Submitted:
23 April 2026
Posted:
27 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods
2.1. Study Cohorts and Exposure Definitions
2.2. Breast-Cancer Ascertainment and Baseline Covariates
2.3. Propensity-Matched Comparative Survival Analyses from Drug Index
2.4. Analyses of Metastatic Disease Burden and Cause of Death
2.5. Dose, weight-Loss, and Persistence One-Year Landmark Analyses
2.6. Oncology-Treatment-Indexed Analyses
2.7. Tumor-Genomic and Preclinical Context Analyses
2.8. Statistical Analysis
2.9. Data Source
2.10. Data Availability
2.11. De-identification and HIPAA compliance certification
2.12. Data Harmonization
2.13. Code Availability.
3. Results
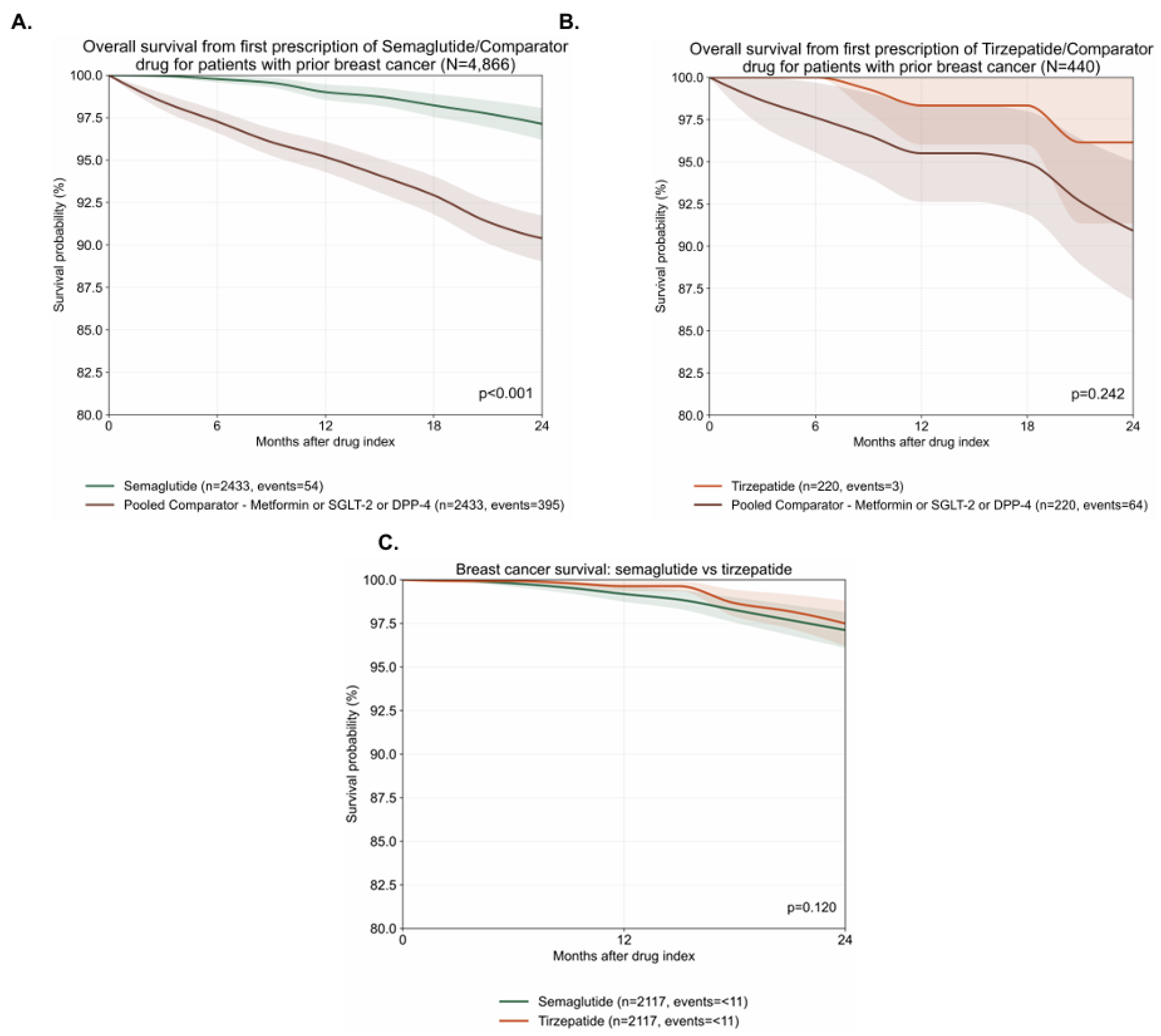
3.1. Semaglutide is Associated with Improved Survival Compared to Other Anti-Diabetic Medications in Patients with Previously Diagnosed Breast Cancer
3.2. Tirzepatide is Associated with a Numerically Lower but Statistically Non-Significant Reduction in Mortality in Patients with Previously Diagnosed Breast Cancer
3.3. Head-to-Head Propensity-Score–Matched Analysis Shows no Significant Overall-Survival Difference Between Semaglutide and Tirzepatide
3.4. Concordant Structured EHR and AI-Derived Clinical Note Analyses Show Significantly Lower Metastatic Burden with Semaglutide Than Matched Pooled Anti-Diabetic Drugs
3.5. LLM-Derived Cause-of-Death Analyses Suggest Fewer Cancer-Associated Deaths in Semaglutide-Treated Patients than in Matched Pooled Anti-Diabetic Comparators
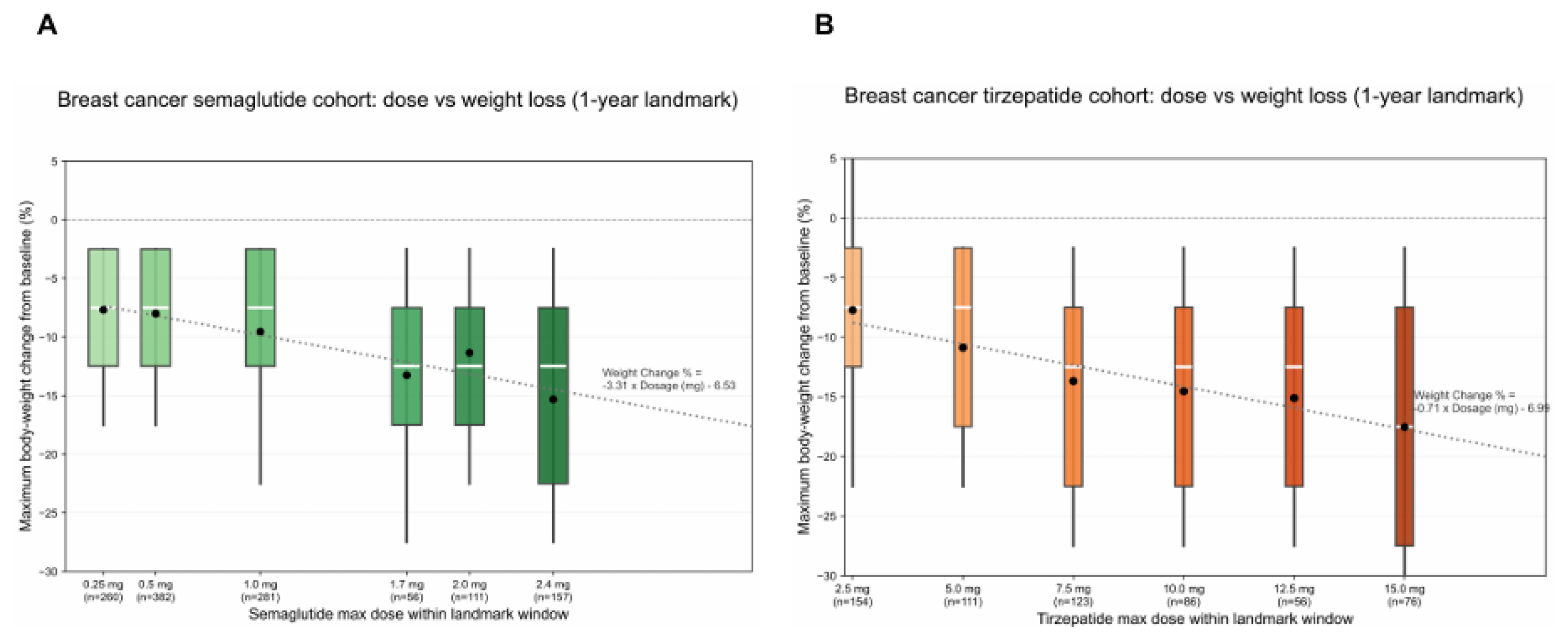
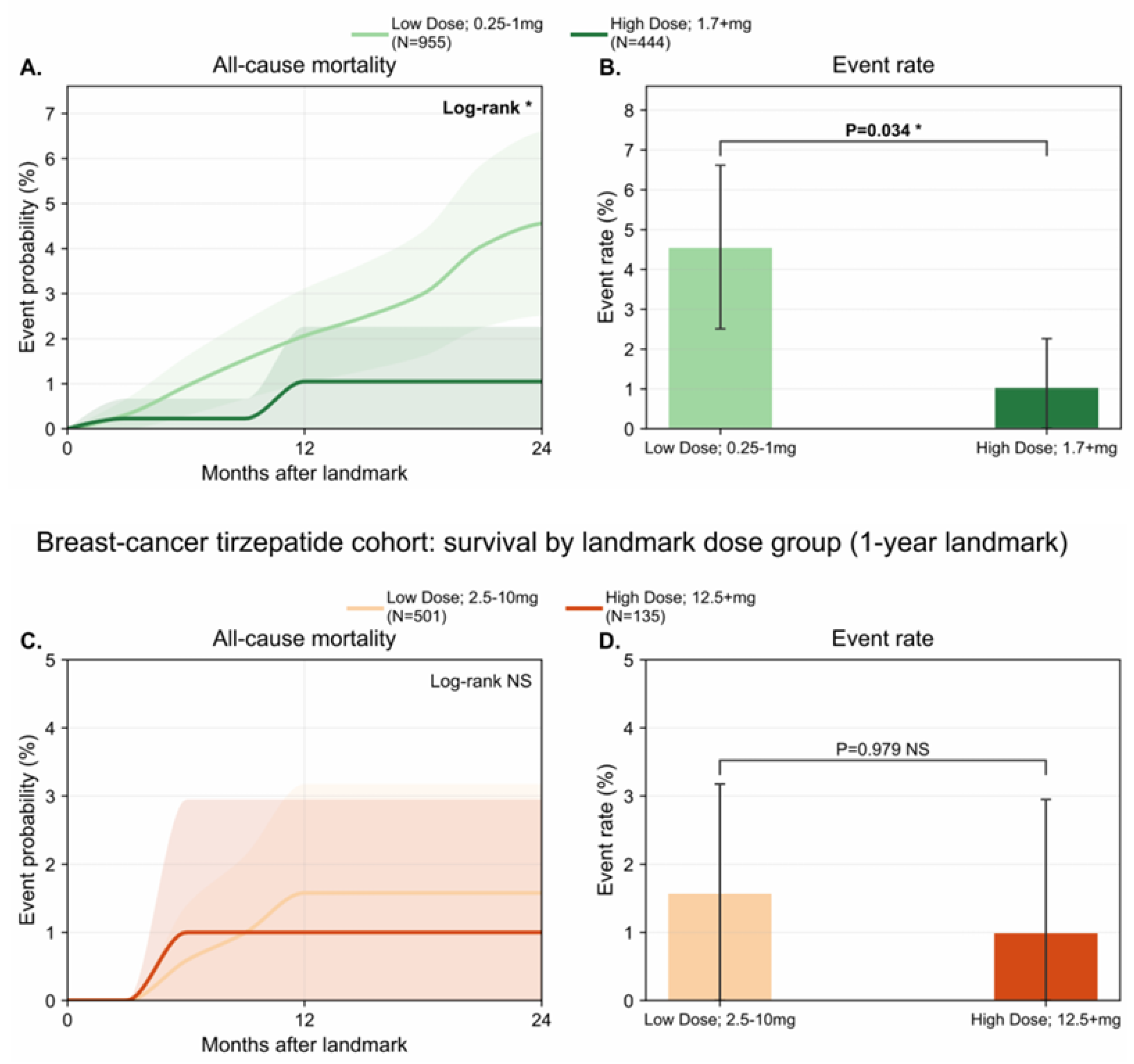
