Submitted:
27 April 2026
Posted:
27 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
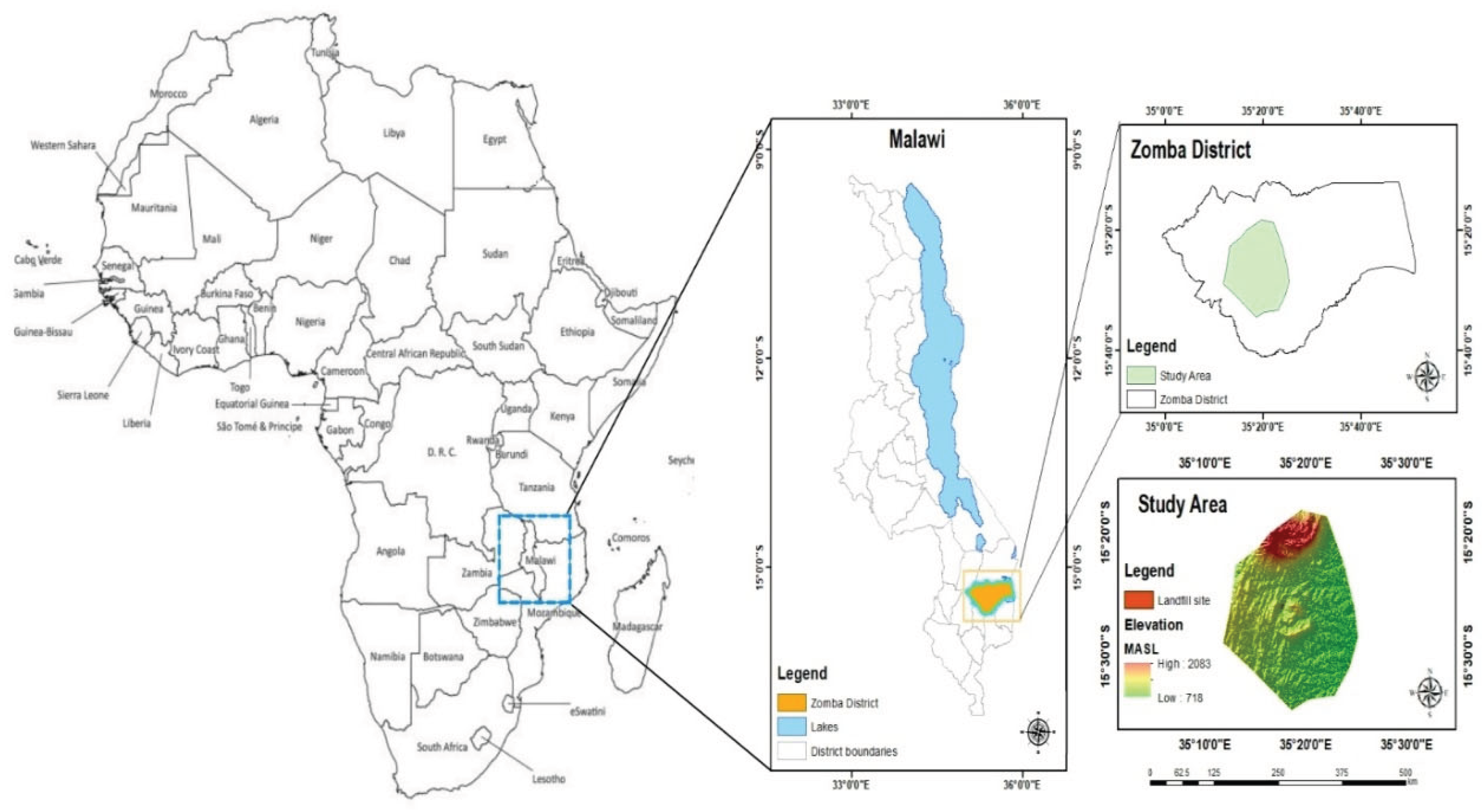
2.1. Description of the Study Area
2.2. Climate
2.3. Collection and Preservation of Water and Soil Samples
2.4. Physicochemical Analysis of Water Samples
2.5. Microbial Analysis of Water Samples
2.5.1. Determination of Total Coliform and E. coli
2.5.2. Preparation of Lauryl Sulphate Broth
2.5.3. Procedure for Microbial Analysis
2.5.3.1. Sample Serial Dilutions and Pour Plating Procedures
2.5.4. Determination of Salmonella
2.5.4.1. Media Preparation
2.5.4.2. Sample preparation
2.5.5. Determination of Vibrio cholerae
2.5.5.1. Sample Preparation
2.5.6. Observation
2.6. Preparation and Determination of Heavy Metals in the Soil Samples
2.7. Data Analysis
2.9. Human Health Risk Assessment of Drinking Water
2.9.1. Chronic Daily Intake
2.9.2. Hazard Quotient
2.9.3. Hazard Index
2.9.4. Carcinogenic Risk
3. Results
3.1. Physicochemical Parameters of Water Samples
3.2. Chemical Parameters of Water Quality
3.3. Microbial Characteristics of the Water Samples
3.4. Chemical Characteristics of the Soil Samples
4. Discussion
4.1. Physicochemical Parameters of Water Samples
4.2. Chemical Parameters of Water Quality
4.3. Microbial Characteristics of the Water Samples
4.4. Pearson Correlation Analysis of Physicochemical and Microbial Parameters of Water Samples
| pH | Temp | TDS | EC | Cr | Cd | Cu | Coliform | E. coli | Shigella | S. aureus | V. cholera | |
| pH | 1 | |||||||||||
| Temp | .638** | 1 | ||||||||||
| TDS | .723** | .938** | 1 | |||||||||
| EC | .717** | .936** | 1.000** | 1 | ||||||||
| Cr | .718** | .966** | .970** | .968** | 1 | |||||||
| Cd | 0.374 | 0.306 | 0.244 | 0.245 | 0.244 | 1 | ||||||
| Cu | .599** | .923** | .832** | .830** | .921** | 0.342 | 1 | |||||
| Coliform | .837** | .843** | .933** | .929** | .930** | 0.110 | .771** | 1 | ||||
| E. coli | .714** | .829** | .887** | .882** | .907** | 0.043 | .748** | .968** | 1 | |||
| Shigella | .805** | .938** | .974** | .972** | .978** | 0.251 | .859** | .971** | .937** | 1 | ||
| S. aureus | .732** | .820** | .909** | .908** | .924** | 0.047 | .819** | .944** | .902** | .910** | 1 | |
| V. cholerae | .761** | .934** | .987** | .985** | .972** | 0.215 | .823** | .964** | .936** | .993** | .903** | 1 |
| pH | Temp | TDS | EC | Cr | Cd | Cu | Coliform | E. coli | Salmonella | Shigella | S. aureus | V. cholera | ||
| pH | 1 | |||||||||||||
| Temp | .565* | 1 | ||||||||||||
| TDS | .613** | 0.073 | 1 | |||||||||||
| EC | .619** | 0.091 | 1.000** | 1 | ||||||||||
| Cr | .316 | 0.196 | -0.418 | 0.415 | 1 | |||||||||
| Cd | .344 | .840** | -0.024 | 0.005 | 0.287 | 1 | ||||||||
