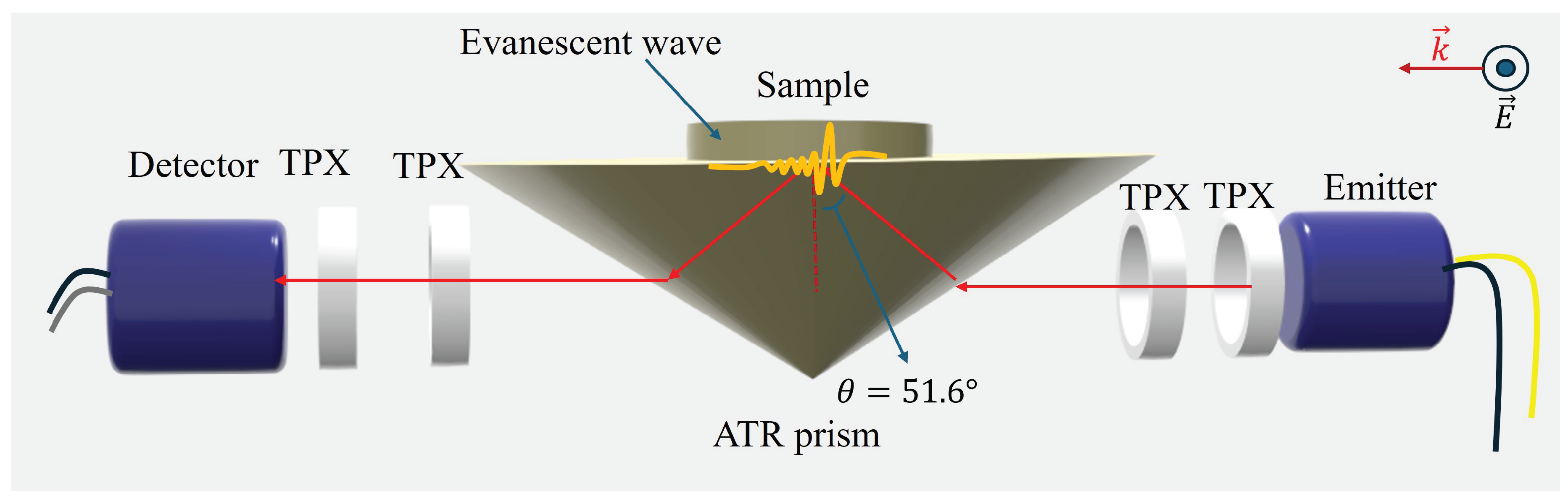
2. Results and Discussion
A set of water–acetone mixtures spanning the full composition range, from pure water to pure acetone, was investigated in order to track the evolution of the dielectric response as a function of acetone molar fraction. As a first step, the reliability of the THz-TDS ATR approach was assessed by measuring the dielectric function of the two pure liquids, whose behavior in the THz range provides a useful reference for the subsequent analysis of the mixtures.
For pure water, the dielectric response in the terahertz region is known to be governed by two characteristic relaxation processes: a slow one,
, generally associated with collective reorientation of molecular dipoles within the hydrogen-bond network, and a faster one,
, related to more localized intermolecular motions, including short-range hydrogen-bond rearrangements and vibrational or librational dynamics [
15,
16]. In this case, the dielectric response is commonly described by a double Debye model:
where
is the high-frequency permittivity and
and
are the dielectric strengths of the two relaxation channels.
The Debye formalism assumes discrete relaxation events characterized by single time constants in a statistically homogeneous environment. While this approximation is often adequate for pure water in the THz range, it becomes less satisfactory for acetone and, even more, for water–acetone mixtures, where overlapping intermolecular contributions, hydrogen-bond disruption, and local structural heterogeneity are expected to produce non-Debye behavior [
8,
17]. For this reason, a broader phenomenological description is required.
To account for these effects, the dielectric spectra were analyzed using a double Cole–Cole model [
18]:
where
and
are empirical broadening parameters that account for a distribution of relaxation times. In physical terms, non-zero
values indicate that the corresponding relaxation process is no longer governed by a single well-defined timescale, but rather by a heterogeneous dynamical environment in which correlated interactions, local disorder, clustering, or structural heterogeneity broaden the dielectric response [
19]. The Debye model is recovered as the limiting case
.