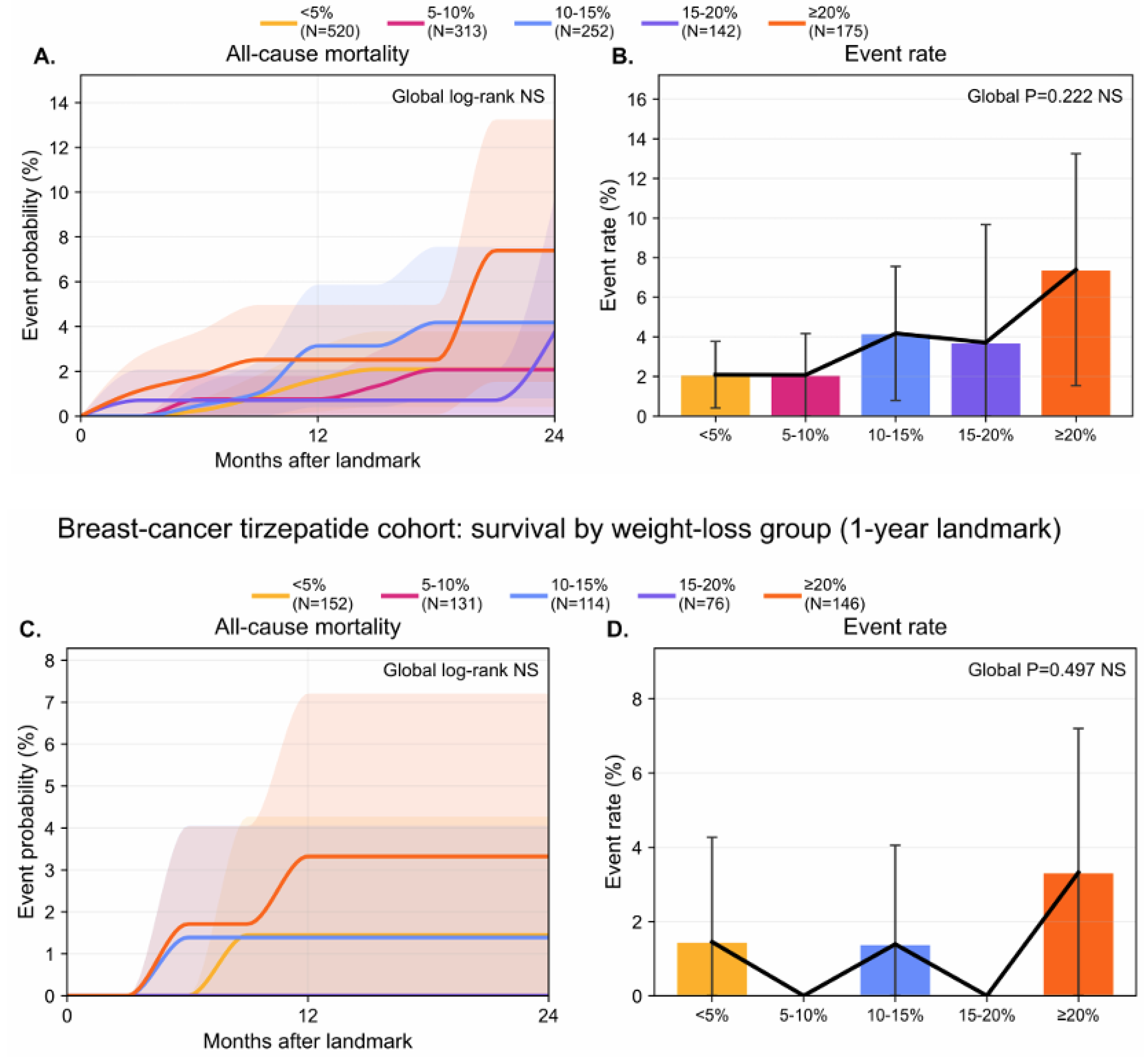
3.6. In Patients with Pre-Existing Breast Cancer, Higher Attained Semaglutide Dose is Associated with Improved All-Cause Mortality After One Year of Semaglutide Exposure
3.7. Tirzepatide Landmark Analyses Do Not Show A Significant Dose- or Weight-Loss–Stratified Survival Difference
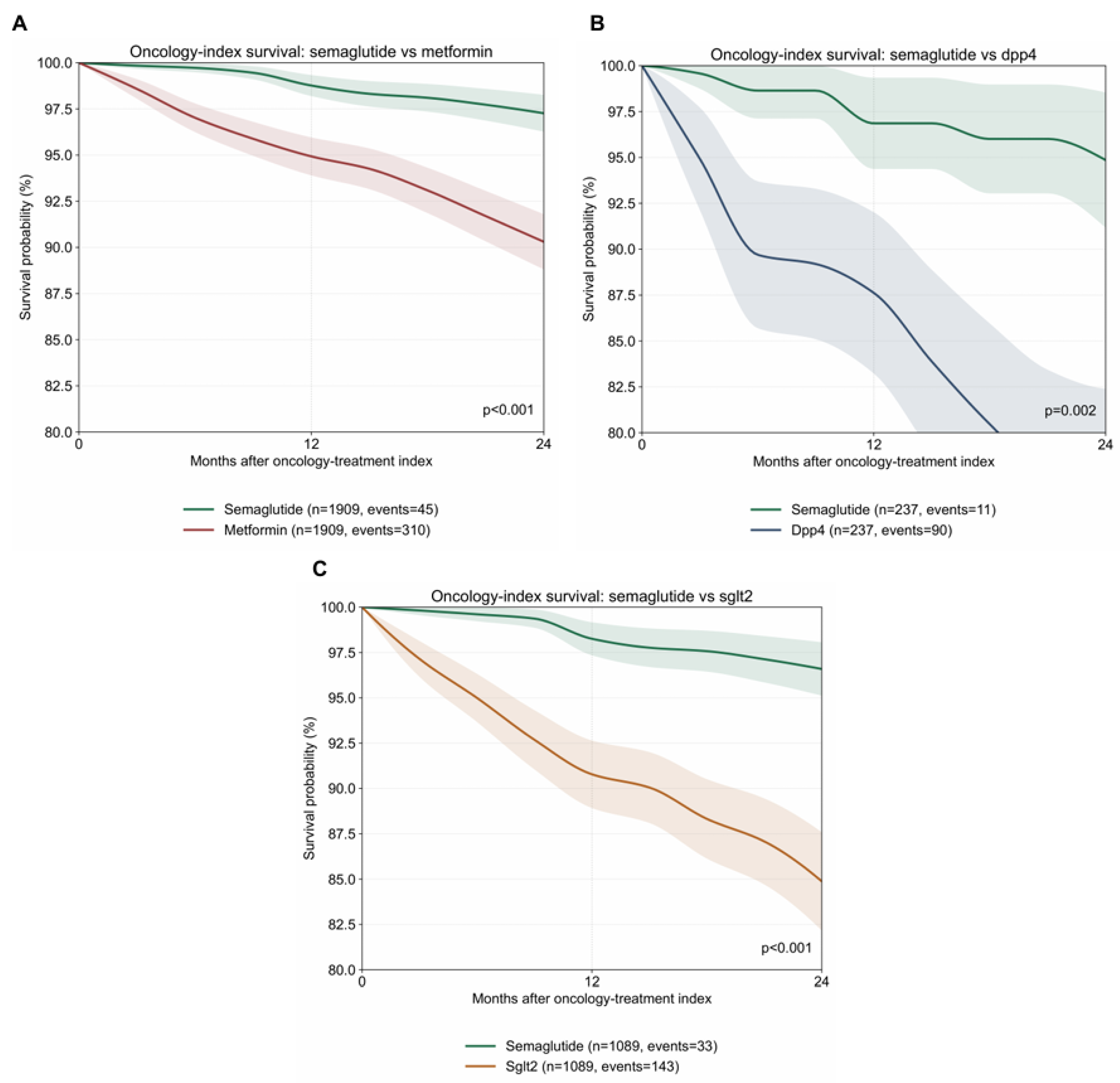
3.8. Semaglutide-Associated Survival Remained Favorable When Follow-up Was Anchored to Oncology Treatment Initiation
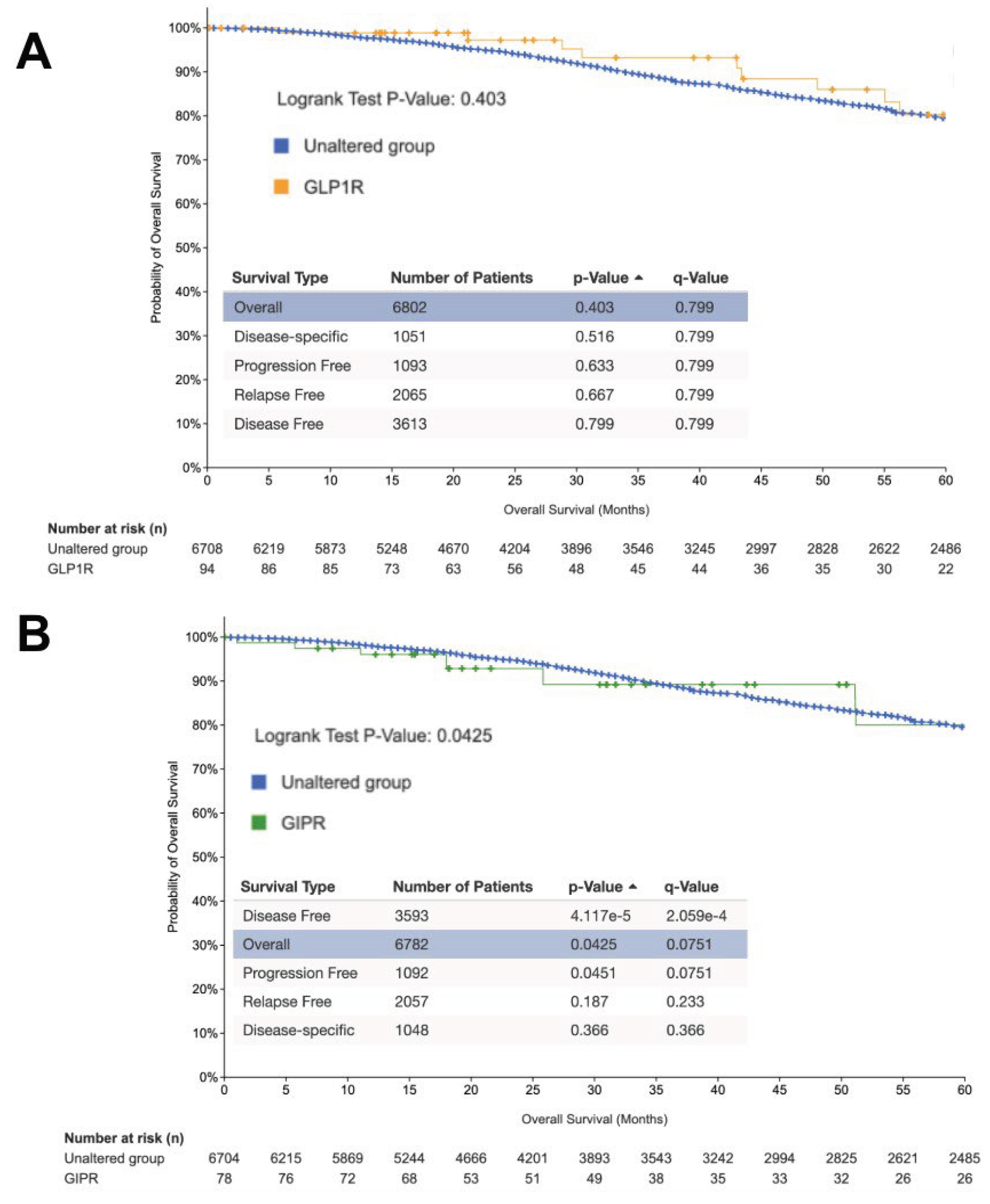
3.9. Genomic and Functional Perturbation Analyses of GLP1R and GIPR in Breast Cancer
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest Statement
References
- Chan, D. S. M. et al. Body mass index and survival in women with breast cancer-systematic literature review and meta-analysis of 82 follow-up studies. Ann Oncol 25, 1901–1914 (2014). [CrossRef]
- Puklin, L. S. et al. Post-diagnosis weight trajectories and mortality among women with breast cancer. NPJ Breast Cancer 9, 98 (2023). [CrossRef]
- Wilding, J. P. H. et al. Once-Weekly Semaglutide in Adults with Overweight or Obesity. N Engl J Med 384, 989–1002 (2021). [CrossRef]
- Lincoff, A. M. et al. Semaglutide and Cardiovascular Outcomes in Obesity without Diabetes. N Engl J Med 389, 2221–2232 (2023). [CrossRef]
- Packer, M. et al. Tirzepatide for Heart Failure with Preserved Ejection Fraction and Obesity. N Engl J Med 392, 427–437 (2025). [CrossRef]
- Aronne, L. J. et al. Tirzepatide as Compared with Semaglutide for the Treatment of Obesity. N Engl J Med 393, 26–36 (2025). [CrossRef]
