Submitted:
24 April 2026
Posted:
27 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
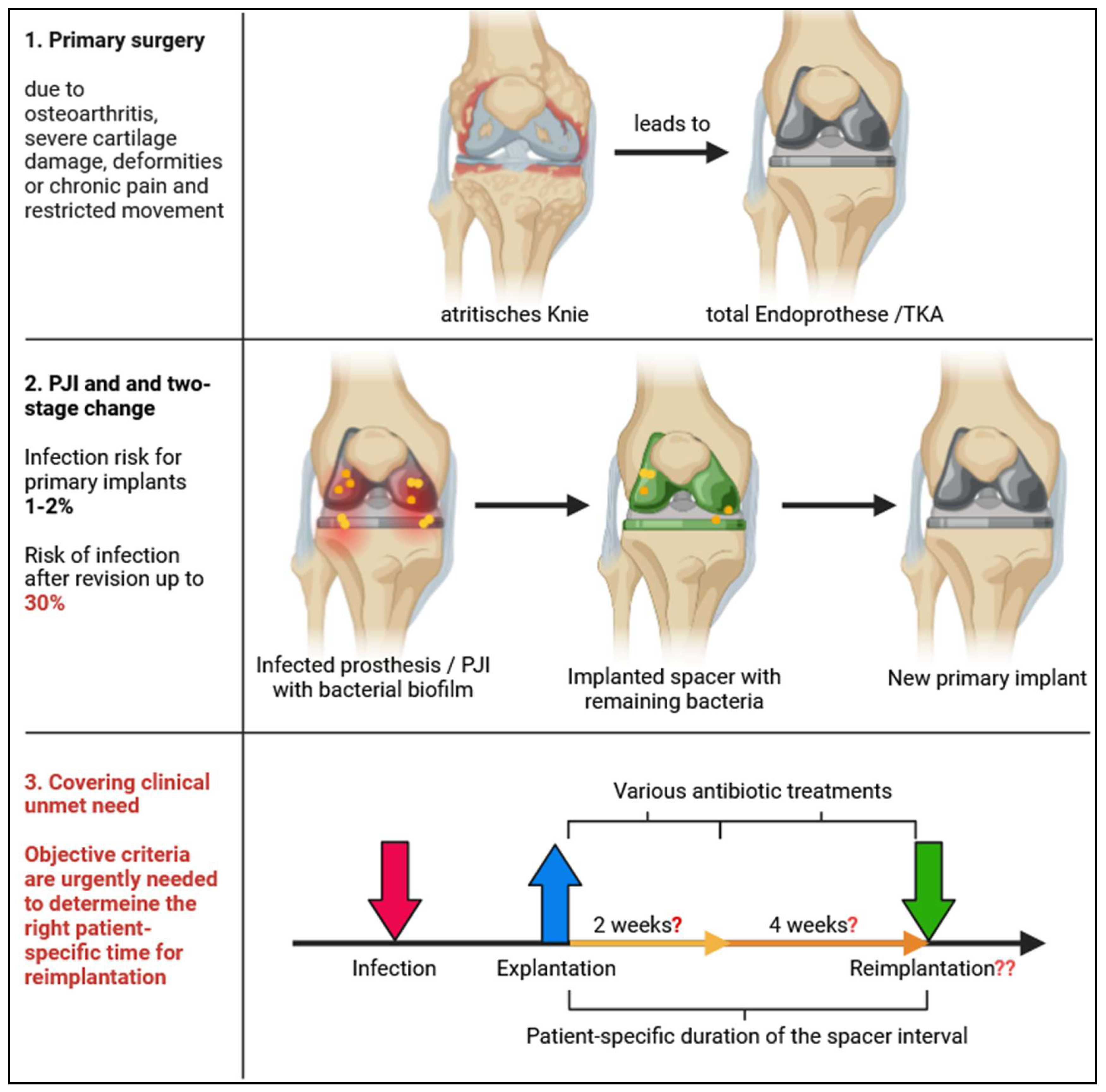
1.1. The Unresolved Clinical Challenge of Periprosthetic Joint Infection
1.2. Failure of the Current Treatment Paradigm
1.3. Light-Based Therapies as a Missing Link for In Vivo Infection Control
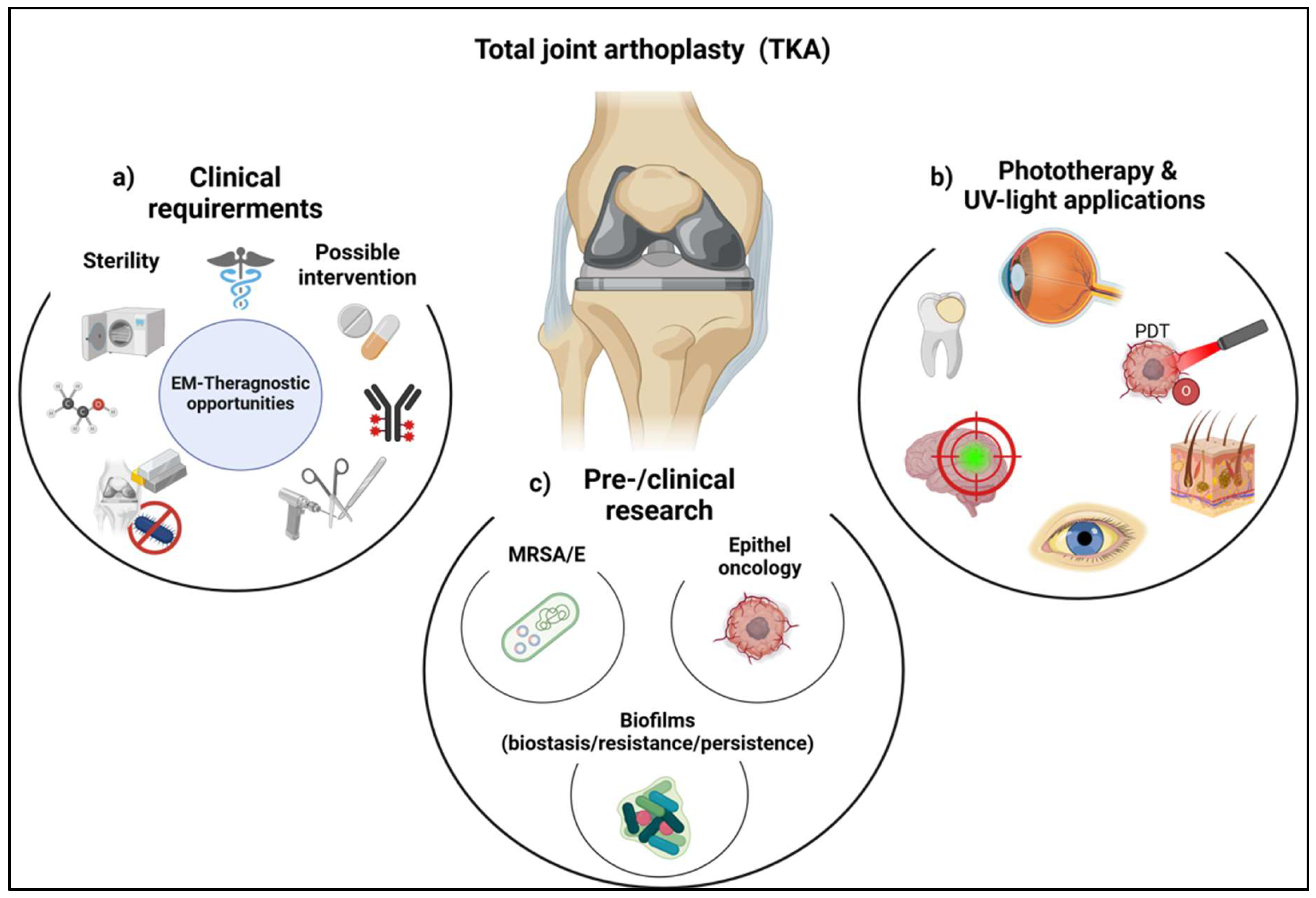
2. Concept of a Theranostic Implant System
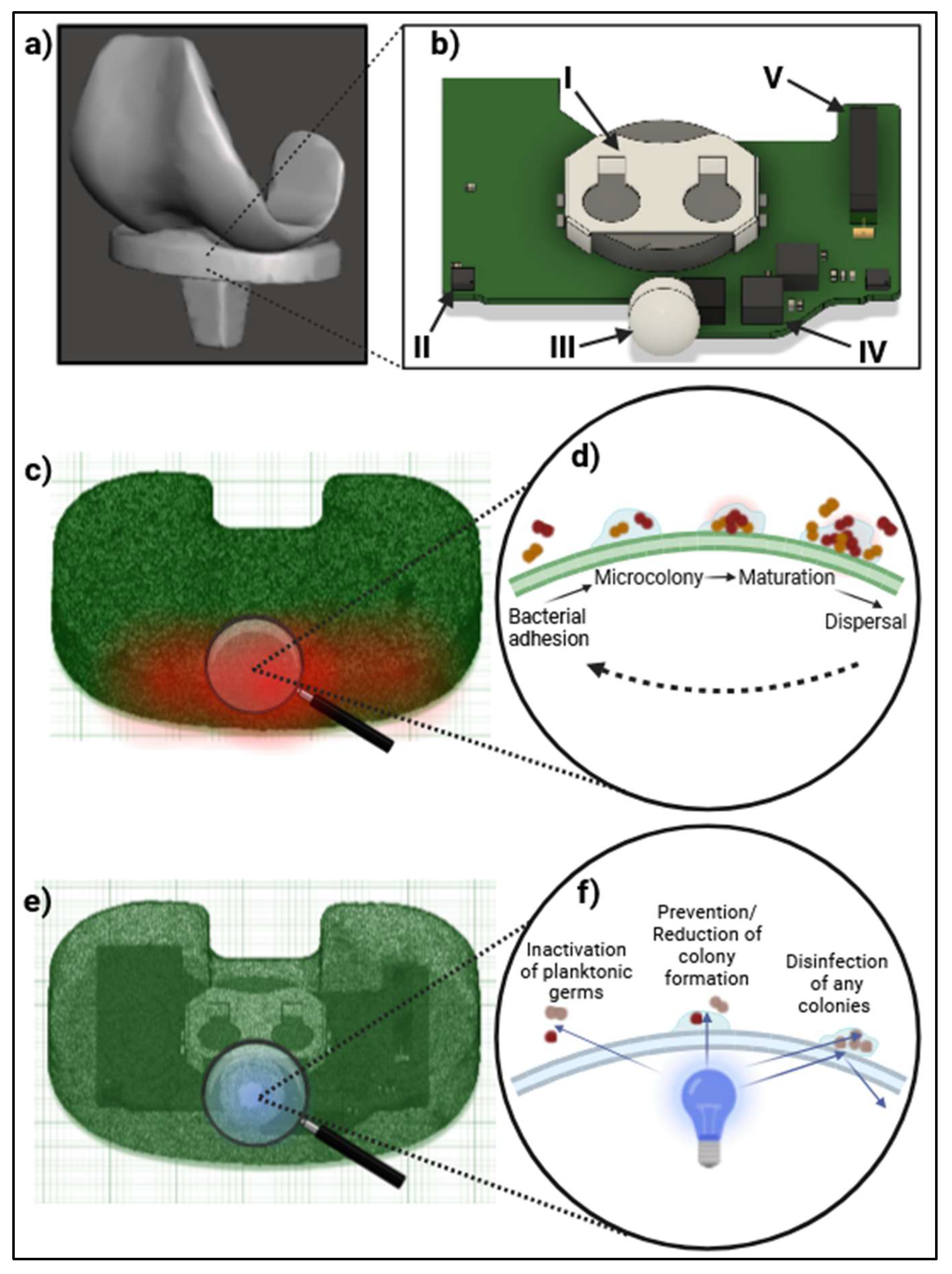
2.1. The SmartSpacer Platform Architecture
2.2. Optical Accessibility as a Unique Advantage
2.3. Enabling Light-Based Therapeutic Modalities
2.4. Engineering Constraints and Design Considerations
2.5. From Concept to Translational Framework
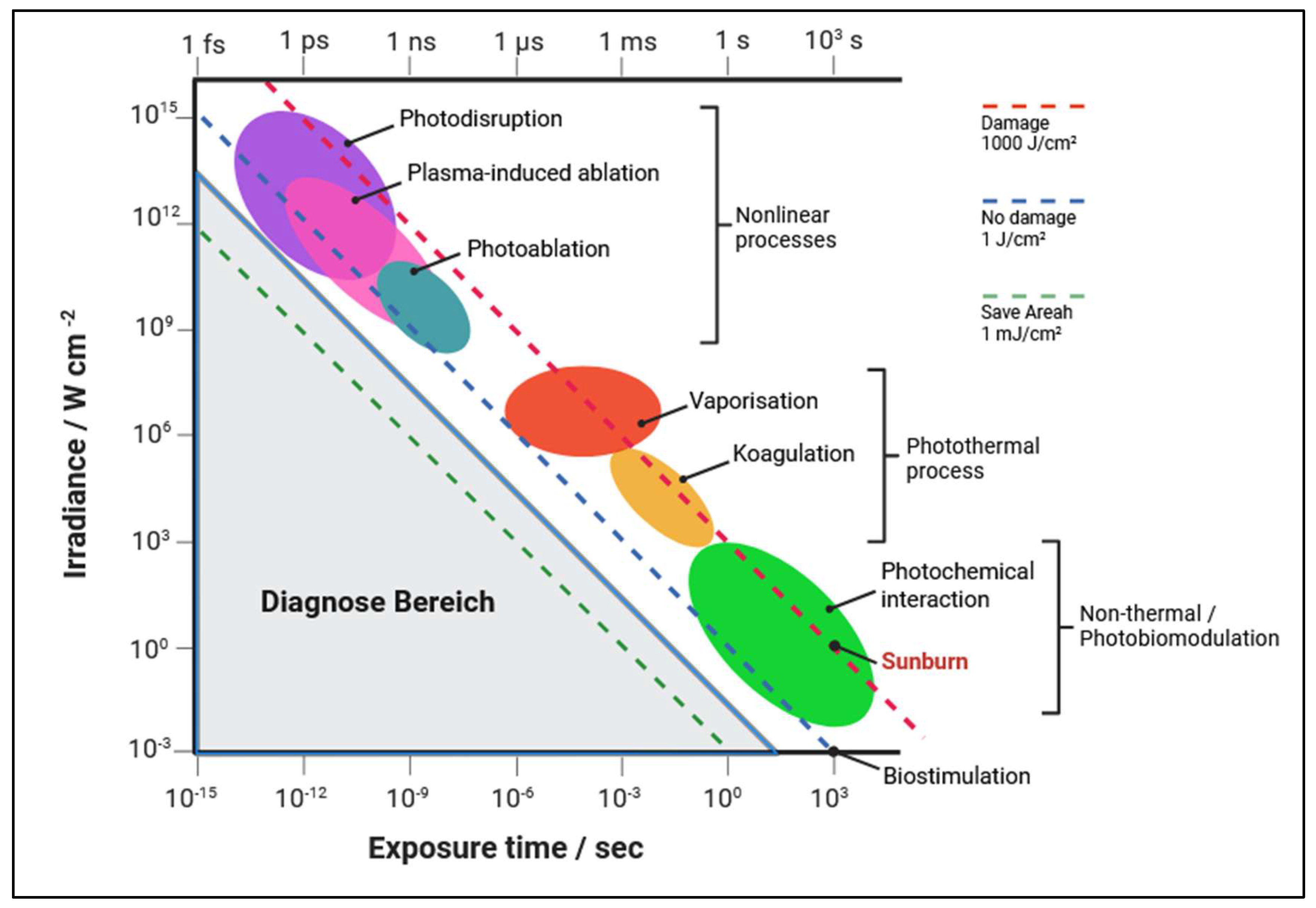
3. From Descriptive Review to Translational Framework
3.1. Translational Selection and Parameter Mapping
3.2. Integration of Engineering Constraints
3.3. From Experimental Evidence to Design Rules
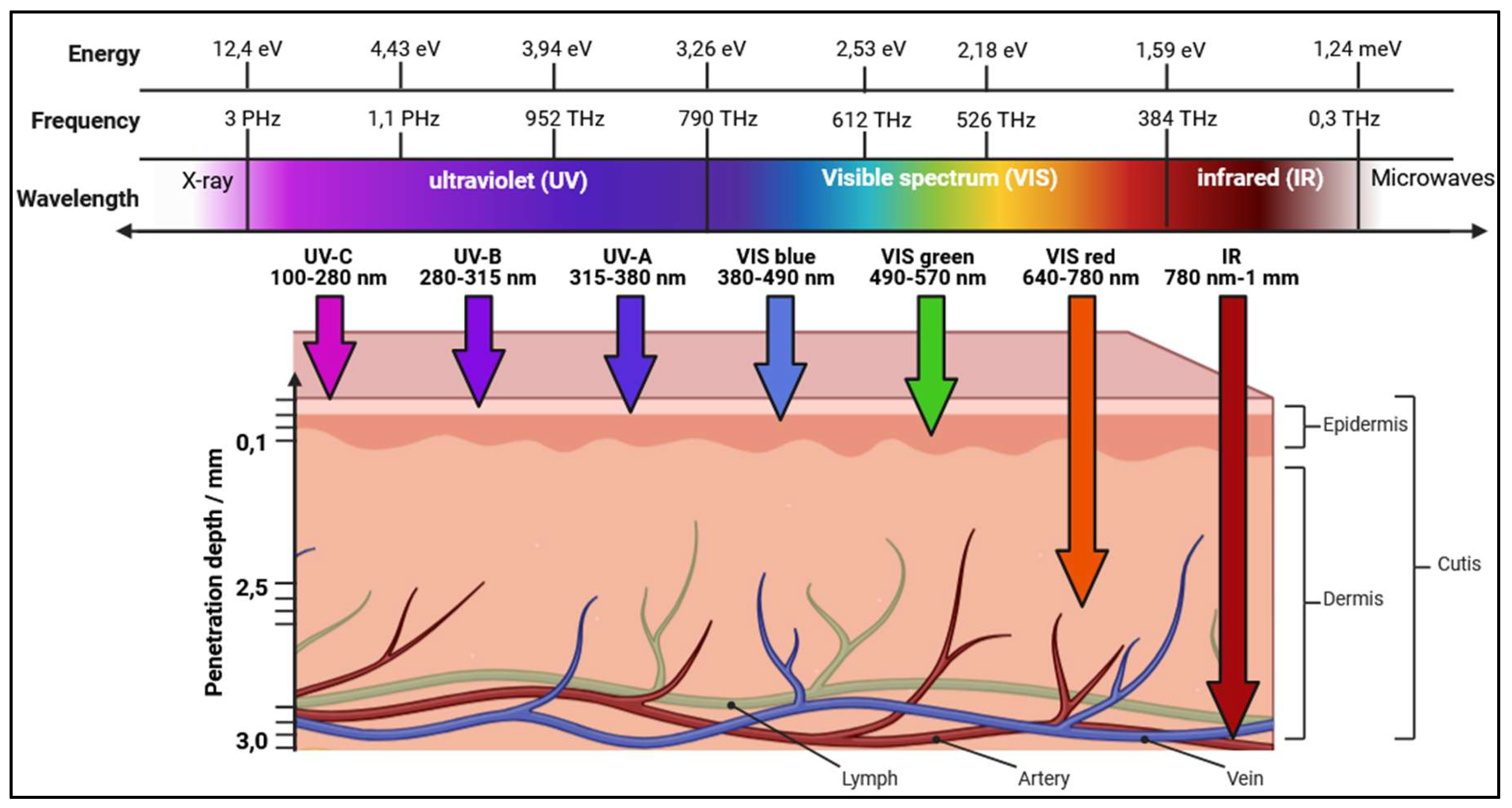
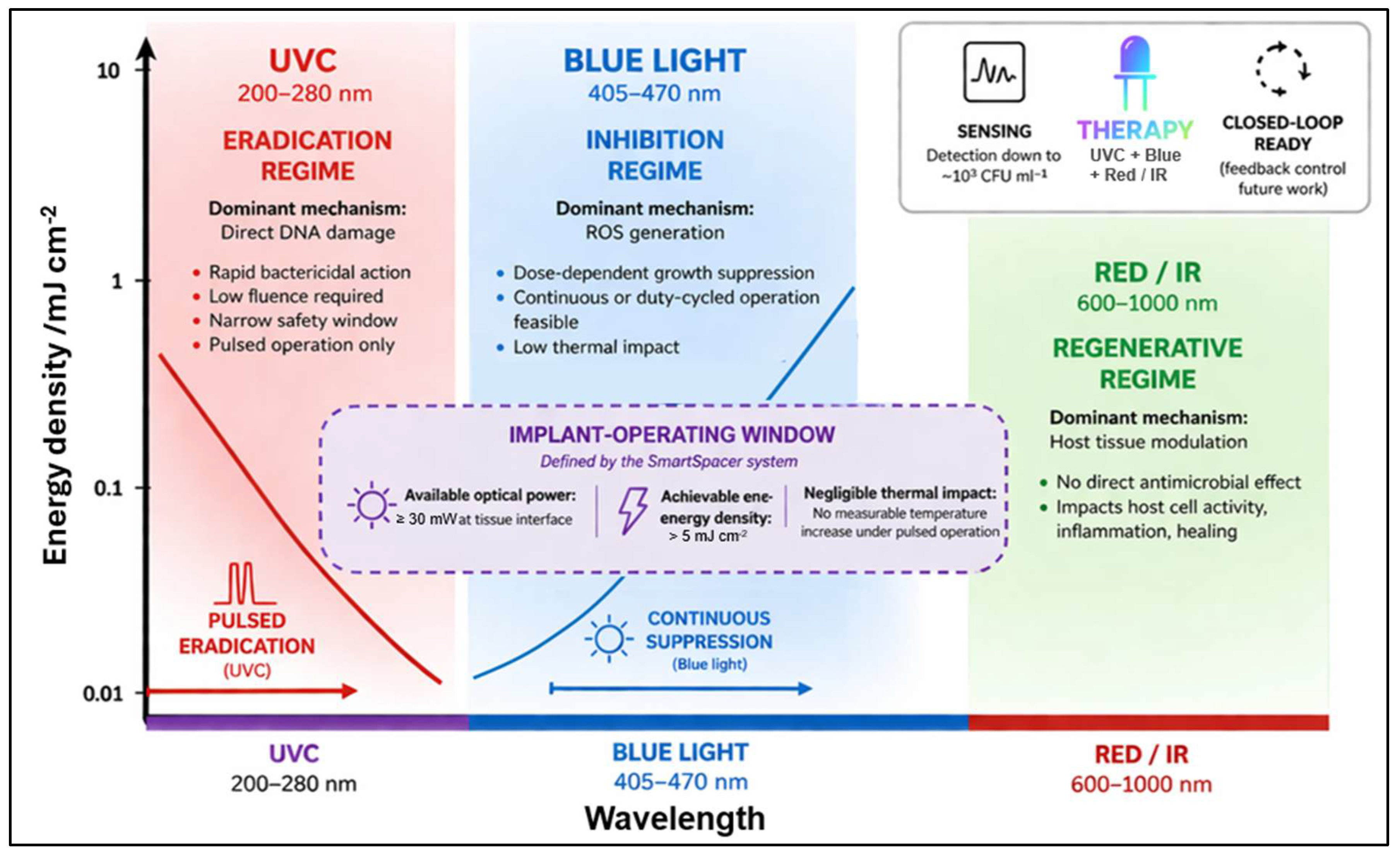
4. Spectral Design Space for Light-Based Antimicrobial Therapy
4.1. Wavelength-Dependent Antimicrobial Mechanisms
| UV-C | UV-B | UV-A | VIS blue | VIS green | VIS red | IR |
| Effect on human cells | ||||||
| Causes DNA changes | Acute damage: main cause of sunburn. | Long-term damage: Causes skin ageing, can increase the risk of skin cancer | Increase in nitric oxide | Improvement of differentiation and matrix mineralisation of osteogenic cells | Enhancement of cell proliferation (by suppressing cell apoptosis) | Induction of regenerative and anti-inflammatory effects |
| Changes the barrier function of cells | Long-term effects: DNA damage and increase the risk of skin cancer | Immediate effects: May cause sunburn, although less energetic | Molecular and cellular changes in fibroblasts | Support of healing processes through mitochondrial stimulation and increase in ATP | Modulation of cell growth | Increase in collagen production in dermal fibroblasts |
| Can lead to cell necrosis | Vitamin D production in the skin, for bone health & immune system | Vitamin D synthesis: Contributes to the production of vitamin D | Premature skin ageing | Hardly harmful Effect on skin and eyes: Compared to blue light | Enhancement of succinic dehydrogenase activity | Change in the collagen balance of the extracellular matrix of the skin |
| Effect on bacteria | ||||||
| Direct DNA destruction of the bacteria leads to immediate cell death | Direct DNA damage to the bacteria prevents correct replication and leads to mutations | Indirekt DNA-Schäden indem es reaktive Sauerstoffspezies (ROS) erzeugt | Strong bactericidal effect | Moderate bactericidal effect | Reduced bactericidal effect | Low direct bactericidal effect |