This generalized description is particularly appropriate for the present system. Water and acetone differ markedly in polarity, hydrogen-bonding capability, and molecular polarizability (
;
) [
20]. Therefore, their mixture cannot be treated as a simple non-interacting combination of the two pure liquids. In addition to self-correlations within each component, the dielectric response of the mixture is influenced by water–acetone hydrogen bonding, dipole–dipole interactions, and local compositional heterogeneity. Within the Cole–Cole framework, the dielectric strengths
and
can be related to effective orientational correlations through the Kirkwood–Fröhlich (KF) factors
and
[
21], according to
where
N is the dipole density and
is the effective dipole moment.
In pure liquids,
indicates a net tendency toward parallel dipolar correlations, whereas
reflects predominantly antiparallel correlations;
corresponds to uncorrelated dipoles [
22]. In binary mixtures, however, the corresponding quantity should be regarded as an effective, composition-dependent parameter embedding both like–like and unlike orientational correlations [
23].
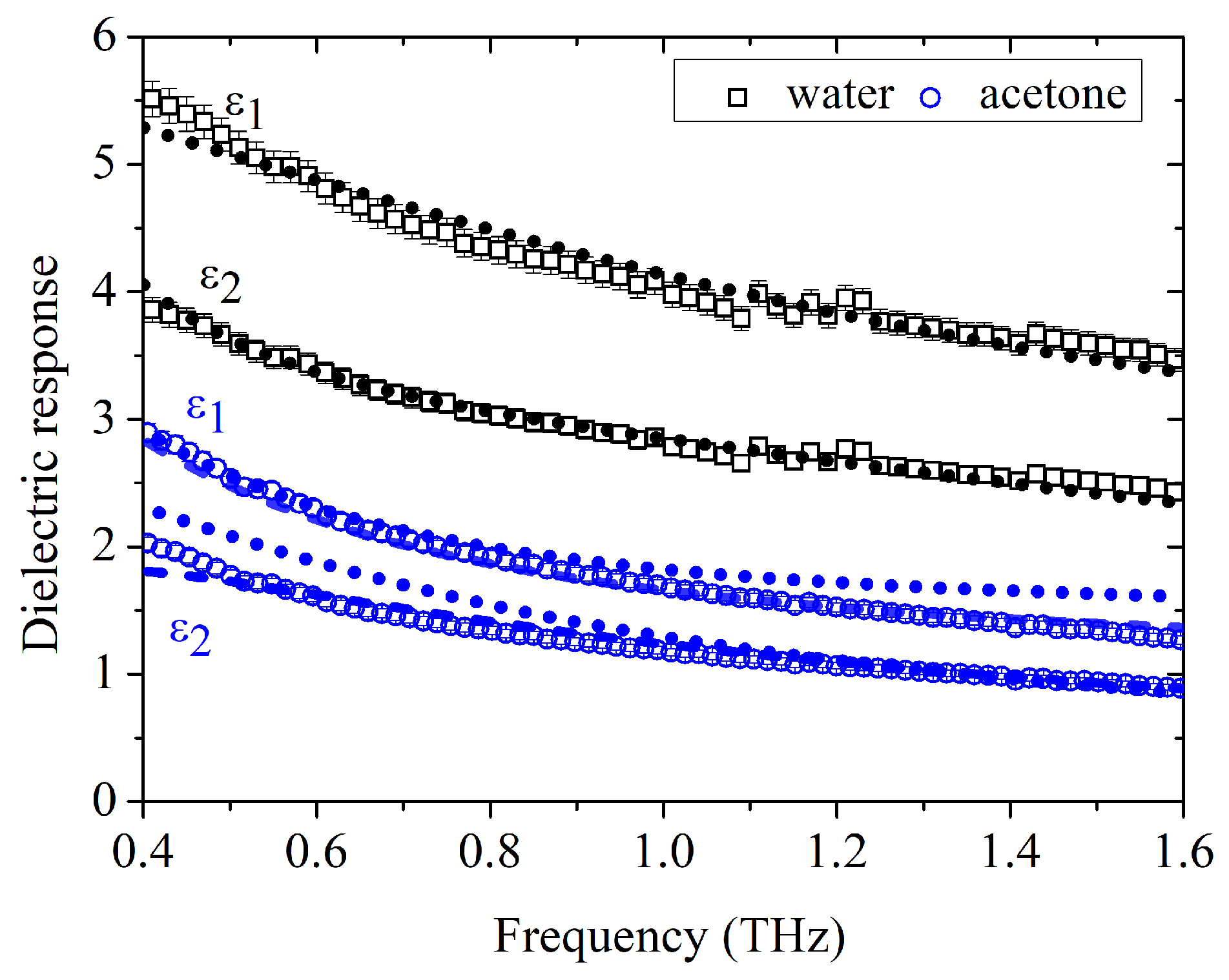
Figure 1 shows the experimentally retrieved complex dielectric function,
, of pure water and pure acetone in the 0.4–1.6 THz range, together with the best fits obtained using the double Debye and double Cole–Cole models. The fitting procedure was based on nonlinear regression simultaneously performed on the real and imaginary parts of the dielectric response, using
,
,
,
, and, for the Cole–Cole model,
and
as free parameters. The high-frequency permittivity
was initialized from the high-frequency behavior of the experimental
data.