| Cu | .608** | .899** | -0.093 | 0.079 | .565* | .835** | 1 | |||||||
| Coliform | .743** | .886** | 0.346 | 0.365 | 0.145 | .705** | .771** | 1 | ||||||
| E. coli | .405 | .770** | 0.030 | 0.049 | 0.017 | .500* | .578* | .852** | 1 | |||||
| Salmo | .747** | .881** | 0.352 | 0.370 | 0.140 | .715** | .778** | .975** | .821* | 1 | ||||
| Shigella | .742** | .890** | 0.330 | 0.349 | 0.151 | .716** | .788** | .980** | .837* | .999** | 1 | |||
| S. aureus | .761** | .918** | 0.227 | 0.244 | 0.276 | .712** | .863** | .970** | .837* | .975** | .981** | 1 | ||
| V. cholerae | .803** | .877** | 0.338 | 0.357 | 0.251 | .691** | .806** | .984** | .813* | .982** | .985** | .983** | 1 | |
| *. Correlation is significant at the 0.05 level (2-tailed). | ||||||||||||||
| **. Correlation is significant at the 0.01 level (2-tailed). | ||||||||||||||
4.5. Chemical Characteristics of the Soil Samples
4.6. Microbial Characteristics of the Soil Samples
4.7. Pearson Correlation Analysis of the Chemical and Microbial Parameters of the Soil Samples
4.8. Suitability of Water for Human and Agricultural Use
4.8.1. Human Health Risk Assessment
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data availability statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ADD AT As CDI Cd CR Cr ED EF HI HHR RfD IR HQ BW MS MSW WHO |
Average Daily Dose Average Time Arsenic Chronic Daily Intake Cadmium Carcinogenic Risk Chromium Duration of Exposure Exposure Frequency Hazard Index Human Health Risk Reference Dosage Rate at which a person drinks water Hazard Quotient Body Weight Malawi Standard Municipal Solid Waste World Health Organization |
References
- M. Sarma Bora et al., Pollution and health risk assessment of toxic metals in solid waste dumping site soil and its impact on groundwater: a case study, Int. J. Environ. Anal. Chem., vol. 103, no. 3, pp. 612–632, Feb. 2023. [CrossRef]
- P. Zhang et al., Water Quality Degradation Due to Heavy Metal Contamination: Health Impacts and Eco-Friendly Approaches for Heavy Metal Remediation, Toxics, vol. 11, no. 10, Sep. 2023. [CrossRef]
- Z. Kazemi, M. Hesami Arani, M. Panahande, M. Kermani, and Z. Kazemi, Chemical quality assessment and health risk of heavy metals in groundwater sources around Saravan landfill, the northernmost province of Iran, Int. J. Environ. Anal. Chem., vol. 103, no. 18, pp. 6577–6595, Dec. 2023. [CrossRef]
- R. S. Malikula, C. C. Kaonga, H. W. Mapoma, F. G. Thulu, and P. Chiipa, Heavy metals and nutrients loads in water, soil, and crops irrigated with effluent from WWTPs in Blantyre City, Malawi, Water, vol. 14, no. 1, p. 121, 2022.
