Submitted:
25 April 2026
Posted:
28 April 2026
You are already at the latest version
Abstract
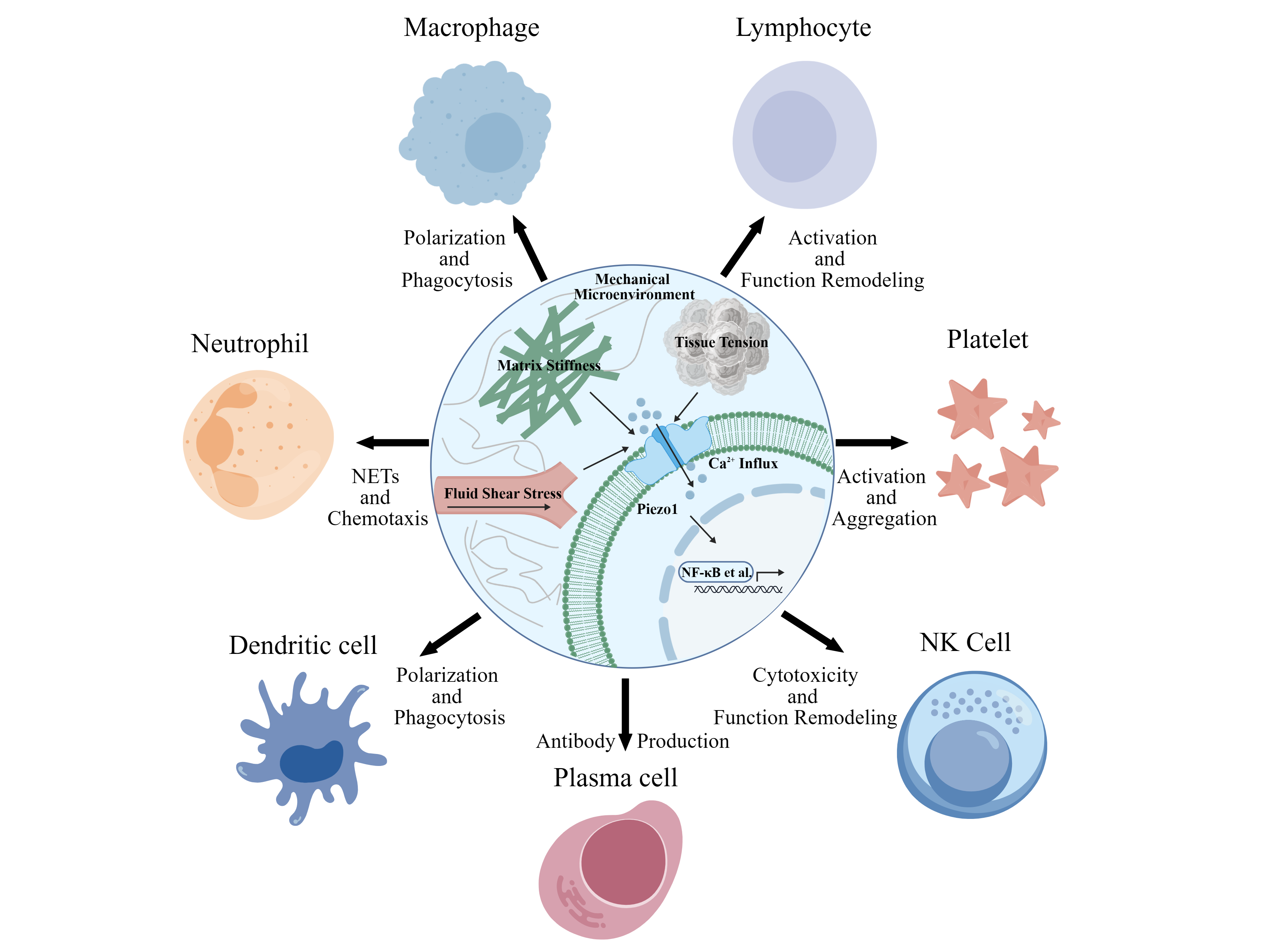
Keywords:
1. Introduction
2. Mechanoresponsive and Functional Regulatory Roles of Piezo1 in Major White Blood Cells
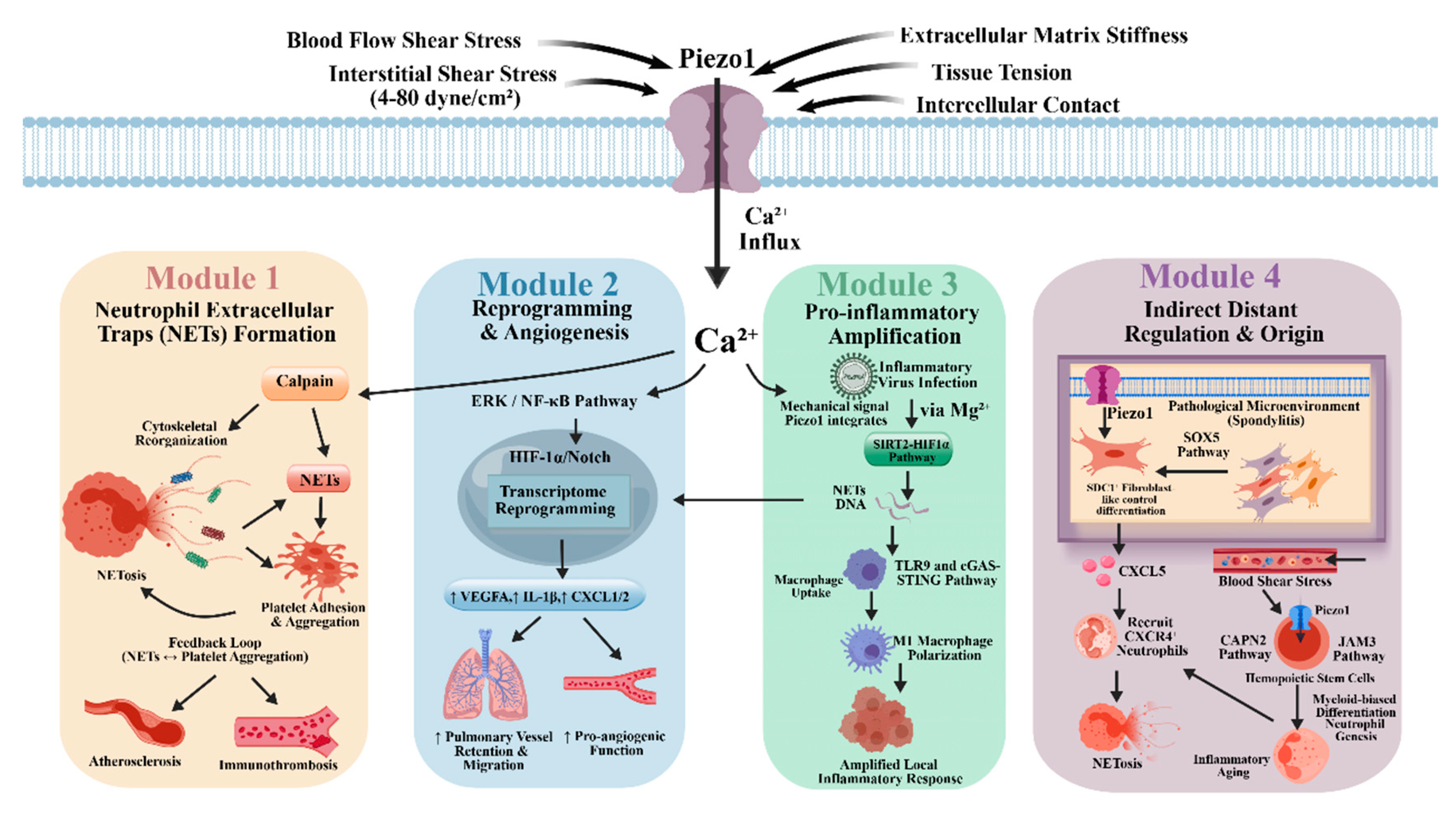
2.1. Piezo1-Mediated Regulation of Neutrophil Activation and Functional Reprogramming
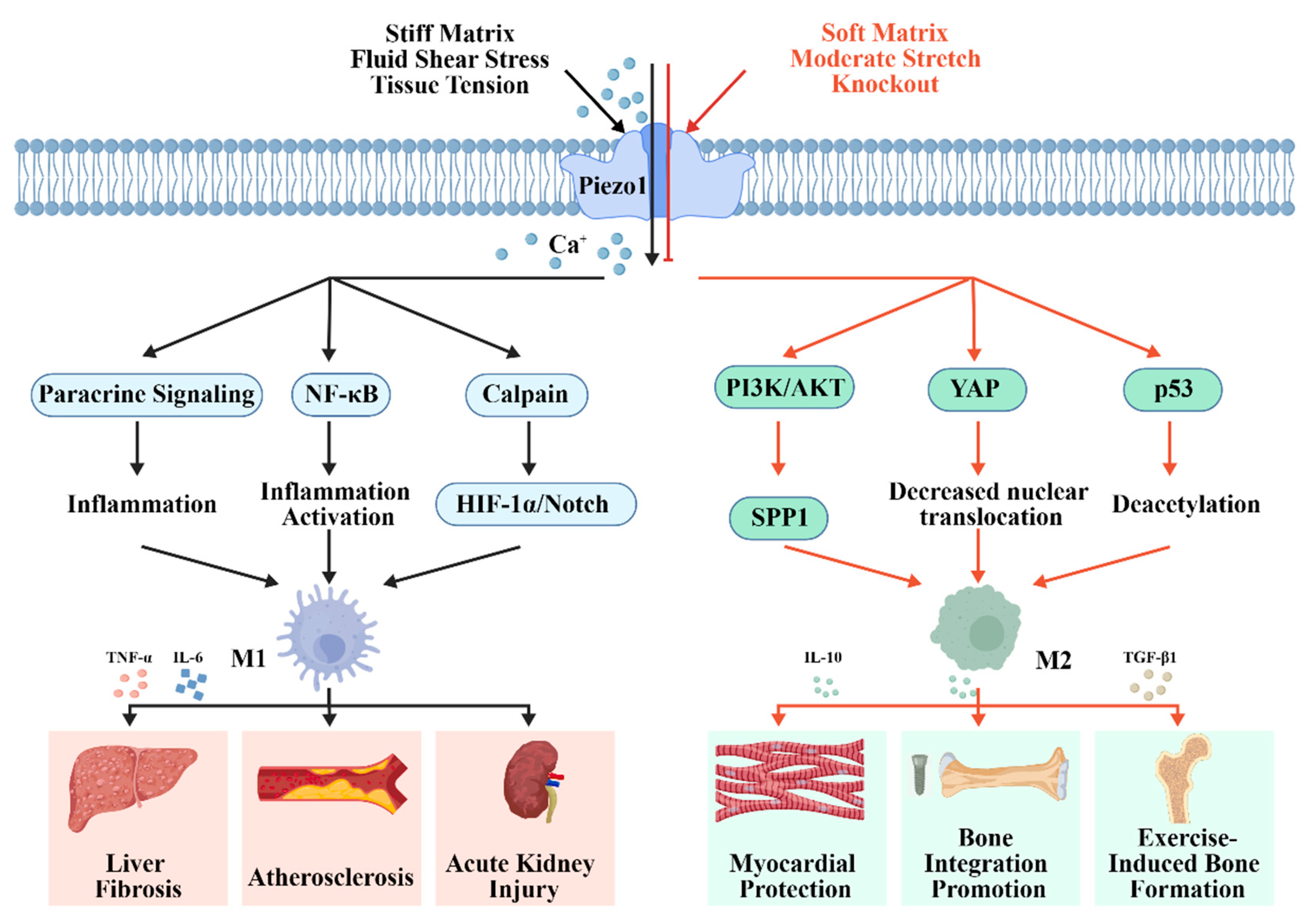
2.2. Piezo1-Dependent Mechanical Signal Regulation of Macrophage Polarization and Inflammatory Phenotype Specialization
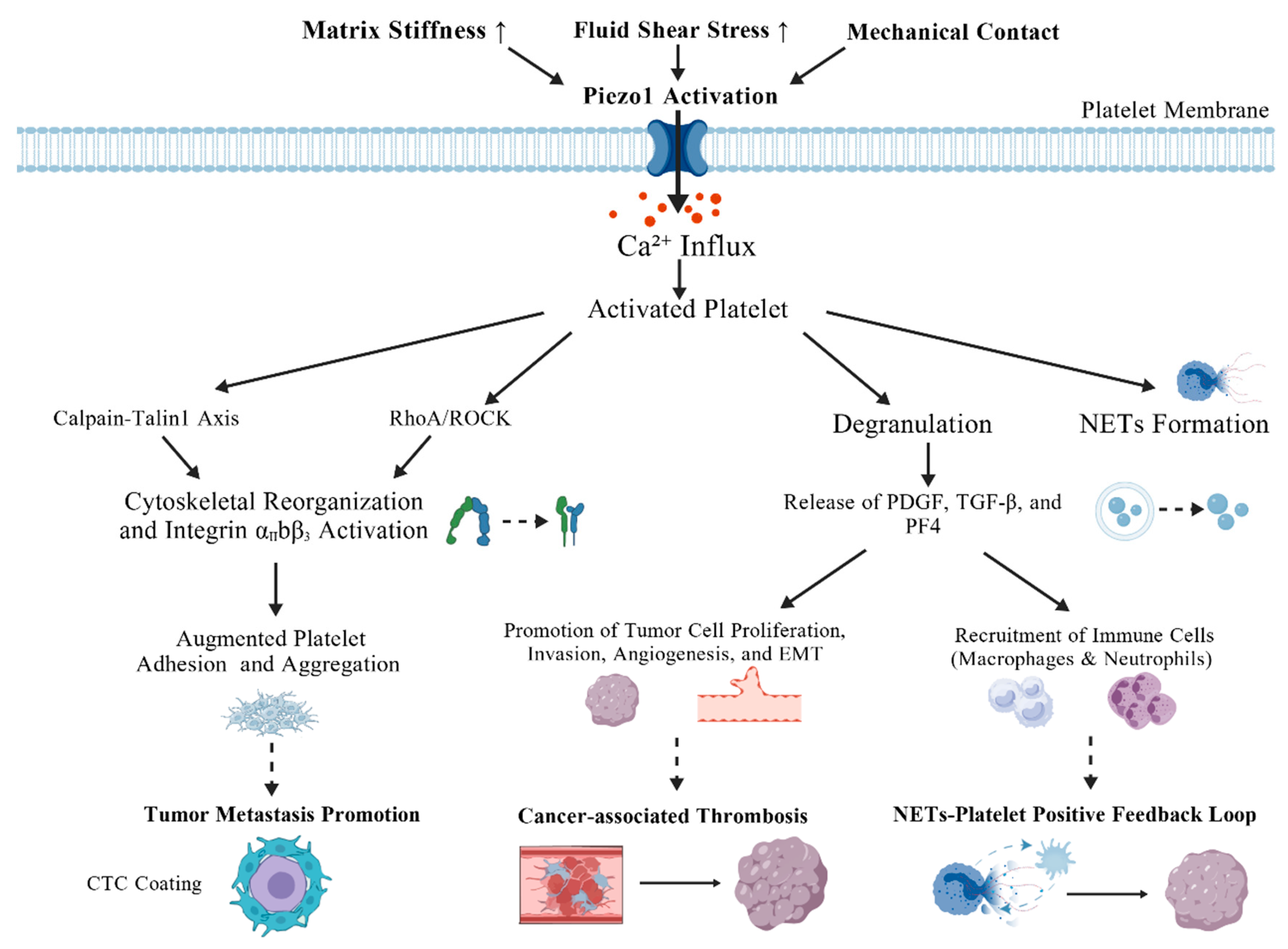
2.3. Piezo1-Dependent Mechanical Signal Regulation of Platelet Mechanosensing and Coagulation Function
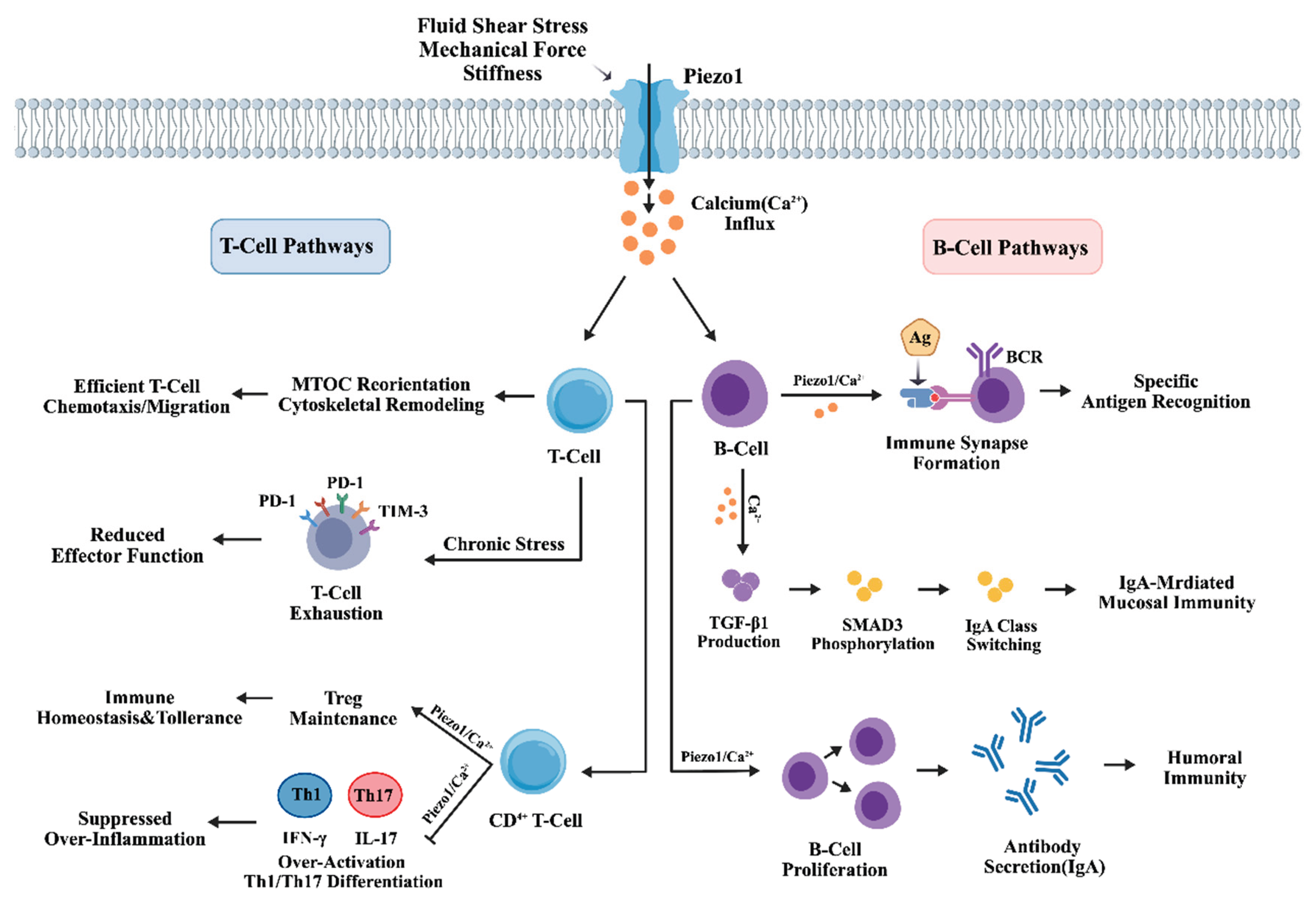
2.4. Piezo1-Dependent Mechanical Signal Regulation of Lymphocyte Immune Activation and Differentiation
2.5. Piezo1 and Other White Blood Cells
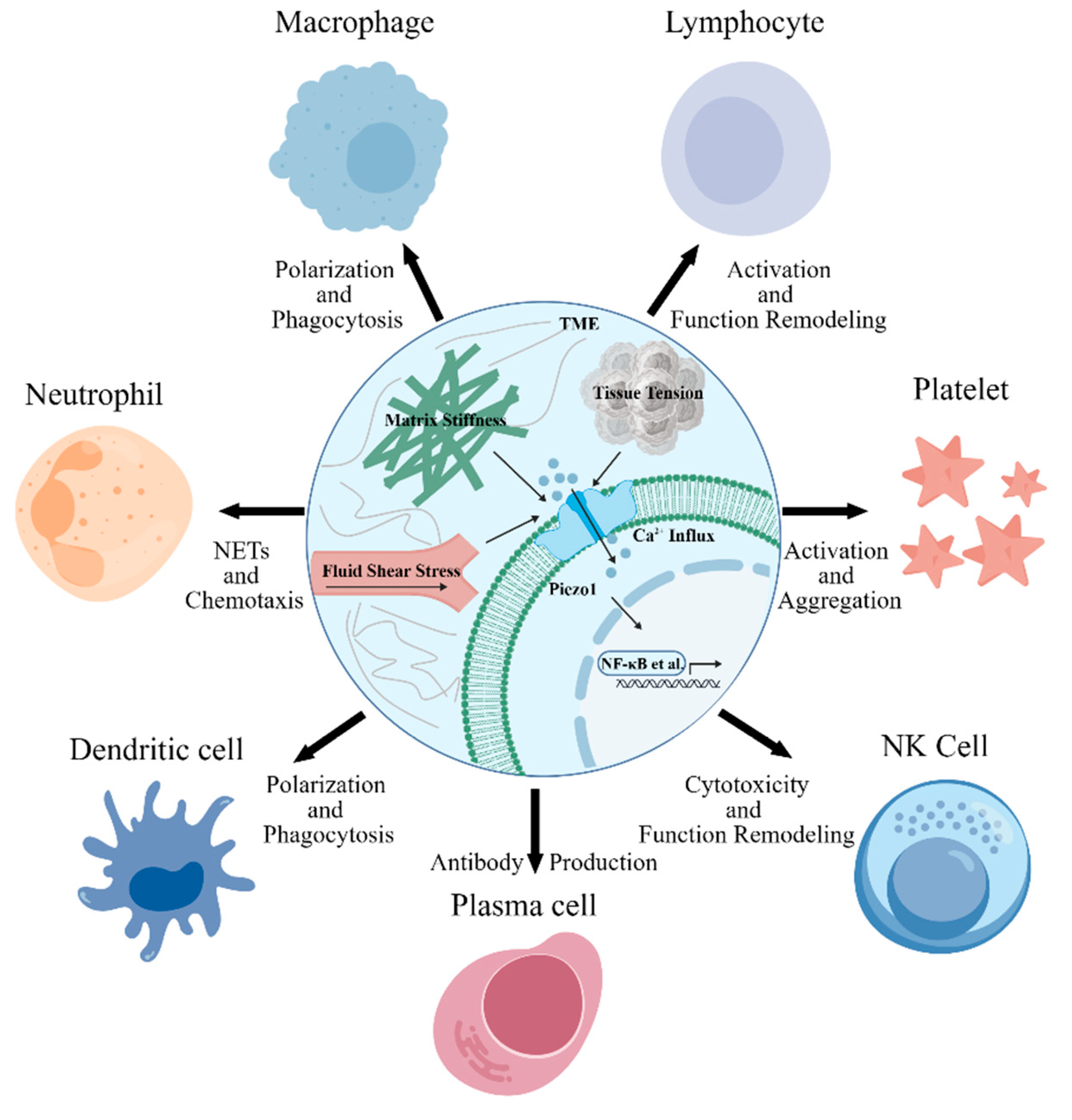
3. Integrated Effects of Piezo1-Mediated Blood Cell Mechanotransduction in Disease Mechanical Microenvironments
3.1. Interactive Regulation Between Disease Mechanical Microenvironment Mechanical Signals and Blood Cell Functions
3.2. Mechanical Synergism Among Different Blood Cells Mediated by Piezo1
4. Potential and Challenges of Piezo1 as a Therapeutic Target in Diseases Associated with Tissue Stiffening
4.1. Therapeutic Strategies Targeting Piezo1-Mediated Blood Cell Functional Regulation
4.2. Challenges and Optimization Directions in Clinical Translation
5. Summary and Perspectives
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mayorca-Guiliani, A. E.; et al. ECM formation and degradation during fibrosis, repair, and regeneration of the article. Npj Metab. Health Dis. 2025, 3, 25. [Google Scholar] [CrossRef]
- Tiskratok, W.; et al. Extracellular matrix stiffness: mechanotransduction and mechanobiological response-driven strategies for biomedical applications targeting fibroblast inflammation of the article. Polymers 2025, 17, 822. [Google Scholar] [CrossRef]
- Ai, J.; et al. Mechanical microenvironment in tumor immune evasion: bidirectional regulation between matrix stiffness and immune cells and its therapeutic implications of the article. Int. J. Biol. Sci. 2026, 22, 280–307. [Google Scholar] [CrossRef]
- Levental, K. R.; et al. Matrix crosslinking forces tumor progression by enhancing integrin signaling of the article. Cell 2009, 139, 891–906. [Google Scholar] [CrossRef] [PubMed]