- Wang, L., Xu, R., Kaelber, D. C. & Berger, N. A. Glucagon-Like Peptide 1 Receptor Agonists and 13 Obesity-Associated Cancers in Patients With Type 2 Diabetes. JAMA Netw Open 7, e2421305 (2024). [CrossRef]
- Dai, H. et al. GLP-1 Receptor Agonists and Cancer Risk in Adults With Obesity. JAMA Oncol 11, 1186–1193 (2025). [CrossRef]
- Radwan, R. M. et al. GLP-1 RA Use and Survival Among Older Adults With Cancer and Type 2 Diabetes. JAMA Netw Open 8, e2521887 (2025). [CrossRef]
- Shen, S. et al. GLP-1 Receptor Agonist Use and Weight Change in Patients With Breast Cancer. Oncology (Williston Park) null, 294–296 (2025).
- Cerami, E. et al. The cBio cancer genomics portal: an open platform for exploring multidimensional cancer genomics data. Cancer Discov 2, 401–404 (2012). [CrossRef]
- Gao, J. et al. Integrative analysis of complex cancer genomics and clinical profiles using the cBioPortal. Sci Signal 6, l1 (2013). [CrossRef]
- de Bruijn, I. et al. Analysis and Visualization of Longitudinal Genomic and Clinical Data from the AACR Project GENIE Biopharma Collaborative in cBioPortal. Cancer Res 83, 3861–3867 (2023). [CrossRef]
- ClinicalTrials.gov. https://clinicaltrials.gov/study/NCT06517212.
- Willard, F. S. et al. Tirzepatide is an imbalanced and biased dual GIP and GLP-1 receptor agonist. JCI Insight 5, (2020). [CrossRef]
- Liu, Q. K. Mechanisms of action and therapeutic applications of GLP-1 and dual GIP/GLP-1 receptor agonists. Front Endocrinol (Lausanne) 15, 1431292 (2024). [CrossRef]