| Cell wall and membrane damage leads to loss of structural integrity and cell death | Protein modification leads to impairment of cell functions | ROS-Angriff auf Zellmembran führt zu erhöhter Permeabilität und Zelltod führen | Formation of reactive oxygen species (ROS) | Destabilisation of the cell membrane | Influencing cell proliferation | Often in combination with photosensitisers |
| Inactivation of enzymes leads to a halt in metabolic processes | Stress response leads to the production of protective proteins and damage is repaired | Stress response leads to the production of protective proteins and damage is being repaired | Damage to the cell membrane and DNA | Inhibition of cell division | Damage to the cell membrane due to thermal effects | Influenced by thermal effects, increase in permeability |
4.2. Visible Light: Inhibition-Dominated Regime
4.3. Low-Fluence Regimes and Continuous Operation
4.4. Ultraviolet Radiation: Eradication-Dominated Regime
4.5. Functional Complementarity Across Spectral Regimes
4.6. Implications for Implant-Based Therapy Design
5. Synergistic Effects and Combination Therapy
6. Toward In Vivo Implementation
7. Future Perspective: Toward Theranostic Infection Control
Funding
Acknowledgements
Data Availability Statement
References
- Tande, A.J.; Patel, R. Prosthetic joint infection. Clin. Microbiol. Rev. 2014, 27, 302–345. [Google Scholar] [CrossRef]
- S.M. Kurtz, I. Exponent, Philadelphia, Pennsylvania, P. Drexel University, Pennsylvania et al. Economic Burden of Periprosthetic Joint Infection in the United States. J. Arthroplast. 2012, 27, 61–65.e61. [CrossRef]
- Zimmerli, W.; Trampuz, A.; Ochsner, P.E. Prosthetic-Joint Infections. N. Engl. J. Med. 2004, 351, 1645–1654. [Google Scholar] [CrossRef]
- Kurtz, S.; Ong, K.; Lau, E.; et al. Projections of Primary and Revision Hip and Knee Arthroplasty in the United States from 2005 to 2030. J. Bone Jt. Surg. 2007, 89, 780–785. [Google Scholar] [CrossRef]
- Sloan, M.; Premkumar, A.; Sheth, N.P. Projected Volume of Primary Total Joint Arthroplasty in the U.S., 2014 to 2030. J. Bone Jt. Surg. 2018, 100, 1455–1460. [Google Scholar] [CrossRef] [PubMed]
- Prince, M.J.; Wu, F.; Guo, Y.; et al. The burden of disease in older people and implications for health policy and practice. The Lancet 2015, 385, 549–562. [Google Scholar] [CrossRef] [PubMed]