- S. W. Rashid, D. M. S. Shwan, and K. A. Rashid, Physicochemical Characterization and Evaluation of Seasonal Variations of Landfill Leachate and Groundwater Quality around Tanjaro Open Dump Area of Sulaymaniyah City, Kurdistan, Iraq, J. Chem., vol. 2022, no. 1, p. 8574935, 2022. [CrossRef]
- L. F. M. Rebellon, Waste Management: An Integrated Vision. BoD – Books on Demand, 2012.
- M. Sarma Bora et al., Pollution and health risk assessment of toxic metals in solid waste dumping site soil and its impact on groundwater: a case study, Int. J. Environ. Anal. Chem., vol. 103, no. 3, pp. 612–632, Feb. 2023. [CrossRef]
- Uzoigwe, C. I., & Agwa, O. K. (2012). Microbiological quality of water collected from boreholes sited near refuse dumpsites in Port Harcourt, Nigeria. African Journal of Biotechnology, 11(13), 3135–3139. [CrossRef]
- F. Afolabi, G. C. Adeyinka, O. O. Ajibola, and B. F. Bakare, Comparative assessment of heavy metal contamination of abandoned and active dumpsite of Osun waste management, Ejigbo Road, Osogbo, Osun State, Nigeria, Int. J. Environ. Anal. Chem., vol. 103, no. 20, pp. 8739–8755, Dec. 2023. [CrossRef]
- R. S. Malikula, C. C. Kaonga, H. W. T. Mapoma, F. G. D. Thulu, and P. Chiipa, Heavy Metals and Nutrients Loads in Water, Soil, and Crops Irrigated with Effluent from WWTPs in Blantyre City, Malawi, Water, vol. 14, no. 1, Jan. 2022. [CrossRef]
- N. Mherzi, F. Lamchouri, M. Y. Lakhloufi, A. Zalaghi, and H. Toufik, Biological treatment of leachate from the uncontrolled landfill: evaluation of toxicity and heavy metals reduction, Chem. Ecol., vol. 39, no. 5, pp. 459–483, May 2023. [CrossRef]
- M. Sarma Bora et al., Pollution and health risk assessment of toxic metals in solid waste dumping site soil and its impact on groundwater: a case study, Int. J. Environ. Anal. Chem., vol. 103, no. 3, pp. 612–632, Feb. 2023. [CrossRef]
- Z. Kazemi, M. Hesami Arani, M. Panahande, M. Kermani, and Z. Kazemi, Chemical quality assessment and health risk of heavy metals in groundwater sources around Saravan landfill, the northernmost province of Iran, Int. J. Environ. Anal. Chem., vol. 103, no. 18, pp. 6577–6595, Dec. 2023. [CrossRef]
- A. A. Ameloko and E. A. Ayolabi, Geophysical assessment for vertical leachate migration profile and physicochemical study of groundwater around the Olusosun dumpsite Lagos, south-west Nigeria, Appl. Water Sci., vol. 8, no. 5, p. 142, Sep. 2018. [CrossRef]
- Orvestedt, S. (2015). Waste management and impact on people’s health when cultivating on sites contaminated with heavy metals-Minor field study made in Zomba, Malawi. http://stud.epsilon.slu.se. Available: https://www.google.com/search?q [Accessed: Feb. 22, 2026].