- Wynn, T. A.; et al. Mechanisms of fibrosis: therapeutic translation for fibrotic disease of the article. Nat. Med. 2012, 18, 1028–1040. [Google Scholar] [CrossRef]
- Paliwal, N.; et al. Presence of left atrial fibrosis may contribute to aberrant hemodynamics and increased risk of stroke in atrial fibrillation patients of the article. Front. Physiol. 2021, 12, 657452. [Google Scholar] [CrossRef]
- Yan, R.; et al. Mechanotransduction in shaping immunity: pathways, crosstalk, and pathophysiological relevance of the article. Adv. Sci. 2025, 12, e12164. [Google Scholar] [CrossRef]
- Pirri, C. PIEZO channels in mechano-inflammation: gatekeepers of neuroimmune crosstalk of the article. Diseases 2025, 13, 263. [Google Scholar] [CrossRef] [PubMed]
- Karkempetzaki, A. I.; et al. Piezo1 and its function in different blood cell lineages of the article. Cells 2024, 13, 482. [Google Scholar] [CrossRef]
- Baratchi, S.; et al. Piezo1 expression in neutrophils regulates shear-induced NETosis of the article. Nat. Commun. 2024, 15, 7023. [Google Scholar] [CrossRef] [PubMed]
- Atcha, H.; et al. Mechanically activated ion channel Piezo1 modulates macrophage polarization and stiffness sensing of the article. Nat. Commun. 2021, 12, 3256. [Google Scholar] [CrossRef]
- Ilkan, Z.; et al. Evidence for shear-mediated Ca2+ entry through mechanosensitive cation channels in human platelets and a megakaryocytic cell line of the article. J. Biol. Chem. 2017, 292, 9204–9217. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; et al. Involvement of Piezo 1 in inhibition of shear-induced platelet activation and arterial thrombosis by ginsenoside Rb1 of the article. Br. J. Pharmacol. 2025, 182, 1957–1974. [Google Scholar] [CrossRef] [PubMed]
- Demagny, J.; et al. Role of the mechanotransductor PIEZO1 in megakaryocyte differentiation of the article. J. Cell. Mol. Med. 2024, 28, e70055. [Google Scholar] [CrossRef] [PubMed]
- Bachelot-Loza, C.; et al. Piezo1 gain of function induces a platelet preactivation state of the article. J. Thromb. Haemost. JTH 2026, 24, 1118–1129. [Google Scholar] [CrossRef]
- Kwak, K.; et al. B cell responses to membrane-presented antigens require the function of the mechanosensitive cation channel Piezo1 of the article. Sci. Signal. 2023, 16, eabq5096. [Google Scholar] [CrossRef]
- Qu, P.; et al. The dual role of Piezo1 in tumor cells and immune cells: a new target for cancer therapy of the article. Front. Immunol. 2025, 16, 1635388. [Google Scholar] [CrossRef]
