Submitted:
23 April 2026
Posted:
28 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Site
2.2. Sampling Design and Macroinvertebrate Collection
2.3. Physicochemical Measurements
2.4. Data Analysis
3. Results
3.1. Physicochemical Characteristics
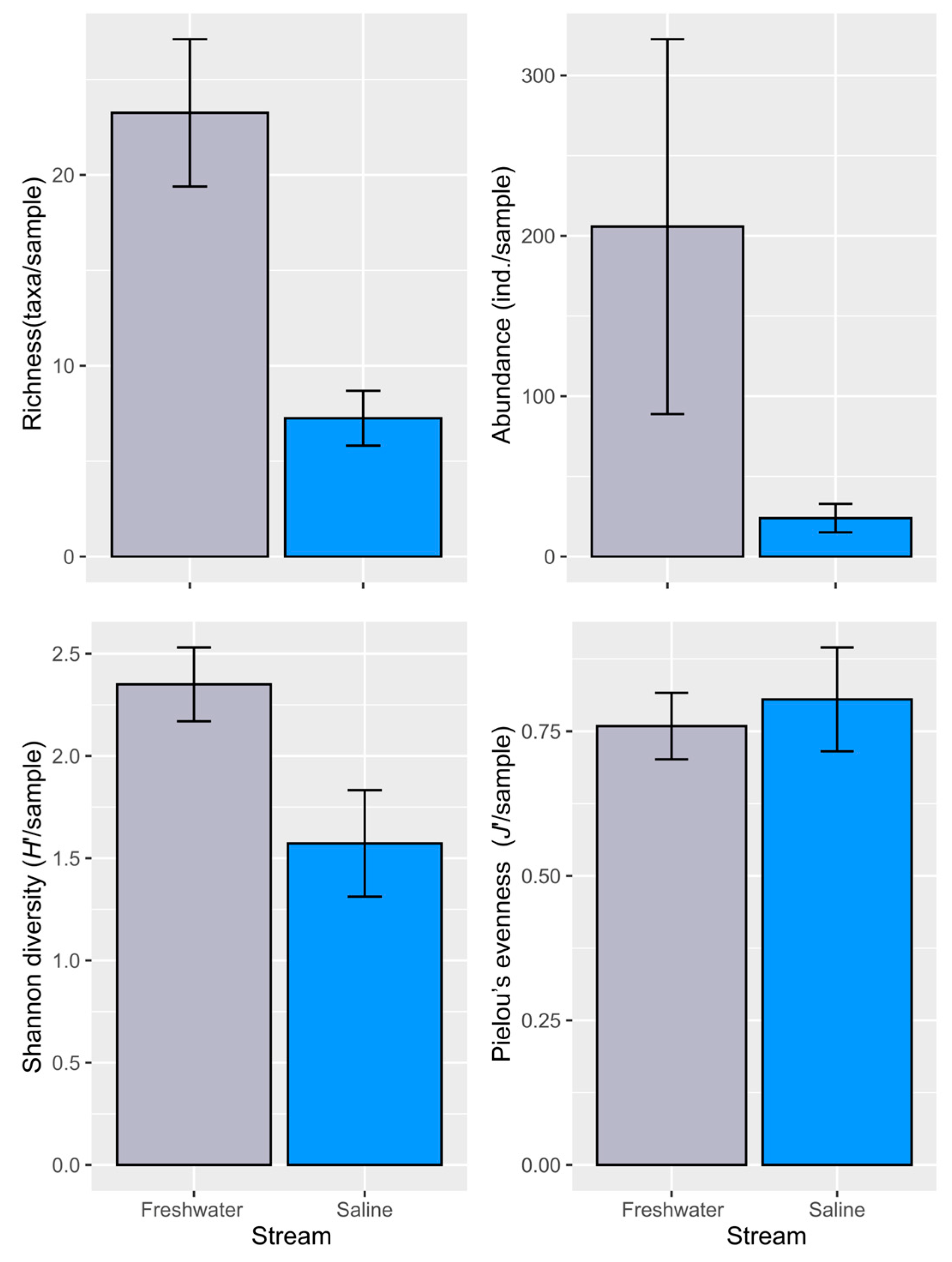
3.2. Macroinvertebrate Diversity
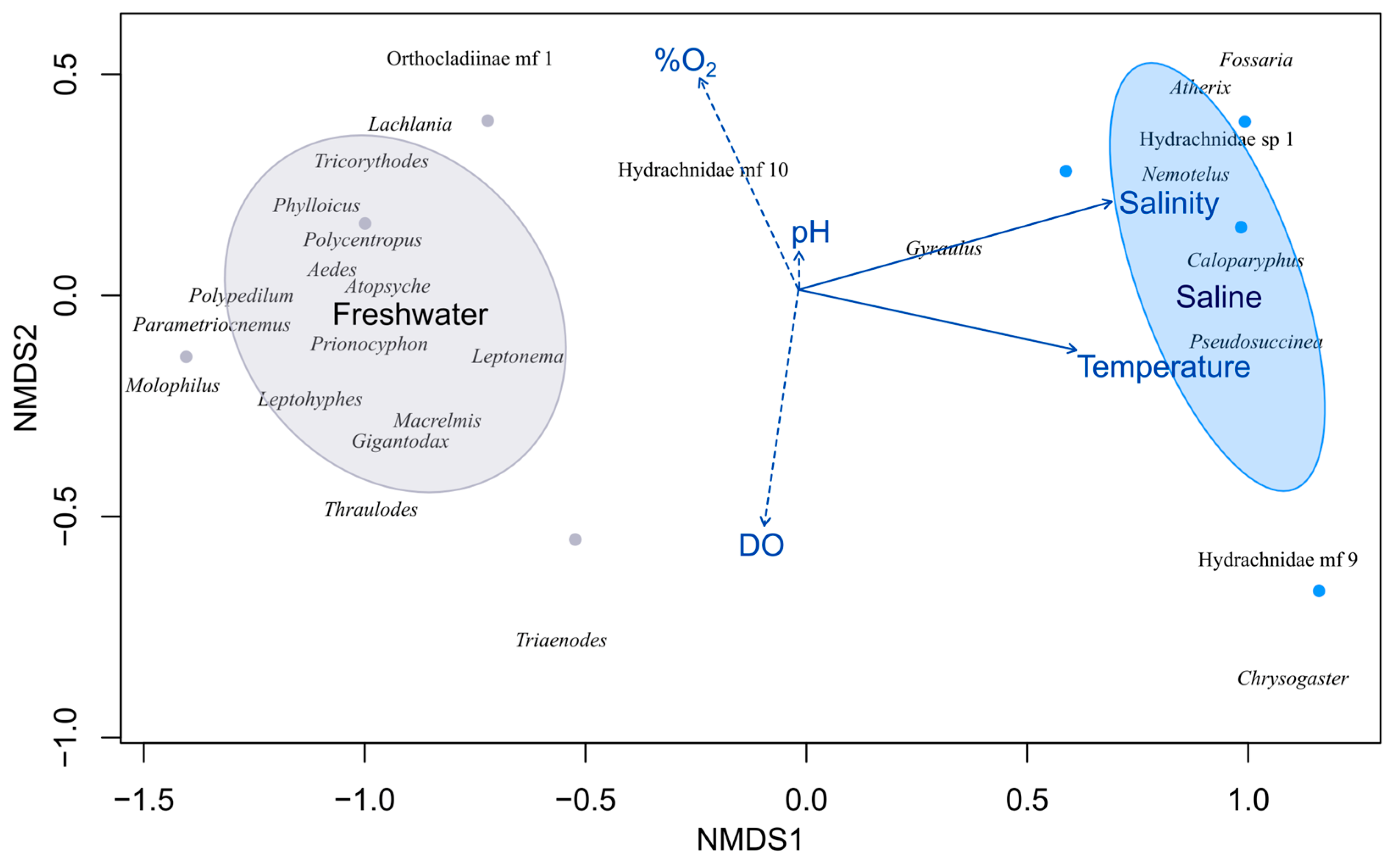
3.3. Community Composition and Environmental Drivers
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Acknowledgments
Conflicts of Interest
References
- Hauer, F.R.; Resh, V.H. Macroinvertebrates. In Methods in Stream Ecology, Volume 1: Ecosystem Structure; Hauer, F.R., Lamberti, G.A., Eds.; Academic Press: London, 2017; pp. 297–319 ISBN 9780124165588.