- APHA_Standard Methods for the Examination of Water and Wastewater (1).
- US Environmental Protection Agency (2002) Method 1604. Total Coliforms and Escherichia coli in Water by Membrane Filtration Using a Simultaneous Detection Technique (MI Medium). Rept. Mo. EPA 821-R-02-024, Environmental Protection Agency, Washington, D.C. Available: https://www.scirp.org/reference/referencespapers?referenceid=1418604 [Accessed: Feb. 28, 2026].
- United States Environmental Protection Agency (USEPA) (2011). Exposure Factors Handbook, Final ed., (EPA/600/R-09/052F). Washington, D.C., U.S. Environmental Protection Agency. Available: https://www.scirp.org/reference/referencespapers?referenceid=2736489 [Accessed: Feb. 28, 2026].
- J. N. Onwualu-John and M. U. Uzoegbu, Physicochemical Characteristics and Heavy Metals Level in Groundwater and Leachate around Solid Waste Dumpsite at Mbodo, Rivers State, Nigeria., J. Appl. Sci. Environ. Manag., vol. 26, no. 12, p. 2107, Dec. 2022. [CrossRef]
- R. Benaddi, Y. Ferkan, A. Bouriqi, and N. Ouazzani, Impact of landfill leachate on groundwater quality–a comparison between three different landfills in Morocco, J. Ecol. Eng., vol. 23, no. 11, pp. 89–94, 2022.
- D. Abiriga, L. S. Vestgarden, and H. Klempe, “Groundwater contamination from a municipal landfill: Effect of age, landfill closure, and season on groundwater chemistry, Sci. Total Environ., vol. 737, p. 140307, 2020.
- D. de C. S. Bacha et al., Evaluation of the contamination of the soil and water of an open dump in the Amazon region, Brazil.,” 2021, Accessed: Feb. 28, 2026. [Online]. Available: https://www.cabidigitallibrary.org/doi/full/10.5555/20210318608.
- H. Kada, A. Demdoum, F. Baali, H. Aouati, and H. D. Eddine, Heavy metal contamination and exposure risk assessment via drinking groundwater in Ain Azel territory, north-eastern Algeria, Sustain. Water Resour. Manag., vol. 8, no. 5, p. 163, Oct. 2022. [CrossRef]
- Vaccari, M., Tudor, T., & Vinti, G. (2019). Characteristics of leachate from landfills and dumpsites in Asia, Africa and Latin America: an overview. Waste Management, 95, 416–431. [CrossRef]
- G. Badmus, Y. Ajiboye, O. Ogungbemi, A. Jita, and O. Adenuga, Assessment of groundwater quality, irrigation suitability, and health risks from radon and heavy metals near Ilokun dumpsite, Ado-Ekiti, Nigeria, J. Water Health, vol. 22, pp. 1725–1742, Sep. 2024. [CrossRef]
- Sankoh, A. A., Amara, J., Komba, T., Laar, C., Sesay, A., Derkyi, N. S., & Frazer-williams, R. (2023). Seasonal assessment of heavy metal contamination of groundwater in two major dumpsites in Sierra Leone. Cogent Engineering, 10(1). Available: https://www.google.com/search?q [Feb. 28, 2026]. [CrossRef]
- Javahershenas, M., Nabizadeh, R., Alimohammadi, M., & Mahvi, A. H. (2022). The effects of Lahijan landfill leachate on the quality of surface and groundwater resources. International Journal of Environmental Analytical Chemistry, 102(2), 558–574. [CrossRef]
- E. Vunain, C. Nkhuzenje, J. Mwatseteza, and S. Sajidu, Groundwater quality assessment from Phalombe Plain, Malawi, 2019.