- Yanamandra, A. K.; et al. PIEZO1-mediated mechanosensing governs NK-cell killing efficiency and infiltration in three-dimensional matrices of the article. Eur. J. Immunol. 2024, 54, e2350693. [Google Scholar] [CrossRef]
- Wang, Y.; et al. Dendritic cell Piezo1 directs the differentiation of TH1 and Treg cells in cancer of the article. eLife 2022, 11, e79957. [Google Scholar] [CrossRef]
- Wu, L.; et al. Neutrophils in the tumor microenvironment of the article. Adv. Exp. Med. Biol. 2020, 1224, 1–20. [Google Scholar]
- Hou, R.; et al. Tumor-associated neutrophils: critical regulators in cancer progression and therapeutic resistance (Review) of the article. Int. J. Oncol. 2025, 66, 28. [Google Scholar] [CrossRef]
- Wang, Y.; et al. Piezo1-directed neutrophil extracellular traps regulate macrophage differentiation during influenza virus infection of the article. Cell. Death Dis. 2025, 16, 60. [Google Scholar] [CrossRef] [PubMed]
- Mukhopadhyay, A.; et al. Trans-endothelial neutrophil migration activates bactericidal function via Piezo1 mechanosensing of the article. Immunity 2024, 57, 52–67.e10. [Google Scholar] [CrossRef] [PubMed]
- Knaus, U. G.; et al. Challenging the role of a NOX4-Piezo1 axis in neutrophil bactericidal function of the article. Immunity 2024, 57, 1716–1718. [Google Scholar] [CrossRef]
- Lightsey, S.; et al. Natural killer cell mechanosensing in solid tumors of the article. Bioengineering 2024, 11, 328. [Google Scholar] [CrossRef]
- Zhu, Y.; et al. Low shear stress exacerbates atherosclerosis by inducing the generation of neutrophil extracellular traps via Piezo1-mediated mechanosensation of the article. Atherosclerosis 2024, 391, 117473. [Google Scholar] [CrossRef]
- Zhao, W.; et al. Mechanotransduction in neutrophil: mechanosensing and immune function regulation of the article. Mechanobiol. Med. 2025, 3, 100157. [Google Scholar] [CrossRef]
- Wang, S.; et al. Mechanosensation by endothelial PIEZO1 is required for leukocyte diapedesis of the article. Blood 2022, 140, 171–183. [Google Scholar] [CrossRef]
- Kim, O. H.; et al. Fluid shear stress facilitates prostate cancer metastasis through Piezo1-Src-YAP axis of the article. Life Sci. 2022, 308, 120936. [Google Scholar] [CrossRef]
