Submitted:
27 April 2026
Posted:
28 April 2026
You are already at the latest version
Abstract
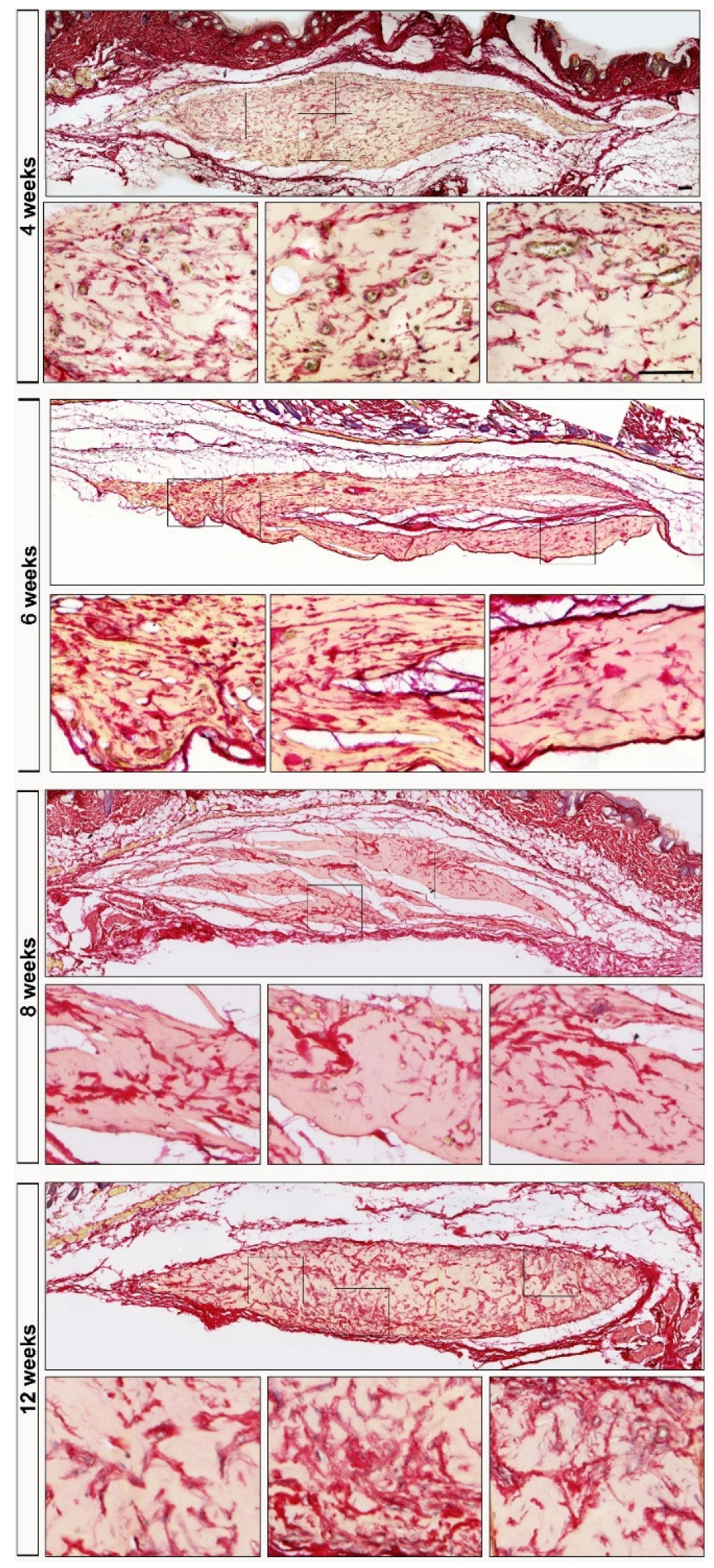
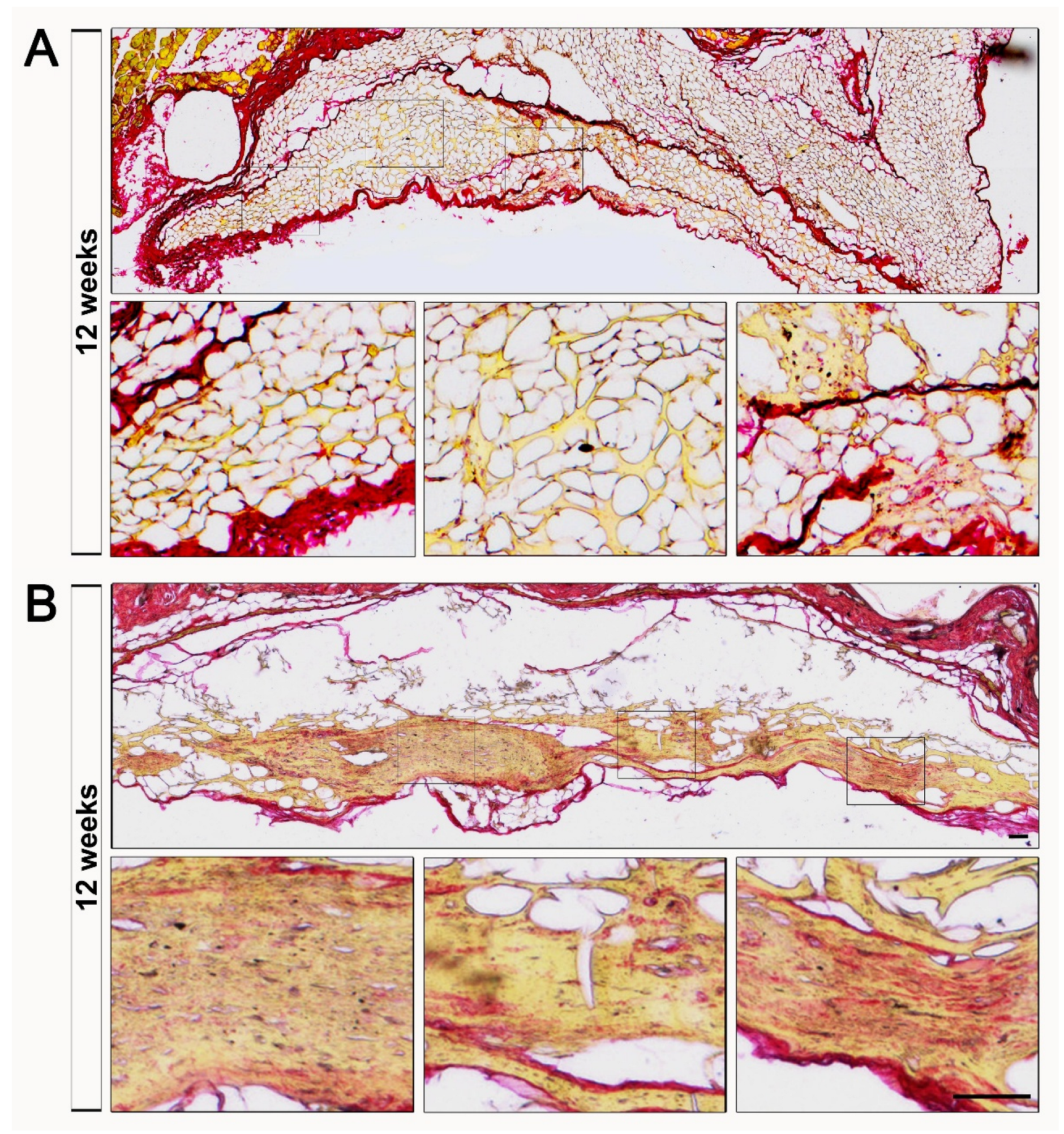
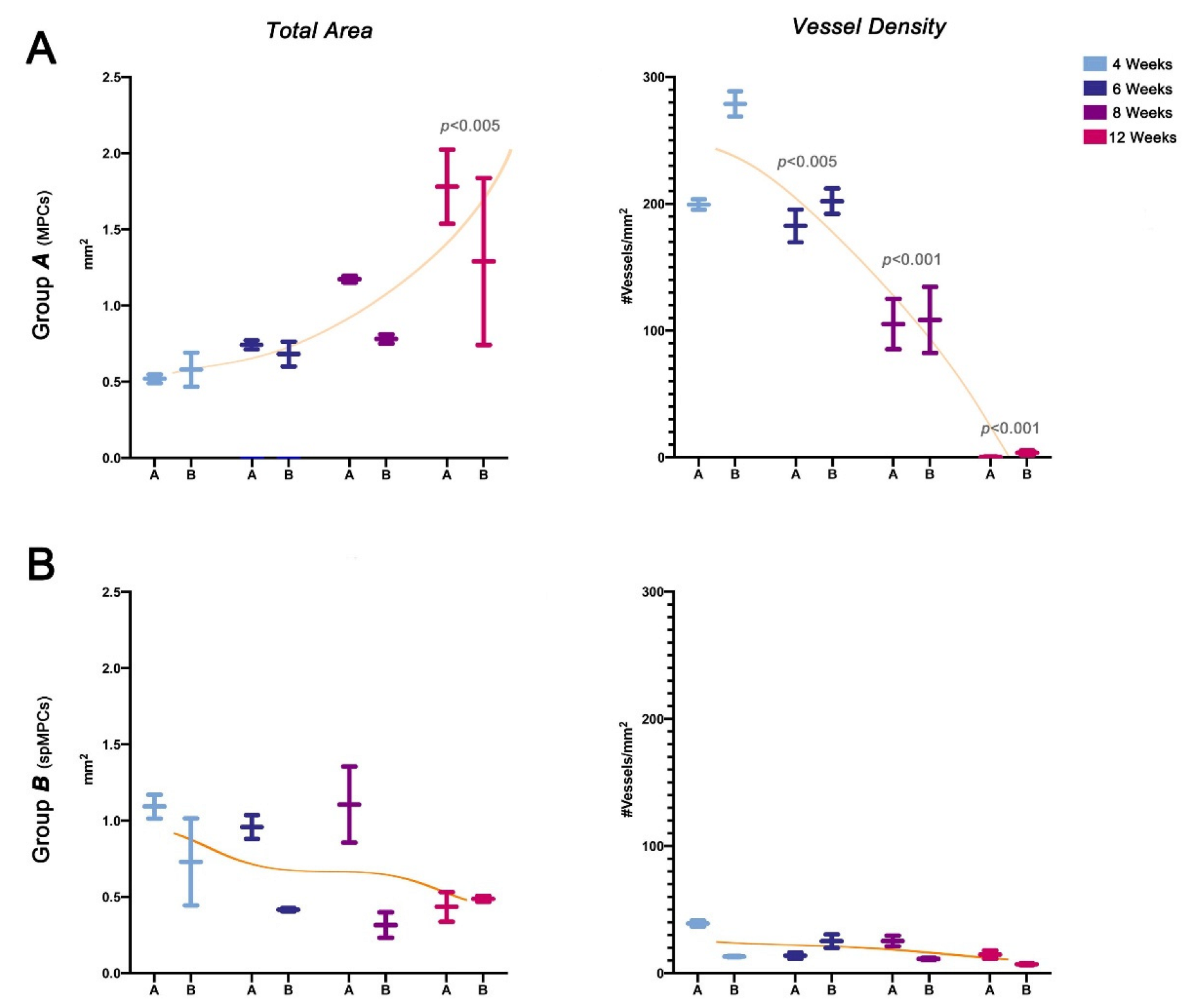
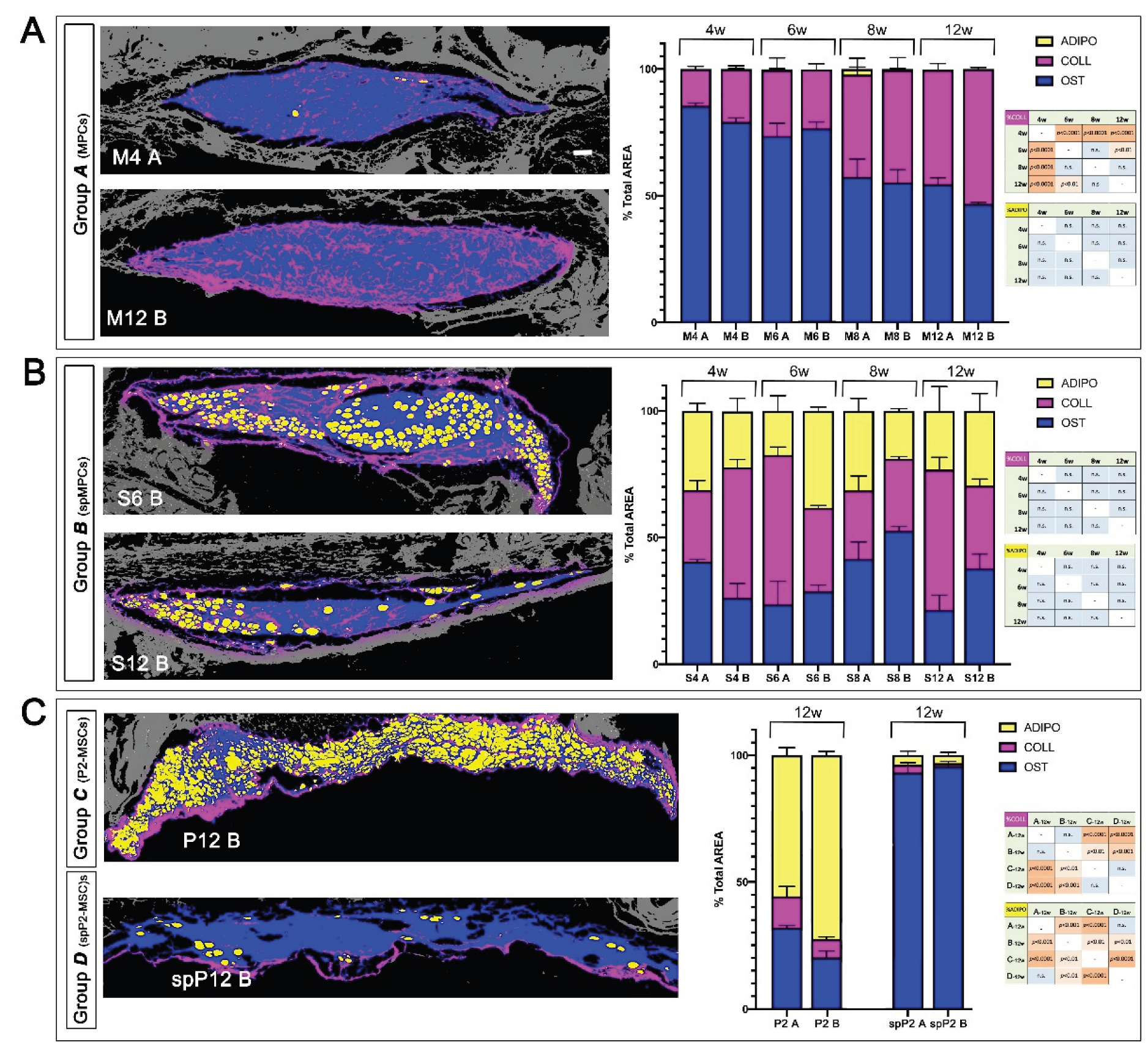
Background: Mesangiogenic Progenitor cells (MPCs) were first described in 2008 in cultures of human bone marrow mononuclear cells (hBM-MNCs) aimed at isolating mesenchymal stromal cells (MSCs) using human autologous serum. A selective culture method was subsequently developed to isolate MPCs with a high degree of purity, yielding approximately 1% of the total plated cells. Since their initial description, MPCs have demonstrated the ability to differentiate into highly clonogenic MSCs while retaining early vasculogenic potential. Gene expression profiling of MPCs revealed constitutive expression of pluripotency-associated transcription factors such as OCT-4 and NANOG, as well as SOX15 instead of SOX2, suggesting a possible molecular mechanism that sustains MPC plasticity, defined as the “adult Oct-4 circuit”. Although the expression of these pluripotency-associated markers has been hypothesized to represent a distinctive adult molecular circuit, concerns regarding the tumorigenic potential of MPCs are reasonable and have not yet explored. Methods: Here, we present data from the tumorigenicity test in partial compliance with WHO recommendations of two different MPC-derived cell products in athymic nude mice. Results: Histomorphometric analysis of nodules excised from animals at 6, 8, or 12 weeks