- Vollset, S.E.; Goren, E.; Yuan, C.-W.; et al. Fertility, mortality, migration, and population scenarios for 195 countries and territories from 2017 to 2100: A forecasting analysis for the Global Burden of Disease Study. The Lancet 2020, 396, 1285–1306. [Google Scholar] [CrossRef]
- Corvec, S.; Portillo, M.E.; Pasticci, B.M.; et al. Epidemiology and new developments in the diagnosis of prosthetic joint infection. Int. J. Artif. Organs 2012, 35, 923–934. [Google Scholar] [CrossRef] [PubMed]
- Ong, K.L.; Kurtz, S.M.; Lau, E.; et al. Prosthetic joint infection risk after total hip arthroplasty in the Medicare population. J. Arthroplast. 2009, 24, 105–109. [Google Scholar] [CrossRef]
- Li, T.; Zhang, H.; Chan, P.K.; et al. Risk factors associated with surgical site infections following joint replacement surgery: A narrative review. Arthroplasty 2026, 4, 11. [Google Scholar] [CrossRef]
- Lazic, I.; Scheele, C.; Pohlig, F.; et al. Treatment options in PJI is two-stage still gold standard? J. Orthop. 2021, 23, 180–184. [Google Scholar] [CrossRef]
- Martino, A.D.; Carlo, G.D.; Pederiva, D.; et al. Which Patients with Chronic Periprosthetic Joint Infection Are Less Suitable to Successful Two Stage Exchange Arthroplasty Surgery? A Retrospective Clinical Trial. Clin. Pract. 2023, 13, 190–199. [Google Scholar] [CrossRef] [PubMed]
- Charette, R.S.; Melnic, C.M. Two-Stage Revision Arthroplasty for the Treatment of Prosthetic Joint Infection. Curr. Rev. Musculoskelet. Med. 2018, 11, 332–340. [Google Scholar] [CrossRef]
- Samelis, P.V.; Papagrigorakis, E.; Sameli, E.; et al. Current Concepts on the Application, Pharmacokinetics and Complications of Antibiotic-Loaded Cement Spacers in the Treatment of Prosthetic Joint Infections. Cureus 2022, 14. [Google Scholar] [CrossRef]
- Lora-Tamayo, J.; Mancheño-Losa, M.; Meléndez-Carmona, M.Á.; et al. Appropriate Duration of Antimicrobial Treatment for Prosthetic Joint Infections: A Narrative Review. Antibiotics 2024, 13, 293. [Google Scholar] [CrossRef]
- Gálvez-López, R.; Peña-Monje, A.; Antelo-Lorenzo, R.; et al. Elution kinetics, antimicrobial activity, and mechanical properties of 11 different antibiotic loaded acrylic bone cement. Diagn. Microbiol. Infect. Dis. 2014, 78. [Google Scholar] [CrossRef] [PubMed]
- Fink, B.; Tetsworth, K.D. Antibiotic Elution from Cement Spacers and Its Influencing Factors. Antibiotics 2025, 14, 705. [Google Scholar] [CrossRef]
- Meng-Lun, T.; Herng-Shouh, H.A.; Cheng-Ta, W.; et al. Optimal reimplantation timing in two-stage exchange for periprosthetic joint infection: An observative cohort study in Asian population. BMC Musculoskelet. Disord. 2024, 25, 28. [Google Scholar] [CrossRef]
