Submitted:
25 April 2026
Posted:
28 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
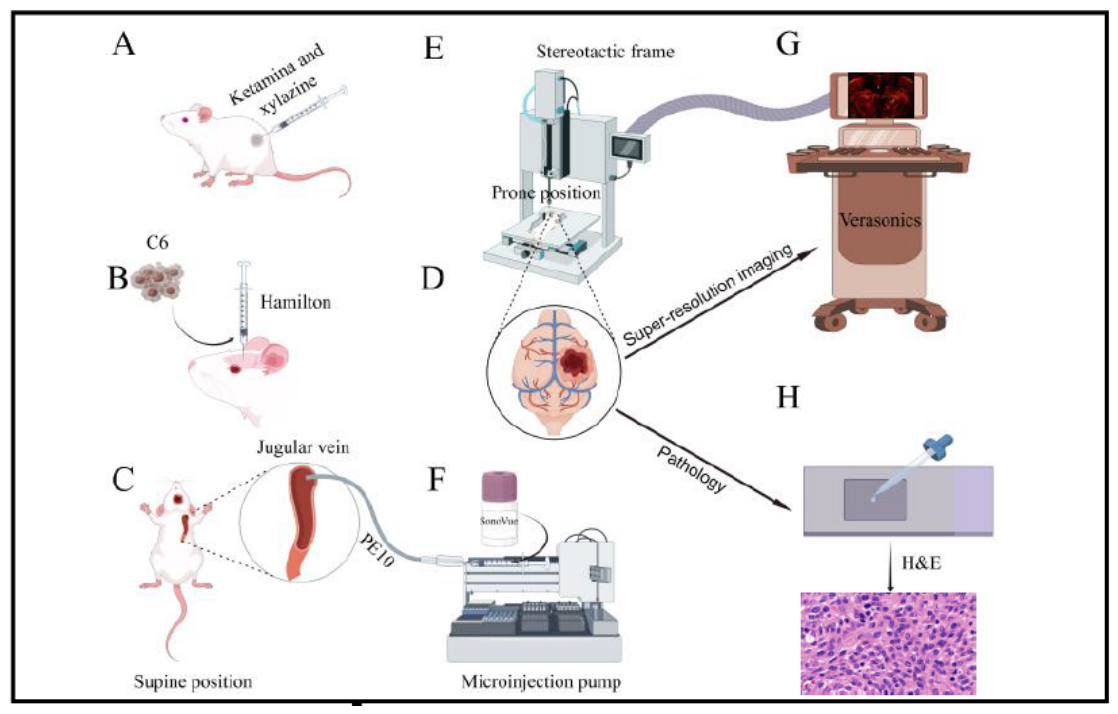
2.1. Ethics Statement
2.2. Animal Model
2.3. Pre-Imaging Preparation
2.4. Multi-Plane RF Data Acquisition
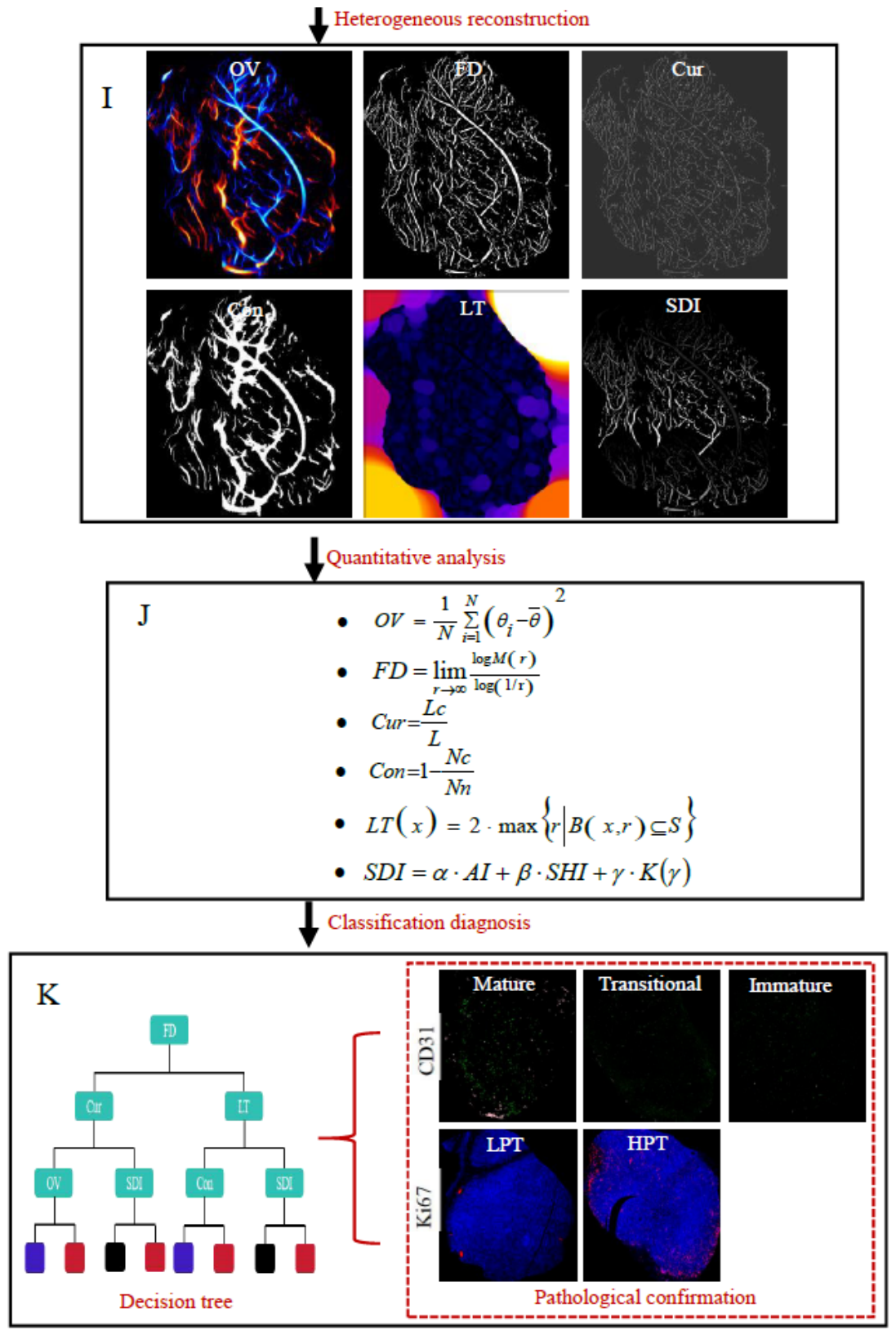
2.5. Image Reconstruction
2.6. Histopathological Analysis
2.7. Imaging-Derived Parameter Analysis
2.8. Statistical Analysis
3. Results
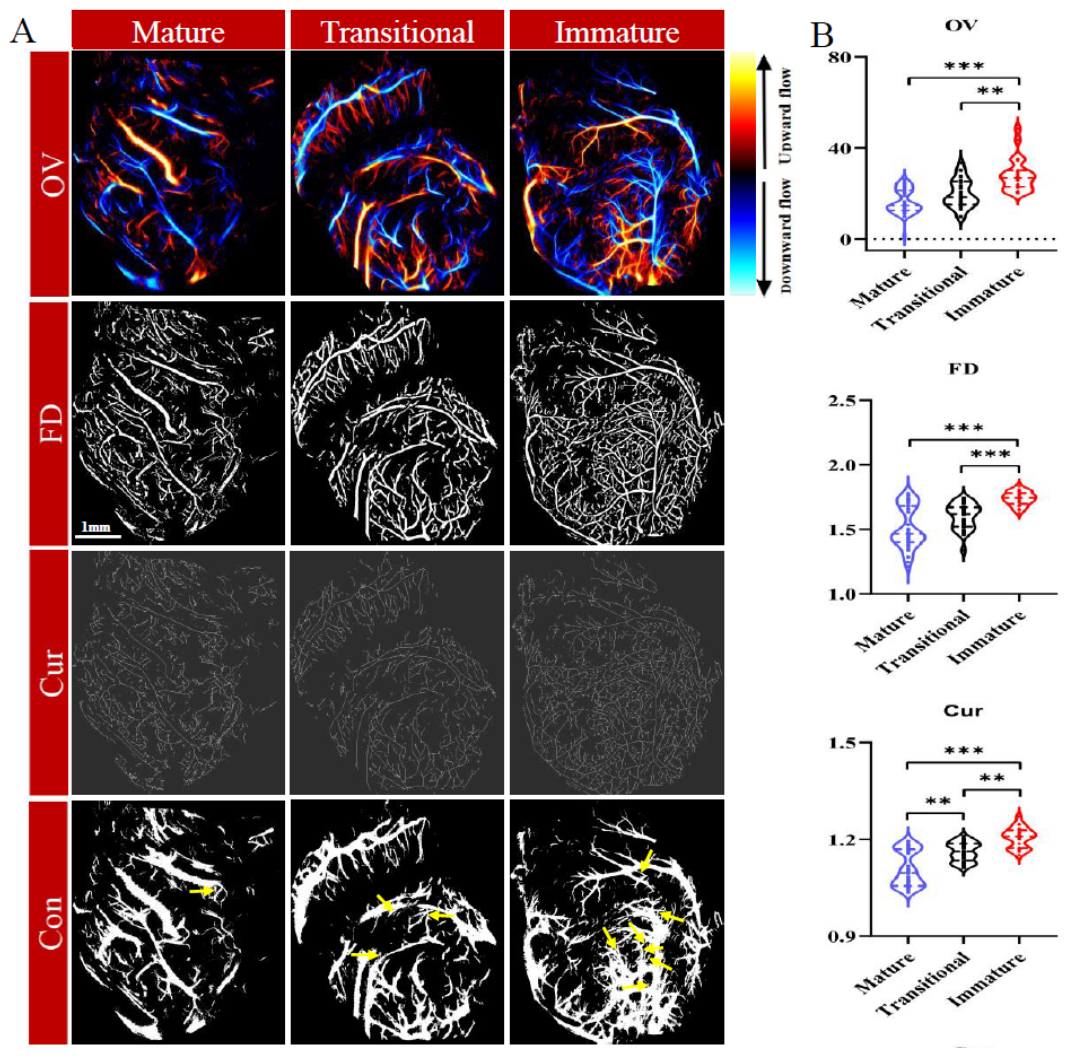
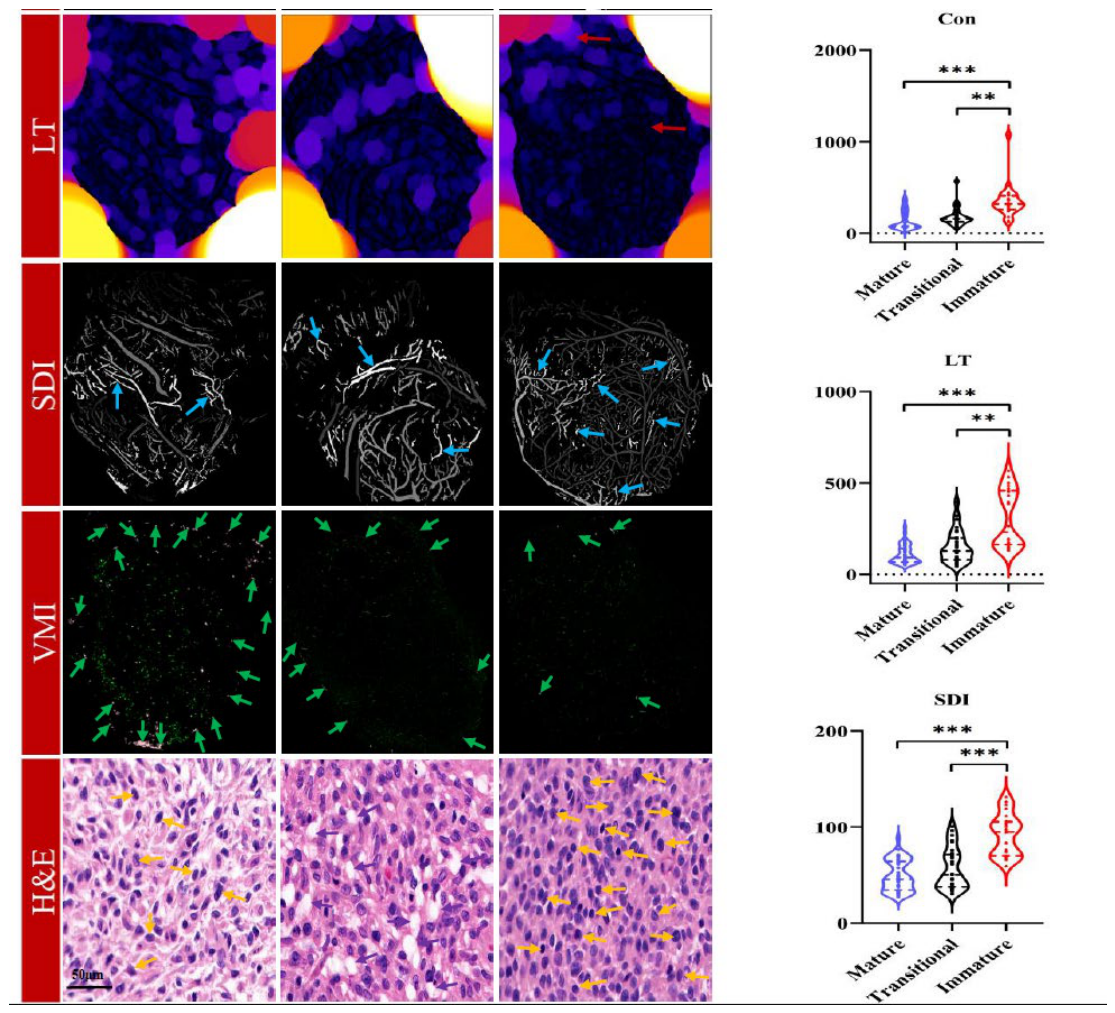
3.1. Associations Between Microvascular Heterogeneity and Pathology
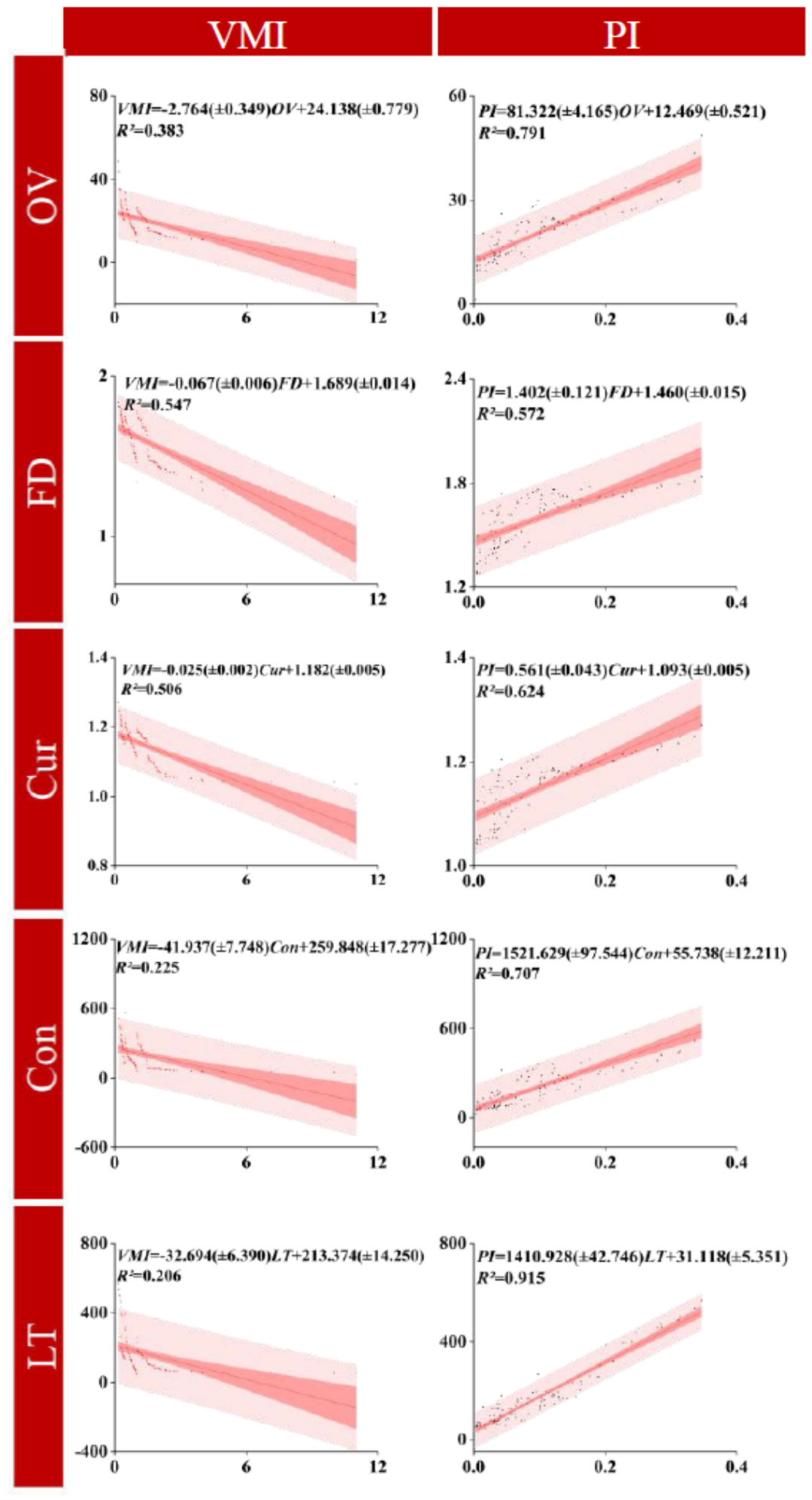
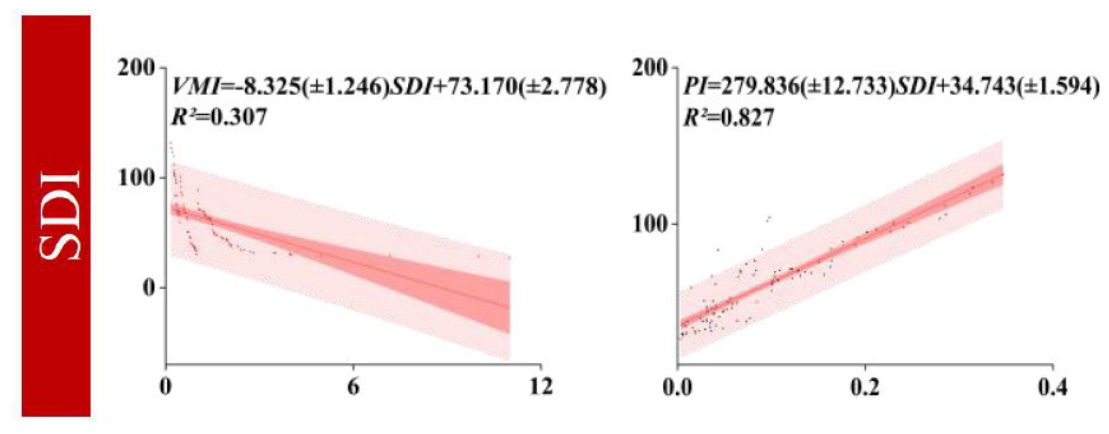
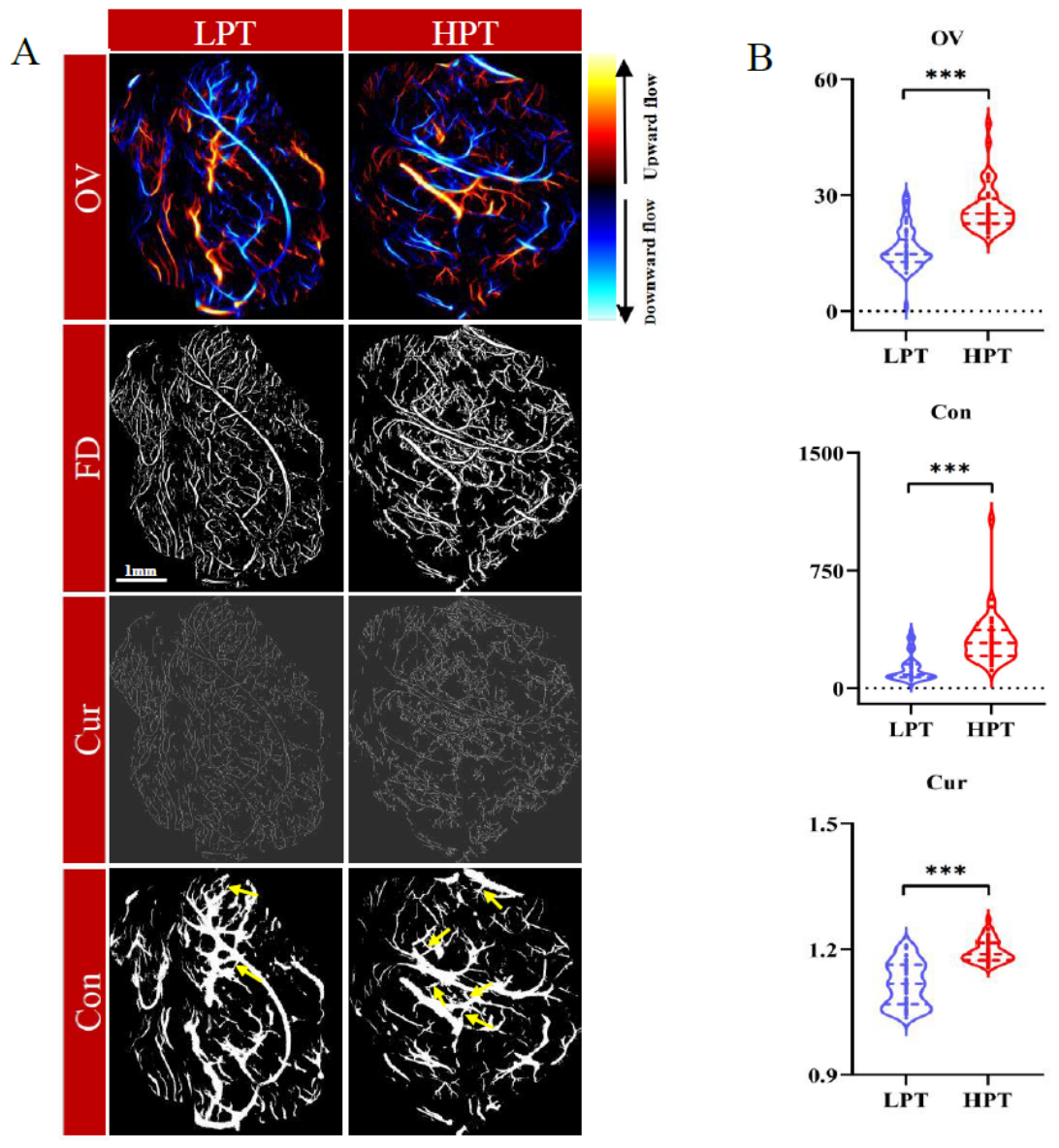
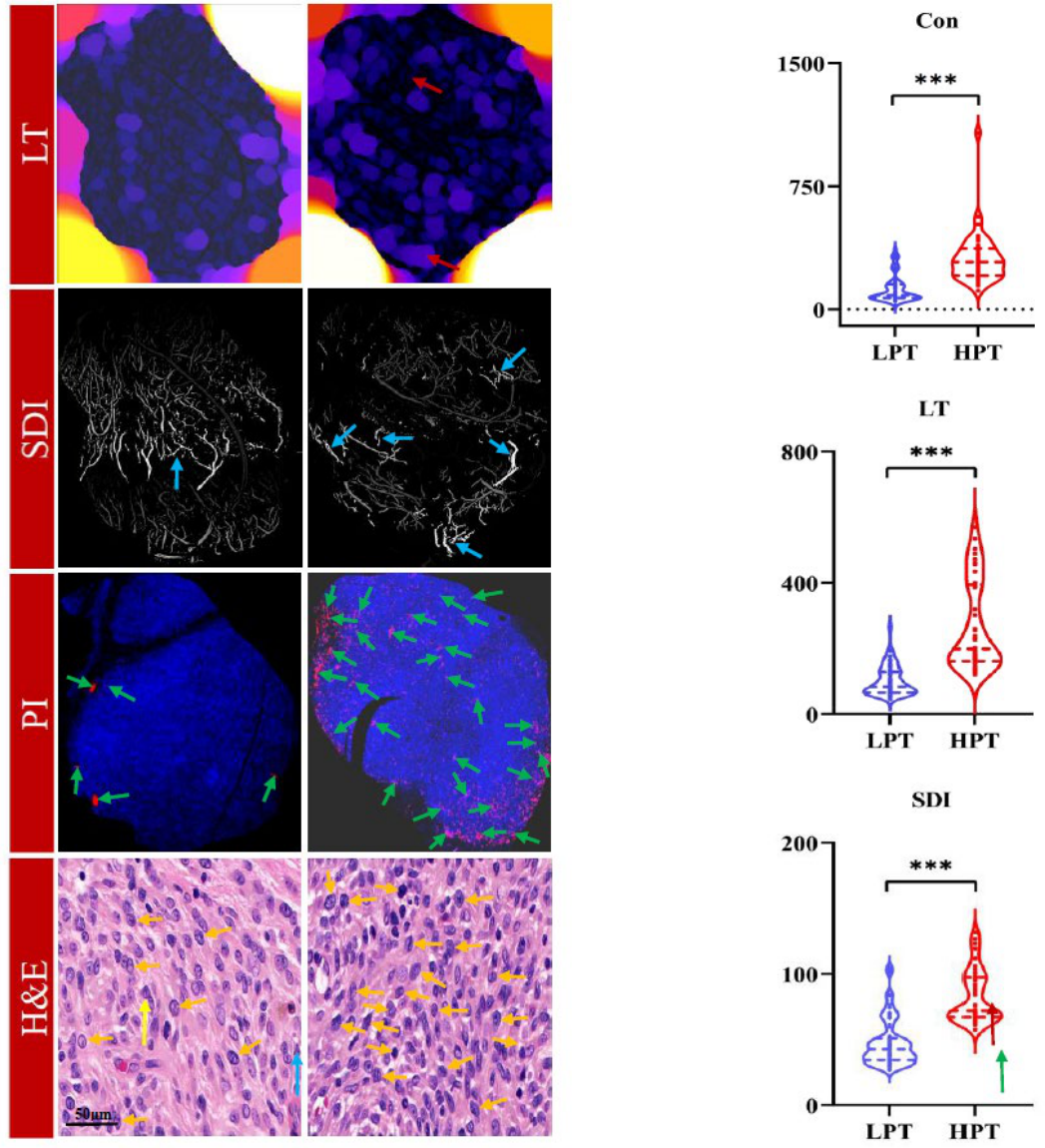
3.2. Microvascular Heterogeneity Across Different VMI Levels
3.3. Microvascular Heterogeneity Across Different PI Levels
3.4. Classification of Microvascular Maturity
3.5. Classification of Cellular Proliferative Status
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of interest
Abbreviations
| CART | classification and regression tree |
| CI | confidence interval |
| FD | fractal dimension |
| GBM | glioblastoma |
| LT | local thickness |
| PI | proliferation index |
| SDI | spatial distribution index |
| ULM | ultrasound localization microscopy |
| VMI | vascular maturity index |
References
- Roncali, L.; Hindré, F.; Samarut, E.; Lacoeuille, F.; Rousseau, A.; Lemée, J.M.; Garcion, E.; Chérel, M. Current landscape and future directions of targeted-alpha-therapy for glioblastoma treatment. Theranostics 2025, 15, 4861–4889. [Google Scholar] [CrossRef]
- Begley, S.L.; O’Rourke, D.M.; Binder, Z.A. CAR T cell therapy for glioblastoma: A review of the first decade of clinical trials. Mol. Ther. 2025, 33, 2454–2461. [Google Scholar] [CrossRef]
- Bayona, C.; Ranđelović, T.; Ochoa, I. Tumor Microenvironment in Glioblastoma: The Central Role of the Hypoxic-Necrotic Core. Cancer Lett. 2026, 639, 218216. [Google Scholar] [CrossRef]
- Jütten K, O.J.; Kernbach, J.M.; Meyer-Baese, A.; Meyer-Baese, U.; Hamou, H.A.; Clusmann, H.; Wiesmann, M.; Bremer, J.; Koch, H.; Bak, A.; et al. High peritumoral network connectedness in glioblastoma reveals a distinct epigenetic signature and is associated with decreased overall survival. Neuro Oncol. 2025, 27, 2564–2573. [Google Scholar] [CrossRef]