- Yang, K., Liu, C., Guo, Q. & Li, Y. GLP-1 Receptor Agonists and Noncardiometabolic Outcomes: An Umbrella Review of Meta-Analyses. JAMA Netw Open 9, e264722 (2026). [CrossRef]
- Wali, A. F. et al. Reassessing cancer risk with GLP-1 receptor agonists: a comprehensive meta-analysis of gastrointestinal malignancies. Front Pharmacol 17, 1736380 (2026). [CrossRef]
- Harris, M., Harvie, M. & Renehan, A. G. Glucagon-like Peptide-1 (GLP-1) Receptor Agonists and Cancer Prevention: Methodological Pitfalls in Observational Studies. Cancers (Basel) 17, (2025). [CrossRef]
- Miousse, I. R. GLP-1 receptor agonists in the context of cancer: the road ahead. Am J Physiol Cell Physiol 328, C1822–C1828 (2025). [CrossRef]
- Figlioli, G. et al. Glucagon-like peptide-1 receptor agonists and risk of gastrointestinal cancers: A systematic review and meta-analysis of randomized controlled trials. Pharmacol Res 208, 107401 (2024). [CrossRef]
- Ungvari, Z. et al. Prognostic impact of glucagon-like peptide-1 receptor (GLP1R) expression on cancer survival and its implications for GLP-1R agonist therapy: an integrative analysis across multiple tumor types. Geroscience 47, 4413–4427 (2025). [CrossRef]
- Stanisavljevic, I. et al. Semaglutide decelerates the growth and progression of breast cancer by enhancing the acquired antitumor immunity. Biomed Pharmacother 181, 117668 (2024). [CrossRef]
- Murugadoss, K. et al. Building a best-in-class automated de-identification tool for electronic health records through ensemble learning. Patterns (N Y) 2, 100255 (2021). [CrossRef]
- Venkatakrishnan, A. J. et al. Clinical nSights: A software platform to accelerate real world oncology analyses. Journal of Clinical Oncology (2024) . [CrossRef]