post-cell transplantation excluded tumor formation and demonstrated the ability of MPCs to generate homogeneous and organized tissue through distinct phases: an “early” vasculogenic phase, followed by remodeling of the newly formed microvascular network and the deposition of structured, aligned collagen fibers. Conclusions: MPCs do not possess intrinsic tumorigenic potential and spontaneously form vascularized xenogenic tissue four weeks after injection into the subcutaneous space.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Human Bone Marrow Collection and Mononuclear Cell Isolation
2.2. Mesangiogenic Progenitor Cells (MPCs) Isolation
2.3. Obtaining MPC-Derived Mesenchymal Stromal Cells (MSCs)
2.4. Flow Cytometry
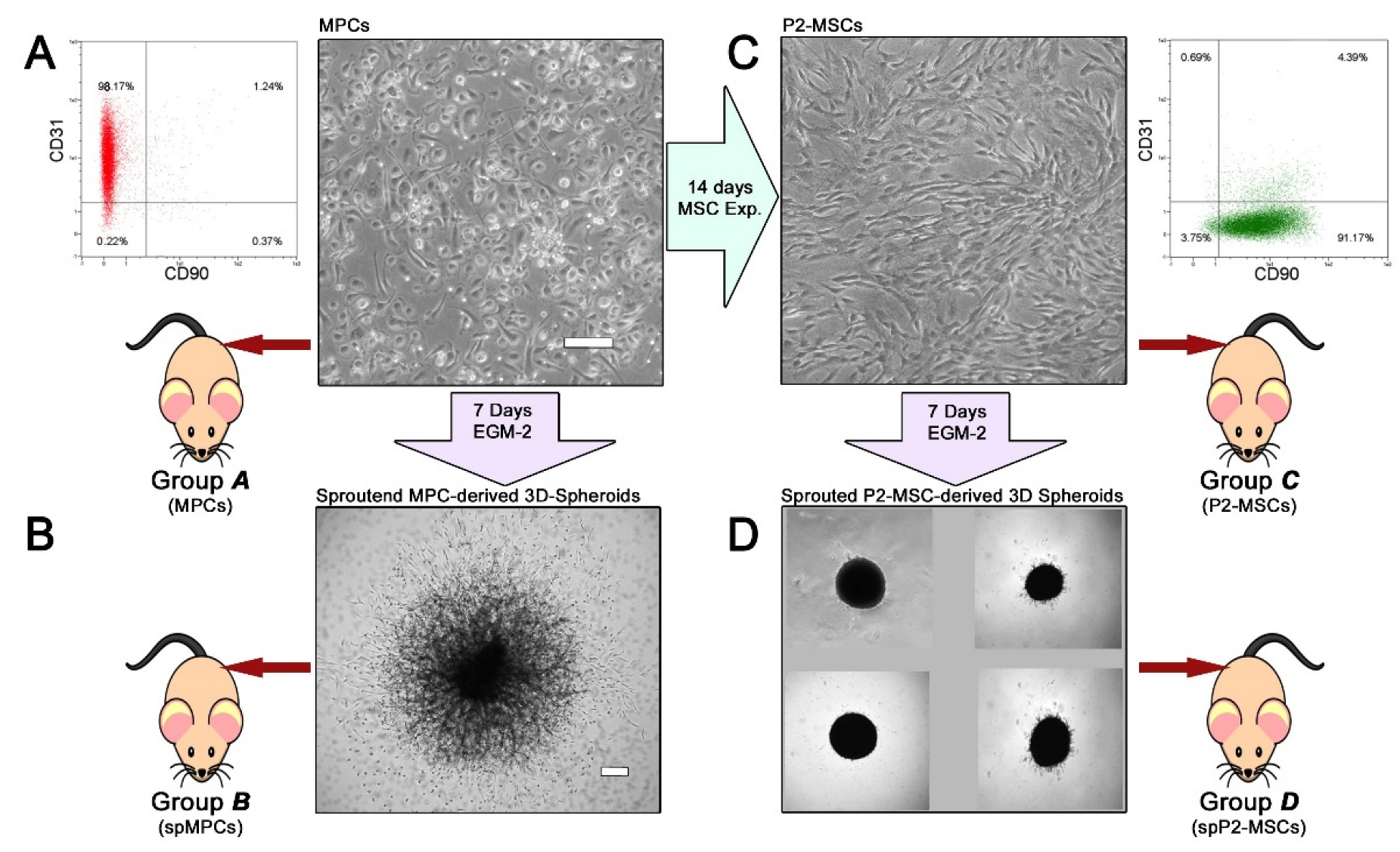
2.5. 3D Spheroids Preparation and Sprouting Induction
2.6. Subcutaneous Implantation in Nude Mice and Clinical Observation
2.7. Histological and Histomorphometric Analysis
3. Results
3.1.1. Characterization of Cell Products
3.1.2. In Vivo Experiments
3.1.3. Biopsy Analysis
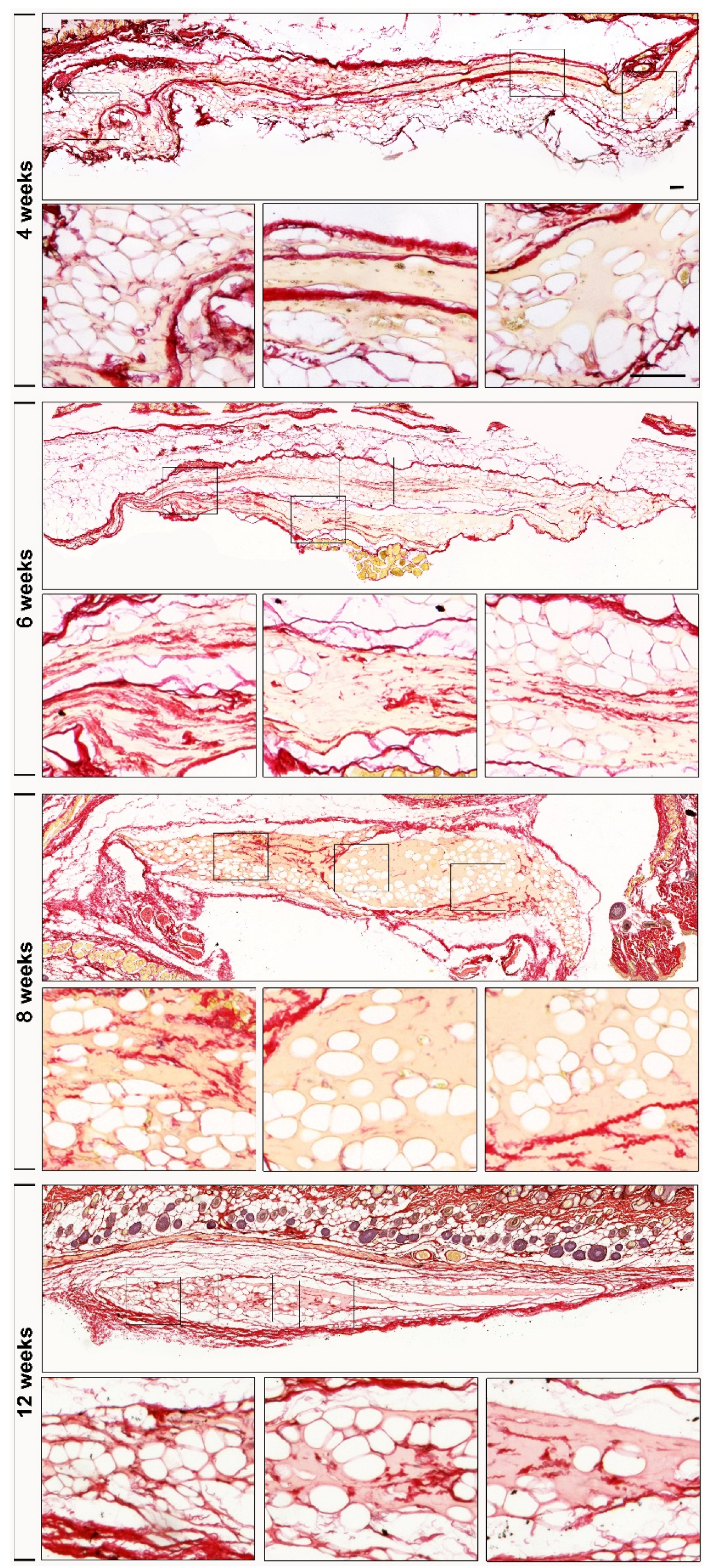
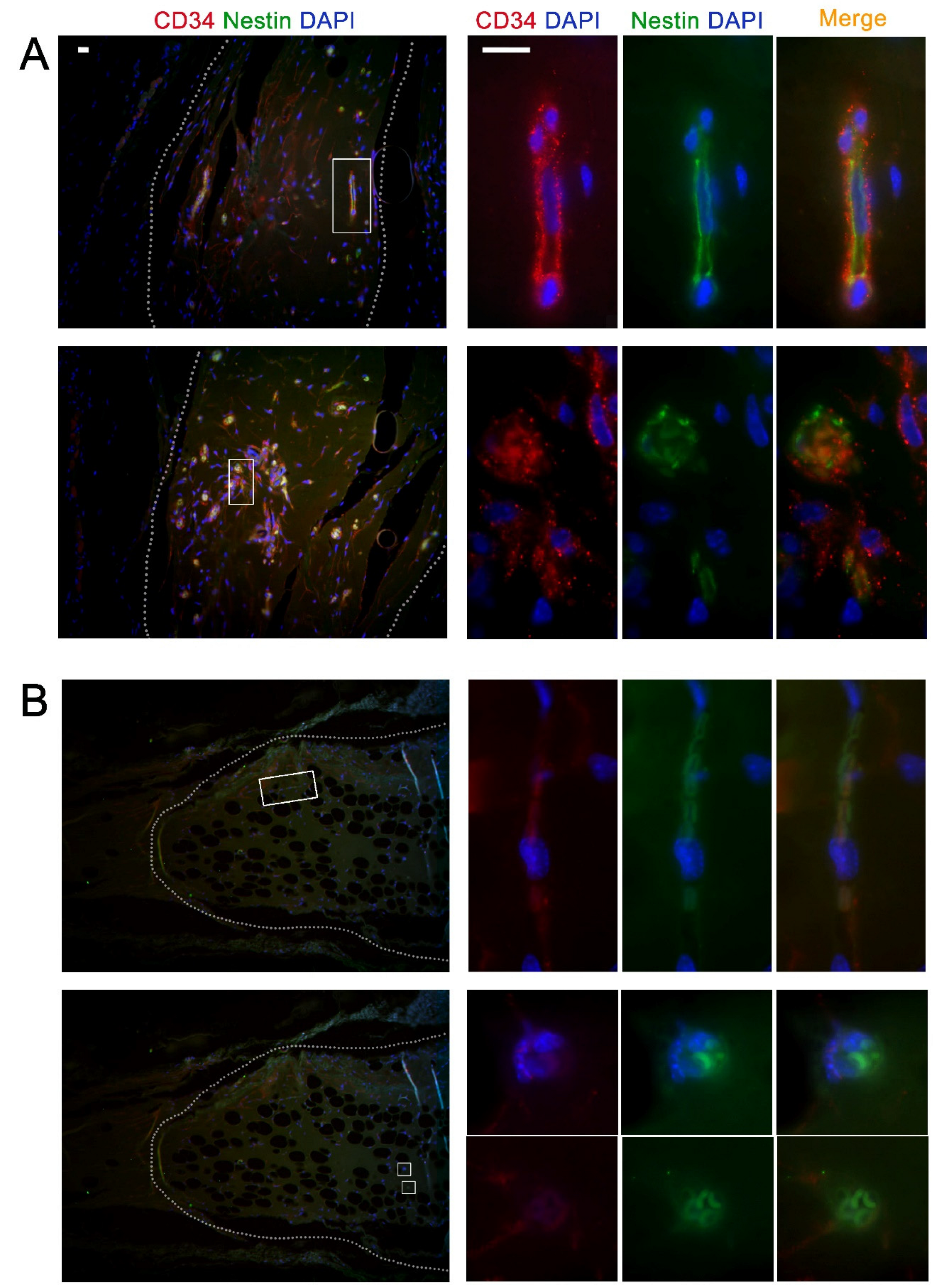
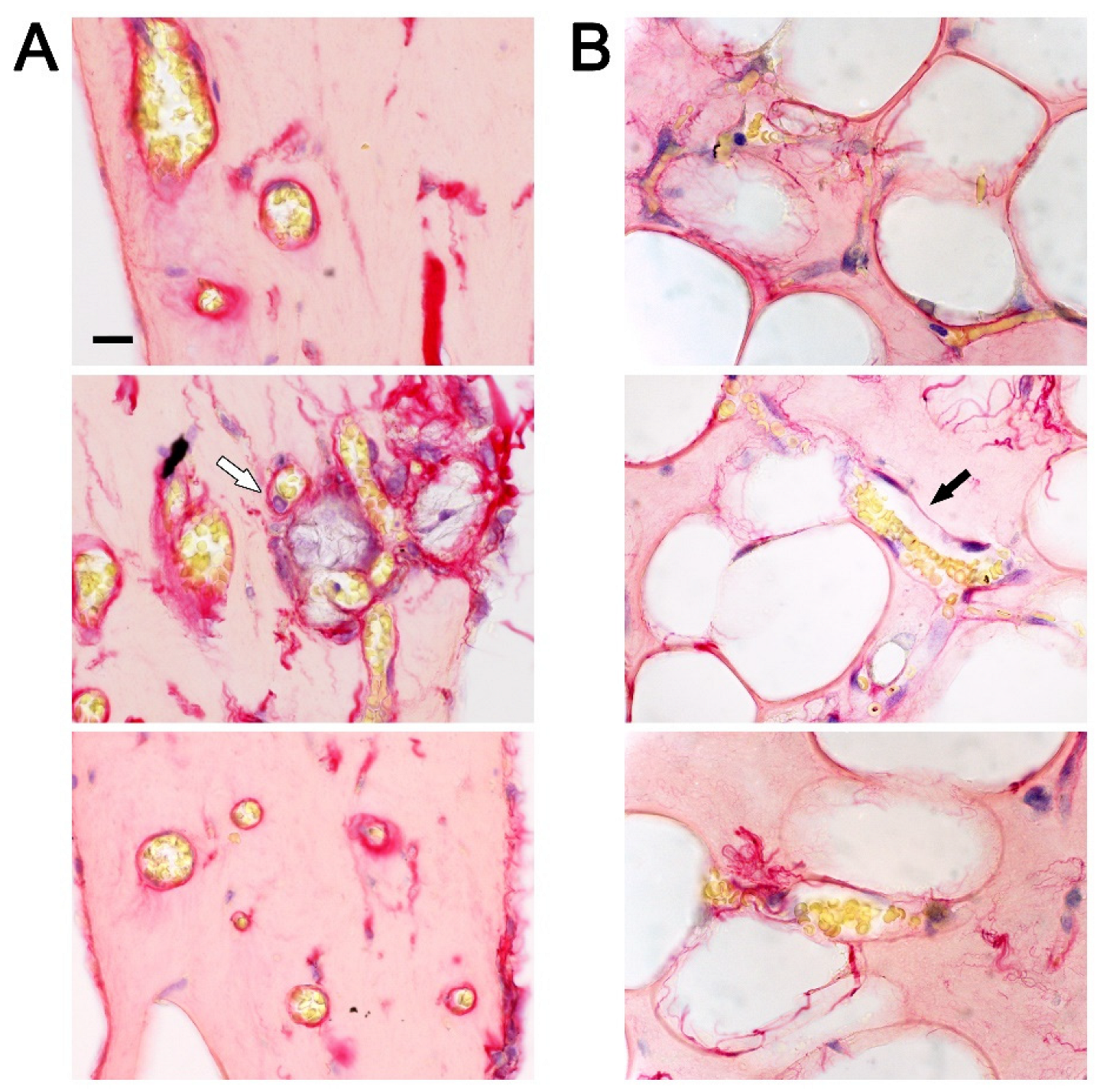
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| MPC | Mesangiogenic Progenitor Cell |
| MSC | Mesenchymal Stromal Cell |
| CBMP | Cell-based Medicinal product |
| BM | Bone Marrow |
| hBM-MNC | Human Bone Marrow Mononuclear Cell |
| hPSC | Human Pluripotent stem cell |
| SE | Standard Error |
| 3D | Three dimensions |
| WHO | World Health Organization |
| OCT-4 | Octamer-binding transcription factor 4 |
