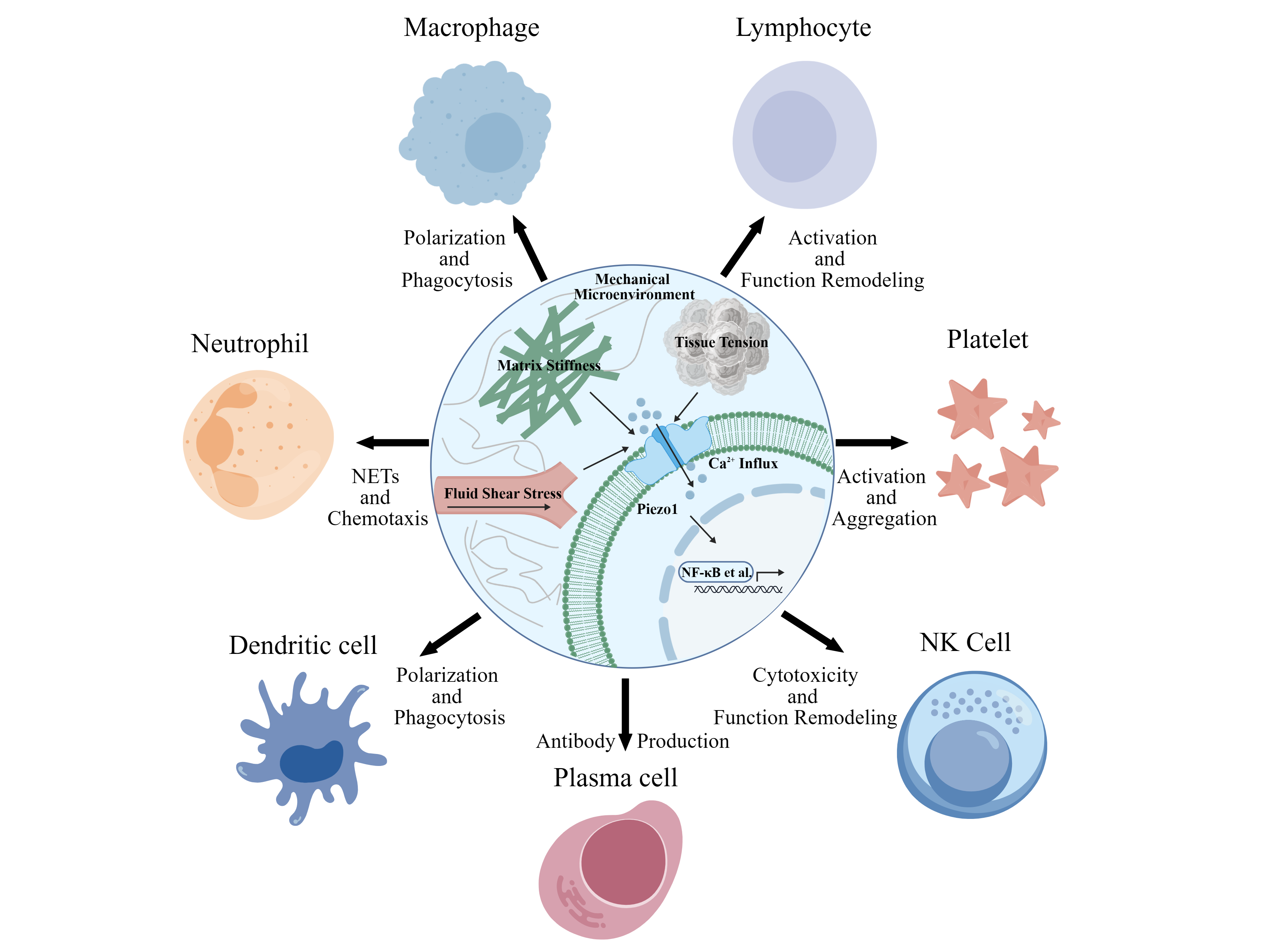
The mechanosensitive ion channel Piezo1 acts as a crucial molecule for cellular mechanical signal sensing and transduction. It transforms physical mechanical cues in the microenvironment, including matrix stiffness, fluid shear stress, and tissue tension, into intracellular biochemical signals through Ca2⁺ influx-mediated mechanisms. Consequently, it regulates the activation, proliferation, differentiation, migration, and effector functions of blood cells. Herein, we review the research progress of Piezo1 in various white blood cells, with a particular emphasis on its functional regulatory mechanisms in neutrophils, macrophages, platelets, and T and B lymphocytes. We briefly summarize its current functional status in natural killer cells, dendritic cells, plasma cells, platelets, and other cell types. We analyze the integrated effects and multi-cellular cooperative interactions of Piezo1-mediated blood cell mechanotransduction across physiological and pathological contexts. We discuss the potential value of Piezo1 as a mechano-immunotherapeutic target, therapeutic strategies, and challenges facing clinical translation. Finally, we provide perspectives on future research directions, offering theoretical references for deepening the understanding of the molecular mechanisms by which mechanical microenvironments regulate white blood cell function and disease progression and for developing novel therapeutic strategies.