| VARIABLE (1:1 propensity matched) | Semaglutide | Pooled comparator |
|---|---|---|
| N patients | 2433 | 2433 |
| Age at index, mean (SD) | 62.2 (11.1) | 62.4 (11.4) |
| Baseline BMI, mean (SD) | 33.9 (6.0) | 34.0 (6.7) |
| Baseline weight, mean (SD) | 92.0 (18.7) | 92.3 (21.1) |
| Index year, mean (SD) | 2023 (1.3) | 2020 (4.8) |
| Days from qualifying prior breast cancer to index, mean (SD) | 2193.7 (1842.7) | 2089.9 (1850.1) |
| Days from most recent oncology treatment to index, mean (SD) | 2653.5 (6536.5) | 2523.3 (6067.7) |
| Female sex, n (%) | 2419 (99.4%) | 2401 (98.7%) |
| T2DM at baseline, n (%) | 449 (18.5%) | 460 (18.9%) |
| Any prior oncology treatment, n (%) | 2418 (99.4%) | 2412 (99.1%) |
| Prior endocrine therapy, n (%) | 1673 (68.8%) | 1667 (68.5%) |
| Prior chemotherapy, n (%) | 666 (27.4%) | 608 (25.0%) |
| Prior HER2-directed therapy, n (%) | 225 (9.2%) | 218 (9.0%) |
| Prior CDK4/6 inhibitor, n (%) | 101 (4.2%) | 81 (3.3%) |
| Prior PARP inhibitor, n (%) | 13 (0.5%) | 3 (0.1%) |
| Prior checkpoint immunotherapy, n (%) | 41 (1.7%) | 33 (1.4%) |
| Prior other targeted therapy, n (%) | 24 (1.0%) | 29 (1.2%) |
| Follow up days, median [IQR] | 421 [182,769] | 665 [223,1,531] |
| VARIABLE | Tirzepatide | Pooled comparator |
|---|---|---|
| N patients | 220 | 220 |
| Age at index, mean (SD) | 63.9 (15.0) | 64.3 (9.7) |
| Baseline BMI, mean (SD) | 35.3 (7.2) | 31.4 (6.2) |
| Baseline weight, mean (SD) | 98.1 (25.0) | 93.8 (18.7) |
| Index year, mean (SD) | 2024 (0.9) | 2023 (4.9) |