- Katanasaka, Y.; Kodera, Y.; Kitamura, Y.; Morimoto, T.; Tamura, T.; Koizumi, F. Epidermal growth factor receptor variant type III markedly accelerates angiogenesis and tumor growth via inducing c-myc mediated angiopoietin-like 4 expression in malignant glioma. Mol. Cancer 2013, 12, 31. [Google Scholar] [CrossRef] [PubMed]
- Stadlbauer, A.; Zimmermann, M.; Kitzwögerer, M.; Oberndorfer, S.; Rössler, K.; Dörfler, A.; Buchfelder, M.; Heinz, G. MR Imaging-derived Oxygen Metabolism and Neovascularization Characterization for Grading and IDH Gene Mutation Detection of Gliomas. Radiology 2017, 283, 799–809. [Google Scholar] [CrossRef]
- Louis, D.N.; Perry, A.; Wesseling, P.; Brat, D.J.; Cree, I.A.; Figarella-Branger, D.; Hawkins, C.; Ng, H.K.; Pfister, S.M.; Reifenberger, G.; et al. The 2021 WHO Classification of Tumors of the Central Nervous System: a summary. Neuro Oncol. 2021, 23, 1231–1251. [Google Scholar] [CrossRef] [PubMed]
- Patel, A.P.; Tirosh, I.; Trombetta, J.J.; Shalek, A.K.; Gillespie, S.M.; Wakimoto, H.; Cahill, D.P.; Nahed, B.V.; Curry, W.T.; Martuza, R.L.; et al. Single-cell RNA-seq highlights intratumoral heterogeneity in primary glioblastoma. Science 2014, 344, 1396–1401. [Google Scholar] [CrossRef]
- Verhaak, R.G.; Hoadley, K.A.; Purdom, E.; Wang, V.; Qi, Y.; Wilkerson, M.D.; Miller, C.R.; Ding, L.; Golub, T.; Mesirov, J.P.; et al. Integrated genomic analysis identifies clinically relevant subtypes of glioblastoma characterized by abnormalities in PDGFRA, IDH1, EGFR, and NF1. Cancer Cell. 2010, 17, 98–110. [Google Scholar] [CrossRef]
- Mathur, R.; Wang, Q.; Schupp, P.G.; Nikolic, A.; Hilz, S.; Hong, C.; Grishanina, N.R.; Kwok, D.; Stevers, N.O.; Jin, Q.; et al. Glioblastoma evolution and heterogeneity from a 3D whole-tumor perspective. Cell. 2024, 187, 446–463.e16. [Google Scholar] [CrossRef]
- Chokshi, C.R.; Shaikh, M.V.; Brakel, B.; Rossotti, M.A.; Tieu, D.; Maich, W.; Anand, A.; Chafe, S.C.; Zhai, K.; Suk, Y.; et al. Targeting axonal guidance dependencies in glioblastoma with ROBO1 CAR T cells. Nat. Med. 2024, 30, 2936–2946. [Google Scholar] [CrossRef]