| SOX15 | SRY-box transcription factor 15 |
References
- Petrini, M.; Pacini, S.; Trombi, L.; Fazzi, R.; Montali, M.; Ikehara, S.; Abraham, N.G. Identification and purification of mesodermal progenitor cells from human adult bone marrow. Stem Cells Dev. 2009, 18, 857–66. [Google Scholar] [CrossRef]
- Montali, M.; Barachini, S.; Pacini, S.; Panvini, F.M.; Petrini, M. Isolating Mesangiogenic Progenitor Cells (MPCs) from Human Bone Marrow. J. Vis. Exp. 2016. [Google Scholar] [CrossRef] [PubMed]
- Fazzi, R.; Pacini, S.; Carnicelli, V.; Trombi, L.; Montali, M.; Lazzarini, E.; Petrini, M. Mesodermal progenitor cells (MPCs) differentiate into mesenchymal stromal cells (MSCs) by activation of Wnt5/calmodulin signalling pathway. PLoS ONE 2011, 6, e25600. [Google Scholar] [CrossRef]
- Montali, M.; Panvini, F.M.; Barachini, S.; Ronca, F.; Carnicelli, V.; Mazzoni, S.; Petrini, I.; Pacini, S. Human adult mesangiogenic progenitor cells reveal an early angiogenic potential, which is lost after mesengenic differentiation. Stem Cell. Res. Ther. 2017, 8, 106. [Google Scholar] [CrossRef]
- Pacini, S.; Barachini, S.; Montali, M.; Carnicelli, V.; Fazzi, R.; Parchi, P.; Petrini, M. Mesangiogenic Progenitor Cells Derived from One Novel CD64(bright)CD31(bright)CD14(neg) Population in Human Adult Bone Marrow. Stem Cells Dev. 2016, 25, 661–73. [Google Scholar] [CrossRef]
- Pacini, S. Deterministic and stochastic approaches in the clinical application of mesenchymal stromal cells (MSCs). Front Cell. Dev. Biol. 2014, 2, 50. [Google Scholar] [CrossRef] [PubMed]