As shown in
Figure 1, the double Debye model accurately reproduces the dielectric response of pure water, consistent with its nearly Debye-like behavior in the investigated spectral range. By contrast, the same model provides only a partial description of pure acetone, whereas the double Cole–Cole expression yields a consistent fit for both liquids. In the case of water, the Cole–Cole model naturally collapses to the Debye limit, with
, while for acetone finite broadening parameters are required. The fitting parameters obtained from the Cole–Cole analysis are summarized in Table 1. The extracted values are in good agreement with literature data [
17,
24] and support the use of the double Cole–Cole formalism as a unified framework for describing both the pure liquids and the intermediate mixtures.
Table 1.
Fitting parameters of pure water and acetone using the Cole–Cole model.
Table 1.
Fitting parameters of pure water and acetone using the Cole–Cole model.
| Liquid |
|
(ps) |
(ps) |
|
|
|
|
| Water |
3.0±0.2 |
11.2±0.9 |
0.20±0.03 |
72.5±0.1 |
3.3±0.1 |
0 |
0 |
| Acetone |
1.3±0.1 |
6.7±0.5 |
0.8±0.4 |
3.1±0.1 |
5.4±1.4 |
0.3±0.1 |
0.10±0.01 |
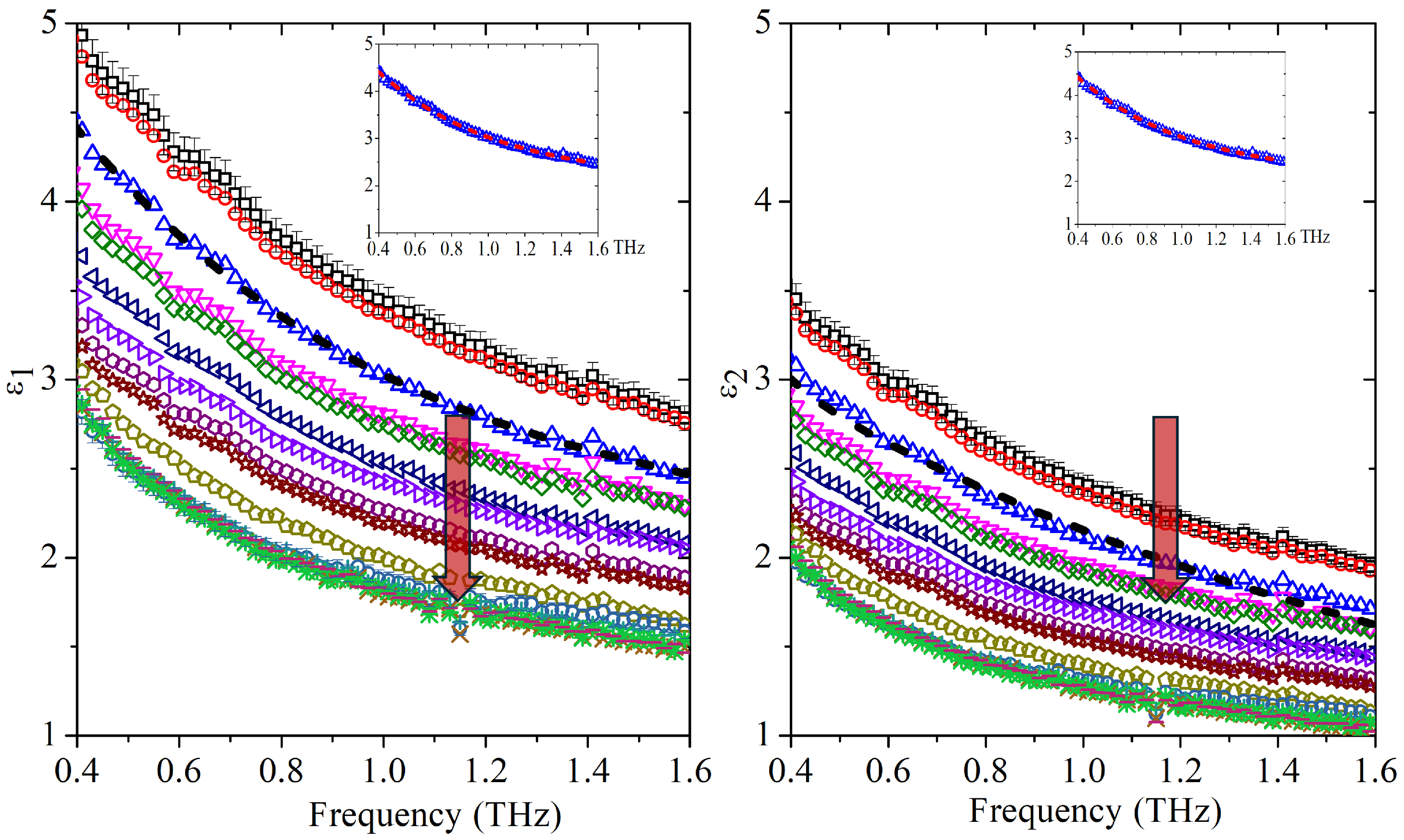
After validating the fitting strategy on the two pure liquids, we extended the analysis to 15 water–acetone mixtures distributed over the full composition range. For each sample, the complex dielectric response was measured as a function of frequency and fitted using the double Cole–Cole model.