- Jan, P.; Marc, H.; Georg, G.; et al. Evaluation of time to reimplantation as a risk factor in two-stage revision with static spacers for periprosthetic knee joint infection. J. Orthop. Traumatol. 2024, 25, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Johnson, N.R.; Rowe, T.M.; Valenzeula, M.M.; et al. Do Pre-Reimplantation Erythrocyte Sedimentation Rate/C-Reactive Protein Cutoffs Guide Decision-Making in Prosthetic Joint Infection? Are We Flying Blind? J. Arthroplast. 2022, 37. [Google Scholar] [CrossRef]
- McNally, M.; Sousa, R.; Wouthuyzen-Bakker, M.; et al. The EBJIS definition of periprosthetic joint infection: A practical guide for clinicians. Bone Jt. J. 2021, 103-B, 18–25. [Google Scholar] [CrossRef] [PubMed]
- Drago, L.; Clerici, P.; Morelli, I.; et al. The World Association against Infection in Orthopaedics and Trauma (WAIOT) procedures for Microbiological Sampling and Processing for Periprosthetic Joint Infections (PJIs) and other Implant-Related Infections. J. Clin. Med. 2019, 8, 933. [Google Scholar] [CrossRef] [PubMed]
- Mitterer, J.A.; Hartmann, S.G.; Simon, S.; et al. Comparison of synovial calprotectin and alpha-defensin for the diagnosis of persistent periprosthetic joint infections at second stage of a two-stage revision arthroplasty. Arch. Orthop. Trauma Surg. 2025, 145, 428. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, F.; Shafiq, B.; Noor, S.S.; et al. Economic Burden of Periprosthetic Joint Infection Following Primary Total Knee Replacement in a Developing Country. Clin. Orthop. Surg. 2020, 12, 470–476. [Google Scholar] [CrossRef]
- Kurz, S.M.; Higgs, G.B.; Lau, E.; et al. Hospital Costs for Unsuccessful Two-Stage Revisions for Periprosthetic Joint Infection. osthetic Joint Infection. J. Arthroplast. 2022, 37, 205–212. [Google Scholar] [CrossRef]
- Zmistowski, B.; Karam, J.A.; Durinka, J.B.; et al. Periprosthetic joint infection increases the risk of one-year mortality. J. Bone Jt. Surgery. Am. Vol. 2013, 95. [Google Scholar] [CrossRef]
- Schwarz, E.M.; Parvizi, J.; Gehrke, T. International Consensus Meeting on Musculoskeletal Infection: Research Priorities from the General Assembly Questions. J. Orthop. Res. 2019, 37, 997–1006. [Google Scholar] [CrossRef]
- Li, C.; Renz, N.; Trampuz, A. Management of Periprosthetic Joint Infection. Hip Pelvis 2018, 30, 138–146. [Google Scholar] [CrossRef]
- Hamblin, M.R. Mechanisms and applications of the anti-inflammatory effects of photobiomodulation. AIMS Biophys. 2017, 4, 337–361. [Google Scholar] [CrossRef]
- Rathod, D.G.; Muneer, H.; Masood, S. StatPearls in Phototherapy Treasure Island (FL) (StatPearls Publishing, 2023).
