Sulfur and Iron Analyses of Marine Archaeological Wood in Shipwrecks from the Baltic Sea and Scandinavian Waters. J. Archaeol. Sci. 2012, 39, 2521–2532. [Google Scholar] [CrossRef]
- Almkvist, G.; Hocker, E.; Sahlstedt, M.; Museums, S.M. Iron Removal from Waterlogged Wood. Swed. Univ. Agric. Sci. SLU Repro Upps. 2013. [Google Scholar]
- Kılıç, A.G.; Kılıç, N.; Arnold, D.C. Analyses of Sulfur and Iron in Waterlogged Archaeological Wood: The Case of Polyethylene-Glycol-Treated Yenikapı 12 Shipwreck. Forests 2023, 14. [Google Scholar] [CrossRef]
- Mortensen, M.N.; Egsgaard, H.; Hvilsted, S.; Shashoua, Y.; Glastrup, J. Characterisation of the Polyethylene Glycol Impregnation of the Swedish Warship Vasa and One of the Danish Skuldelev Viking Ships. J. Archaeol. Sci. 2007, 34, 1211–1218. [Google Scholar] [CrossRef]
- Pearson, C. Conservation of Marine Archaeological Objects; 1988; ISBN 978-1-4832-9465-0. [Google Scholar]
- Hoffmann, P. To Be and to Continue Being a Cog: The Conservation of the Bremen Cog of 1380. Int. J. Naut. Archaeol. 2001, 30, 129–140. [Google Scholar] [CrossRef]
- Glastrup, J.; Shashoua, Y.; Egsgaard, H.; Nordvig Mortensen, M. Formic and Acetic Acids in Archaeological Wood. A Comparison between the Vasa Warship, the Bremen Cog, the Oberländer Boat and the Danish Viking Ships. Holzforschung 2006, 60, 259–264. [Google Scholar] [CrossRef]
- Dedic, D.; Iversen, T.; Ek, M. Cellulose Degradation in the Vasa: The Role of Acids and Rust. Stud. Conserv. 2013, 58, 308–313. [Google Scholar] [CrossRef]
- Baird, J.A.; Olayo-Valles, R.; Rinaldi, C.; Taylor, L.S. Effect of Molecular Weight, Temperature, and Additives on the Moisture Sorption Properties of Polyethylene Glycol. J. Pharm. Sci. 2010, 99, 154–168. [Google Scholar] [CrossRef] [PubMed]
- Majka, J.; Zborowska, M.; Fejfer, M.; Waliszewska, B.; Olek, W. Dimensional Stability and Hygroscopic Properties of PEG Treated Irregularly Degraded Waterlogged Scots Pine Wood. J. Cult. Herit. 2018, 31, 133–140. [Google Scholar] [CrossRef]
- Pecoraro, E.; Pizzo, B.; Salvini, A.; Macchioni, N. Dynamic Mechanical Analysis (DMA) at Room Temperature of Archaeological Wood Treated with Various Consolidants. Holzforschung 2019, 73, 757–772. [Google Scholar] [CrossRef]
- Pizzo, B.; Li, R.; Macchioni, N.; Sozzi, L.; Yin, Y.; Musina, G.; Novello, M.; Pecoraro, E. Diagnostic Characterisation of Wood from Archaeological Artefacts Already Treated with Polyethylene Glycols. in preparation.