| Days from qualifying prior breast cancer to index, mean (SD) | 2739.1 (2871.0) | 1378.4 (1641.9) |
| Days from most recent oncology treatment to index, mean (SD) |
2120.0 (3201.0) | 2200.0 (3100.0) |
| Female sex, n (%) | 219 (99.5%) | 218 (99.1%) |
| T2DM at baseline, n (%) | 23 (10.5%) | 25 (11.4%) |
| Any prior oncology treatment, n (%) | 213 (96.8%) | 213 (96.8%) |
| Prior endocrine therapy, n (%) | 147 (66.8%) | 147 (66.8%) |
| Prior chemotherapy, n (%) | 57 (25.9%) | 57 (25.9%) |
| Prior HER2-directed therapy, n (%) | 19 (8.6%) | 19 (8.6%) |
| Prior CDK4/6 inhibitor, n (%) | <11 (N/A) | <11 (N/A) |
| Prior PARP inhibitor, n (%) | <11 (N/A) | <11 (N/A) |
| Prior checkpoint immunotherapy, n (%) | <11 (N/A) | <11 (N/A) |
| Prior other targeted therapy, n (%) | <11 (N/A) | <11 (N/A) |
| VARIABLE | Semaglutide | Tirzepatide |
|---|---|---|
| N patients | 2117 | 2117 |
| Age at index, mean (SD) | 61.4 (11.4) | 60.7 (10.8) |
| Baseline BMI, mean (SD) | 33.8 (6.0) | 34.1 (6.0) |
| Baseline weight, mean (SD) | 92.2 (19.1) | 93.7 (20.2) |
| Index year, mean (SD) | 2023.6 (1.2) | 2024.4 (0.8) |
| Days from qualifying prior breast cancer to index, mean (SD) | 2201.7 (1892.8) |
2111.9 (1859.7) |
| Female sex, n (%) | 2107 (99.5%) | 2100 (99.2%) |
| T2DM at baseline, n (%) | 361 (17.1%) | 299 (14.1%) |
| Not used for matching | ||
| Days from most recent oncology treatment to index, mean (SD) | 2763.1 (6658.8) |
2190.4 (6201.8) |
| Any prior oncology treatment, n (%) | 2106 (99.5%) | 1886 (89.1%) |
| Prior endocrine therapy, n (%) | 1457 (68.8%) | 1249 (59.0%) |