- H. A. Emmanuel-Akerele and F. I. Peter, Microbial and physico-chemical assessment of soil and water around waste dump sites in Lagos, Int. J. Appl. Biol., vol. 5, no. 1, pp. 37–82, 2021.
- A. Kashtanjeva, I. Vehapi, K. Kurteshi, and M. Paçarizi, Determining the Physico-Chemical and Microbiological Parameters of the Water Quality in the Batllava Lake, Kosovo, J. Ecol. Eng., vol. 23, no. 9, 2022. https://yadda.icm.edu.pl/baztech/element/bwmeta1 [Accessed: Feb. 28, 2026].
- N. Khatoon, A. H. Khan, M. Rehman, and V. Pathak, Correlation study for the assessment of water quality and its parameters of Ganga River, Kanpur, Uttar Pradesh, India, IOSR J. Appl. Chem., vol. 5, no. 3, pp. 80–90, 2013.
- R. Saxena et al., Correlation coefficient for physico-chemical parameters to assess the quality of tannery effluents at Kanpur. 2024. Available: http://nanobioletters.com/wp-content/uploads/2024/06/LIANBS131.047.pdf [Accessed: Feb. 28, 2026].
- Y. El Mourabit, A. Assabbane, and M. Hamdani, Study of correlations between microbiological and physicochemical parameters of drinking water quality in El kolea city (Agadir, Morocco): Using multivariate statistical methods, J Mater Env. Sci, vol. 11, no. 2, pp. 310–317, 2020.
- M. N. Ifeoma, N. Nwawuike, F. N. Chidimma, A. O. Okereke, U. I. Asomugha, E. E. Eches, Spatial Assessment of Some Selected Heavy Metals and Soil Physicochemical Properties in Open Dumpsites in Owerri North Nigeria., African Journal of Environment and Natural Science Research (AJENSR). Available: https://abjournals.org/ajensr/papers/volume [Accessed: Feb. 28, 2026].
- N. S. Soumahoro, N. L. B. Kouassi, K. M. Yao, E. K. Kwa-Koffi, A. M. Kouassi, and A. Trokourey, Impact of municipal solid waste dumpsites on trace metal contamination levels in the surrounding area: a case study in West Africa, Abidjan, Cote d’Ivoire, Environ. Sci. Pollut. Res., vol. 28, no. 24, pp. 30425–30435, Jun. 2021. [CrossRef]
- Y. Pu et al., Pollution Characteristics and Risk Assessment of Heavy Metals in the Soil of a Municipal Solid Waste Landfill Site, Soil Sediment Contam. Int. J., vol. 34, no. 6, pp. 1116–1133, Aug. 2025. [CrossRef]
- F. Afolabi, G. C. Adeyinka, O. O. Ajibola, and B. F. Bakare, Comparative assessment of heavy metal contamination of abandoned and active dumpsite of Osun waste management, Ejigbo Road, Osogbo, Osun State, Nigeria, Int. J. Environ. Anal. Chem., vol. 103, no. 20, pp. 8739–8755, Dec. 2023. [CrossRef]
- E. O. Igwe, C. O. Ede, P. N. Nnabo, and S. N. Ukpai, “Impact of heavy metals dispersion on water supplies around Oshiri and Ishiagu mine districts of Southern Benue Trough, Nigeria,” Model. Earth Syst. Environ., vol. 7, no. 3, pp. 2015–2030, Sep. 2021. [CrossRef]
- G. D. Dzinjalamala et al., Human health risk assessment of microbial contamination and trace metals in water and soils of Chileka Township, Blantyre, Malawi, Discov. Environ., vol. 2, no. 1, p. 62, Jun. 2024. [CrossRef]
- I. A. Simon-Oke, O. O. Oladele, and O. E. Dada, Microorganisms in Soil and Groundwater of Epe and Laje Solid Waste Dumpsites in Ondo Town, Nigeria., J. Appl. Sci. Environ. Manag., vol. 27, no. 2, 2023, Available: https://search.ebscohost.com/login.aspx?direct [Accessed: Feb. 28, 2026].
- A. Arthur and D. Okae-Anti, Variations in Soil Physico-Chemical Properties as Influenced by Landuse in a Toposequence, J. Geosci. Environ. Prot., vol. 10, no. 8, pp. 98–121, 2022.
