Submitted:
23 April 2026
Posted:
24 April 2026
You are already at the latest version
Abstract
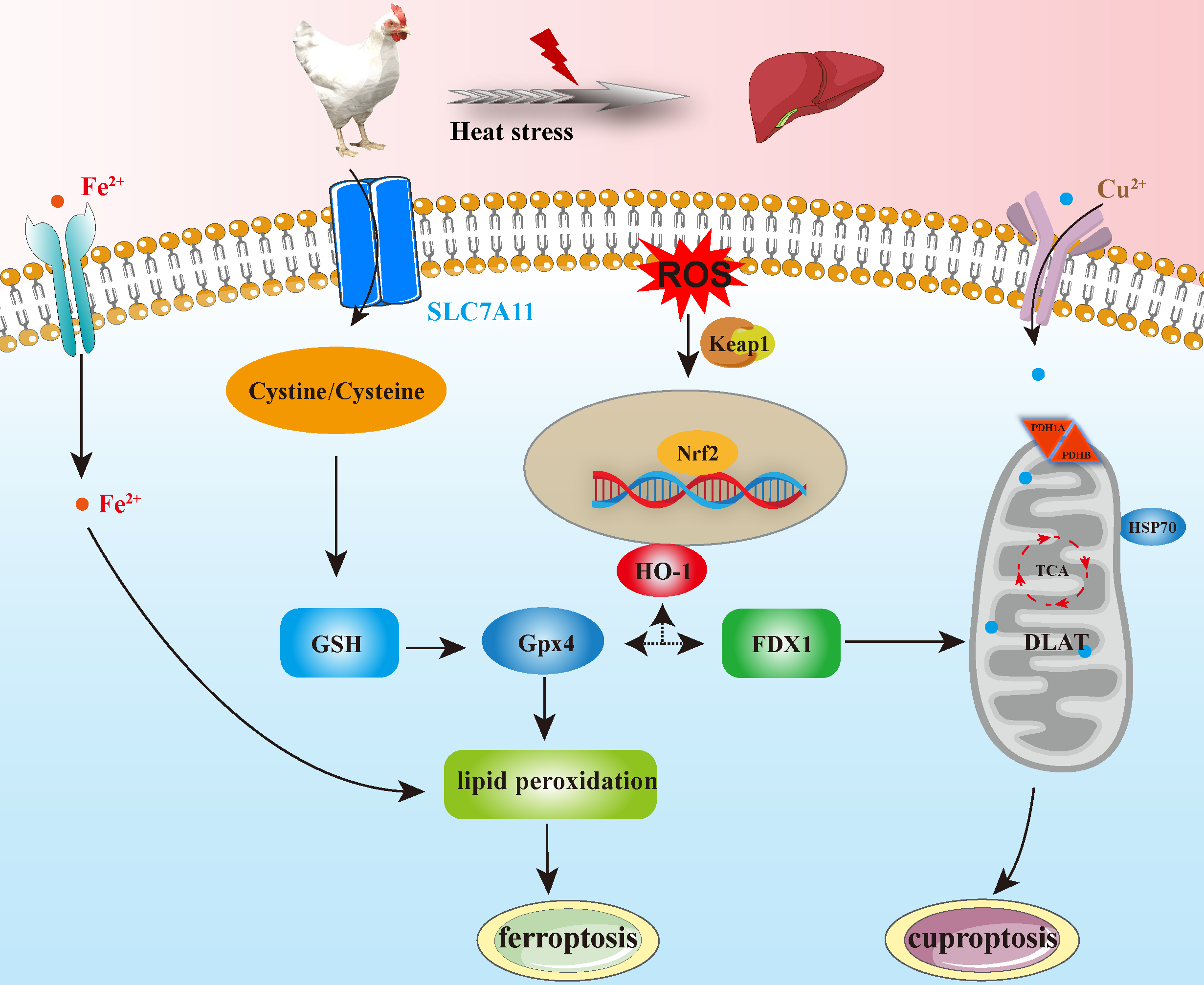
Keywords:
1. Introduction
2. Materials and Methods
2.1. Animals and Treatment
2.2. Biochemical Analysis
2.3. Histopathological Analysis
2.4. Determination of Oxidative Stress Level
2.5. Determination of Cu and Iron Levels
2.6. Immunohistochemical Assay
2.7. Immunofluorescence Assay
2.8. Western Blot Analysis
2.9. Molecular Docking
2.10. Statistical Analysis
3. Results
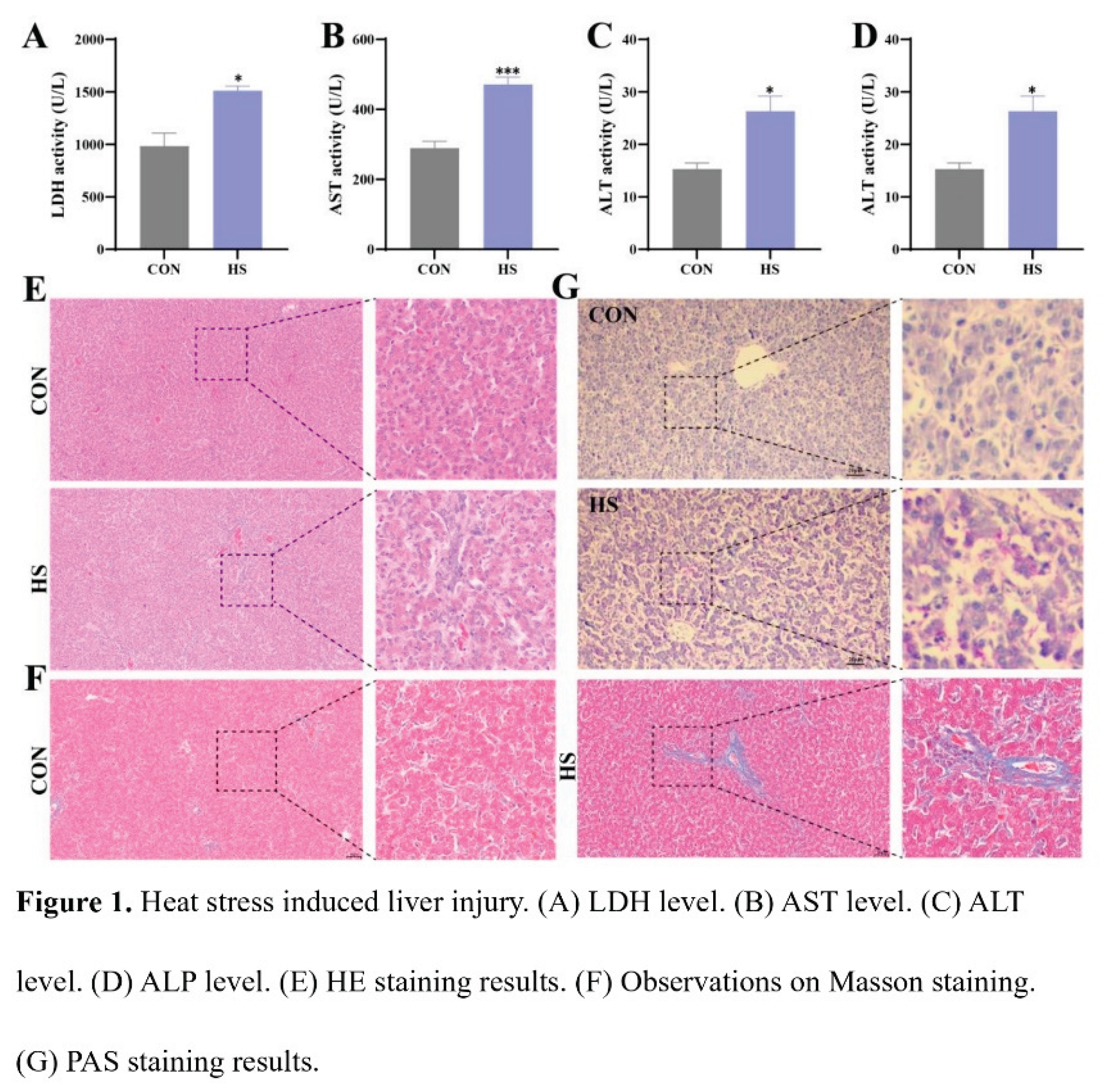
3.1. Heat Stress Induced Liver Injury in Chicken
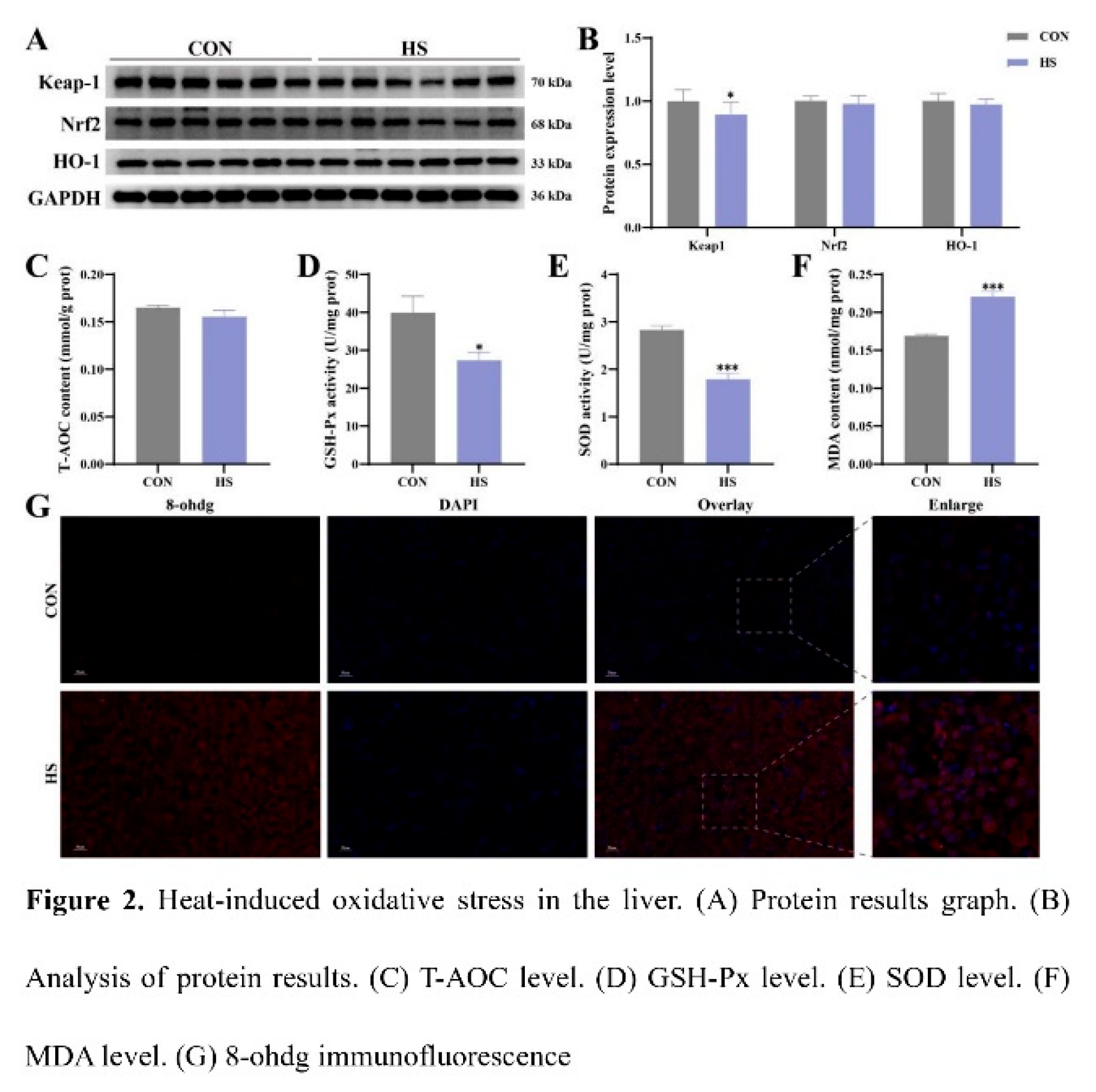
3.2. Effect of Heat Stress on Oxidative Stress in Chicken Liver
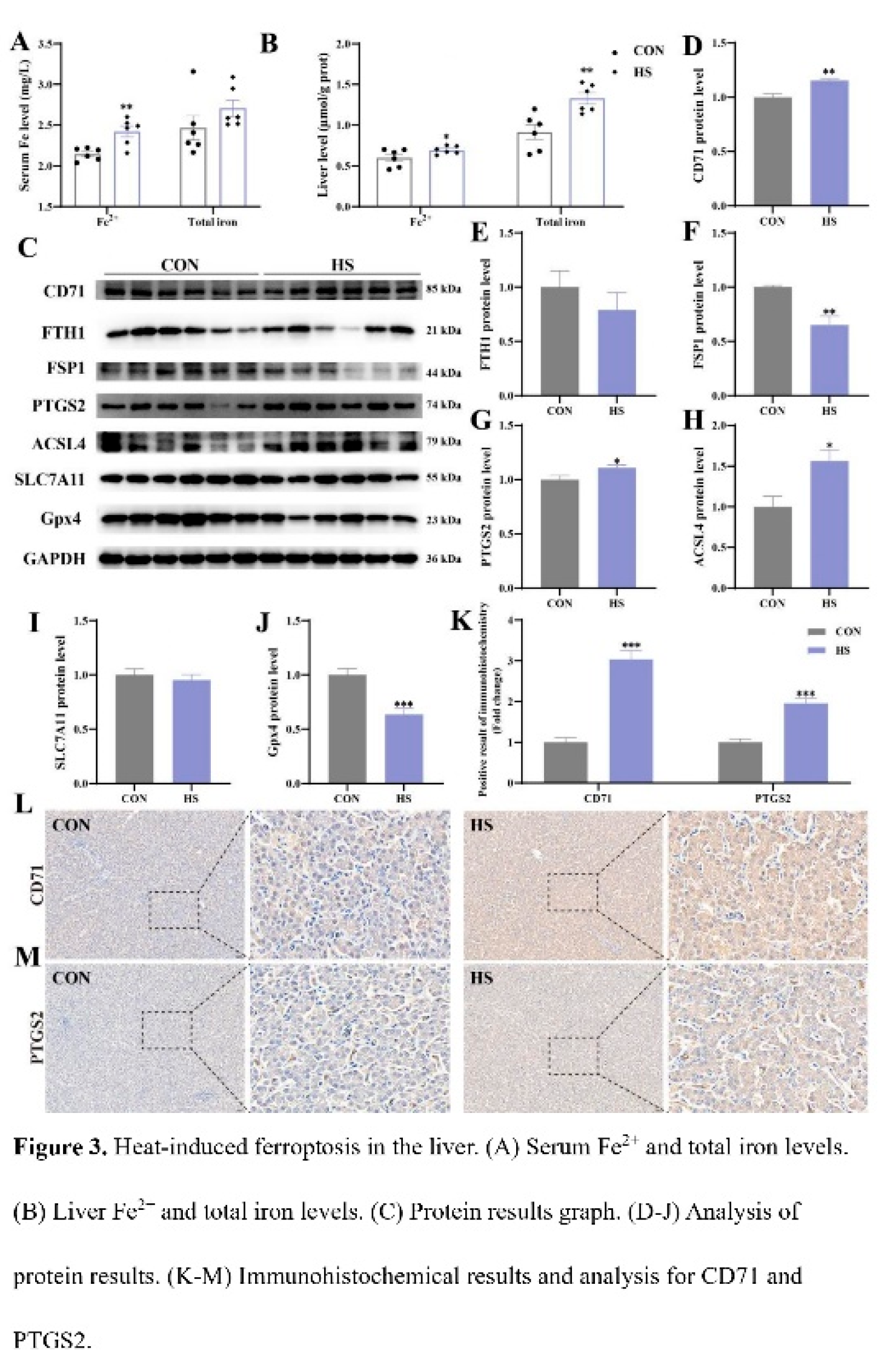
3.3. Effect of Heat Stress on Ferroptosis in Chicken Liver
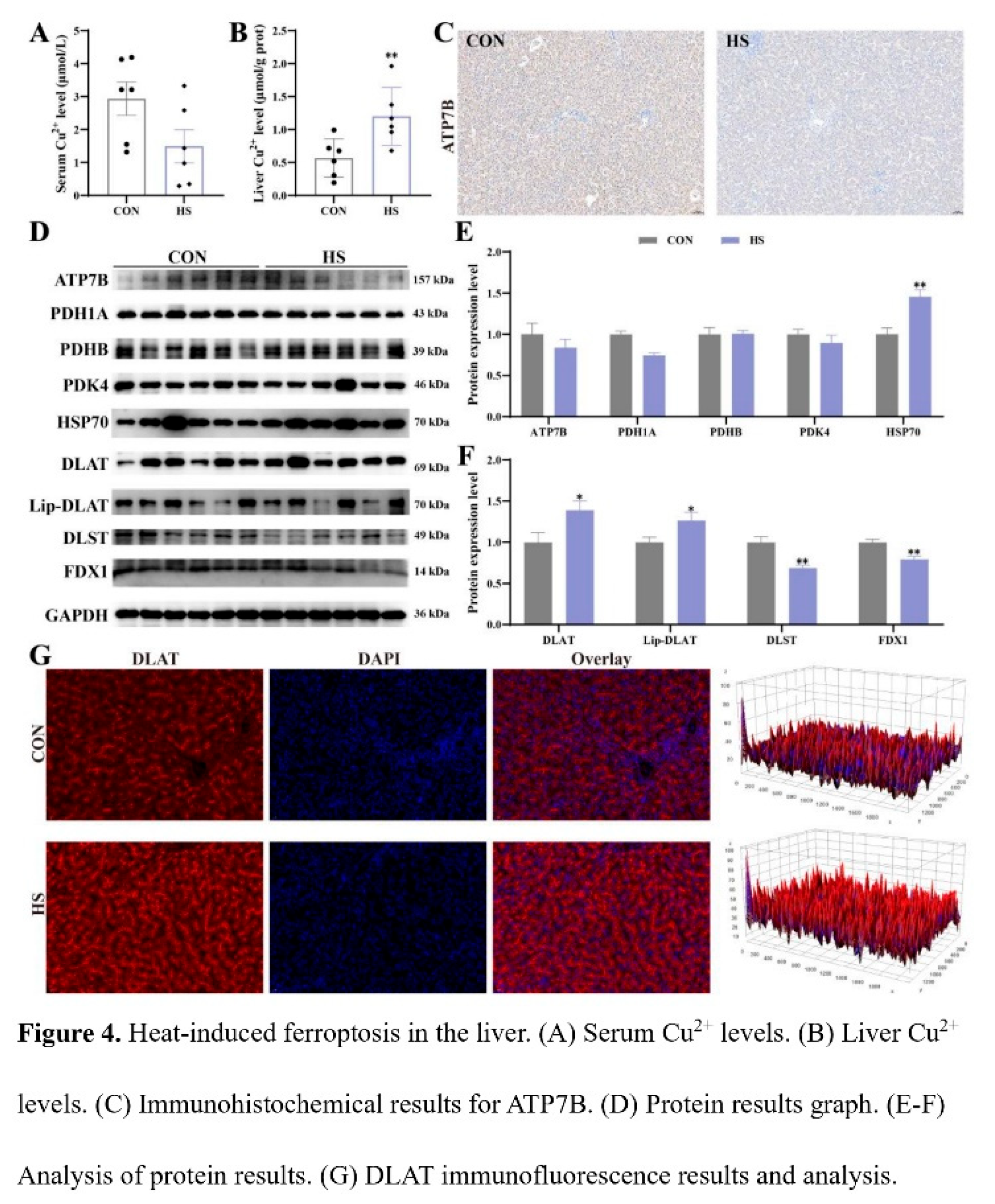
3.4. Effect of Heat Stress on Cuproptosis in Chicken Liver
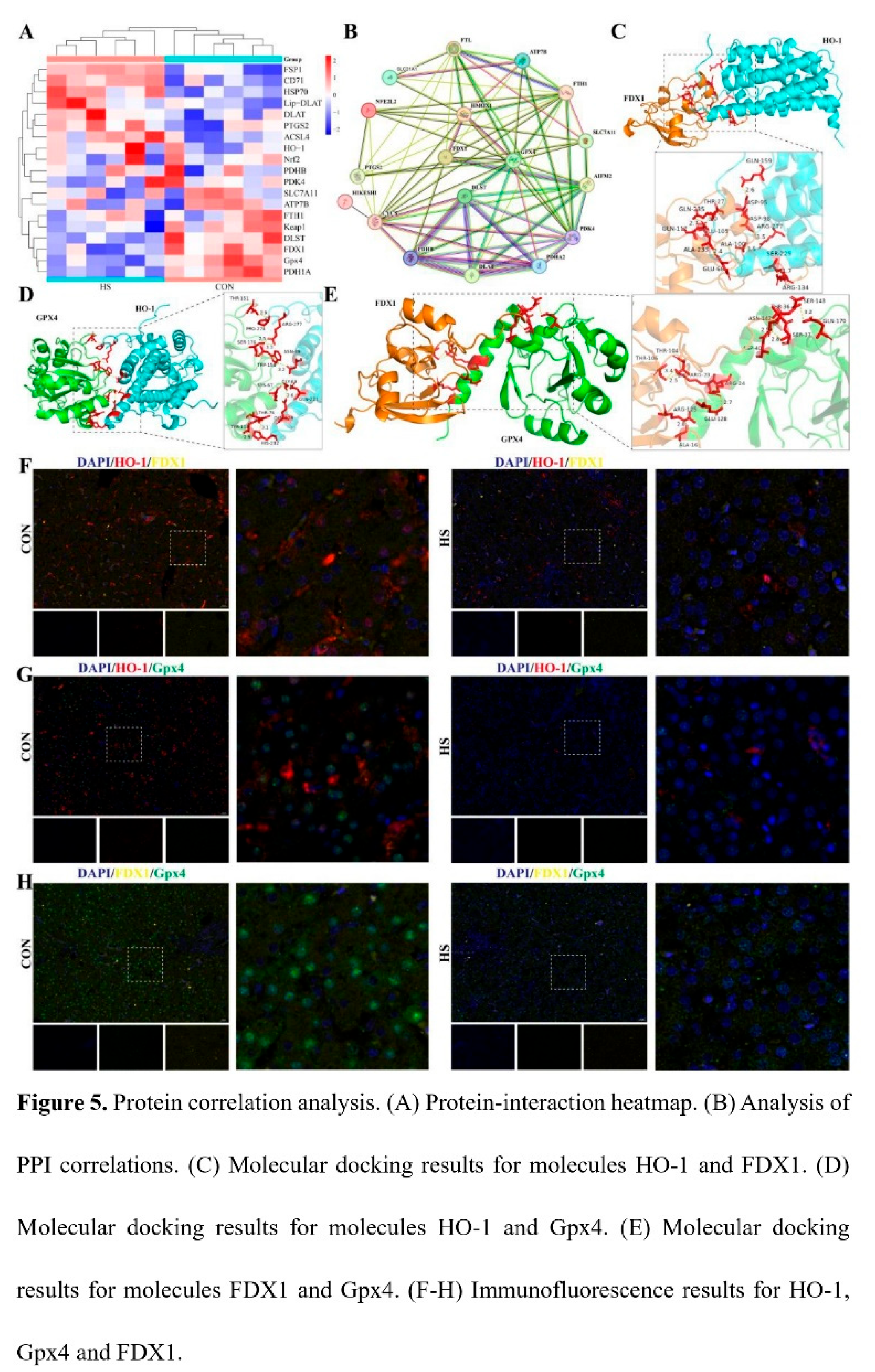
3.5. Correlation Analysis of HO-1, FDX1 and Gpx4
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Hossain, M.M.; Ahn, J.; Choi, S.Y.; Hur, S.P.; Lim, D.; Shin, D.; Lee, S.; Park, J.E. Thermal stress responses and heat stress resilience genes in chickens are revealed through genomic and transcriptomic insights. Journal of animal science and biotechnology 2026, 17, 42. [CrossRef]