- Gholami, L.; Shahabi, S.; Jazaeri, M.; et al. Clinical applications of antimicrobial photodynamic therapy in dentistry. Front. Microbiol. 2023, 13, 1020995. [Google Scholar] [CrossRef]
- Woodgate, P.; Jardine, L.A. Neonatal jaundice: Phototherapy. BMJ Clin. Evid. 2015, 2015, 0319. [Google Scholar] [PubMed]
- Mazloumi, M.; Dalvin, L.A.; Abtahi, S.-H.; et al. Photodynamic Therapy in Ocular Oncology. J. Ophthalmic Vis. Res. 2020, 15, 547. [Google Scholar] [CrossRef] [PubMed]
- Jiang, W.; Liang, M.; Lei, Q.; et al. The Current Status of Photodynamic Therapy in Cancer Treatment. Cancers 2023, 15, 585. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Qin, R.; Zaat, S.A.J.; et al. Antibacterial photodynamic therapy: Overview of a promising approach to fight antibiotic-resistant bacterial infections. J. Clin. Transl. Res. 2015, 1, 140–167. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wang, Y.; Wang, Y.; et al. Antimicrobial blue light inactivation of pathogenic microbes: State of the art. Drug. Resist. Updat. 2017, 33–35, 1–22. [Google Scholar] [CrossRef]
- BoneSens-Our Progress. Available online: https://bonesens.com/Our-Progress (accessed on 28 November 2025).
- Dillitzer, C.; Tran, N.B.; Morandell, P.; et al. in International Conference on Engineering for Life Sciences: ENROL 2025.
- C. Dillitzer, N.B. Tran and O. Hayden. Scattering spectra acquisition and algorithmic analysis to distinguish between different bacterial concentrations of Staphylococcus Aureus as an application of a SmartSpacer system for two-stage knee total endoprosthesis (KTEP) revision. Vol. PC12837, San Francisco, SPIE.
- N.B. Tran, V. Bub and C. Dillitzer. Discrimination of bacterial concentrations of Staphylococcus Aureus based on statistical spectral analysis using a smart-spacer prototype with a low-resolution spectrometer unit as an application during two-stage total endoprosthesis (TEP) revision. Vol. 12387, SPIE.
- T. German Federal Ministry of Education and Research (BMBF) since 2025 Federal Ministry of Research, and Space (BMFTR). “SWi2M.; Intelligentes drahtloses Implantat zur Infektionsüberwachung - Entwicklung eines miniaturisierten Sensorsystems. Report No. FKZ: 01EK2107A, (BMBF now BMFTR, 2022). Available online: https://bonesens.com/ (accessed on 20 April 2026).
- H.K. Walker and V.E. Del-Bene. in Clinical Methods: The History, Physical, and Laboratory Examinations Ch. 218, (Butterworths, 1990). Available online: https://www.ncbi.nlm.nih.gov/pubmed/ (accessed on 20 April 2026).
- Zhang, S.; Li, P.; Xu, X.; et al. No-Reference Image Blur Assessment Based on Response Function of Singular Values. Symmetry 2018, 10, 304. [Google Scholar] [CrossRef]
- Glänzer, L.; Masalkhi, H.E.; Roeth, A.A.; et al. Vessel Delineation Using U-Net: A Sparse Labeled Deep Learning Approach for Semantic Segmentation of Histological Images. Cancers 2023, 15, 3773. [Google Scholar] [CrossRef]
- Chen, X.; Zhang, Q.; Lin, M. No-Reference Color Image Quality Assessment: From Entropy to Perceptual Quality. EURASIP J. Image Video Process. 2019, 77, 2019. [Google Scholar] [CrossRef]
- Krauß, F. “BlaVeS: Bladder Vessel Segmentation,” v 1 (2025). [CrossRef]
- Galo, I.D.C.; Prado, R.P.; Santos, W.G.D. Blue and red light photoemitters as approach to inhibit Staphylococcus aureus and Pseudomonas aeruginosa growth. Braz. J. Biol. 2021, 82. [Google Scholar] [CrossRef]
- Galo, I.D.C.; Lima, B.E.; Santos, T.G.; et al. Staphylococcus aureus growth delay after exposure to low fluencies of blue light (470 nm). Braz. J. Biol. 2021, 81. [Google Scholar] [CrossRef]
- Dai, T.; Murray, C.K.; Vrahas, M.S.; et al. Ultraviolet C light for Acinetobacter baumannii wound infections in mice: Potential use for battlefield wound decontamination? J. Trauma Acute Care Surg. 2012, 73, 661–667. [Google Scholar] [CrossRef]
- Costerton, J.W.; Stewart, P.S.; Greenberg, E.P. Bacterial biofilms: A common cause of persistent infections. Science 1999, 284, 1318–1322. [Google Scholar] [CrossRef]
- Hamblin, M.R.; Hasan, T. Photodynamic therapy: A new antimicrobial approach to infectious disease? Photochemical & photobiological sciences: Official journal of the European Photochemistry Association and the European Society for Photobiology 2004, 3, 436–450. [Google Scholar] [CrossRef]
- Ou, Y.; Petersen, P.M. Application of ultraviolet light sources for in vivo disinfection - IOPscience. Jpn. J. Appl. Phys. 2021, 100501. [Google Scholar] [CrossRef]
- International Commission on Radiological Protection. The 2007 Recommendations of the International Commission on Radiological Protection. ICRP publication 103. Report No. 0146-6453, (ICRP, 2007). Available online: https://www.ncbi.nlm.nih.gov/pubmed/18082557.
- Union, E. “Directive 2013/59/Euratom - protection against ionising radiation. (2013). Available online: https://osha.europa.eu/en/legislation/directives/directive-2013-59-euratom-protection-against-ionising-radiation (accessed on 20 April 2026).
- Commission, I.E. “IEC 60601-1-2: Medical electrical equipment – Part 1-2: General requirements for basic safety and essential performance – Collateral standard: Electromagnetic disturbances – Requirements and tests. Report No. IEC 60601-1-2, (International Electrotechnical Commission, 2014). Available online: https://www.vde-verlag.de/iec-normen/249094/iec-60601-1-2-2014-amd1-2020-csv.html (accessed on 19 April 2026).
- Commission, I.E. “IEC 60601-2-33: Medical electrical equipment – Part 2-33: Particular requirements for the basic safety and essential performance of magnetic resonance equipment for medical diagnosis. (International Electrotechnical Commission, 2015). Available online: https://www.vde-verlag.de/iec-normen/251063/iec-60601-2-33-2022.html (accessed on 19 April 2026).
- Yokomizo, S.; Roessing, M.; Morita, A.; et al. Near-infrared II photobiomodulation augments nitric oxide bioavailability via phosphorylation of endothelial nitric oxide synthase. FASEB J. 2022, 36. [Google Scholar] [CrossRef] [PubMed]
- Cadet, J.; Douki, T. Formation of UV-induced DNA damage contributing to skin cancer development. Photochem. Photobiol. Sci. 2018, 17, 1816–1841. [Google Scholar] [CrossRef]
- Kowalski, W. Ultraviolet Germicidal Irradiation Handbook: UVGI for Air and Surface Disinfection. Berlin/Heidelberg, Springer, ISBN 978-3-642-01998-2.