- Remazeilles, C.; Leveque, F.; Minjacq, M.; Refait, P.; Sanchez, C.; Jézégou, M.-P. Characterisation of Iron (II) Sulfides in Wet Archaeological Woods: The Wreck of Mandirac (IV Th Century, Antique Ports of Narbonne, France). 2016. [Google Scholar]
- Rémazeilles, C.; Saheb, M.; Neff, D.; Guilminot, E.; Tran, K.; Bourdoiseau, J.-A.; Sabot, R.; Jeannin, M.; Matthiesen, H.; Dillmann, P.; et al. Microbiologically Influenced Corrosion of Archaeological Artefacts: Characterisation of Iron(II) Sulfides by Raman Spectroscopy. J. Raman Spectrosc. 2010, 41, 1425–1433. [Google Scholar] [CrossRef]
- KILIÇ, A.G.; Kilic, N.; Akgün, C. The Importance of Using Multiple Analyses Techniques to Determine the Physical Condition of the Waterlogged Wood Near the Corroded Parts. Wood Res. 2021, 66, 1046–1054. [Google Scholar] [CrossRef]
- Fors, Y.; Jalilehvand, F.; Sandström, M. Analytical Aspects of Waterlogged Wood in Historical Shipwrecks. Anal. Sci. 2011, 27, 785–792. [Google Scholar] [CrossRef] [PubMed]
- Rouchon, V.; Badet, H.; Belhadj, O.; Bonnerot, O.; Lavédrine, B.; Michard, J.; Miska, S. Raman and FTIR Spectroscopy Applied to the Conservation Report of Paleontological Collections: Identification of Raman and FTIR Signatures of Several Iron Sulfate Species Such as Ferrinatrite and Sideronatrite. J. Raman Spectrosc. 2012, 43, 1265–1274. [Google Scholar] [CrossRef]
- Song, K.; Yin, Y.; Salmén, L.; Xiao, F.; Jiang, X. Changes in the Properties of Wood Cell Walls during the Transformation from Sapwood to Heartwood. J. Mater. Sci. 2014, 49, 1734–1742. [Google Scholar] [CrossRef]
- Almkvist, G.; Persson, I. Extraction of Iron Compounds from Wood from the Vasa. Holzforschung 2006, 60, 678–684. [Google Scholar] [CrossRef]
- Fors, Y.; Sandström, M. Sulfur and Iron in Shipwrecks Cause Conservation Concerns. Chem. Soc. Rev. 2006, 35, 399–415. [Google Scholar] [CrossRef]
- Pelé-Meziani, C.; Macchioni, N.; Sozzi, L.; Guilminot, E.; Lemoine, G.; Pizzo, B.; Mevellec, J.Y.; Pecoraro, E.; Monachon, M. Assessment of Various Iron Extraction Treatments on Waterlogged Archaeological Oak. Forests 2023, 14, 1834. [Google Scholar] [CrossRef]
- Sandström, M.; Jalilehvand, F.; Persson, I.; Gelius, U.; Frank, P.; Hall-Roth, I. Deterioration of the Seventeenth-Century Warship Vasa by Internal Formation of Sulphuric Acid. Nature 2002, 415, 893–897. [Google Scholar] [CrossRef]
- Pecoraro, E.; Pelé-Meziani, C.; Macchioni, N.; Lemoine, G.; Guilminot, E.; Shen, D.; Pizzo, B. The Removal of Iron from Waterlogged Archaeological Wood: Efficacy and Effects on the Room Temperature Wood Properties. Wood Mater. Sci. Eng. 2023, 18, 672–689. [Google Scholar] [CrossRef]
- Monachon, M.; Albelda-Berenguer, M.; Pelé, C.; Cornet, E.; Guilminot, E.; Rémazeilles, C.; Joseph, E. Characterization of Model Samples Simulating Degradation Processes Induced by Iron and Sulfur Species on Waterlogged Wood. Microchem. J. 2020, 155, 104756. [Google Scholar] [CrossRef]
- Brogly, M.; Bistac, S.; Bindel, D. Adsorption and Structuration of PEG Thin Films: Influence of the Substrate Chemistry. Polymers 2024, 16, 1244. [Google Scholar] [CrossRef] [PubMed]
- Peak, D.; Ford, R.G.; Sparks, D.L. An in Situ ATR-FTIR Investigation of Sulfate Bonding Mechanisms on Goethite. J. Colloid Interface Sci. 1999, 218, 289–299. [Google Scholar] [CrossRef]