- Perez, J.R.; Kouroupis, D.; Li, D.J.; Best, T.M.; Kaplan, L.; Correa, D. Tissue Engineering and Cell-Based Therapies for Fractures and Bone Defects. Front Bioeng. Biotechnol. 2018, 6, 105. [Google Scholar] [CrossRef]
- Ilic, D.; Ogilvie, C. Pluripotent Stem Cells in Clinical Setting-New Developments and Overview of Current Status. Stem Cells 2022, 40, 791–801. [Google Scholar] [CrossRef]
- Pacini, S.; Carnicelli, V.; Trombi, L.; Montali, M.; Fazzi, R.; Lazzarini, E.; Giannotti, S.; Petrini, M. Constitutive expression of pluripotency-associated genes in mesodermal progenitor cells (MPCs). PLoS ONE 2010, 5, e9861. [Google Scholar] [CrossRef] [PubMed]
- World Health Organizzation. Recommendations for the evaluation of animal cell cultures as substrates for the manufacture of biological medicinal products and for the characterization of cell banks. 2013, Annex 3. [Google Scholar]
- Sato, Y.; Bando, H.; Di, P.M.; Gowing, G.; Herberts, C.; Jackman, S.; Leoni, G.; Libertini, S.; MacLachlan, T.; McBlane, J.W.; et al. Tumorigenicity assessment of cell therapy products: The need for global consensus and points to consider. Cytotherapy 2019, 21, 1095–1111. [Google Scholar] [CrossRef]
- Delgado, J.A.; Guillén-Grima, F.; Moreno, C.; Panizo, C.; Pérez-Robles, C.; Mata, J.J.; Moreno, L.; Arana, P.; Chocarro, S.; Merino, J. A simple flow-cytometry method to evaluate peripheral blood contamination of bone marrow aspirates. J. Immunol. Methods 2017, 442, 54–58. [Google Scholar] [CrossRef] [PubMed]