- Buss, D.F.; Carlisle, D.M.; Chon, T.-S.; Culp, J.; Harding, J.S.; Keizer-Vlek, H.E.; Robinson, W.A.; Strachan, S.; Thirion, C.; Hughes, R.M. Stream Biomonitoring Using Macroinvertebrates around the Globe: A Comparison of Large-Scale Programs. Environ. Monit. Assess. 2015, 187, 4132. [CrossRef]
- Ramírez, A.; Gutiérrez-Fonseca, P.E. Functional Feeding Groups of Aquatic Insect Families in Latin America: A Critical Analysis and Review of Existing Literature. Rev. Biol. Trop. 2014, 62, 155–167.
- Millán, A.; Velasco, J.; Gutiérrez-Cánovas, C.; Arribas, P.; Picazo, F.; Sánchez-Fernández, D.; Abellán, P. Mediterranean Saline Streams in Southeast Spain: What Do We Know? J. Arid Environ. 2011, 75, 1352–1359. [CrossRef]
- Velasco, J.; Millán, A.; Hernández, J.; Gutiérrez, C.; Abellán, P.; Sánchez, D.; Ruiz, M. Response of Biotic Communities to Salinity Changes in a Mediterranean Hypersaline Stream. Saline Systems 2006, 2, 12. [CrossRef]
- Arribas, P.; Gutiérrez-Cánovas, C.; Abellán, P.; Sánchez-Fernández, D.; Picazo, F.; Velasco, J.; Millán, A. Tipificación de Los Ríos Salinos Ibéricos. Ecosistemas 2009, 18, 1–13.
- Gandin, A.; Capezzuoli, E. Travertine: Distinctive Depositional Fabrics of Carbonates from Thermal Spring Systems. Sedimentology 2014, 61, 264–290. [CrossRef]
- Pentecost, A. Travertine; Springer: Berlin, 2005; ISBN 1402035233.
- Minissale, A.; Kerrick, D.M.; Magro, G.; Murrell, M.T.; Paladini, M.; Rihs, S.; Sturchio, N.C.; Tassi, F.; Vaselli, O. Geochemistry of Quaternary Travertines in the Region North of Rome (Italy): Structural, Hydrologic and Paleoclimatic Implications. Earth Planet. Sci. Lett. 2002, 203, 709–728. [CrossRef]
- Leiva-Tafur, D.; Manco Perez, H.G.; Rascón, J.; Culqui, L.; Gamarra-Torres, O.A.; Oliva-Cruz, M. Hydrogeochemical Characterization of Mineral Springs in Peruvian Tropical Highlands. Water 2025, 17, 2539. [CrossRef]
- De Grys, A.; Vera, J.; Goossens, P. A Note on the Hot Springs of Ecuador. Geothermics 1970, 2, 1400–1404. [CrossRef]
- Quezada, P.; Fadel Cury, L.; Calderón, M.; Henríquez, C.; Mancini, L.; Micheletto, J.; Barbosa Athayde, G.; Bahniuk Rumbelsperger, A. Similar Sources but Distinct Δ13C Signatures in Adjacent Low-Temperature Travertines from Laguna Amarga (Southern Patagonian Andes). Sediment. Geol. 2024, 473, 106758. [CrossRef]
- Chrobak, A.; Ugolini, F.; Pearlmutter, D.; Raschi, A. Thermal Tourism and Geoheritage: Examining Visitor Motivations and Perceptions. Resources 2020, 9, 58. [CrossRef]
- Myers, N.; Mittermeier, R.A.; Mittermeier, C.G.; da Fonseca, G.A.; Kent, J. Biodiversity Hotspots for Conservation Priorities. Nature 2000, 403, 853–858. [CrossRef]
- Encalada, A.C.; Flecker, A.S.; Poff, N.L.R.; Suárez, E.; Herrera-R, G.A.; Ríos-Touma, B.; Jumani, S.; Larson, E.I.; Anderson, E.P. A Global Perspective on Tropical Montane Rivers. Science (80-. ). 2019, 365, 1124–1129.
- Jacobsen, D.; Schultz, R.; Encalada, A.C. Structure and Diversity of Stream Invertebrate Assemblages : The Influence of Temperature with Altitude and Latitud. Freshw. Biol. 1997, 38, 247–261.