- Zhao, F.; et al. Mechanosensitive ion channel PIEZO1 signaling in the hall-marks of cancer: structure and functions of the article. Cancers 2022, 14, 4955. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; et al. PIEZO1 mediates mechanical reprogramming of neutrophils for proangiogenic specialization in the lung of the article. J. Clin. Investig. 2025, 135. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.; et al. Mechanical strain-programmed SDC1+ sheath fibroblasts trigger CXCR4hi neutrophil-mediated enthesitis in ankylosing spondylitis of the article. Adv. Sci. 2026, 13, e20617. [Google Scholar] [CrossRef]
- Shang, T.; et al. Shear stress governs hematopoietic stem cell fate to promote inflammation-induced aging of the article. Nat. Aging 2026, 6, 88–107. [Google Scholar] [CrossRef]
- Aykut, B.; et al. Targeting Piezo1 unleashes innate immunity against cancer and infectious disease of the article. Sci. Immunol. 2020, 5, eabb5168. [Google Scholar] [CrossRef]
- Murray, P. J.; et al. Protective and pathogenic functions of macrophage subsets of the article. Nat. Rev. Immunol. 2011, 11, 723–737. [Google Scholar] [CrossRef]
- Xiong, J.; et al. Matrix stiffness affects tumor-associated macrophage functional polarization and its potential in tumor therapy of the article. J. Transl. Med. 2024, 22, 85. [Google Scholar] [CrossRef]
- Mei, F.; et al. Matrix stiffness regulates macrophage polarisation via the Piezo1-YAP signalling axis of the article. Cell. Prolif. 2024, 57, e13640. [Google Scholar] [CrossRef] [PubMed]
- Cai, G.; et al. Piezo1-mediated M2 macrophage mechanotransduction enhances bone formation through secretion and activation of transforming growth factor-β1 of the article. Cell. Prolif. 2023, 56, e13440. [Google Scholar] [CrossRef]
- Zhang, Y.; et al. Piezo1 knockdown activates PI3K/AKT and enhances SPP1 to drive M2 macrophage polarization and reduce cardiac inflammation of the article. Sci. Rep. 2026, 16, 4879. [Google Scholar] [CrossRef]
- Fish, A.; et al. Flow-induced shear stress primes NLRP3 inflammasome activation in macrophages via Piezo1 of the article. ACS Appl. Mater. Interfaces 2024, 16, 4505–4518. [Google Scholar] [CrossRef] [PubMed]
- Luo, S.; et al. Piezo1 specific deletion in macrophage protects the progression of liver fibrosis in mice of the article. Theranostics 2023, 13, 5418–5434. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; et al. Nano-mechanical immunoengineering: nanoparticle elasticity reprograms tumor-associated macrophages via Piezo1 of the article. ACS Nano 2024, 18, 21221–21235. [Google Scholar] [CrossRef]
- Ezzo, M.; et al. Acute contact with profibrotic macrophages mechanically activates fibroblasts via αvβ3 integrin-mediated engagement of Piezo1 of the article. Sci. Adv. 2024, 10, eadp4726. [Google Scholar] [CrossRef]