- Aryal, B.; Kwakye, J.; Ariyo, O.W.; Ghareeb, A.F.A.; Milfort, M.C.; Fuller, A.L.; Khatiwada, S.; Rekaya, R.; Aggrey, S.E. Major oxidative and antioxidant mechanisms during heat stress-induced oxidative stress in chickens. Antioxidants (Basel) 2025, 14, 471. [CrossRef]
- Chen, S.; Liu, H.; Zhang, J.; Zhou, B.; He, X.; Wang, T.; Wang, C. Dietary rutin improves breast meat quality in heat-stressed broilers and protects mitochondria from oxidative attack via the AMPK/PINK1-Parkin pathway. J. Sci. Food Agric. 2023, 103, 2367-2377. [CrossRef]
- Ma, F.; Wang, M.; Zhong, G.; Liao, J.; Huo, Y.; Wang, Z.; He, S. The impact of copper-induced oxidative damage on the endoplasmic reticulum quality control system in broiler kidneys. BioMetals 2025, 38, 1131-1143. [CrossRef]
- Alqhtani, H.A.; Elbaz, A.M.; Hegazy, S.A.; Abdelhady, A.Y.; Safhi, F.A.; Marzok, M.; Rizk, M.A.; Al-Rasheed, M.; Mohamed, M.H.; Abdel-Raheem, S.M.; et al. Dietary Combined Thyme Meal and Bacillus subtilis to Promote Growth Performance, Immune Function, Gene Expression, Antioxidant Defense, and Cecal Microbiota in Growing Rabbits Under Heat Stress Conditions. Veterinary Sciences 2026, 13. [CrossRef]
- Ji, L.; Yu, H.; Wang, R.; Yan, H.; Yin, X.; Guo, S.; Wang, R. Targeting magnesium homeostasis: a novel therapeutic strategy for liver diseases. Frontiers in nutrition 2026, 13, 1709477. [CrossRef]