Figure 2a,b show the frequency-dependent real and imaginary parts of the dielectric function respectively for all investigated mixtures. In both plots,
and
decrease monotonically with increasing frequency over the whole concentration range. As the acetone content increases, the dielectric response progressively shifts toward lower values, consistent with the lower static permittivity and reduced absorption of acetone compared with water. In the inset, the fit (dashed curve) obtained for the 4% molar fraction together with the experimental data is shown as a representative example, illustrating the ability of the double Cole–Cole model to reproduce the measured spectral broadening.
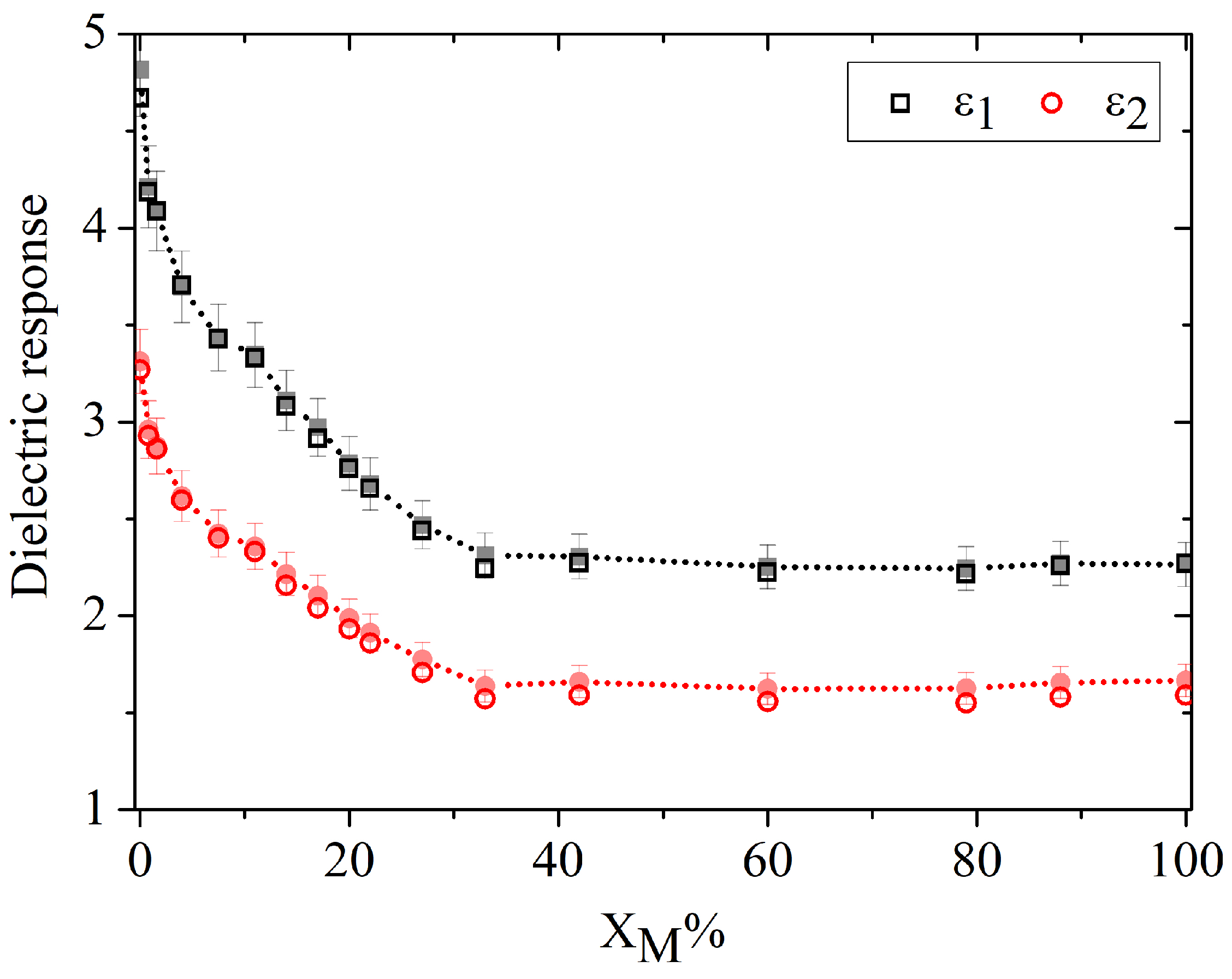
To better visualize the concentration dependence of the dielectric response,
Figure 3 reports the experimental values of
and
at 0.6 THz (open symbols), together with the corresponding values obtained from the fitting procedure (filled symbols). This frequency was selected because it provides the highest signal-to-noise ratio within the investigated spectral window. The comparison shows excellent agreement between experiment and model over the whole molar fraction range, confirming the robustness of the fitting procedure. A marked and strongly nonlinear decrease of both
and
is observed already at very low acetone content, with measurable variations appearing for concentrations as low as 0.8 mol%. This behavior reflects the abrupt perturbation of the extended hydrogen-bond network of bulk water induced by the addition of an aprotic hydrogen-bond acceptor [
3,
5,
6]. The pronounced initial slope of both dielectric components in the water-rich region also explains the particularly high sensitivity of the method at low solute concentration.
The uncertainty associated with the ATR-TDS extraction of the dielectric function is approximately 5%. On this basis, the limit of detection (LOD) for acetone can be estimated as
where
, and
is the local slope of the
and
curves. In the water-rich region, where the concentration dependence is steepest, the estimated LOD is approximately 0.5% from
and 0.6% from
. These values indicate that THz-TDS ATR is highly sensitive to small perturbations in hydrogen-bond connectivity and local dipolar organization, making it particularly effective for detecting acetone in aqueous environments at low concentration.
While the concentration dependence of and already reveals the strong sensitivity of the THz response to acetone addition, a more detailed physical interpretation requires analysis of the parameters extracted from the double Cole–Cole fits.