- Jiang, J.; et al. Mobile mechanical signal generator for macrophage polarization of the article. Exploration 2023, 3, 20220147. [Google Scholar] [CrossRef] [PubMed]
- Jiang, T.; et al. Synergistic effect of ultrasound and reinforced electrical environment by bioinspired periosteum for enhanced osteogenesis via immunomodulation of macrophage polarization through Piezo1 of the article. Mater. Today. Bio 2024, 27, 101147. [Google Scholar] [CrossRef]
- Chen, X.; et al. Piezo1 aggravates ischemia/reperfusion-induced acute kidney injury by Ca2+-dependent calpain/HIF-1α/Notch signaling of the article. Ren. Fail. 2025, 47, 2447801. [Google Scholar] [CrossRef]
- Mammadova-Bach, E.; et al. Platelet mechanotransduction: Regulatory cross talk between mechanosensitive receptors and calcium channels of the article. Arterioscler. Thromb. Vasc. Biol. 2023, 43, 1339–1348. [Google Scholar] [CrossRef]
- Sforna, L.; et al. Piezo1 controls cell volume and migration by modulating swelling-activated chloride current through Ca2+ influx of the article. J. Cell. Physiol. 2022, 237, 1857–1870. [Google Scholar] [CrossRef]
- Liang, P.; et al. Targeting PIEZO1-TMEM16F Coupling to Mitigate Sickle Cell Disease Complications of the article. Am. J. Hematol. 2025, 100, 2261–2275. [Google Scholar] [CrossRef]
- Zhu, W.; et al. PIEZO1 mediates a mechanothrombotic pathway in diabetes of the article. Sci. Transl. Med. 2022, 14, eabk1707. [Google Scholar] [CrossRef] [PubMed]
- Podoplelova, N. A.; et al. Coagulation factors bound to procoagulant platelets concentrate in cap structures to promote clotting of the article. Blood 2016, 128, 1745–1755. [Google Scholar] [CrossRef]
- Zhang, M.; et al. Extracellular matrix stiffness: mechanisms in tumor progression and therapeutic potential in cancer of the article. Exp. Hematol. Oncol. 2025, 14, 54. [Google Scholar] [CrossRef]
- Zhang, C.; et al. Mechanoadaptation via myosin cytoplasmic redistribution protects circulating tumor cells from shear-induced death during hematogenous dissemination of the article. Adv. Sci. 2026, e23112. [Google Scholar] [CrossRef] [PubMed]
- Cheng, C. K.; et al. Biophysical and biochemical roles of shear stress on endothelium: a revisit and new insights of the article. Circ. Res. 2025, 136, 752–772. [Google Scholar] [CrossRef]