- Ma, F.; Huo, Y.; Liao, J.; Xu, G.; Wang, Z.; He, S. The role of lipid metabolism imbalance in copper-induced PANoptosis in broiler kidney. Poult. Sci. 2025, 104, 105549. [CrossRef]
- Qin, W.; Sheng, H.; Hu, X.; Burden, R.E.; Martin, S.L.; Wu, H. Cuproptosis in cancer: emerging mechanism and therapeutic opportunities. Trends Pharmacol. Sci. 2026, 47, 386-402. [CrossRef]
- Siquier-Coll, J.; Bartolomé, I.; Perez-Quintero, M.; Grijota, F.J.; Arroyo, J.; Muñoz, D.; Maynar-Mariño, M. Serum, erythrocyte and urinary concentrations of iron, copper, selenium and zinc do not change during an incremental test to exhaustion in either normothermic or hyperthermic conditions. J. Therm. Biol. 2019, 86, 102425. [CrossRef]
- Tsvetkov, P.; Coy, S.; Petrova, B.; Dreishpoon, M.; Verma, A.; Abdusamad, M.; Rossen, J.; Joesch-Cohen, L.; Humeidi, R.; Spangler, R.D.; et al. Copper induces cell death by targeting lipoylated TCA cycle proteins. Science 2022, 375, 1254-1261. [CrossRef]
- Xiong, W.; Luo, Z.; Wang, H.; Su, Q.; Wang, H.; Xu, J.; Zheng, W. Amyloid beta 42 disrupts cardiac function in Alzheimer's disease mice via SLC31A1 upregulation-mediated cuproptosis. Basic Res. Cardiol. 2026, 121, 285-299. [CrossRef]
- Cui, S.; Ye, J. Ferroptosis: the demise of cells through phospholipid peroxidation. Advanced science (Weinheim, Baden-Wurttemberg, Germany) 2026, 20, e24387. [CrossRef]