| Prior chemotherapy, n (%) | 595 (28.1%) | 430 (20.3%) |
| Prior HER2-directed therapy, n (%) | 200 (9.4%) | 114 (5.4%) |
| Prior CDK4/6 inhibitor, n (%) | 99 (4.7%) | 51 (2.4%) |
| Prior PARP inhibitor, n (%) | 12 (0.6%) | <11 (N/A) |
| Prior checkpoint immunotherapy, n (%) | 41 (1.9%) | 19 (0.9%) |
| Prior other targeted therapy, n (%) | 26 (1.2%) | <11 (N/A) |
| Metastasis Finding |
Semaglutide pre-tx, n (%) | Comparator, pre-tx, n (%) | Semaglutide, post-tx, n (%) | Comparator post-tx, n (%) | Rate Ratio (post-tx) |
P-value (post-tx) |
|---|---|---|---|---|---|---|
| Structured ICD Codes | ||||||
| Lung metastasis [C78.00, C78.01, C78.02] |
<11 (<0.5%) | 23 (0.9%) | <11 (<0.5%) | 51 (2.1%) | <0.20 | <0.001 |
| Pleural metastasis [C78.2] |
- | - | <11 (<0.5%) | 17 (0.7%) | <0.59 | 0.01 |
| Skin metastasis [C79.2] |
<11 (<0.5%) | <11 (<0.5%) | <11 (<0.5%) | 15 (0.6%) | <0.67 | 0.025 |
| Metastatic cancer (unspecified site) [C79.9] |
<11 (<0.5%) | 13 (0.5%) | <11 (<0.5%) | 15 (0.6%) | <0.67 | 0.025 |
| LLM Extraction | ||||||
| Any metastasis | 177 (3.1%) | 281 (11.5%) | 171 (7.0%) | 365 (15.0%) | 0.47 | <0.001 |
| Bone metastasis | 15 (0.6%) | 71 (2.9%) | 24 (1.0%) | 127 (5.2%) | 0.19 | <0.001 |
| Liver metastasis | <11 (<0.5%) | 13 (0.5%) | <11 (<0.5%) | 62 (2.5%) | <0.16 | <0.001 |
| Lung metastasis | - | - | <11 (<0.5%) | 59 (2.4%) | <0.17 | <0.001 |
| Brain metastasis | - | - | <11 (<0.5%) | 47 (1.9%) | <0.21 | <0.001 |
| Cause of Death | Semaglutide, n (%) | Comparator, n (%) | Rate Ratio | p-value |
|---|---|---|---|---|