- Zhao, W.; et al. Piezo1 initiates platelet hyperreactivity and accelerates thrombosis in hypertension of the article. J. Thromb. Haemost. JTH 2021, 19, 3113–3125. [Google Scholar] [CrossRef] [PubMed]
- Tsuchiya, M.; et al. Cell surface flip-flop of phosphatidylserine is critical for PIEZO1-mediated myotube formation of the article. Nat. Commun. 2018, 9, 2049. [Google Scholar] [CrossRef] [PubMed]
- Leng, L.; et al. RhoA and the function of platelet integrin alphaIIbbeta3 of the article. Blood 1998, 91, 4206–4215. [Google Scholar] [CrossRef]
- Rab, S. O.; et al. Targeting platelet-tumor cell interactions: a novel approach to cancer therapy of the article. Med. Oncol. 2025, 42, 232. [Google Scholar] [CrossRef]
- Li, Y.; et al. Effects of the interactions between platelets with other cells in tumor growth and progression of the article. Front. Immunol. 2023, 14, 1165989. [Google Scholar]
- Palumbo, J. S.; et al. Platelets and fibrin(ogen) increase metastatic potential by impeding natural killer cell-mediated elimination of tumor cells of the article. Blood 2005, 105, 178–185. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; et al. Hypoxia stimulates CTC-platelet cluster formation to promote breast cancer metastasis of the article. iScience 2024, 27, 109547. [Google Scholar] [CrossRef]
- Schmied, L.; et al. Platelet-mediated protection of cancer cells from immune surveillance - possible implications for cancer immunotherapy of the article. Front. Immunol. 2021, 12, 640578. [Google Scholar] [CrossRef]
- Abbonante, V.; et al. Newly identified roles for PIEZO1 mechanosensor in controlling normal megakaryocyte development and in primary myelofibrosis of the article. Am. J. Hematol. 2024, 99, 336–349. [Google Scholar] [CrossRef]
- Kim, S. J.; et al. Role of platelets in neutrophil extracellular trap (NET) production and tissue injury of the article. Semin. Immunol. 2016, 28, 546–554. [Google Scholar] [CrossRef]
- Caudrillier, A.; et al. Platelets induce neutrophil extracellular traps in transfusion-related acute lung injury of the article. J. Clin. Investig. 2012, 122, 2661–2671. [Google Scholar] [CrossRef]
- Li, J.; et al. Pathological roles of NETs-platelet synergy in thrombotic diseases: from molecular mechanisms to therapeutic targeting of the article. Int. Immunopharmacol. 2025, 159, 114934. [Google Scholar] [CrossRef] [PubMed]
