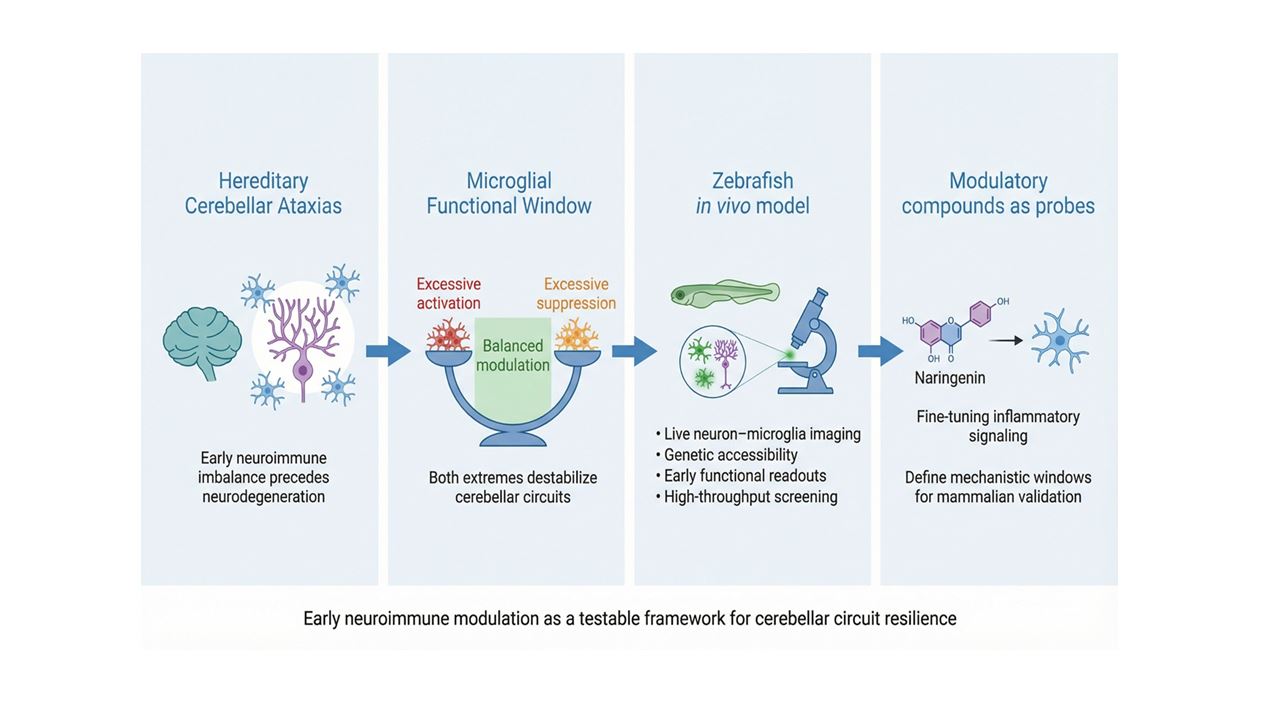
Hereditary cerebellar ataxias are progressive neurodegenerative disorders for which diseasemodifying treatments are still lacking. Although these conditions have traditionally been studied from a neuron-centered perspective, evidence from several ataxia models indicates that changes in the cerebellar immune microenvironment can arise before overt neuronal loss and may contribute to early circuit dysfunction. This review examines hereditary cerebellar ataxias through the lens of early neuroimmune regulation, with particular attention to the region-specific properties of cerebellar microglia and their roles in synaptic refinement and circuit homeostasis. We also discuss zebrafish as a useful experimental system for this question, because they combine in vivo imaging, genetic manipulation, and scalable functional assays in an intact vertebrate model. In this context, flavonoids—and especially naringenin—are considered not as immediate therapeutic candidates, but as experimental tools to investigate how modulation of inflammatory balance affects disease-relevant phenotypes in vivo. By integrating genetic ataxia models with dynamic neuroimmune readouts, zebrafish-based approaches can help identify early windows in which neuroimmune signalling influences cerebellar vulnerability and can guide subsequent validation in mammalian systems.