Submitted:
24 April 2026
Posted:
27 April 2026
You are already at the latest version
Abstract
Keywords:
Introduction
Detailed Patients ‘Description
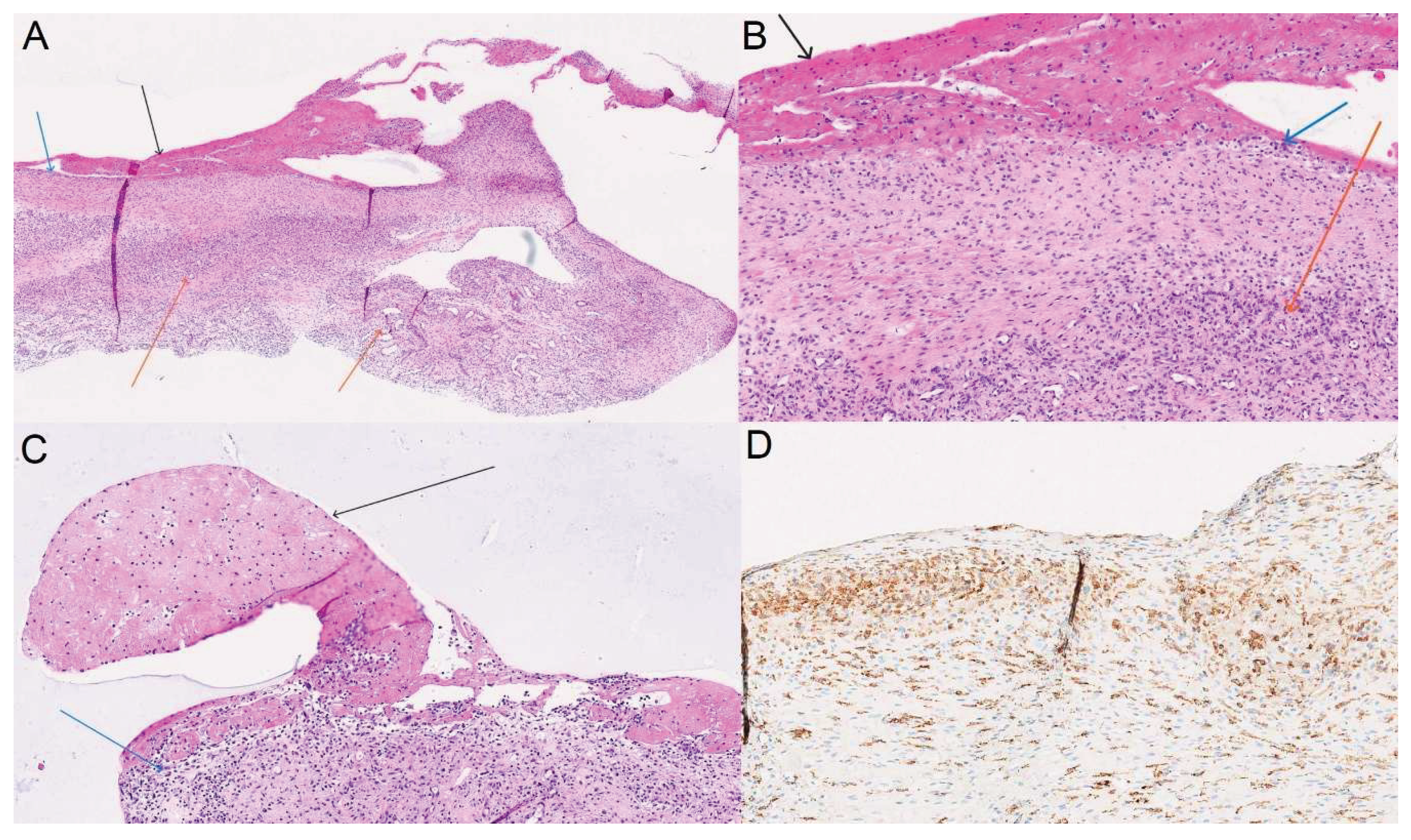
Results
Discussion
Conclusion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
References
- Polak, F.P.; Thomas, S.J.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; et al. Safety and Efficacy of the BNT162b2 mRNA Covid-19 Vaccine. N Engl. J. Med. 2020, 383, 2603–2615. [Google Scholar] [CrossRef]
- Baden, L.R.; El Sahly, H.M.; Essink, B.; Kotloff, K.; Frey, S.; Novak, R.; et al. Efficacy and Safety of the mRNA-1273 SARS-CoV-2 Vaccine. N Engl. J. Med. 2021, 384, 403–416. [Google Scholar] [CrossRef]
- Nazar, W.; Romantowski, J.; Nazar, G.; Niedoszytk, M.; Braun-Dullaeus, R.; Daniłowicz-Szymanowicz, L. Serious Adverse Drug Reactions associated with anti-SARS-CoV-2 vaccines and their reporting trends in the Eudra Vigilance database. Sci. Rep. 2025, 15, 18582. [Google Scholar] [CrossRef]
- Unal Enginar, A. Arthritis following Covid-19 vaccination: report of two cases. Int. Immunopharmacol. 2021, 101, 108256. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.-C.; Chen, C.-J. new onset inflammatory arthritis after Covid-19vaccination: A systematic review. Int. J. Rheum. Dis. 2022, 00, 1–11. [Google Scholar]
- Ana, Q.-J.; Qin, D.-A.; Pei, J.-X. Reactive arthritis after Covid-19 vaccination. Hum. Vaccine Immunother. 2021, 17, 2954–2956. [Google Scholar] [CrossRef] [PubMed]
- Vanaskova E, Kelbich P, Novotny T; reactive synovitis of the k; 2022;00:1–4. Knee joint after Covid-19 vaccination: the first ultrastructural analysis of synovial fluid; int J Rheum Dis; 2022;00:1-4. [CrossRef]
- Golstein, M.-A. post-covid19 vaccine hyperproduction of anti-Spike antibodies and rheumatological manifestations. Vaccines 2025, 13, 1028. [Google Scholar] [CrossRef]
- Sun, Z.; Brodsky, J.L. Protein quality control in the secretory pathway. J. Cell. Biol. 2019, 218, 3171–3187. [Google Scholar] [CrossRef]
- Abhishek, A.; de Pablo, P.; Cader, M.Z.; Buckley, C.D.; Raza, K.; Filer, A. Diagnostic outcomes associated with ankle synovitis in early inflammatory arthritis: a cohort study. Clin. Exp. Rheumatol. 2014, 32, 533–538. [Google Scholar]