- Wu, Y.; Fu, L.; Xu, X.; Chan, P.S.; Wu, R. Systematic and quantitative investigation of newly synthesized proteins reveals distinct ion homeostasis and mitochondrial changes between cuproptosis and ferroptosis in human cells. Anal. Chem. 2026, 98, 8309-8320. [CrossRef]
- Hao, L.; Yu, P.; Yan, R.; Li, K.; Luo, Z.; Xiang, S.; Wang, Y.; Fang, C.; Wang, G.; Ma, S.; et al. Transdermal siRNA delivery via biomineralized nanoparticle-incorporated microneedles modulates cuproptosis-ferroptosis interaction for psoriasis therapy. Materials today. Bio 2026, 37, 102991. [CrossRef]
- Zhang, Y.; Li, Y.; Wang, R.; Wang, S.; Sun, B.; Cao, D.; Sun, Z.; Lv, W.; Ma, B.; Zhang, Y. Chronic heat stress caused lipid metabolism disorder and tissue injury in the lliver of Huso dauricus via oxidative-stress-mediated ferroptosis. Antioxidants (Basel) 2025, 14, 92. [CrossRef]
- Huang, X.; Yu, S.; Wei, W.; Tao, W.; Cai, T.; Wen, L.; Ye, J.; Zhang, C.; Feng, H.; Cao, S.; et al. Fe-S protein FDX1 triggers tumor-intrinsic innate immunity via mitochondrial nucleic acids release to orchestrate ferroptosis in CCRCC. Advanced science (Weinheim, Baden-Wurttemberg, Germany) 2026, 13, e18323. [CrossRef]
- Ding, J.; Miao, J.; Zhang, X.; Xiong, Y.; Ma, F.; He, S. Total saponins of Gynostemma pentaphyllum mitigate chronic heat stress-induced thymus and spleen inflammation in broilers via NF-κB pathway activation. Poult. Sci. 2026, 105, 106797. [CrossRef]