- Mikolajewicz, N.; Tatari, N.; Wei, J.; Savage, N.; Granda, F.A.; Dimitrov, V.; Chen, D.; Zador, Z.; Dasgupta, K.; Aguilera-Uribe, M.; et al. Functional profiling of murine glioma models highlights targetable immune evasion phenotypes. Acta Neuropathol. 2024, 148, 74. [Google Scholar] [CrossRef]
- Hanahan, D. Hallmarks of Cancer: New Dimensions. Cancer Discov. 2022, 12, 31–46. [Google Scholar] [CrossRef] [PubMed]
- Shamshiripour, P.; Rahnama, M.; Nikoobakht, M.; Rad, V.F.; Moradi, A.R.; Ahmadvand, D. Extracellular vesicles derived from dendritic cells loaded with VEGF-A siRNA and doxorubicin reduce glioma angiogenesis in vitro. J. Control Release 2024, 369, 128–145. [Google Scholar] [CrossRef]
- Jain, R.K.; Duda, D.G.; Willett, C.G.; Sahani, D.V.; Zhu, A.X.; Loeffler, J.S.; Batchelor, T.T.; Sorensen, A.G. Biomarkers of response and resistance to antiangiogenic therapy. Nat. Rev. Clin. Oncol. 2009, 6, 327–338. [Google Scholar] [CrossRef]
- Kuczynski, E.A.; Reynolds, A.R. Vessel co-option and resistance to anti-angiogenic therapy. Angiogenesis 2020, 23, 55–74. [Google Scholar] [CrossRef]
- Lu, K.; Zhang, R.; Wang, H.; Li, C.; Yang, Z.; Xu, K.; Cao, X.; Wang, N.; Cai, W.; Zeng, J.; et al. PEGylated Ultrasmall Iron Oxide Nanoparticles as MRI Contrast Agents for Vascular Imaging and Real-Time Monitoring. ACS Nano 2025, 19, 3519–3530. [Google Scholar] [CrossRef] [PubMed]
- Kang, C.K.; Park, C.A.; Lee, D.S.; Lee, Y.B.; Park, C.W.; Kim, Y.B.; Cho, Z.H. Velocity measurement of microvessels using phase-contrast magnetic resonance angiography at 7 Tesla MRI. Magn. Reson Med. 2016, 75, 1640–1646. [Google Scholar] [CrossRef]
- Cho, Z.H.; Lee, Y.B.; Kang, C.K.; Yang, J.W.; Jung, I.H.; Park, C.A.; Park, C.W.; Kim, Y.B. Microvascular imaging of asymptomatic MCA steno-occlusive patients using ultra-high-field 7T MRI. J. Neurol. 2013, 260, 144–150. [Google Scholar] [CrossRef]
- Shi, Z.; Zhao, X.; Zhu, S.; Miao, X.; Zhang, Y.; Han, S.; Wang, B.; Zhang, B.; Ye, X.; Dai, Y.; et al. Time-of-Flight Intracranial MRA at 3 T versus 5 T versus 7 T: Visualization of Distal Small Cerebral Arteries. Radiology 2023, 306, 207–217. [Google Scholar] [CrossRef]