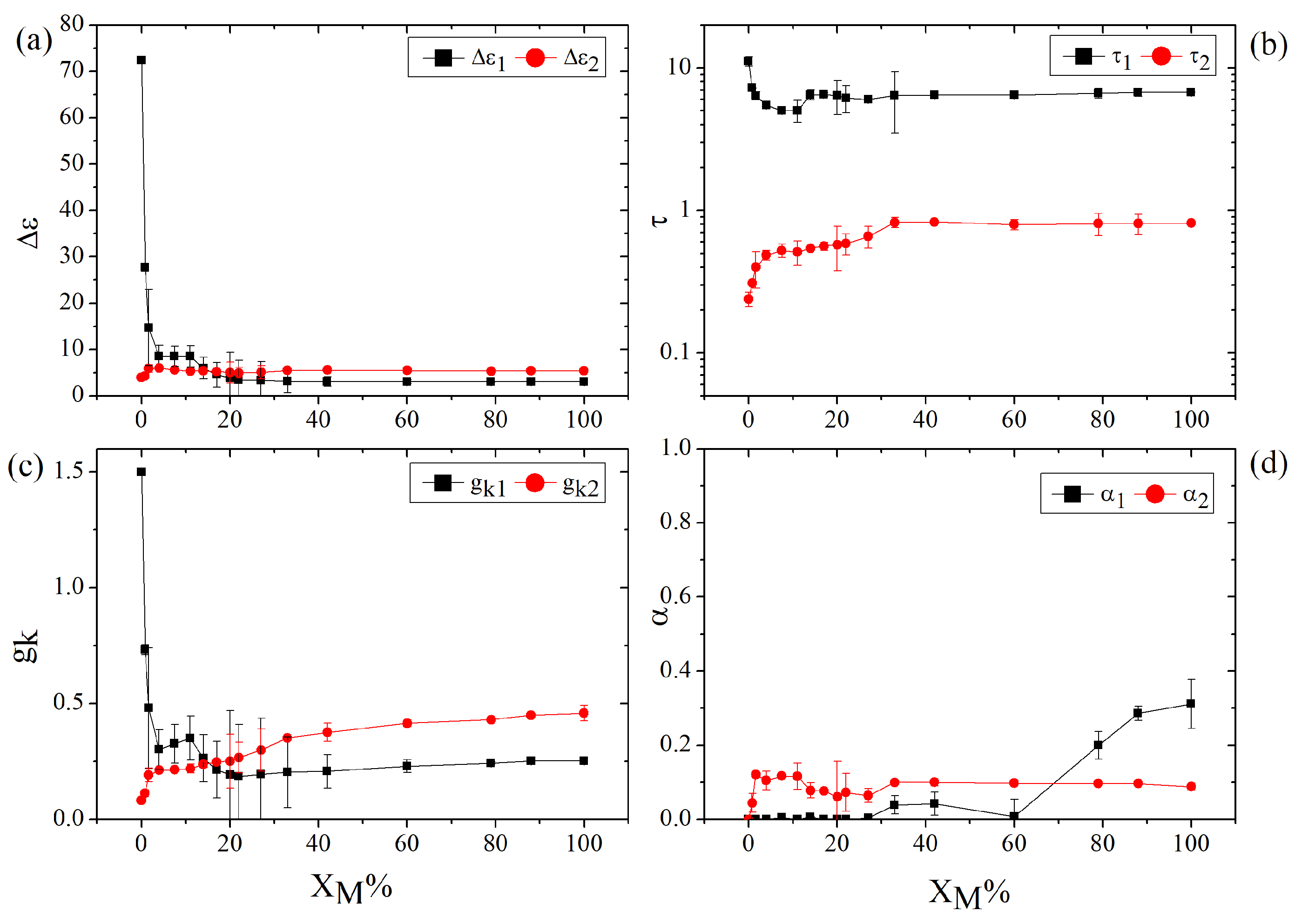
Figure 4 summarizes the parameters extracted from the double Cole–Cole analysis for all investigated mixtures as a function of the acetone molar fraction
. In addition to the dielectric strengths
and
, the corresponding relaxation times
and
, and the broadening parameters
and
, the figure also reports the effective Kirkwood–Fröhlich correlation factors
and
calculated from the retrieved dielectric strengths using eq.(
3).
The behavior of the dielectric strengths, shown in
Figure 4(a), immediately reveals the markedly different sensitivity of the two relaxation channels to acetone addition. Even the smallest molar fraction of acetone produces a sharp decrease in
, whereas
remains nearly constant over the whole concentration range. The strong suppression of the slow dielectric strength indicates that acetone primarily disrupts the cooperative reorganization of the extended hydrogen-bond network of water. In pure water, the slow relaxation process, characterized by
on the order of 10 ps, is generally associated with large-scale collective orientational rearrangements involving strongly correlated molecules within the hydrogen-bond network [
15,
16]. Its dielectric intensity therefore depends critically on the continuity and cooperativity of the water–water hydrogen-bond structure. The addition of acetone, an aprotic molecule that can accept but not donate hydrogen bonds, perturbs this network by interrupting its three-dimensional connectivity and reducing the number of configurations able to sustain such collective relaxation [
3,
5,
6]. As a consequence, the dielectric strength of the slow mode decreases sharply as the acetone content increases. By contrast, the faster relaxation channel, associated with
on the order of 0.1 ps, appears much less sensitive to the presence of acetone. This suggests that the fast process is governed by more local intermolecular dynamics, such as libration-assisted orientational fluctuations, transient hydrogen-bond rearrangements, or short-range cage motions, which do not rely as strongly on the existence of an extended and percolating hydrogen-bond network [
15,
16].