- Xu, G.; Ma, F.; Li, Y.; Miao, J.; Zhang, X.; Xiong, Y.; He, S. Puerarin alleviates liver injury induced by heat-stress through regulating oxidative stress and apoptosis in broilers. J. Therm. Biol. 2025, 134, 104317. [CrossRef]
- Zhang, T.; Dai, X.; Wang, H.; Hu, Y.; Gao, X.; Shu, X.; Cao, H.; Hu, G.; Xing, C.; Yang, F. Copper-induced duodenal injury: Unveiling the dual role of cuproptosis and ferroptosis via the FDX1/GPX4 axis. J. Nutr. Biochem. 2026, 149, 110181. [CrossRef]
- Tolba, S.A.; Amer, S.A.; Abdel-Warith, A.A.; Younis, E.M.; Gouda, A.; Elshopakey, G.E.; Abd-Allah, N.A.; Reda, R.; Hamed, R.I.; Saleh, G.K.; et al. Fennel essential oil as a natural feed additive to mitigate heat stress in broilers: Effects on growth, physiological and immunological responses, and intestinal morphology. J. Therm. Biol. 2026, 137, 104437. [CrossRef]
- Zendeli, D.; Colaninno, N.; van Esch, M.; Eldesoky, A.H.; Morello, E.; van Timmeren, A. Urban heat stress and health: A systematic literature review of dimensions and indicators for planning and design. Health Place 2026, 99, 103643. [CrossRef]
- Pradhan, P.; Foresti, R.; Motterlini, R.; Immenschuh, S. Regulatory interplay between nitric oxide and heme in redox signaling and inflammation. Redox Biol 2026, 92, 104126. [CrossRef]