- Porter, J.C.; Ganeshan, B.; Win, T.; Fraioli, F.; Khan, S.; Rodriguez-Justo, M.; Endozo, R.; Shortman, R.I.; Hoy, L.R.; Maher, T.M.; et al. [18F]FDG PET/CT Signal Correlates with Neoangiogenesis Markers in Patients with Fibrotic Interstitial Lung Disease Who Underwent Lung Biopsy: Implication for the Use of PET/CT in Diffuse Lung Diseases. J. Nucl. Med. 2024, 65, 617–622. [Google Scholar] [CrossRef]
- Harat, M.; Rakowska, J.; Harat, M.; Szylberg, T.; Furtak, J.; Miechowicz, I.; Małkowski, B. Combining amino acid PET and MRI imaging increases accuracy to define malignant areas in adult glioma. Nat. Commun. 2023, 14, 4572. [Google Scholar] [CrossRef]
- Zhang, Y.; Bander, E.D.; Lee, Y.; Muoser, C.; Schaffer, C.B.; Nishimura, N. Microvessel occlusions alter amyloid-beta plaque morphology in a mouse model of Alzheimer’s disease. J. Cereb. Blood Flow. Metab. 2020, 40, 2115–2131. [Google Scholar] [CrossRef]
- Yang, L.; Chen, P.; Wen, X.; Zhao, Q. Optical coherence tomography (OCT) and OCT angiography: Technological development and applications in brain science. Theranostics 2025, 15, 122–140. [Google Scholar] [CrossRef]
- Errico, C.; Pierre, J.; Pezet, S.; Desailly, Y.; Lenkei, Z.; Couture, O.; Tanter, M. Ultrafast ultrasound localization microscopy for deep super-resolution vascular imaging. Nature 2015, 527, 499–502. [Google Scholar] [CrossRef]
- Demené, C.; Robin, J.; Dizeux, A.; Heiles, B.; Pernot, M.; Tanter, M.; Perren, F. Transcranial ultrafast ultrasound localization microscopy of brain vasculature in patients. Nat. Biomed. Eng. 2021, 5, 219–228. [Google Scholar] [CrossRef]
- Renaudin, N.; Demené, C.; Dizeux, A.; Ialy-Radio, N.; Pezet, S.; Tanter, M. Functional ultrasound localization microscopy reveals brain-wide neurovascular activity on a microscopic scale. Nat. Methods 2022, 19, 1004–1012. [Google Scholar] [CrossRef]
- Couture, O.; Hingot, V.; Heiles, B.; Muleki-Seya, P.; Tanter, M. Ultrasound Localization Microscopy and Super-Resolution: A State of the Art. IEEE Trans. Ultrason Ferroelectr. Freq. Control. 2018, 65, 1304–1320. [Google Scholar] [CrossRef]
- Yan, J.; Huang, B.; Tonko, J.; Toulemonde, M.; Hansen-Shearer, J.; Tan, Q.; Riemer, K.; Ntagiantas, K.; Chowdhury, R.A.; Lambiase, P.D.; et al. Transthoracic ultrasound localization microscopy of myocardial vasculature in patients. Nat. Biomed. Eng. 2024, 8, 689–700. [Google Scholar] [CrossRef]