- Doello, K. A new pentachrome method for the simultaneous staining of collagen and sulfated mucopolysaccharides. Yale J. Biol. Med. 2014, 87, 341–7. [Google Scholar]
- IARC/WHO. Classification of Tumours Editorial Board. WHO Classification of Tumours Online. 5th Edition; International Agency for Research on Cancer, 2019–2026. [Google Scholar]
- Leung, S.C.Y.; Nielsen, T.O.; Zabaglo, L.A.; Arun, I.; Badve, S.S.; Bane, A.L.; Bartlett, J.M.S.; Borgquist, S.; Chang, M.C.; Dodson, A.; et al. Analytical validation of a standardised scoring protocol for Ki67 immunohistochemistry on breast cancer excision whole sections: an international multicentre collaboration. Histopathology 2019, 75, 225–235. [Google Scholar] [CrossRef] [PubMed]
- Schmitz, F.; Voigtländer, H.; Strauss, D.; Schlemmer, H.P.; Kauczor, H.U.; Jang, H.; Sedaghat, S. Differentiating low- and high-proliferative soft tissue sarcomas using conventional imaging features and radiomics on MRI. BMC Cancer 2024, 30(24(1)), 1589. [Google Scholar] [CrossRef]
- Botquin, V.; Hess, H.; Fuhrmann, G.; Anastassiadis, C.; Gross, M.K.; Vriend, G.; Schöler, H.R. New POU dimer configuration mediates antagonistic control of an osteopontin preimplantation enhancer by Oct-4 and Sox-2. Genes. Dev. 1998, 12, 2073–90. [Google Scholar] [CrossRef]
- Ng, C.K.; Li, N.X.; Chee, S.; Prabhakar, S.; Kolatkar, P.R.; Jauch, R. Deciphering the Sox-Oct partner code by quantitative cooperativity measurements. Nucleic Acids Res. 2012, 40, 4933–41. [Google Scholar] [CrossRef]