| Cancer Progression / Metastasis | <11 (19%) | 186 (47%) | 0.39 | <0.001 |
| Cardiac Events | 16 (30%) | 55 (14%) | 2.13 | 0.003 |
| Sepsis / Septic Shock | 12 (22%) | 43 (11%) | 2.04 | 0.017 |
| Respiratory Failure | <11 (11%) | 63 (16%) | 0.70 | 0.351 |
| Hemorrhage / Stroke | <11 (7%) | 20 (5%) | 1.46 | 0.474 |
| Other | <11 (11%) | 28 (7%) | 1.57 | 0.296 |
| VARIABLE | Semaglutide | Pooled comparator |
|---|---|---|
| N patients | 1909 | 1909 |
| Age at index, mean (SD) | 62.3 (11.0) | 62.5 (11.4) |
| Baseline BMI, mean (SD) | 34.1 (6.1) | 34.2 (6.6) |
| Baseline weight, mean (SD) | 92.7 (18.8) | 92.6 (21.0) |
| Index year, mean (SD) | 2023.7 (1.2) | 2020.4 (4.5) |
| Days from qualifying prior breast cancer to index, mean (SD) | 2299.2 (1792.1) | 2213.1 (1801.5) |
| Days from most recent oncology treatment to index, mean (SD) | 56.0 (108.0) | 59.5 (120.7) |
| Female sex, n (%) | 1907 (99.9%) | 1895 (99.3%) |
| T2DM at baseline, n (%) | 367 (19.2%) | 380 (19.9%) |
| Any prior oncology treatment, n (%) | 1909 (100.0%) | 1909 (100.0%) |
| Prior endocrine therapy, n (%) | 1333 (69.8%) | 1329 (69.6%) |
| Prior chemotherapy, n (%) | 548 (28.7%) | 527 (27.6%) |
| Prior HER2-directed therapy, n (%) | 191 (10.0%) | 165 (8.6%) |
| Prior CDK4/6 inhibitor, n (%) | 84 (4.4%) | 80 (4.2%) |
| Prior PARP inhibitor, n (%) | 8 (0.4%) | 6 (0.3%) |
| Prior checkpoint immunotherapy, n (%) | 32 (1.7%) | 27 (1.4%) |
| Prior other targeted therapy, n (%) | 20 (1.0%) | 25 (1.3%) |
| Follow up days, median [IQR] (approx.) | 437 [210,776] | 793 [341,1,917] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