- Gruijl, F.R.D. Photocarcinogenesis: UVA vs UVB. Methods Enzymol. 2000, 319. [Google Scholar] [CrossRef]
- Ishida, K.; Matsubara, M.; Nagahashi, M.; et al. Efficacy of ultraviolet-light emitting diodes in bacterial inactivation and DNA damage via sensitivity evaluation using multiple wavelengths and bacterial strains. Arch. Microbiol. 2025, 207, 130. [Google Scholar] [CrossRef]
- Montero, P.; Roger, I.; Milara, J.; et al. Damaging effects of UVA, blue light, and infrared radiation: In vitro assessment on a reconstructed full-thickness human skin. Front. Med. 2023, 10, 1267409. [Google Scholar] [CrossRef]
- Chen, H.; Cheng, Y.; Moraru, C.I. Blue 405 nm LED light effectively inactivates bacterial pathogens on substrates and packaging materials used in food processing. Sci. Rep. 2023, 13, 15472. [Google Scholar] [CrossRef]
- Tamimi, R.; Mahmoodi, N.M.; Samadikhah, H.R.; et al. Anti-inflammatory effect of green photobiomodulation in human adipose-derived mesenchymal stem cells. Lasers Med. Sci. 2022, 37, 3693–3703. [Google Scholar] [CrossRef]
- Kang, Y.T.; Tri, T.T.; Jo, D.S.; et al. Impact of Red and Red/NIR OLEDs photobiomodulation effects towards promoting ADMSCs chondrogenic differentiation. Tissue Cell. 2025, 96, 102948. [Google Scholar] [CrossRef] [PubMed]
- Kharkwal, G.B.; Sharma, S.K.; Huang, Y.-Y.; et al. Photodynamic Therapy for Infections: Clinical Applications. Lasers Surg. Med. 2011, 43, 755. [Google Scholar] [CrossRef] [PubMed]
- Chung, H.; Dai, T.; Sharma, S.K.; et al. The nuts and bolts of low-level laser (light) therapy. Ann. Biomed. Eng. 2012, 40. [Google Scholar] [CrossRef] [PubMed]
- Ailioaie, L.M.; Litscher, G. Probiotics, Photobiomodulation, and Disease Management: Controversies and Challenges. Int. J. Mol. Sci. 2021, 22, 4942. [Google Scholar] [CrossRef]
- Karu, T.I. Mitochondrial signaling in mammalian cells activated by red and near-IR radiation. Photochem. Photobiol. 2008, 84, 1091–1099. [Google Scholar] [CrossRef]
- Silveira, P.V.D.; Panariello, B.H.D.; Araújo-Costa, C.A.G.D.; et al. Twice-daily red and blue light treatment for Candida albicans biofilm matrix development control. Lasers Med. Sci. 2019, 34, 441–447. [Google Scholar] [CrossRef]
- Wang, C.; Yang, Z.; Peng, Y.; et al. Application of 460 nm visible light for the elimination of Candida albicans in vitro and in vivo. Mol. Med. Rep. 2018, 18, 2017–2026. [Google Scholar] [CrossRef]
- Makdoumi, K.; Hedin, M.; Bäckman, A. Different photodynamic effects of blue light with and without riboflavin on methicillin-resistant Staphylococcus aureus (MRSA) and human keratinocytes in vitro. Lasers Med. Sci. 2019, 34, 1799–1805. [Google Scholar] [CrossRef]
- Makdoumi, K.; Goodrich, R.; Bäckman, A. Photochemical eradication of methicillin-resistant Staphylococcus aureus by blue light activation of riboflavin. Acta Ophthalmol. 2017, 95, 498–502. [Google Scholar] [CrossRef]
- Bauer, R.; Hoenes, K.; Meurle, T.; et al. The effects of violet and blue light irradiation on ESKAPE pathogens and human cells in presence of cell culture media. Sci. Rep. 2021, 11, 24473. [Google Scholar] [CrossRef] [PubMed]
- Rutala, W.A.; Kanamori, H.; Gergen, M.F.; et al. Antimicrobial activity of a continuous visible light disinfection system. Infect. Control Hosp. Epidemiol. 2018, 39, 1250–1253. [Google Scholar] [CrossRef]
- Maclean, M.; Anderson, J.G.; MacGregor, S.J.; et al. A New Proof of Concept in Bacterial Reduction: Antimicrobial Action of Violet-Blue Light (405 nm) in Ex Vivo Stored Plasma. J. Blood Transfus. 2016, 2016, 2920514. [Google Scholar] [CrossRef]
- Maclean, M.; McKenzie, K.; Anderson, J.G.; et al. 405 nm light technology for the inactivation of pathogens and its potential role for environmental disinfection and infection control. J. Hosp. Infect. 2014, 88, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Maclean, M.; MacGregor, S.J.; Anderson, J.G.; et al. High-intensity narrow-spectrum light inactivation and wavelength sensitivity of Staphylococcus aureus. FEMS Microbiol. Lett. 2008, 285, 227–232. [Google Scholar] [CrossRef] [PubMed]
- Tomb, R.M.; White, T.A.; Coia, J.E.; et al. Review of the Comparative Susceptibility of Microbial Species to Photoinactivation Using 380-480 nm Violet-Blue Light. Photochem. Photobiol. 2018, 94, 445–458. [Google Scholar] [CrossRef]
- Enwemeka, C.S.; Baker, T.L.; Bumah, V.V. The role of UV and blue light in photo-eradication of microorganisms. J. Photochem. Photobiol. 2021, 8, 100064. [Google Scholar] [CrossRef]
- Rullan, P.P. A Handheld Broadband UV Phototherapy Module for the Treatment of Patients With Psoriasis and Vitiligo. Cutis 2010, 86, 321–326. [Google Scholar]
- Robatto, M.; Pavie, M.C.; Garcia, I.; et al. Ultraviolet A/blue light-emitting diode therapy for vulvovaginal candidiasis: A case presentation. Lasers Med. Sci. 2019, 34, 1819–1827. [Google Scholar] [CrossRef]
- Rezaie, A.; Leite, G.G.S.; Melmed, G.Y.; et al. Ultraviolet A light effectively reduces bacteria and viruses including coronavirus. PLoS ONE 2020, 15, E0236199. [Google Scholar] [CrossRef]
- Rezaie, A.; Melmed, G.Y.; Leite, G.; et al. Endotracheal Application of Ultraviolet A Light in Critically Ill Patients with Severe Acute Respiratory Syndrome Coronavirus 2: A First-in-Human Study. Adv. Ther. 2021, 38, 4556–4568. [Google Scholar] [CrossRef] [PubMed]
- Anderson, R.R.; Parrish, J.A. The optics of human skin. J. Investig. Dermatol. 1981, 77, 13–19. [Google Scholar] [CrossRef]
- Gläser, R.; Navid, F.; Schuller, W.; et al. UV-B radiation induces the expression of antimicrobial peptides in human keratinocytes in vitro and in vivo. J. Allergy Clin. Immunol. 2009, 123, 1117–1123. [Google Scholar] [CrossRef]
- Mallbris, L.; Edström, D.W.; Sundblad, L.; et al. UVB Upregulates the Antimicrobial Protein hCAP18 mRNA in Human Skin. J. Investig. Dermatol. 2005, 125, 1072–1074. [Google Scholar] [CrossRef]
- Hong, S.P.; Kim, M.J.; Jung, M.; et al. Biopositive Effects of Low-Dose UVB on Epidermis: Coordinate Upregulation of Antimicrobial Peptides and Permeability Barrier Reinforcement. J. Investig. Dermatol. 2008, 128, 2880–2887. [Google Scholar] [CrossRef] [PubMed]
- T. Gambichler, R.U.o.B. Department of Dermatology, Gudrunstr. 56, D-44791 Bochum, Germany, M. Skrygan et al. Changes of antimicrobial peptide mRNA expression in atopic eczema following phototherapy. Br. J. Dermatol. 2024, 155, 1275–1278. [CrossRef]
- Vähävihu, K.; Ala-Houhala, M.; Peric, M.; et al. Narrowband ultraviolet B treatment improves vitamin D balance and alters antimicrobial peptide expression in skin lesions of psoriasis and atopic dermatitis. Br. J. Dermatol. 2010, 163, 321–328. [Google Scholar] [CrossRef]
- Dotterud, L.K.; Wilsgaard, T.; Vorland, L.H.; et al. The effect of UVB radiation on skin microbiota in patients with atopic dermatitis and healthy controls. Int. J. Circumpolar Health 2008, 67, 254–260. [Google Scholar] [CrossRef] [PubMed]
- Silva, S.; Guedes, A.; Gontijo, B.; et al. Influence of narrow-band UVB phototherapy on cutaneous microbiota of children with atopic dermatitis. J. Eur. Acad. Dermatol. Venereol. 2006, 20, 1114–1120. [Google Scholar] [CrossRef]
- Shimoda, H.; Matsuda, J.; Iwasaki, T.; et al. Efficacy of 265-nm ultraviolet light in inactivating infectious SARS-CoV-2. J. Photochem. Photobiol. 2021, 7, 100050. [Google Scholar] [CrossRef] [PubMed]