- D.-H. Jeong, Y.-B. Yun, H.-J. Son, Y. Um, J.-H. Song, and J. Kim, Correlation analysis of soil microbial communities and physicochemical properties with growth characteristics of Sageretia thea across different habitats, Plants, vol. 13, no. 23, p. 3310, 2024.
- P. Heepngoen, A. Thoumazeau, M.-S. Renevier, K. Sajjaphan, F. Gay, and A. Brauman, Relationships between physico-chemical, biological and functional approaches for soil quality assessment. A case study along a gradient of disturbance, Eur. J. Soil Biol., vol. 104, p. 103300, 2021.
- A. Ghosh, S. Kumar, and J. Das, Impact of leachate and landfill gas on the ecosystem and health: Research trends and the way forward towards sustainability, J. Environ. Manage., vol. 336, p. 117708, 2023.
- H. T. Abadi, T. Alemayehu, and B. A. Berhe, Heavy metal’s pollution health risk assessment and source appraisal of groundwater and surface water in Irob catchment, Tigray, Northern Ethiopia, Appl. Water Sci., vol. 14, no. 9, p. 201, Aug. 2024. [CrossRef]
- M. Rahman et al., Groundwater contamination and health risk evaluation of naturally occurring potential toxic metals of Hatiya Island, Bangladesh, J. Ecol. Eng., vol. 23, no. 6, 2022, Available: https://yadda.icm.edu.pl/baztech/element/bwmeta1. [Accessed: Feb. 28, 2026].
| Soil sampling point | Calculated distance from the dumpsite | Borehole and well | Calculated distance from the dumpsite |
| Sampling point 1 | At the centre of the dumpsite | Well 1 | Within the dumpsite (6 m from the north east corner of the dumpsite |
| Sampling point 2 | 50 m | Well 2 | 120 m |
| Sampling point 3 | 150 m | Borehole 1 | 140 m |
| Sampling point 4 | 300 m | Well 3 | 170 m |
| Sampling point 5 | 2200 m (control sampling point) | Well 4 | 260 m |
| Borehole 2 | 2200 m (control water sampling point) |
| Sample ID | Wet Season | Dry Season | ||||||||||
| Temperature (°C) |
Ph | TDS (mg/L) |
EC (µs/cm) |
Temperature (°C) |
pH | TDS (mg/L) |
EC (µs/cm) |
|||||
| WI | 28.60 ± 0.10a | 7.25 ± 0.02a | 1366.33 ± 2.08a | 1965.00 ± 3.00a | 28.20 ±0.24a | 7.29 ± 0.03a | 155.13 ± 0.01c | 311.13 ± 0.00b | ||||
| W2 | 25.20 ± 0.52b | 6.33 ± 0.02c | 46.16 ± 0.04f | 74.41 ± 0.61f | 25.43b ± 0.59 | 6.38 ± 0.02c | 47.73 ± 0.02f | 93.23 ± 0.06f | ||||
| W3 | 25.23 ± 0.12b | 5.75 ± 0.01d | 247.80 ± 0.30b | 385.70 ± 1.15b | 25.40 ± 0.10b | 5.63 ± 0.08d | 81.81 ± 0.02d | 164.61 ± 0.01d | ||||
| W4 | 23.83 ± 0.06d | 6.59 ± 0.05b | 66.44 ± 0.05e | 107.70 ± 0.10e | 23.70 ± 0.10d | 6.56 ± 0.06b | 156.32 ± 0.02b | 310.13 ± 0.06c | ||||
| B1 | 24.70 ± 0.26c | 5.57 ± 0.03e | 109.40 ± 0.20d | 176.07 ± 0.10d | 24.70 ± 0.26c | 5.55 ± 0.04d | 58.80 ± 0.01e | 118.13 ± 0.06e | ||||