- Ríos-Touma, B.; Encalada, A.C.; Prat Fornells, N. Macroinvertebrate Assemblages of an Andean High-Altitude Tropical Stream: The Importance of Season and Flow. Int. Rev. Hydrobiol. 2011, 96, 667–685. [CrossRef]
- Barbour, M.T.; Gerritsen, J.; Snyder, B.D.; Stribling, J.B. Rapid Bioassessment Protocols for Use in Streams and Wadeable Rivers: Periphyton, Benthic Macroinvertebrates, and Fish; Second.; US Environmental Protection Agency, Office of Water: Washington DC, 1999;
- Wantzen, K.M.; Rueda-Delgado, G. Técnicas de Muestreo de Macroinvertebrados Bentónicos. In Macroinvertebrados bentónicos sudamericanos. Sistemática y biología; Domínguez, E., Fernández, H.R., Eds.; Fundación Miguel Lillo: Tucumán, 2009; pp. 17–46.
- R Development Core Team R: A Language and Environment for Statistical Computing, Vienna, Austria 2025.
- Hothorn, T.; Horni, K.; van de Wiel, M.A.; Zeileis, A. Implementing a Class of Permutation Tests: The Coin Package. J. Stat. Softw. 2008, 28, 1–23, doi:doi:10.18637/jss.v028.i08.
- Oksanen, J.; Blanchet, F.G.; Kindt, R.; Legendre, P.; Minchin, P.R.; O’Hara, R.B.; Simpson, G.L.; Solymos, P.; Stevens, H.H.; Wagner, H. Vegan: Community Ecology Package. R Package Version 2.6-4 2022.
- Torchiano, M. Effsize: Efficient Effect Size Computation. R Package Version 0.8.1 2020.
- Naimi, B. Usdm: Uncertainty Analysis for Species Distribution Models. R Package Version 1.1-18 2015.
- Kefford, B.J.; Buchwalter, D.; Cañedo-Argüelles, M.; Davis, J.; Duncan, R.P.; Hoffmann, A.; Thompson, R. Salinized Rivers: Degraded Systems or New Habitats for Salt-Tolerant Faunas? Biol. Lett. 2016, 12, 20151072. [CrossRef]
- Washko, S.; Bogan, M. Ability of Aquatic Stratiomyidae (Diptera) Larvae to Survive Stream Drying Using Dormancy. Aquat. Insects 2024, 45, 402–415. [CrossRef]
- Golovatyuk, L. V.; Zinchenko, T.D.; Sushchik, N.N.; Kalachova, G.S.; Gladyshev, M.I. Biological Aspects of the Associations of Biting Midges (Diptera: Ceratopogonidae) in Two Saline Rivers of the Elton Lake Basin, Russia. Mar. Freshw. Res. 2018, 69, 906–916. [CrossRef]
- Goldschmidt, T. Water Mites (Acari, Hydrachnidia): Powerful but Widely Neglected Bioindicators – a Review. Neotrop. Biodivers. 2016, 2, 12–25. [CrossRef]
- Smith, I.M.; Cook, D.R.; Smith, B.P. Water Mites (Hydrachnidiae) and Other Arachnids. In Ecology and Classification of North American Freshwater Invertebrates; Thorp, J.H., Covich, A.P., Eds.; Academic Press: New York, 2010; pp. 485–586 ISBN 9780123748553.