- Feng, Y.N.; Xu, Z.H.; Liu, J.T.; Sun, X.L.; Wang, D.Q.; Yu, Y. Intelligent prediction of RBC demand in trauma patients using decision tree methods. Mil. Med. Res. 2021, 8, 33. [Google Scholar] [CrossRef]
- Montorsi, F.; Oelke, M.; Henneges, C.; Brock, G.; Salonia, A.; d’Anzeo, G.; Rossi, A.; Mulhall, J.P.; Büttner, H. Exploratory Decision-Tree Modeling of Data from the Randomized REACTT Trial of Tadalafil Versus Placebo to Predict Recovery of Erectile Function After Bilateral Nerve-Sparing Radical Prostatectomy. Eur. Urol. 2016, 70, 529–537. [Google Scholar] [CrossRef]
- Thangaraj, J.W.V.; Krishna, N.S.; Devika, S.; Egambaram, S.; Dhanapal, S.R.; Khan, S.A.; Srivastava, A.K.; Mishra, A.; Shrinivasa, B.; Gour, D.; et al. Estimates of the burden of human rabies deaths and animal bites in India, 2022-23: a community-based cross-sectional survey and probability decision-tree modelling study. Lancet Infect. Dis. 2025, 25, 126–134. [Google Scholar] [CrossRef]
- Rodríguez, A.H.; Avilés-Jurado, F.X.; Díaz, E.; Schuetz, P.; Trefler, S.I.; Solé-Violán, J.; Cordero, L.; Vidaur, L.; Estella, Á.; Pozo, L.J.C.; et al. Procalcitonin (PCT) levels for ruling-out bacterial coinfection in ICU patients with influenza: A CHAID decision-tree analysis. J. Infect. 2016, 72, 143–151. [Google Scholar] [CrossRef]
- Lowerison, M.R.; Vaithiyalingam, C.S.N.; Dong, Z.; Chen, X.; You, Q.; Llano, D.A.; Song, P. Super-Resolution Ultrasound Reveals Cerebrovascular Impairment in a Mouse Model of Alzheimer’s Disease. J. Neurosci. 2024, 44, e1251232024. [Google Scholar] [CrossRef]
- Yan, S.; Shou, J.; Yu, J.; Song, J.; Mao, Y.; Xu, K. Ultrafast Ultrasound Vector Doppler for Small Vasculature Imaging. IEEE Trans. Ultrason Ferroelectr. Freq. Control. 2023, 70, 613–624. [Google Scholar] [CrossRef]
- Nyúl-Tóth, Á.; Negri, S.; Sanford, M.; Jiang, R.; Patai, R.; Budda, M.; Petersen, B.; Pinckard, J.; Chandragiri, S.S.; Shi, H.; et al. Novel intravital approaches to quantify deep vascular structure and perfusion in the aging mouse brain using ultrasound localization microscopy (ULM). J. Cereb. Blood Flow. Metab. 2024, 44, 1378–1396. [Google Scholar] [CrossRef]