| B2 | 24.67 ± 0.06c | 6.28 ± 0.01c | 197.37 ± 0.46c | 311.10 ± 0.10c | 24.70 ± 0.10c | 6.36 ± 0.1c | 181.27 ± 0.07a | 357.32 ± 0.03a | ||||
| WHO MS |
- - |
6.5 - 8.5 6 - 9.5 |
1000 2000 |
1000 3500 |
- - |
6.5 - 8.5 6 - 9.5 |
1000 2000 |
1000 3500 |
||||
| Wet Season | Dry Season | |||||||||
| ID | As (mg/L) |
Cd (mg/L) |
Cr (mg/L) |
Cu (mg/L) |
Pb (mg/L) |
As (mg/L) |
Cd (mg/L) |
Cr (mg/L) |
Cu (mg/L) |
Pb (mg/L) |
| WI | BDL | 0.013 ± 0.00a | 0.41 ± 0.03a | 0.24 ± 0.01a | BDL | BDL | 0.08 ± 0.00a | 0.08 ± 0.00a | 0.19 ± 0.00a | BDL |
| W2 | BDL | 0.014 ± 0.00a | 0.13 ± 0.01b | 0.12 ± 0.01b | BDL | BDL | 0.02 ± 0.00c | 0.02 ± 0.00c | 0.12 ± 0.01b | BDL |
| W3 | BDL | 0.012 ± 0.00a | 0.14 ± 0.00b | 0.07 ± 0.00c | BDL | BDL | 0.06 ± 0.00b | 0.06 ± 0.00b | 0.07 ± 0.00c | BDL |
| W4 | BDL | 0.012 ± 0.00a | 0.08 ± 0.00c | BDL | BDL | BDL | BDL | BDL | BDL | BDL |
| B1 | BDL | 0.011 ± 0.00a | 0.08 ± 0.00c | BDL | BDL | BDL | BDL | BDL | BDL | BDL |
| B2 WHO MS |
BDL 0.01 0.01 |
0.014 ± 0.02a 0.03 0.01 |
0.07 ± 0.00c 0.003 - |
BDL 2.0 2.0 |
BDL 0.05 0.01 |
BDL 0.01 0.01 |
BDL 0.03 0.01 |
BDL 0.003 - |
BDL 2.0 2.0 |
BDL 0.05 0.01 |
| Wet | Season | Dry | Season | |||||||||||
| Total Coliform CFU/100 mL |
E. coli CFU/100 mL |
Salmonella CFU/100 mL |
Shigella CFU/100 mL |
S. aureus CFU/100 mL |
V. cholerae CFU/100 mL |
Total Coliform CFU/100 mL |
E. coli CFU/100 mL |
Salmonella CFU/100 mL |
Shigella CFU/100 mL |
S aureus CFU/100 mL |
V cholerae CFU/100 mL |
|||
| WI | 20900 | 3300 | 0 | TNTC | 2500 | 5900 | TNTC | 3400 | 400 | TNTNC | TNTC | 12300 | ||
| W2 | 1700 | 700 | 0 | 900 | 200 | 0 | 1000 | 300 | 0 | 1100 | TNTC | 1300 | ||
| W3 | 1300 | 500 | 0 | 200 | 900 | 0 | 1900 | 100 | 0 | 200 | 0 | 0 | ||
| W4 | 6000 | 1100 | 0 | 400 | 800 | 0 | 5700 | 0 | 0 | 0 | 0 | 1300 | ||
| B1 | 1400 | 1000 | 0 | 0 | 100 | 200 | 9800 | 2000 | 0 | 1200 | 0 | 200 | ||
| B2 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||
| MS | <1 | <1 | <10 | <1 | <1 | <1 | <1 | <1 | <10 | <1 | <1 | <1 | ||
| Wet Season | Dry Season | |||||||||
| ID | As (mg/kg) |
Cd (mg/kg) |
Cr (g/kg) |
Cu (g/kg) |
Pb (mg/kg) |
As (mg/kg) |
Cd (mg/kg) |
Cr (mg/kg) |
Cu (mg/kg) |
Pb (mg/kg) |
| P1 | 0.14 ± 0.00a | 0.89 ± 0.01a | 0.91 ± 0.14a | 24.76 ± 0.23a | 57.85 ± 0.55a | 1.88 ± 0.04a | 0.43 ± 0.01b | 0.51 ± 0.01b | 94.86 ± 0.37a | 224.05 ± 5.7a |
| P2 | 0.06 ± 0.00 | 0.49 ± 0.01b | 0.56 ± 0.05b | 11.69 ± 0.42b | 23.96 ± 0.49b | 0.42 ± 0.02b | 0.63 ± 0.10b | 0.55 ± 0.01a | 60.65 ± 0.99b | 54.76 ± 0.74b |
| P3 | BDL | 0.44 ± 0.01c | 0.22 ± 0.02c | 14.04 ± 0.23c | 5.72 ± 0.19c | 0.03 ± 0.00c | 0.09 ± 0.01c | 0.19 ± 0.01c | 12.94 ± 0.43c | 2.24 ± 0.10c |
| P4 | BDL | 0.14 ± 0.01e | 0.14 ± 0.05cd | 4.39 ± 0.34d | 0.51 ± 0.01d | BDL | BDL | 0.19 ± 0.01c | 8.04 ± 0.37d | 0.16 ± 0.00d |