- Zhao, R.; et al. T cell polarization and NFAT activation are stiffness dependent and differentially regulated by the channels PIEZO1 and ORAI1 of the article. Sci. Signal. 2026, 19, eadt9566. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; et al. Osr2 functions as a biomechanical checkpoint to aggravate CD8+ T cell exhaustion in tumor of the article. Cell 2024, 187, 3409–3426.e3424. [Google Scholar] [CrossRef]
- Pang, R.; et al. PIEZO1 mechanically regulates the antitumour cytotoxicity of T lymphocytes of the article. Nat. Biomed. Eng. 2024, 8, 1162–1176. [Google Scholar] [CrossRef] [PubMed]
- Jung, Y.; et al. Piezo1 selectively enhances TGF-β1-induced IgA class switching by B cells of the article. Cell. Mol. Life Sci. CMLS 2025, 82, 243. [Google Scholar] [CrossRef]
- Liu, C. S. C.; et al. Piezo1 mechanosensing regulates integrin-dependent chemotactic migration in human T cells of the article. eLife 2024, 12, RP91903. [Google Scholar] [CrossRef] [PubMed]
- Choi, S. H.; et al. Piezo1 restrains proinflammatory response but is essential in T-cell-mediated immunopathology of the article. J. Leukoc. Biol. 2025, 117, qiae242. [Google Scholar] [CrossRef]
- Jairaman, A.; et al. Piezo1 channels restrain regulatory T cells but are dispensable for effector CD4+ T cell responses of the article. Sci. Adv. 2021, 7, eabg5859. [Google Scholar] [CrossRef]
- Chang, J. E.; et al. Mechanosensing by Peyer’s patch stroma regulates lymphocyte migration and mucosal antibody responses of the article. Nat. Immunol. 2019, 20, 1506–1516. [Google Scholar] [CrossRef]
- Abiff, M.; et al. Piezo1 facilitates optimal T cell activation during tumor challenge of the article. Oncoimmunology 2023, 12, 2281179. [Google Scholar] [CrossRef]
- Chakraborty, M.; et al. Mechanical stiffness controls dendritic cell metabolism and function of the article. Cell. Rep. 2021, 34, 108609. [Google Scholar] [CrossRef]
- Pattanayak, S.; et al. Spatiotemporal regulation of ligand trafficking and TLR9 activation by PIEZO1 in human plasmacytoid dendritic cells of the article. Research 2026, 9, 1067. [Google Scholar]
- Friedrich, E. E.; et al. Endothelial cell Piezo1 mediates pressure-induced lung vascular hyperpermeability via disruption of adherens junctions of the article. Proceedings of the National Academy of Sciences of the United States of America 2019, 116, 12980–12985. [Google Scholar] [CrossRef]
- Albarrán-Juárez, J.; et al. Piezo1 and Gq/G11 promote endothelial inflammation depending on flow pattern and integrin activation of the article. J. Exp. Med. 2018, 215, 2655–2672. [Google Scholar] [CrossRef] [PubMed]