- Costil, K.; Dussart, G.B.J.; Daguzan, J. Biodiversity of Aquatic Gastropods in the Mont St-Michel Basin (France) in Relation to Salinity and Drying of Habitats. Biodivers. Conserv. 2001, 10, 1–18. [CrossRef]
- Griffith, M.B. Toxicological Perspective on the Osmoregulation and Ionoregulation Physiology of Major Ions by Freshwater Animals: Teleost Fish, Crustacea, Aquatic Insects, and Mollusca. Environ. Toxicol. Chem. 2017, 36, 576–600. [CrossRef]

| Parameter | Unit | Freshwater stream | Saline stream | p-value |
|---|---|---|---|---|
| Temperature | °C | 13.7 ± 0.9 | 17.5 ± 3.3 | 0.028 |
| pH | 7.4 ± 0.7 | 7.4 ± 0.9 | 0.532 | |
| Conductivity | mS/cm | 0.1 ± 0.0 | 20.9 ± 0.4 | < 0.001 |
| Dissolved oxygen | Mg/L | 7.7 ± 0.4 | 6.5 ± 4.0 | 0.901 |
| O2 saturation | % | 102.2 ± 3.4 | 90.7 ± 37.5 | 0.805 |
| Total dissolved solids | g/L | 0.1 ± 0.0 | 21.0 ± 0.3 | < 0.001 |
| Salinity | g/L | 0.0 ± 0.0 | 12.5 ± 0.2 | < 0.001 |
| Stream | Class | Order | Family | Genus | Ind. |
|---|---|---|---|---|---|
| Freshwater | Malacostraca | Amphipoda | Hyalellidae | Hyallela | 17 |
| Oligochaeta | Arhynchobdellida | Cylicobdellidae | Cylicobdellidae mf 1 | 47 | |
| Insecta | Coleoptera | Elmidae | Austrolimnius | 1 | |
| Heterelmis | 25 | ||||
| Hexacylloepus | 1 | ||||
| Macrelmis | 4 | ||||
| Neoelmis | 8 | ||||
| Onychelmis | 1 | ||||
| Psephenidae | Pheneps | 1 | |||
| Scirtidae | Prionocyphon | 31 | |||
| Diptera | Blephariceridae | Paltostoma | 2 | ||
| Chironomidae | Aechnida | 3 | |||
| Chironomus | 49 | ||||
| Diamesa | 3 | ||||
| Larsia | 23 | ||||
| Metriocnemus | 4 | ||||
| Orthocladiinae mf 1 | 1 | ||||
| Parametriocnemus | 15 | ||||
| Polypedilum | 3 | ||||
| Culicidae | Aedes | 7 | |||
| Limoniidae | Molophilus | 1 | |||
| Polymera | 2 | ||||
| Simuliidae | Gigantodax | 38 | |||
| Tipulidae | Hexatoma | 1 | |||
| Ephemeroptera | Baetidae | Andesiops | 84 | ||
| Baetodes | 220 | ||||
| Hydrobiosidae | Atopsyche | 20 | |||
| Leptohyphidae | Haplohyphes | 52 | |||
| Leptohyphes | 53 | ||||
| Thraulodes | 8 | ||||
| Tricorythodes | 12 | ||||
| Oligoneuriidae | Lachlania | 6 | |||
| Trichoptera | Anomalopsyche | Contulma | 4 | ||
| Brachycentridae | Brachycentrus | 1 | |||
| Calamoceratidae | Heteropletron | 1 | |||
| Phylloicus | 1 | ||||
| Hydropsychidae | Leptonema | 12 | |||
| Macronema | 2 | ||||
| Hydroptilidae | Ochrotrichia | 26 | |||
| Leptoceridae | Triaenodes | 1 | |||
| Polycentropodidae | Polycentropus | 10 | |||
| Plecoptera | Perlidae | Anacroneuria | 13 | ||
| Gastropoda | Gastropoda | Gastropoda | Gastropoda mf 1 | 1 | |
| Malacostraca | Isopoda | Isopoda | Isopoda mf 1 | 1 | |
| Odanata | Aeshnidae | Allopetalia | 1 | ||
| Ironoquia | 1 | ||||
| Gastropoda | Pulmonata | Planorbidae | Gyraulus | 1 | |
| Arachnida | Trombidiformes | Hydrachnidae | Hydrachnidae mf 10 | 3 | |
| Hydrachnidae mf 14 | 1 | ||||
| Saline | Insecta | Diptera | Athericidae | Atherix | 4 |
| Ceratopogonidae | Stilobezzia | 29 | |||
| Tabanidae | Tabanus | 11 | |||
| Stratiomyidae | Caloparyphus | 16 | |||
| Nemotelus | 2 | ||||
| Syrphidae | Chrysogaster | 1 | |||
| Arachnida | Trombidiformes | Hydrachnidae | Hydrachnidae mf 10 | 2 | |
| Hydrachnidae mf 20 | 4 | ||||
| Hydrachnidae mf 22 | 1 | ||||
| Hydrachnidae mf 9 | 15 | ||||
| Hydrachnidae mf 1 | 4 | ||||
| Gastropoda | Pulmonata | Lymnaeidae | Fossaria | 1 | |
| Pseudosuccinea | 5 | ||||
| Planorbidae | Gyraulus | 1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