| P5 | BDL | 0.16 ± 0.01d | 0.07 ± 0.00d | 3.03 ± 0.13e | 0.05 ± 0.00d | BDL | BDL | 0.03 ± 0.00d | 4.93 ± 0.39e | BDL |
| Wet | Season | Dry | Season | ||||||||||
| Coliform CFU/100 g |
E. coli CFU/100 g |
Salmonella CFU/100 g |
Shigella CFU/100 g |
S. aureus CFU/100 g |
V. cholerae CFU/100 g |
Coliform CFU/100 g |
E. coli CFU/100 g |
Salmonella CFU/100 g |
Shigella CFU/100 g |
S. aureus CFU/100 g |
V. cholerae CFU/100 g |
||
| P1 | 101 | 58 | 2 | 39 | 84 | 28 | 290 | 39 | 0 | 2 | 119 | 1 | |
| P2 | 24 | 10 | 0 | 3 | 43 | 6 | 225 | 31 | 0 | 0 | 68 | 1 | |
| P3 | 15 | 8 | 0 | 1 | 30 | 1 | 121 | 31 | 0 | 0 | 19 | 0 | |
| P4 | 12 | 2 | 0 | 2 | 24 | 0 | 102 | 3 | 0 | 0 | 22 | 0 | |
| P5 | 2 | 0 | 0 | 4 | 14 | 0 | 24 | 0 | 0 | 1 | 8 | 0 |
| Pb | Cd | Cr | Cu | As | T. Coliform | E. coli | Salmonella | Shigella | S. aureus | ||
| Pb | 1 | ||||||||||
| Cd | .954** | 1 | |||||||||
| Cr | .968** | .935** | 1 | ||||||||
| Cu | .906** | .983** | .880** | 1 | |||||||
| As | .994** | .921** | .961** | .858** | 1 | ||||||
| Total Coliform | .967** | .921** | .903** | .907** | .949** | 1 | |||||
| E. coli | .960** | .929** | .899** | .917** | .935** | .995** | 1 | ||||
| Salmonella | .918** | .855** | .829** | .844** | .900** | .979** | .984** | 1 | |||
| Shigella | .918** | .844** | .821** | .822** | .907** | .972** | .976** | .997** | 1 | ||
| S. aureus | .985** | .957** | .963** | .933** | .969** | .974** | .966** | .917** | .907** | 1 | |
| **. Correlation is significant at the 0.01 level (2-tailed). | |||||||||||
| Lead | Cd | Cr | Cu | As | Coliform | E. coli | Shigella | S. aureus | V. cholerea | ||
| Lead | 1 | ||||||||||
| Cadmium | .599* | 1 | |||||||||
| Chromium | .710** | .947** | 1 | ||||||||
| Copper | .934** | .843** | .902** | 1 | |||||||
| Arsenic | .999** | .579* | .694** | .926** | 1 | ||||||
| Coliform | .853** | .848** | .956** | .955** | .844** | 1 | |||||
| E. coli | .668** | .740** | .792** | .779** | .663** | .860** | 1 | ||||
| Shigella | .807** | 0.164 | 0.202 | .602* | .816** | 0.389 | 0.239 | 1 | |||
| S. aureus | .958** | .785** | .879** | .992** | .952** | .955** | .752** | .630* | 1 | ||
| V. cholera | .785** | .959** | .951** | .950** | .769** | .912** | .724** | 0.408 | .915** | 1 | |
| *. Correlation is significant at the 0.05 level (2-tailed). | |||||||||||
| **. Correlation is significant at the 0.01 level (2-tailed). | |||||||||||
| Wet Season | Dry Season | ||||||
| Adults | Children | Infants | Adults | Children | Infants | ||
| HI range | 14.48-70.48 | 21.80-105.07 | 20.9 –111.89 | 4.80-17.20 | 12.50-43.13 | 13.3-46.15 | |
| CR range | 0.00130-0.0120 | 0.00037-0.00399 | 0.000039-0.00043 | 0-0.00087 | 0-0.00228 | 0-0.00243 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