- Conroy, S.; Wagemakers, M.; Walenkamp, A.M.; Kruyt, F.A.; denDunnen, W.F. Novel insights into vascularization patterns and angiogenic factors in glioblastoma subclasses. J. Neurooncol 2017, 131, 11–20. [Google Scholar] [CrossRef]
- Ullrich, R.; Backes, H.; Li, H.; Kracht, L.; Miletic, H.; Kesper, K.; Neumaier, B.; Heiss, W.D.; Wienhard, K.; Jacobs, A.H. Glioma proliferation as assessed by 3’-fluoro-3’-deoxy-L-thymidine positron emission tomography in patients with newly diagnosed high-grade glioma. Clin. Cancer Res. 2008, 14, 2049–2055. [Google Scholar] [CrossRef]
- Schindelin, J.; Arganda-Carreras, I.; Frise, E.; Kaynig, V.; Longair, M.; Pietzsch, T.; Preibisch, S.; Rueden, C.; Saalfeld, S.; Schmid, B.; et al. Fiji: an open-source platform for biological-image analysis. Nat. Methods. 2012, 9, 676–682. [Google Scholar] [CrossRef]
- Wissel, T.; Stüber, P.; Wagner, B.; Bruder, R.; Schweikard, A.; Ernst, F. Enriching 3D optical surface scans with prior knowledge: tissue thickness computation by exploiting local neighborhoods. Int. J. Comput Assist Radiol. Surg. 2016, 11, 569–579. [Google Scholar] [CrossRef]
- Schmidt, A.J.; Wright, G.D.; Ronchese, F.; Price, K.M. Skin Whole-Mount Immunofluorescent Staining Protocol, 3D Visualization, and Spatial Image Analysis. Curr. Protoc. 2023, 3, e820. [Google Scholar] [CrossRef]
- Park, J.S.; Choi, S.H.; Sohn, C.H.; Park, J. Joint Reconstruction of Vascular Structure and Function Maps in Dynamic Contrast Enhanced MRI Using Vascular Heterogeneity Priors. IEEE Trans. Med. Imaging 2022, 41, 52–62. [Google Scholar] [CrossRef]
- Sweeney, A.; Langley, A.; Xavierselvan, M.; Shethia, R.T.; Solomon, P.; Arora, A.; Mallidi, S. Vascular regional analysis unveils differential responses to anti-angiogenic therapy in pancreatic xenografts through macroscopic photoacoustic imaging. Theranostics 2025, 15, 2649–2671. [Google Scholar] [CrossRef]
- Pitre-Champagnat, S.; Leguerney, I.; Bosq, J.; Peronneau, P.; Kiessling, F.; Calmels, L.; Coulot, J.; Lassau, N. Dynamic contrast-enhanced ultrasound parametric maps to evaluate intratumoral vascularization. Invest Radiol. 2015, 50, 212–217. [Google Scholar] [CrossRef]