- Buonanno, M.; Stanislauskas, M.; Ponnaiya, B.; et al. 207-nm UV Light—A Promising Tool for Safe Low-Cost Reduction of Surgical Site Infections. II: In-Vivo Safety Studies. PLoS ONE 2016, 11, E0138418. [Google Scholar] [CrossRef]
- Dai, T.; Kharkwal, G.B.; Zhao, J.; et al. Ultraviolet-C Light for Treatment of Candida albicans Burn Infection in Mice. Photochem. Photobiol. 2011, 87, 342–349. [Google Scholar] [CrossRef] [PubMed]
- Dai, T.; Garcia, B.; Murray, C.K.; et al. UVC Light Prophylaxis for Cutaneous Wound Infections in Mice. Antimicrob. Agents Chemother. 2012. [Google Scholar] [CrossRef] [PubMed]
- Thai, T.P.; Houghton, P.E.; Campbell, K.E.; et al. Ultraviolet light C in the treatment of chronic wounds with MRSA: A case study. Ostomy/wound Manag. 2002, 48, 52–60. [Google Scholar]
- Ponnaiya, B.; Buonanno, M.; Welch, D.; et al. Far-UVC light prevents MRSA infection of superficial wounds in vivo. PLoS ONE 2018, 13, E0192053. [Google Scholar] [CrossRef]
- Glaab, J.; Lobo-Ploch, N.; Cho, H.K.; et al. Skin tolerant inactivation of multiresistant pathogens using far-UVC LEDs. Sci. Rep. 2021, 11, 1–11. [Google Scholar] [CrossRef]
- Zwicker, P.; Schleusener, J.; Lohan, S.B.; et al. Application of 233 nm far-UVC LEDs for eradication of MRSA and MSSA and risk assessment on skin models. Sci. Rep. 2022, 12, 1–15. [Google Scholar] [CrossRef]
- Schleusener, J.; Lohan, S.B.; Busch, L.; et al. Irradiation of human oral mucosa by 233 nm far UV-C LEDs for the safe inactivation of nosocomial pathogens. Sci. Rep. 2023, 13, 1–12. [Google Scholar] [CrossRef]
- Narita, K.; Asano, K.; Morimoto, Y.; et al. Disinfection and healing effects of 222-nm UVC light on methicillin-resistant Staphylococcus aureus infection in mouse wounds. J. Photochem. Photobiol. B Biol. 2018, 178, 10–18. [Google Scholar] [CrossRef]
- Fukui, T.; Niikura, T.; Oda, T.; et al. Exploratory clinical trial on the safety and bactericidal effect of 222-nm ultraviolet C irradiation in healthy humans. PLoS ONE 2020, 15, E0235948. [Google Scholar] [CrossRef] [PubMed]
- Buonanno, M.; Ponnaiya, B.; Welch, D.; et al. Germicidal Efficacy and Mammalian Skin Safety of 222-nm UV Light. Radiat. Res. 2017, 187, 493–501. [Google Scholar] [CrossRef] [PubMed]
| Detector modality | Measured signal | Biomarker class | Biological interpretation | Diagnostic role |
| Temperature sensor | Local temperature | Inflammatory biomarker | Local immune response, perfusion changes, infection onset | Early detection, trend monitoring |
| Optical imaging (camera) | Tissue morphology, turbidity, structure | Structural / morphological | Vascularization, biofilm presence, tissue alteration | Spatial assessment of infection environment |
| Spectral sensor | Absorption / scattering signatures | Microbial / metabolic | Bacterial load, metabolic activity, chromophore response | Quantification of infection state |
| Multimodal integration | Temporal signal correlation | Dynamic biomarker (trend) | Evolution of infection vs. physiological baseline | Predictive diagnostics, therapy guidance |
| IR (>780 nm) | ||||||
| IR 1064 nm |
~10 mW/cm2 | Endothelial cells | higher NO release, higher Akt & eNOS phosphorylation, mitochondrial involvement | In-vitro | [57] | |
| IR 980 nm |
~5–50 J/cm2 | Muscle tissue / inflammation | Anti-inflammatory effects, reduced oxidative stress, improved tissue regeneration | In-vivo | [29] | |
| IR 830 nm |
1–20 J/cm2 | Wound healing (fibroblasts, keratinocytes) | Enhanced proliferation, collagen synthesis, accelerated wound healing | In-vivo | [67] | |
| IR 808 nm |
~1–10 J/cm2, | Human fibroblasts / mitochondrial activity | Increased ATP production, enhanced proliferation, activation of cytochrome c oxidase | In-vitro | [69] | |
| VIS (380-780 nm) | ||||||
| VIS 470 nm 660 nm |
5 mW, 6 hours 284.90 J/cm2, 603.44 J/cm2 |
Inhibition of Staphylococcus aureus and Pseudomonas aeruginosa growth | Blue light, but not red light, can temporarily inhibit the growth of gram-negative and gram-positive bacteria. | In-vitro | [47] | |
| VIS 420 nm 635 nm |
Twice daily 72 J/cm2 43.8, 87.6, and 175.5 J/cm2 |
Candida albicans biofilm matrix development | Affects biofilm development and physiology of polysaccharide production | In-vitro | [70] | |
| VIS blue only (400-500 nm) | ||||||
| VIS 470 nm |
16.29, 27.16 and 54.32 J/cm2 |
Staphylococcus aureus growth delay | Inhibitory effect but not absolute antibacterial | In-vitro | [48] | |
| VIS 450-470 nm |
60–240 J/cm2 | Candida albicans elimination | 460 nm eradicates planktonic and biofilm Candida albicans | In-vitro, In-vivo Animal |
[71] | |
| VIS 450 nm |
With riboflavin: 15, 30, 56, and 84 J/cm2 |
Effects on MRSA and human keratinocytes | ● High doses blue light could treat infections without harming skin cells, ● PDT with riboflavin is promising |
In-vitro | [72] | |
| VIS 412 nm, 450 nm |
With riboflavin 5.4 J/cm2 and 28.5 J/cm2 |
MRSA eradication | Riboflavin enhances the antibacterial effect of blue light | In-vitro | [73] | |
| VIS 405 nm, 450 nm |
300 J/cm2 400 J/cm2 (1000 J/cm2) |
Effect on ESKAPE pathogens and human cells | Significant reduction in viable bacteria | In-vitro | [74] | |
| VIS 405 nm ± 3 nm |
Continuous visible light over 72 hours 0.34–0.44 mW/cm2 |
Antimicrobial activity against ● C. difficile ● MRSA ● VRE ● MDR Acinetobacter baumannii |
● The high irradiant light significantly reduced both vegetative bacteria and spores ● Over a 72-hour exposure period. |
In-vitro | [75] | |
| VIS 405 nm |
144 J/cm2 |
Antimicrobial action in stored plasma | ● Decontaminates pre-bagged plasma without photosensitizing agents ● 99.9% reduction of low-density bacterial populations ● 405 nm light application provides new proof of concept for bacterial reduction in biological fluids |
In-vitro | [76] | |
| VIS 405 nm |
● Overview of the mechanisms of 405 nm for environmental decontamination ● Different energy densities |
Pathogen inactivation Environmental disinfection |
● Significant antimicrobial properties for environmental disinfection ● Even in crowded working areas |
In-vitro | [77] | |
| VIS 400-420 nm |
350–400 mW/cm2 | Staphylococcus aureus inactivation | Maximum inactivation at 405±5 nm | In-vitro | [78] | |
| VIS 380-480 nm |
Up to >1500 J/cm2 | Review of the last thirty years Microbial species susceptibility |
Higher doses needed for longer wavelengths | In-vitro | [79] | |
| UVA (380–315 nm) | ||||||
| UV-A, B, C, blue light pulsed blue light (PBL) |
Different UV doses and PBL |
Overview of various methods for photo eradication of microorganisms (SARS-CoV-2) | ● UV-C is highly effective, followed by UV-B, UV-A, ● UV-C minimal depth penetration in humans ● The damaging effect of UV was more significant with UV-B than with UV-C ● High potency of PBL at each of the violet-blue wavelengths makes it significantly more germicidal at lower irradiances and fluences than CW light |
In-vitro, In-vivo Clinical & animal |
[80] | |
| UVA: 343-375 nm UVB: 280-313 nm UVC: 222-254 nm |
Various applications and energy densities | Comparison and overview of possible wavelength ranges and energy densities for in vivo disinfection | ● UV irradiation shows a significant potential of being an effective therapy method for infectious diseases. ● UV irradiation in different wavelength bands can inactivate microbes based on different mechanisms |