- García-Yagüe Á, J.; Cazalla, E.; Cuadrado, A. Therapeutic potential of isothiocyanates by targeting the NRF2 pathway. Free Radic. Biol. Med. 2026, 248, 554-596. [CrossRef]
- Lan, T.; Zhou, K.; Duan, G.; Zhang, Y.; Ma, Y.; Fan, G.; Qi, Y. Lycium L. flavonoids: extraction, purification, signal transduction pathways, and interactions with intestinal microbiota. NPJ science of food 2026, 10.1038/s41538-026-00784-w. [CrossRef]
- Durço, A.O.; da Silva Alcântara, F.; Rocha, C.S.G.; Mota, K.O.; Conceição, L.S.R.; Orts, D.; Sousa, G.M.; Santos, M.R.V.; Souza, D.S.; Roman-Campos, D. Pyrethroid-induced cardiac dysfunction: A systematic review and meta-analysis of preclinical evidence. Chem. Biol. Interact. 2026, 432, 112030. [CrossRef]
- Ding, K.N.; Lu, M.H.; Guo, Y.N.; Liang, S.S.; Mou, R.W.; He, Y.M.; Tang, L.P. Resveratrol relieves chronic heat stress-induced liver oxidative damage in broilers by activating the Nrf2-Keap1 signaling pathway. Ecotoxicol. Environ. Saf. 2023, 249, 114411. [CrossRef]
- Yuan, J.; Li, Y.; Miao, J.; Zhang, X.; Xiong, Y.; Ma, F.; Ding, J.; He, S. Bamboo leaf flavonoids ameliorate cyclic heat stress-induced oxidative damage in broiler liver through activation of Keap1-Nrf2 signaling pathway. Poult. Sci. 2025, 104, 104952. [CrossRef]