- Tulessin, M.; Sarker, R.S.J.; Griger, J.; Leibing, T.; Geraud, C.; Weichert, W.; Steiger, K.; Mogler, C. Vascular Remodeling Is a Crucial Event in the Early Phase of Hepatocarcinogenesis in Rodent Models for Liver Tumorigenesis. Cells 2022, 11, 2129. [Google Scholar] [CrossRef]
- Calvisi, D.F.; Pinna, F.; Ladu, S.; Pellegrino, R.; Simile, M.M.; Frau, M.; De Miglio, M.R.; Tomasi, M.L.; Sanna, V.; Muroni, M.R.; et al. Forkhead box M1B is a determinant of rat susceptibility to hepatocarcinogenesis and sustains ERK activity in human HCC. Gut 2009, 58, 679–687. [Google Scholar] [CrossRef]
- Velez, E.; Goldberg, S.N.; Kumar, G.; Wang, Y.; Gourevitch, S.; Sosna, J.; Moon, T.; Brace, C.L.; Ahmed, M. Hepatic Thermal Ablation: Effect of Device and Heating Parameters on Local Tissue Reactions and Distant Tumor Growth. Radiology 2016, 281, 782–792. [Google Scholar] [CrossRef]
- Díaz-Flores, L.; Gutiérrez, R.; González-Gómez, M.; García, M.D.; Díaz-Flores, L.J.; González-Marrero, I.; Ávila, J.; Martín-Vasallo, P. Disproportion in Pericyte/Endothelial Cell Proliferation and Mechanisms of Intussusceptive Angiogenesis Participate in Bizarre Vessel Formation in Glioblastoma. Cells 2021, 10, 2625. [Google Scholar] [CrossRef]
- Eisenbarth, D.; Wang, Y.A. Glioblastoma heterogeneity at single cell resolution. Oncogene 2023, 42, 2155–2165. [Google Scholar] [CrossRef]
- Liu, Z.L.; Chen, H.H.; Zheng, L.L.; Sun, L.P.; Shi, L. Angiogenic signaling pathways and anti-angiogenic therapy for cancer. Signal Transduct. Target Ther. 2023, 8, 198. [Google Scholar] [CrossRef]
- Ho, R.L.Y.; Ho, I.A.W. Recent Advances in Glioma Therapy: Combining Vascular Normalization and Immune Checkpoint Blockade. Cancers 2021, 13, 3686. [Google Scholar] [CrossRef]
- Mosteiro, A.; Pedrosa, L.; Ferrés, A.; Diao, D.; Sierra, À.; González, J.J. The Vascular Microenvironment in Glioblastoma: A Comprehensive Review. Biomedicines 2022, 10, 1285. [Google Scholar] [CrossRef]
- Liu, L.; Yu, J.; Liu, Y.; Xie, L.; Hu, F.; Liu, H. Hypoxia-driven angiogenesis and metabolic reprogramming in vascular tumors. Front Cell. Dev. Biol. 2025, 13, 1572909. [Google Scholar] [CrossRef]