- Choi, E.B.; Vodnala, M.; Saini, P.; Anugula, S.; Zerbato, M.; Ho, J.J.; Wang, J.; Ho, S.S.J.; Yoon, J.; Roels, M.; et al. Transcription factor SOX15 regulates stem cell pluripotency and promotes neural fate during differentiation by activating the neurogenic gene Hes5. J. Biol. Chem. 2023, 299, 102996. [Google Scholar] [CrossRef] [PubMed]
- Tokuzawa, Y.; Kaiho, E.; Maruyama, M.; Takahashi, K.; Mitsui, K.; Maeda, M.; Niwa, H.; Yamanaka, S. Fbx15 is a novel target of Oct3/4 but is dispensable for embryonic stem cell self-renewal and mouse development. Mol. Cell. Biol. 2003, 23, 2699–708. [Google Scholar] [CrossRef]
- Pacini, S. Mesangiogenic progenitor cells: a mesengenic and vasculogenic branch of hemopoiesis? A story of neglected plasticity. Front Cell. Dev. Biol. 2025, 13, 1513440. [Google Scholar] [CrossRef]
- Dou, D.; Lu, J.; Dou, J.; Huo, Y.; Gong, X.; Zhang, X.; Chen, X. Global regulatory considerations and practices for tumorigenicity evaluation of cell-based therapy. Regul. Toxicol. Pharmacol. 2025, 156, 105769. [Google Scholar] [CrossRef]
- Kleinman, H.K.; McGarvey, M.L.; Hassell, J.R.; Star, V.L.; Cannon, F.B.; Laurie, G.W.; Martin, G.R. Basement membrane complexes with biological activity. Biochemistry 1986, 28(25(2)), 312–8. [Google Scholar] [CrossRef] [PubMed]
- Fridman, R.; Giaccone, G.; Kanemoto, T.; Martin, G.R.; Gazdar, A.F.; Mulshine, J.L. Reconstituted basement membrane (matrigel) and laminin can enhance the tumorigenicity and the drug resistance of small cell lung cancer cell lines. Proc. Natl. Acad. Sci. U S A 1990, 87(17), 6698–702. [Google Scholar] [CrossRef]