- Lei, Y.; Liu, H.; Xiang, Q.; Liu, Y.; Wu, D.; Zhang, J.; Lin, Y. Dietary lycopene mitigates reproductive impairment in heat-stressed rongchang boars: roles of antioxidant, anti-inflammatory and Nrf2 pathway. Antioxidants (Basel) 2026, 15, 245. [CrossRef]
- Kuninaka, Y.; Ishida, Y.; Grimaldi, F.; Irmici, M.; Nosaka, M.; Munekawa, M.; Pelotti, S.; Fais, P.; Kondo, T. 8-OHdG and NT-proBNP as complementary biomarkers in the postmortem diagnosis of acute ischemic heart disease. Sci. Rep. 2026, 16, 6154. [CrossRef]
- Liu, Y.; Chang, Y.Z. The regulation of mitochondrial ferritin on mitochondrial redox balance is essential to cell fate decision. Redox Biol 2026, 92, 104124. [CrossRef]
- McKay, A.K.A.; McCormick, R.; Tee, N.; Peeling, P. Exercise and heat stress: inflammation and the iron regulatory response. Int. J. Sport Nutr. Exerc. Metab. 2021, 31, 460-465. [CrossRef]
- An, Q.; Wei, R.; Huang, Z.; Tang, Y.; Wang, M.; He, S.; Huang, K.; Liu, Z.; Zhang, M.; Li, R.; et al. Palmitoylation of Tfr1 enhances platelet ferroptosis and liver injury in heat stroke. Acta pharmaceutica Sinica. B 2026, 16, 270-286. [CrossRef]
- Caverzan, M.D.; Ibarra, L.E. Advancing glioblastoma treatment through iron metabolism: A focus on TfR1 and Ferroptosis innovations. Int. J. Biol. Macromol. 2024, 278, 134777. [CrossRef]
- Wang, J.; Guo, D.; Jiang, S.; Wu, W.; Gao, X. Targeting ferroptosis in cancer: from mechanistic insights to therapeutic approaches. Molecular biomedicine 2026, 7, 20. [CrossRef]