- Pal, A.; Roongta, R.; Mondal, S.; Sinha, D.; Sinhamahapatra, P.; Ghosh, A.; Chattopadhyay, A. Does post-COVID reactive arthritis exist? Experience of a tertiary care centre with a review of the literature. Rheumatol. Clin. 2023, 19, 67–73. [Google Scholar]
- Jarrot, P.-A.; Mirouse, A.; Ottaviani, S. Polymyalgia rheumatica and giant cell arteritis following COVID-19 vaccination: Results from a nationwide survey. Hum. Vaccines Immunother. 2024, 20, 2334084. [Google Scholar] [CrossRef] [PubMed]
- Tokumasu, K.; Fujita-Yamashita, M.; Sunada, N.; Sakurada, Y.; Yamamoto, K.; Nakano, Y.; Matsuda, Y.; Otsuka, Y.; Hasegawa, T.; Hagiya, H.; Honda, H.; Otsuka, F. Characteristics of Persistent Symptoms Manifested after SARS-CoV-2 Vaccination: An Observational Retrospective Study in a Specialized Clinic for Vaccination-Related Adverse Events. Vaccines 2023, 11, 1661. [Google Scholar] [CrossRef]
- Golstein, M.A.; Fagnard, O.; Steinfeld, S.D. Reactive arthritis after Covid 19 vaccination: 17 cases. Rheumatology 2023, 62, 3706–3709. [Google Scholar] [CrossRef]
- Soroosh, S.G.; et al. synovial biopsy for establishing a definite diagnosis in undifferentiated chronic knee mono arthritis. BMC Musculoskelet. Disord. 2023, 24–23, 1–7. [Google Scholar] [CrossRef]
- de Sousa, P.M.B.; Silva, E.A.; Campos, M.A.G.; Lages, J.S.; da Graça Carvalhal Frazao Correa, R.; Silva, G.E.B. Fatal Myocarditis Following COVID-19 mRNA immunization: A Case Report and Differnetiel Diagnosis Review. Vaccines 2024, 12, 194. [Google Scholar] [CrossRef]
- Schwab, C.; Domke, L.M.; Hartmann, L.; Stenzinger, A.; Longerich, T.; Schirmacher, P. Autopsy-based histopathological characterization of myocarditis after anti-SARS-CoV-2-vaccination. Clin. Res. Cardiol. 2023, 112, 431–440. [Google Scholar] [CrossRef]
- Yonker, L.M.; Swank, Z.; Bartsch, Y.C.; Burns, M.D.; Kane, A.; Boribong, B.; et al. Circulating Spike Protein Detected in Post-COVID-19 mRNA Vaccine Myocarditis. Circulation 2023, 47, 867–876 jan4. [Google Scholar] [CrossRef]
- Khoury, J.; Najjar-Debbiny, R.; Hanna, A.; JabbourA, *!!! REPLACE !!!*; Abu Ahmad, Y.; Saffuri, A.; et al. Vaccine long term immune decline and breakthrough infections. Vaccine 2021, 39, 6984–6989. [Google Scholar] [CrossRef] [PubMed]