The corresponding relaxation times are reported in
Figure 4(b), on a semi-log scale for the sake of clarity. In both cases, only a relatively weak dependence on acetone concentration is observed. This suggests that the addition of the aprotic solute does not primarily modify the intrinsic timescale of the underlying molecular motions. Rather, acetone acts mainly by reducing the statistical weight and cooperativity of the relaxing environments, especially those associated with the slow hydrogen-bond-driven process. In this picture, the spatial continuity of the water network is progressively disrupted, thereby suppressing the amplitude of the slow mode without substantially altering the characteristic relaxation time of the residual water-rich structures that are still able to sustain it. A similar argument applies to the fast relaxation channel: if this process originates from local motions, such as libration-assisted fluctuations or short-range hydrogen-bond rearrangements, its characteristic timescale may remain close to that of pure water even in the presence of significant structural perturbation [
15,
16].
Figure 4(c) reports the effective Kirkwood–Fröhlich factors
and
. Because these correlation factors are directly proportional to the dielectric strengths of the two relaxation channels, their compositional evolution largely follows that of
and
. In the present case, however, the most informative aspect is not simply their variation with acetone molar fraction, but rather the absolute values they assume. Within the standard Kirkwood–Fröhlich framework, values greater than unity are generally associated with predominantly parallel dipolar correlations, whereas values below unity suggest a tendency toward antiparallel orientational arrangements [
22]. Strictly speaking, this interpretation is rigorously defined for pure liquids; nevertheless, when applied with due caution, it can still provide useful qualitative insight into the local dipolar organization of binary mixtures [
23]. From this perspective, the value of
close to 2 in pure water is fully consistent with the well-established picture of a highly correlated hydrogen-bond network and agrees well with values reported in classical molecular dynamics studies at room temperature [
25]. The sharp decrease of
observed upon the first addition of acetone points to a rapid breakdown of the cooperative dipolar correlations associated with the slow relaxation channel. At intermediate compositions, the more gradual variation of
suggests that some hydrogen-bonded water motifs may still survive, although in a progressively more fragmented and less cooperative environment. At higher acetone contents, where a continuous water network can no longer be maintained, the fact that
remains below unity indicates that the residual dipolar correlations are qualitatively different from those of pure water and may reflect a more locally compensated and weakly cooperative arrangement. A similar argument applies to
, whose values remain below unity over the whole concentration range. Although any structural interpretation must remain qualitative in a mixed liquid, this behavior suggests that the fast relaxation channel is associated with local molecular configurations that do not exhibit strong cooperative alignment and may instead favor partial dipolar compensation. In this sense, the persistently low values of the corresponding correlation factor are compatible with the presence of small water aggregates or locally compensated dipolar configurations, in which local dipole compensation is statistically favored [
26,
27].
This distinction is further supported by the behavior of the broadening parameters, shown in
Figure 4(d), which provide complementary information on the dynamical heterogeneity of the same two relaxation channels. The parameter
becomes non-zero immediately after the addition of acetone and rapidly saturates around 0.1, indicating that the fast relaxation channel is highly sensitive to the onset of local heterogeneity, although its ultrafast dynamics remain distributed over a relatively narrow range of timescales [
19]. By contrast,
remains essentially zero, within experimental uncertainty, up to
, and increases only at higher acetone concentrations. This delayed broadening suggests that the slow relaxation process retains an almost Debye-like character as long as residual water-rich regions are still able to sustain collective hydrogen-bond network dynamics. In this regime, acetone mainly reduces the dielectric strength of the slow mode without substantially broadening its time distribution. Only when the extended water network is no longer maintained does a significant distribution of slow relaxation times emerge.
Overall, the concentration dependence of the dielectric strengths, relaxation times, effective correlation factors, and broadening parameters converges toward a coherent molecular picture: acetone acts primarily as a network breaker, strongly suppressing the cooperative hydrogen-bond network of water while leaving the faster and more local relaxation channel comparatively less affected. The THz response of the mixtures is therefore governed not by a simple interpolation between the two pure liquids, but by the concentration-dependent reorganization of hydrogen-bond connectivity, dipolar correlations, and local dynamical heterogeneity.