- Kusakawa, S.; Machida, K.; Yasuda, S.; Takada, N.; Kuroda, T.; Sawada, R.; Okura, H.; Tsutsumi, H.; Kawamata, S.; Sato, Y. Characterization of in vivo tumorigenicity tests using severe immunodeficient NOD/Shi-scid IL2Rγnull mice for detection of tumorigenic cellular impurities in human cell-processed therapeutic products. Regen. Ther. 2015, 19(1), 30–37. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z. Assessing Tumorigenicity in Stem Cell-Derived Therapeutic Products: A Critical Step in Safeguarding Regenerative Medicine. Bioengineering 2023, 19(10(7)), 857. [Google Scholar] [CrossRef]
- Itkin, T.; Gur-Cohen, S.; Spencer, J.A.; Schajnovitz, A.; Ramasamy, S.K.; Kusumbe, A.P.; Ledergor, G.; Jung, Y.; Milo, I.; Poulos, M.G.; et al. Distinct bone marrow blood vessels differentially regulate haematopoiesis. Nature 2016, 21(532(7599)), 323–8. [Google Scholar] [CrossRef]
- Panvini, F.M.; Pacini, S.; Montali, M.; Barachini, S.; Mazzoni, S.; Morganti, R.; Ciancia, E.M.; Carnicelli, V.; Petrini, M. High NESTIN Expression Marks the Endosteal Capillary Network in Human Bone Marrow. Front Cell. Dev. Biol. 2020, 8, 596452. [Google Scholar] [CrossRef] [PubMed]
- Boueya, I.L.; Sandhow, L.; Albuquerque, J.R.P.; Znaidi, R.; Passaro, D. Endothelial heterogeneity in bone marrow: insights across development, adult life and leukemia. Leukemia 2025, 39(1), 8–24. [Google Scholar] [CrossRef]
- Rao, R.R.; Stegemann, J.P. Cell-based approaches to the engineering of vascularized bone tissue. Cytotherapy 2013, 15, 1309. [Google Scholar] [CrossRef]