- Sousa Silva, M.F.; Matias Dinelly Pinto, A.C.; de Cassia Evangelista de Oliveira, F.; Freire Caetano, L.; Montenegro de Carvalho Araujo, F.; Gambim Fonseca, M.H. Antibody response 6 months after booster dose of Pfizer in previous recipients of CoronaVac. J. Med. Virol. 2022, 1–6. [Google Scholar] [CrossRef]
- Crowley, S.D.; Rudemiller, N.P. Immunologic Effects of Renin-Angiotensin System. J. Am. Soc. Nephrol. 2017, 28, 1350–1361. [Google Scholar] [CrossRef] [PubMed]
- Aboudounya, M.M.; Heads, R.J. COVID-19 and Toll-Like Receptor 4 (TLR4): SARS-CoV-2 May Bind and Activate TLR4 to Increase ACE2 Expression, Facilitating Entry and Causing Hyperinflammation. Mediat. Inflamm. 2021, 2021, 8874339. [Google Scholar] [CrossRef]
- Cosentino, M.; Marino, F. understanding the pharmacology of Covid-19 mRNA vaccines: playing dice with Spike? Int. J. Mol. Sci. 2022, 23, 10881. [Google Scholar] [CrossRef]
- Wang, S.-F.; Tseng, S.-P.; Yen, C.-H.; Yang, J.-Y.; Tsao, C.-H.; Shen, C.-W.; Chen, K.-H.; Liu, F.-T.; Liu, W.-T.; Chen, Y.-M.A.; Huang, J.C. Antibody-dependent SARS coronavirus infection is mediated by antibodies against spike proteins. Biochem Biophys. Res. Commun. 2014, 451, 208–214. [Google Scholar] [CrossRef]
- Hasan, A.; Al-Mullab, M.R.; Abubakerc, J.; Al-Mulla, F. Early insight into antibody-dependent enhancement after SARS-CoV-2 m RNA vaccination. Hum. VACCINES Immunother. 2021, 17, 4121–4125. [Google Scholar] [CrossRef] [PubMed]
- Timofeeva, A.M.; Aulova, K.S.; Mustaev, E.A.; Nevinsky, G. Sars-Cov-2 Spike protein and molecular mimicry: an immunoinformatic screen for cros-reactive autoantigen candidates. Int. J. Mol. Sci. 2025, 26, 8793. [Google Scholar] [CrossRef] [PubMed]
- Silvia Fanti , PhD; Carlene Dyer , PhD; Inga J.na Ingimarsd.ttir , MD, PhD; Daniel Harding , MD, PhD; Guosu Wang, MSc;Antonio D’Amati, MD, PhD; Eriomina Shahaj, PhD; Adalbjorg .r Sigurbergsd.ttir, MD; Helga Th.rsd.ttir, MD;Oddný Brattberg Gunnarsd.ttir , MD; Stavroula Kanoni , PhD; Paul Wright , MPharm; John Martin , MD; Jamie Chorlton, PhD;Zoe Hollowood , PhD; Siggeir Fannar Brynj.lfsson , PhD; Bjorn R.nar L.dv.ksson , MD, PhD; Egle Solito , PhD; Serena Bert, PhD; Jack M. Keane, MRes; Saidi A. Mohiddin, MD†; M. Paula Longhi, PhD†; Federica M. Marelli-Berg, MD, PhD†; Combined Adaptive Immune Mechanisms Mediate Cardiac Injury After COVID-19Vaccination. Circulation 2025, 152, 1485–1500. [CrossRef]