- Lacalamita, M.; Ventruti, G.; Della Ventura, G.; Radica, F.; Mauro, D.; Schingaro, E. In Situ High-Temperature X-Ray Powder Diffraction and Infrared Spectroscopic Study of Melanterite, FeSO4·7H2O. Minerals 2021, 11, 392. [Google Scholar] [CrossRef]
- Myneni, S.C.B. X-Ray and Vibrational Spectroscopy of Sulfate in Earth Materials. Rev. Mineral. Geochem. 2000, 40, 113–172. [Google Scholar] [CrossRef]
- J. Saikia, B. Spectroscopic Estimation of Geometrical Structure Elucidation in Natural SiO2 Crystal. J. Mater. Phys. Chem. 2014, 2, 28–33. [Google Scholar] [CrossRef]
- Rémazeilles, C.; Tran, K.; Guilminot, E.; Conforto, E.; Refait, P. Study of Fe(II) Sulphides in Waterlogged Archaeological Wood. Stud. Conserv. 2013, 58, 297–307. [Google Scholar] [CrossRef]
- Grattan, D.W. A Practical Comparative Study of Several Treatments for Waterlogged Wood. Stud. Conserv. 1982, 27, 124. [Google Scholar] [CrossRef]
- Christensen, M.; Kutzke, H.; Hansen, F.K. New Materials Used for the Consolidation of Archaeological Wood–Past Attempts, Present Struggles, and Future Requirements. J. Cult. Herit. 2012, 13, S183–S190. [Google Scholar] [CrossRef]
- Li, R.; Wu, Y.; Bai, Z.; Guo, J.; Chen, X. Effect of Molecular Weight of Polyethylene Glycol on Crystallization Behaviors, Thermal Properties and Tensile Performance of Polylactic Acid Stereocomplexes. RSC Adv. 2020, 10, 42120–42127. [Google Scholar] [CrossRef]
- Jo, A.H.; Lee, K.-H.; Choi, T.-H.; Go, I.H.; Seo, J.-W. Changes in the Amount of PEG Free-Flowing Back from PEG-Treated Waterlogged Archaeological Wood and the Compressive Strength According to Relative Humidity Conditions. J. Conserv. Sci. 2020, 36, 225–235. [Google Scholar] [CrossRef]


| Sample ID | Element type (where available) and Museum reference number | Wood species |
|---|---|---|
| 6 | Secondary keelson - E PE 10/11 | Pinus pinea |
| 8 | Belaying pin | Olea europaea |
| 9 | Splinter | Pinus pinea |
| 11 | Bow - T6 E (2) | Pinus sylvestris |
| 12 | Stem - M34 W ST | Pinus pinea |
| 13 | Partial frame - M34 E SE | Pinus pinea |
| 14 | Stern wheel (keelson fragment) | Larix decidua/Picea abies |
| 15 | Upper planking /spare - T6 E | Pinus pinea |
| 16 | Frame floor - M47 CW M | Pinus pinea |
| 17 | Partial frame - M47 E SE | Pinus pinea |
| 18 | Partial frame - M47 W SE | Pinus pinea |
| 20 | T8 E | Pinus sylvestris |
| 21 | T7 W | Pinus sylvestris |
| 22 | Bow - T7 E | Larix decidua/Picea abies |
| 23 | Keel | Ulmus spp. |
| 24 | F14 E PP | Larix decidua/Picea abies |
| 25 | T4 W | Pinus sylvestris |
| Museum reference number | Element type from which the samples were taken (where available) | photo |
|---|---|---|
| T6 E | Bow |  |
| M48 E SE | Partial frame |  |
| F14 E PP | - |  |
| M12 W ST | Stem |  |
| Sample | Na | Si | S | K | Ca | Fe |
|---|---|---|---|---|---|---|
| T6 E | x | x | x | x | x | x |
| M48 E SE | x | x | x | x | x | x |
| F14 E PP | x | x | x | x | x | |
| M12 W ST | x | x | x |
| Sample | Compounds identified | Chemical formula |
|---|---|---|
| T6 E | Ferrous sulphate tetrahydrate (Rozenite) | FeSO4∙4H2O |
| Hydrated sulphate of Fe, K and Na | K2Na6Fe7(SO4)12O2∙(H2O)18 | |
| Gypsum | CaSO4∙2H2O | |
| Iron oxide hydrate | Fe2O3∙H2O | |
| M48 E SE | Ferrous sulphate tetrahydrate (Rozenite) | FeSO4∙4H2O |
| Hydrated sulphate of Fe, K and Na | K2Na6Fe7(SO4)12O2∙(H2O)18 | |
| Gypsum | CaSO4∙2H2O | |