- Takebe, T.; Kobayashi, S.; Suzuki, H.; Mizuno, M.; Chang, Y.M.; Yoshizawa, E.; Kimura, M.; Hori, A.; Asano, J.; Maegawa, J.; et al. Transient vascularization of transplanted human adult-derived progenitors promotes self-organizing cartilage. J. Clin. Invest 2014, 124, 4325–34. [Google Scholar] [CrossRef]
- Cong, B.; Zhang, F.H.; Zhang, H.G. Stem cell-based cartilage regeneration: Biological strategies, engineering innovations, and clinical translation. World J. Stem Cells 2025, 26(17(9)), 108523. [Google Scholar] [CrossRef]
- Chu, G.; Zhang, W.; Han, F.; Li, K.; Liu, C.; Wei, Q.; Wang, H.; Liu, Y.; Han, F.; Li, B. The role of microenvironment in stem cell-based regeneration of intervertebral disc. Front Bioeng. Biotechnol. 2022, 9(10), 968862. [Google Scholar] [CrossRef] [PubMed]
- Decarli, M.C.; Amaral, R.; Santos, D.P.dos; Tofani, L.B.; Katayama, E.; Rezende, R.A.; Silva, J.V.L.da; Swiech, K.; Suazo, C.A.T.; Mota, C.; et al. Cell spheroids as a versatile research platform: formation mechanisms, high throughput production, characterization and applications. Biofabrication 2021, 13. [Google Scholar] [CrossRef] [PubMed]
- Vakhrushev, I.V.; Nezhurina, E.K.; Karalkin, P.A.; Tsvetkova, A.V.; Sergeeva, N.S.; Majouga, A.G.; Yarygin, K.N. Heterotypic Multicellular Spheroids as Experimental and Preclinical Models of Sprouting Angiogenesis. Biology 2021, 11. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Kusumbe, A.P.; Cai, H.; Wan, Q.; Chen, J. Type H blood vessels in coupling angiogenesis-osteogenesis and its application in bone tissue engineering. J. Biomed. Mater. Res. Part B Appl. Biomater. 2023, 111. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