| Patients | Age | Gender | Vaccines | Delay post vaccine | Initial symptoms | Medical history |
Arthroscopy Biopsy |
MRI knee | |||||
| DNA | RNA | Arthritis | |||||||||||
| Doses | Doses | ||||||||||||
| 1 | 2 | 1 | 2 | 3-4 | |||||||||
| 1 | caucasian | 65 | F | AZ | AZ | Pf | 1 day | 2 knees | Diabetes HT |
- | Synovitis | ||
| 2 | caucasian | 61 | M | Pf | Pf | Mod | 1 day | 2 knees 2 ankles |
Diabetes HT |
- | - | ||
| 3 | African black | 51 | M | AZ | AZ | Pf | 1 month | 2 knees | HT | + | Synovitis | ||
| 4 | Caucasian | 85 | M | Pf | Pf | 2 months | Left knee | - | + | Synovitis | |||
| 5 | Caucasian | 51 | M | Mod | Mod | Mod | >6 months | Left knee | HLAb27+ |
- | - | ||
| 6 | Caucasian | 69 | M | AZ | AZ | Pf | Pf | 1.5 month | Right knee 2 ankles |
- | + | Synovitis osteochondroma |
|
| 7 | Caucasian | 31 | F | Pf | Pf | Pf Mod | >6 months | Right knee Left wrist |
- | + | - | ||
| Age/gender | VS/CRP |
Anti-Spike antibodies Blood BAU/ml delay post vaccine |
Anti Spike Synovial Fluid BAU/ml delay post vaccine |
Articular Fluid | Biopsy | ||||||||
| 1 | 2 | 3 | 1 | 2 | 3 | 1 | 2 | ||||||
| 1 | 65 | F | 90/65 | 16/2 | 1700 6months |
>2080 9months |
1680 13months |
270 10months |
456 18months |
Rare WC RC- |
Rare WC RC- |
- | |
| 2 | 62 | M | 37/94 | >2080 11months | >2080 15months |
>2080 17months |
1190 11months |
>2080 13mont |
WC 4150 65% PNN RC+ |
WC 27100 79% PNN RC+ |
- | ||
| 3 | 51 | M | 113/94 | 41/4 | 34/14 | >2080 13 months |
1050 24mois |
346 34months |
>2080 13months |
126 32months |
WC 300 60 Mono Mac RC+ |
WC 266 67% L RC + |
+ |
| 4 | 85 | M | 18/1 | 6/3 | >2080 9 months |
>2080 21months |
>2080 (PCR-) 9months |
Rare WC RC- |
+ | ||||
| 5 | 51 | M | 30/3 | >2080 13 months |
>2080 13months |
Rare WC RC- |
- | ||||||
| 6 | 69 | M | 6/2 | 10/9 | 2/1 | >2080 1.5months |
641 (6mois) |
596 10months |
1425 1.5 month |
WC 146 RC 16 |
+ | ||
| 7 | 31 | F | 13/6 | >2080 7months |
>2080 7 months |
WC 3500 79% L RC- |
+ | ||||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