In-vivo Clinical & animal |
[52] | |
| UV-A, B 300-380 nm |
Depending on Fitzpatrick skin types Up to 5000 mJ/cm2 |
Psoriasis and Vitiligo treatment | ● Effective for treating dermatoses | In-vivo Clinical |
[81] | |
| UVA peak at 400 nm |
3 mW/cm2, 30 + 30 min | Vulvovaginal candidiasis | Patients with vulvovaginal candidiasis present a reduction of inflammation and disappearance of symptoms of pruritus and burning after a treatment. | In-vivo Clinical |
[82] | |
| UVA peak at 343 nm |
2.0 mW/cm2, 30 min, or 3.0–3.5 mW/cm2, 20 min daily for 2 d |
● Multiple bacteria, yeast, and viruses (in vitro) ● Safety study (in vivo) |
● UVA exposure demonstrates effective inactivation of various bacteria, yeast, and viruses ● Single and repeated UVA irradiation is safe for mice based on the endoscopic examination and full-thickness pathologic assessment results |
In-vivo Animal |
[83] | |
| UVA peak at 343 nm |
2 mW/cm2, 20 min daily for 5 d | SARS-CoV-2 infection | ● Four out of five subjects clinically improved on day 30 after the treatment. ● Endotracheal UVA phototherapy under specific conditions was safe for humans with a significant reduction in respiratory SARS-CoV-2 viral load |
In-vivo Clinical |
[84] | |
| UVB (315–280 nm) | ||||||
| UVB 300 nm |
300 nm | Epidermal absorbers | Major absorbers include aromatic amino acids, nucleic acids, urocanic acid, and melanin. | In-vivo Clinical |
[85] | |
| UVB peak at 313 nm |
250–1500 J/cm2 | AMP expression | ● Expression of AMPs were detected on all subjects ● hBD-2 expression was detected only on one subject |
In-vivo Clinical |
[86] | |
| UVB 280-315 nm |
2.3 mW/cm2 | AMP expression | hCAP18 was found to be expressed significantly in the human skin | In-vivo Clinical |
[87] | |
| UVB peak at 313 nm |
40 mJ/cm2 daily for 1 or 3 d | AMP expression | Expression of mBD-3, cathelin-related antimicrobial peptide, and LL-37 were observed | In-vivo Animal |
[88] | |
| UVB peak at 311 nm |
Up to 1.6 J/cm2 | Atopic eczema | UVB irradiation can modulate the expression of hBD-1 and hBD-2 for atopic eczema | In-vivo Clinical |
[89] | |
| UVB peak at 311 nm |
0.13–8.88 J/cm2 | Atopic dermatitis and psoriasis | Enhanced expression of LL-37 and decreased expression of hBD-2 after UVB treatment, significant improvement of psoriasis and atopic dermatitis | In-vivo Clinical |
[90] | |
| UVB peak at 306 nm |
1.68 J/cm2 | Atopic dermatitis with S. aureus and S. epidermidis | ● The UVB irradiation shows an excellent antimicrobial effect against S. aureus, ● But no effect on S. epidermidis |
In-vivo Clinical |
[91] | |
| UVB 4.3 J accumulated |
4.3 J accumulated | Atopic dermatitis with S. aureus | UVB irradiation demonstrates a significant suppression of superantigen production from S. aureus in patients with atopic dermatitis | In-vivo Clinical |
[92] | |
| UVC (280–100 nm) | ||||||
| UVC 254 nm |
30 minutes after the inoculation 3.24 J/cm2 for abrasions and 2.59 J/cm2 for burns |
Acinetobacter baumannii wound infections | ● DNA lesions were observed immediately after a UVC exposure ● The lesions were extensively repaired within 72 hours. |
In-vivo Animal |
[49] | |
| UVC 254 nm 265 nm 280 nm |
4, 8,11,14 mJ/cm2 1,3, 2,5 3,8, 5,1 mJ/cm2 15,30,45,60 mJ/cm2 |
SARS-CoV-2 inactivation via different sources | ● 265-nm deep UV light-emitting diode (DUV-LED) lamp efficiently inactivated SARS-CoV-2 ● At a similar level as a 254-nm ● And at a higher level than a 280-nm |
In-vitro | [93] | |
| UVC 207 nm |
157 mJ/cm2 | ● infection reduction ● Safety study |
Potential use for antimicrobial properties without hazards to skin | In-vivo Animal |
[94] | |
| UVC peak at 254 nm |
2.7 mW/cm2, 18 min on day 0, or 40 min on day 1 |
● C. albicans burn infection in mice ● Safety study |
● Exposure on day 0 gives a 99.2% reduction of fungal burden, exposure on day 1 gives a 95.8% reduction of fungal burden ● CPD were observed by immunofluorescence in normal mouse skin immediately after UV irradiation, and the damage was extensively repaired within 24 h |
In-vivo Animal |
[95] | |
| UVC peak at 254 nm |
2.7 mW/cm2, 16 min | ● P. aeruginosa and S. aureus cutaneous wound infections in mice ● Safety study |
● A 10-fold reduction of the P. aeruginosa and S. aureus burden on mice skin ● CPD-positive nuclei were observed in the immunofluorescence micrograph of mouse skin, and the damage was extensively repaired within 48 h |
In-vivo Animal |
[96] | |
| UVC peak at 254 nm |
180 s; 7 treatments over 14 d + 4 treatments over 1 month | MRSA-infected chronic ulcer on human | ● UVC irradiation reduced wound bacterial burden and facilitated wound healing for all three patients ● Two patients had complete wound closure following 1 week of UVC irradiation |
In-vivo Clinical |
[97] | |
| UVC peak at 222 nm & peak at 254 nm |
40 mJ/cm2 and 300 mJ/cm2 | ● MRSA infected superficial skin wound of mice ● Safety study |
● Both 222 and 254 nm irradiation show a statistically significant reduction of bacteria counts on days 2 and 7 ● 222 nm light showed the same bactericidal properties of 254 nm light but without the associated skin damage (CPD formation) |
In-vivo Animal |
[98] | |
| UVC peak at 233 nm |
44 µW/cm2 15–40 mJ/cm2 |
● A skin tolerant far-UVC (< 240 nm) irradiation system ● Inactivation of MRSA |
● Porcine skin showed only 3.7% CPD and 2.3% 6-4PP DNA damage ● Corresponding irradiation at 254 nm caused 11–14 times higher damage ● skin damage is so small that it can be expected to be compensated by natural repair mechanisms |
In-vitro | [99] | |
| UVC peak at 233 nm |
40 mJ/cm2 | ● Eradication of MRSA, MSSA, SE and risk assessment on skin | ● 5 log10 levels reduction without no soil load ● 1.5–3.3 with soil load ● DNA damage was far below the damage evoked by 0.1 UVB minimal erythema dose ● far lower than 20 min outdoor visible light |
In-vitro | [100] | |
| UVC peak at 233 nm |
60 mJ/cm2 | ● Safe inactivation of multi-resistant nosocomial pathogens | ● reduced by ≈ 5 log10 for 60 mJ/cm2 ● DNA damage occurred only superficially and decreased after 24 h ● < 10% of keratinocytes were affected |
In-vitro | [101] | |
| UVC peak at 222 nm |
75, 150, and 450 mJ/cm2 | ● MRSA-infected skin wound of mice ● Safety study |
● 222 nm irradiation shows a significant bactericidal effect, which was equal to or more effective than 254 nm irradiation ● CPD-expressing cells were found in both epidermis and dermis with 254 nm |
In-vivo Animal |
[102] | |
| UVC peak at 222 nm |
50–500 mJ/cm2 | ● Bactericidal effect on healthy human skin ● Safety study |
● The number of bacterial colonies in the skin swab culture was reduced significantly ● The CPD amount produced in the irradiated region was slightly but significantly higher than that of the non-irradiated region |
In-vivo Clinical |
[103] | |
| UVC peak at 222 nm & peak at 254 nm |
157 mJ/cm2, delivered in 7 h | ● MRSA (in vitro) ● Safety study (in vivo) |
● Both 254 and 222 nm irradiation can kill MRSA efficiently ● ,Unlike 254 nm, 222 nm UVC irradiation is safe for mouse skin, and no detectable formation of mutagenic CPDs was observed |
In-vivo Animal |
[104] | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