- Chen, Z.; Han, F.; Du, Y.; Shi, H.; Zhou, W. Hypoxic microenvironment in cancer: molecular mechanisms and therapeutic interventions. Signal Transduct. Target Ther. 2023, 8, 70. [Google Scholar] [CrossRef]
- Ghalehbandi, S.; Yuzugulen, J.; Pranjol, M.Z.I.; Pourgholami, M.H. Pourgholami, The role of VEGF in cancer-induced angiogenesis and research progress of drugs targeting VEGF. Eur. J. Pharmacol. 2023, 949, 175586. [Google Scholar] [CrossRef]
- Schwarz, S.; Denis, L.; Nedoschill, E.; Buehler, A.; Danko, V.; Hilger, A.C.; Brevis, N.F.; Dürr, N.R.; Schlunz-Hendann, M.; Brassel, F.; et al. Ultrasound Super-Resolution Imaging of Neonatal Cerebral Vascular Reorganization. Adv. Sci. (Weinh) 2025, 12, e2415235. [Google Scholar] [CrossRef]
- Jones, R.M.; DeRuiter, R.M.; Deshmukh, M.; Dayton, P.A.; Pinton, G.F. Non-invasive volumetric ultrasound localization microscopy detects vascular changes in mice with Alzheimer’s disease. Theranostics 2025, 15, 1110–1121. [Google Scholar] [CrossRef]
- Lin, H.; Wang, Z.; Liao, Y.; Yu, Z.; Xu, H.; Qin, T.; Tang, J.; Yang, X.; Chen, S.; Chen, X.; et al. Super-resolution ultrasound imaging reveals temporal cerebrovascular changes with disease progression in female 5×FAD mouse model of Alzheimer’s disease: correlation with pathological impairments. EBioMedicine 2024, 108, 105355. [Google Scholar] [CrossRef]
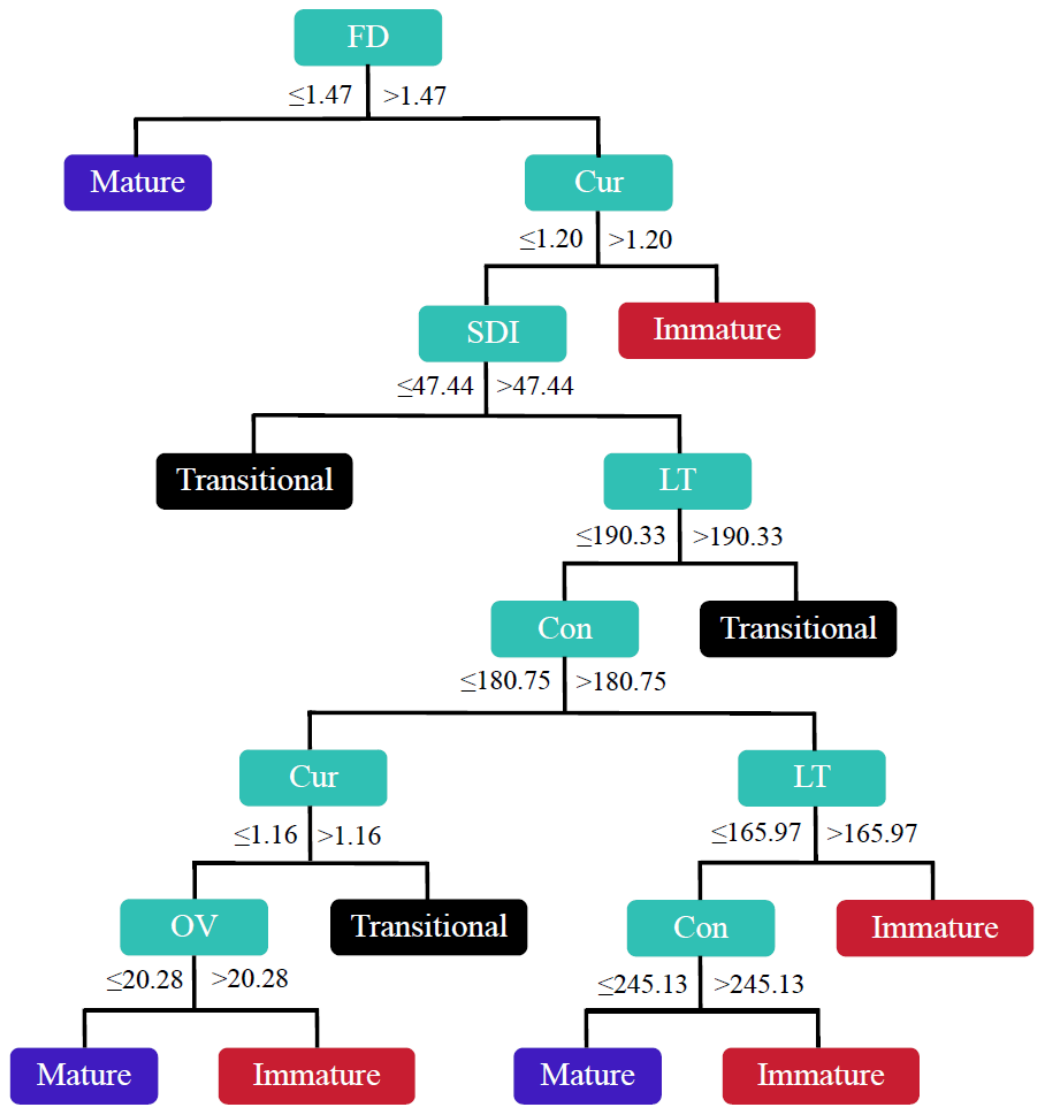
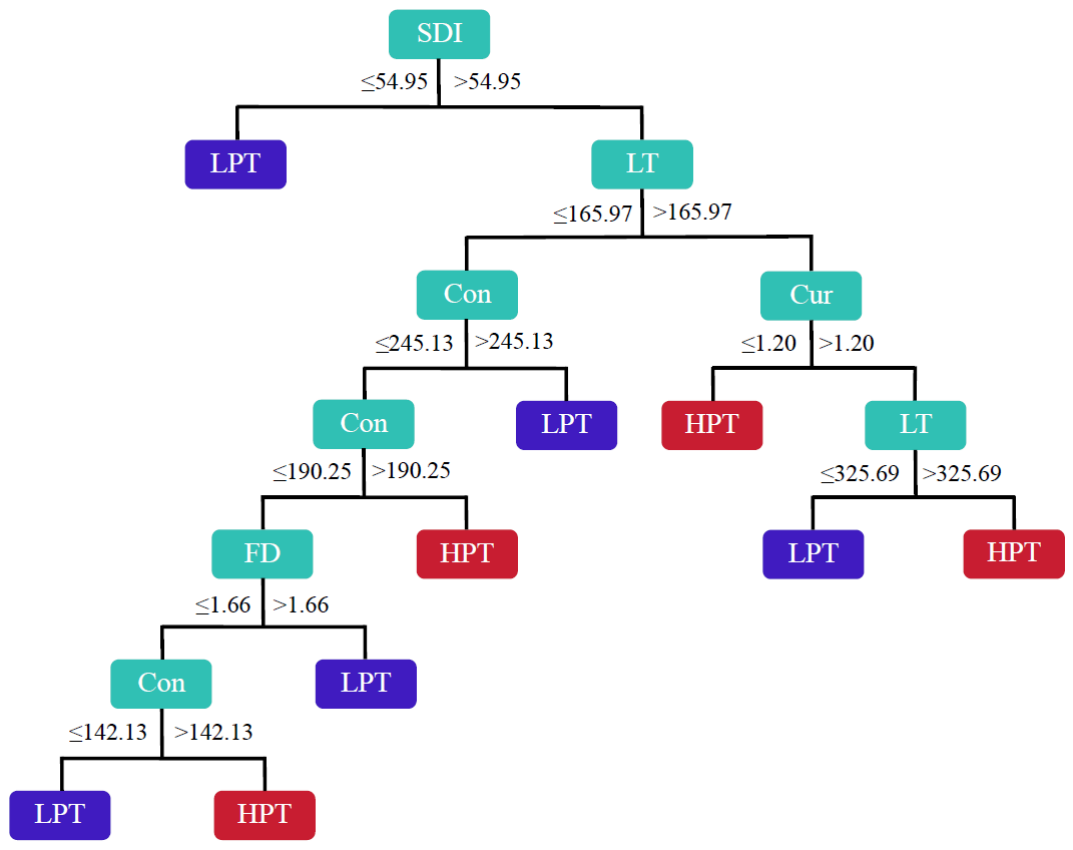
| Classification | VMI | PI | ||
| Mature | Transitional | Immature | ||
| Sen | 94.00% (90.76%-97.27%) | 86.21% (83.79%-90.74%) | 95.45% (91.91%-98.38%) | 90.00% (81.76%-96.25%) |
| Spe | 88.68% (85.71%-94.36%) | 95.95% (91.07%-98.69%) | 96.30% (93.18%-99.60%) | 93.65% (89.51%-97.45%) |
| Acc | 92.16% (88.61%-95.67%) | 93.20% (89.29%-97.05%) | 96.12% (93.29%-99.73%) | 92.23% (87.53%-96.48%) |
| Pre | 88.68% (83.94%-95.67%) | 89.29% (81.34%-96.87%) | 95.45% (90.38%-98.41%) | 93.65% (87.68%-98.25%) |
| Recall | 94.00% (89.37%-97.49%) | 89.29% (84.67%-93.26%) | 87.50% (84.91%-92.53%) | 93.65% (90.68%-97.24%) |
| F1-Score | 91.26% (87.83%-94.35%) | 89.29% (85.29%-93.67%) | 91.44% (88.94%-95.07%) | 93.65% (89.63%-96.89%) |
| p | 0.029 | 0.026 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