| Iron oxide hydrate | Fe2O3∙H2O | |
| F14 E PP | Ferrous sulphate tetrahydrate (Rozenite) | FeSO4∙4H2O |
| Hydrated sulphate of Fe, K and Na | K2Na6Fe7(SO4)12O2∙(H2O)18 | |
| Gypsum | CaSO4∙2H2O | |
| Quartz | SiO2 | |
| M12 W ST | Ferrous sulphate tetrahydrate (Rozenite) | FeSO4∙4H2O |
| Hydrated sulphate of Fe, K and Na | K2Na6Fe7(SO4)12O2∙(H2O)18 | |
| Gypsum | CaSO4∙2H2O | |
| Quartz | SiO2 | |
| Iron oxide hydrate | Fe2O3∙H2O |
| Sample ID | Mi, g | Ai, mm2 | Hi, mm | M35, g | A35, mm2 | H35, mm | , % | , % | , % |
|---|---|---|---|---|---|---|---|---|---|
| 6 | 0.51 | 61.0 | 7.91 | 0.51 | 61.2 | 7.86 | -0.98 | 0.4 | -0.63 |
| 8 | 1.22 | 139.5 | 7.51 | 1.21 | 141.4 | 7.51 | -0.76 | 1.4 | 0.00 |
| 9 | 1.23 | 166.9 | 7.71 | 1.21 | 164.8 | 7.52 | -1.03 | -1.3 | -2.46 |
| 11 | 0.38 | 40.9 | 7.88 | 0.38 | 40.5 | 7.87 | -0.81 | -1.1 | -0.13 |
| 12 | 1.02 | 101.9 | 7.91 | 1.00 | 100.2 | 7.88 | -1.01 | -1.7 | -0.38 |
| 13 | 0.50 | 68.9 | 6.45 | 0.50 | 69.2 | 6.62 | -0.88 | 0.5 | 2.64 |
| 14 | 3.23 | 266.3 | 10.25 | 3.21 | 267.5 | 10.21 | -0.73 | 0.5 | -0.39 |
| 15 | 5.93 | 667.8 | 7.7 | 5.88 | 669.0 | 7.72 | -0.90 | 0.2 | 0.26 |
| 16 | 1.49 | 199.3 | 6.58 | 1.48 | 199.6 | 6.59 | -0.93 | 0.1 | 0.15 |
| 17 | 0.86 | 124.3 | 6.18 | 0.85 | 123.3 | 6.18 | -0.83 | -0.8 | 0.00 |
| 18 | 3.07 | 348.1 | 7.2 | 3.04 | 341.1 | 7.26 | -0.92 | -2.0 | 0.83 |
| 20 | 2.73 | 326.0 | 7.71 | 2.71 | 324.8 | 7.84 | -0.80 | -0.3 | 1.69 |
| 21 | 1.24 | 160.4 | 7.36 | 1.23 | 159.8 | 7.33 | -0.93 | -0.4 | -0.41 |
| 22 | 1.39 | 148.3 | 7.32 | 1.38 | 146.4 | 7.31 | -0.85 | -1.3 | -0.14 |
| 23 | 1.09 | 183.6 | 7.63 | 1.08 | 184.5 | 7.62 | -1.23 | 0.5 | -0.13 |
| 24 | 0.75 | 84.0 | 7.33 | 0.74 | 83.6 | 7.33 | -0.74 | -0.5 | 0.00 |
| 25 | 1.64 | 179.8 | 7.71 | 1.62 | 176.9 | 7.71 | -0.82 | -1.7 | 0.00 |
| average | -0.89 | -0.4 | 0.05 | ||||||
| std. dev. | 0.12 | 1.0 | 1.05 | ||||||
| Sample ID | Mi, g | Ai, mm2 | Hi, mm | M35, g | A35, mm2 | H35, mm | , % | , % | |
|---|---|---|---|---|---|---|---|---|---|
| 6 | 0.40 | 43.0 | 7.83 | 0.44 | broken | 7.90 | 11.80 | – | 0.89 |
| 8 | 1.12 | 129.9 | 7.48 | 1.22 | 131.4 | 7.41 | 9.56 | 1.1 | -0.94 |
| 9 | 1.19 | 154.4 | 6.96 | 1.32 | 165.2 | 6.96 | 11.09 | 7.0 | 0.00 |
| 11 | 0.42 | 45.2 | 7.85 | 0.46 | 47.2 | 7.85 | 10.18 | 4.4 | 0.00 |
| 12 | 1.05 | 111.5 | 7.91 | 1.18 | broken | 7.89 | 12.29 | – | -0.25 |
| 13 | 0.51 | 65.7 | 6.59 | 0.56 | 71.1 | 6.66 | 11.42 | 8.2 | 1.06 |
| 14 | 3.02 | 258.9 | 10.09 | 3.35 | 265.5 | 10.09 | 10.70 | 2.6 | 0.00 |
| 15 | 5.26 | 671.8 | 7.8 | 5.93 | 712.7 | 7.86 | 12.71 | 6.1 | 0.77 |
| 16 | 1.52 | 182.4 | 6.53 | 1.71 | broken | 6.53 | 12.13 | – | 0.00 |
| 17 | 0.72 | 104.4 | 6.56 | 0.79 | 109.4 | 6.72 | 10.71 | 4.7 | 2.44 |
| 18 | 2.02 | 236.5 | 7.3 | 2.24 | broken | 7.27 | 11.20 | – | -0.41 |
| 20 | 2.99 | 306.4 | 7.87 | 3.31 | 318.3 | 7.88 | 10.69 | 3.9 | 0.13 |
| 21 | 1.59 | 177.8 | 7.35 | 1.77 | broken | 7.34 | 11.21 | – | -0.14 |
| 22 | 1.16 | 144.0 | 7.48 | 1.30 | 153.5 | 7.42 | 11.50 | 6.6 | -0.80 |
| 23 | 0.97 | 187.4 | 7.78 | 1.09 | 194.2 | 7.78 | 11.74 | 3.6 | 0.00 |
| 24 | 0.71 | 79.9 | 7.33 | 0.79 | 83.1 | 7.28 | 11.47 | 4.0 | -0.68 |
| 25 | 1.37 | 169.8 | 7.63 | 1.52 | 179.6 | 7.48 | 10.66 | 5.8 | -1.97 |
| average | 11.24 | 4.8 | 0.01 | ||||||
| std. dev. | 0.79 | 2.0 | 0.95 | ||||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