- Gu, Z.; et al. Multimodal regulation of dendritic cells via mineralized vaccines for postsurgical tumor relapse prevention of the article. ACS Nano 2025, 19, 19901–19917. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; et al. Role of the mechanical microenvironment in cancer development and progression of the article. Cancer Biol. Med. 2020, 17, 282–292. [Google Scholar] [CrossRef]
- Bi, Y.; et al. Mechanical models and measurement methods of solid stress in tumors of the article. Appl. Microbiol. Biotechnol. 2024, 108, 363. [Google Scholar] [CrossRef]
- Liu, Y.; et al. Mechanosensitive channel Piezo1 in calcium dynamics: structure, function, and emerging therapeutic strategies of the article. Front. Mol. Biosci. 2025, 12, 1693456. [Google Scholar]
- Deng, J.; et al. Piezo1 regulates actin cytoskeleton remodeling to drive EMT in cervical cancer through the RhoA/ROCK1/PIP2 signaling pathway of the article. Discov. Oncol. 2025, 16, 787. [Google Scholar] [CrossRef] [PubMed]
- Obermann, W. M. J.; et al. Platelets, constant and cooperative companions of sessile and disseminating tumor cells, crucially contribute to the tumor microenvironment of the article. Front. Cell. Dev. Biol. 2021, 9, 674553. [Google Scholar] [CrossRef]
- Cui, X.; et al. Piezo1 as a mechanical checkpoint in T cell immunotherapy for solid tumors of the article. Front. Pharmacol. 2026, 17, 1722027. [Google Scholar] [CrossRef]
- Cui, X.; et al. Mechanotransduction of Piezo1 in the cancer microenvironment: implications for NK cell-based immunotherapy of the article. Front. Oncol. 2025, 15, 1729588. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; et al. Tumor mechanobiology-inspired nanomechanics: Piezo1 mediated tumor-associated macrophages reprogramming and exosome-driven immune amplification of the article. ACS Nano 2025, 19, 41746–41764. [Google Scholar] [CrossRef]
- Xu, J.; et al. Dual roles and therapeutic targeting of tumor-associated macrophages in tumor microenvironments of the article. Signal Transduct. Target. Ther. 2025, 10, 268. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; et al. Tumor-associated macrophages remodel the suppressive tumor immune microenvironment and targeted therapy for immunotherapy of the article. J. Exp. Clin. Cancer Res. CR 2025, 44, 145. [Google Scholar]
- Wang, Y.; et al. Immunoregulatory role of the mechanosensitive ion channel Piezo1 in inflammation and cancer of the article. Molecules 2022, 28, 213. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.