- Jiang, M.; Zhang, L.; Lin, Q.; Guo, D.; Jin, Q.; Xu, J.; Guo, M.; Peng, S.; Sun, Q.; Liu, N.; et al. Endoplasmic reticulum-targeting prodrug nanoassemblies for potent ferroptosis therapy via concurrent glutathione depletion and arachidonic acid accumulation. Acta Biomater. 2026, 214, 542-556. [CrossRef]
- Chen, H.; Dai, X.; Xiong, Z.; Cao, H.; Xing, C.; Li, H.; Gao, X.; Hu, M.; Yang, F. Dual-pathway mechanism of vanadium-induced hepatotoxicity in ducks: Synergistic crosstalk between glucose homeostasis disruption and NADH/FSP1/COQ10 axis-driven ferroptosis. Int. J. Biol. Sci. 2026, 22, 43-59. [CrossRef]
- Liang, Q.H.; Liu, Q.Q.; Tian, S.Z.; Yao, Q.H.; Ye, X.Q.; Liu, W.C. Dietary fucoidan supplementation ameliorates heat stress-induced liver injury in broilers via modulating peroxidation, lipid metabolism, and ferroptosis. Poult. Sci. 2026, 105, 106489. [CrossRef]
- Zhao, Z.X.; Yuan, Y.M.; Zhao, Z.H.; Yao, Q.H.; Ye, X.Q.; Wang, Y.Y.; Liu, H.M.; Jha, R.; Balasubramanian, B.; Liu, W.C. Phlorotannin alleviates liver injury by regulating redox balance, apoptosis, and ferroptosis of broilers under heat stress. Antioxidants (Basel) 2024, 13, 1048. [CrossRef]
- Yan, L.; Zheng, H.; Chen, K.; Fang, M.; Ren, F.; Huang, T.; Cai, W.; Huang, K.; Wu, X.; Zheng, F. The copper transporter protein SLC31A1-mediated copper metabolism imbalance drives cuproptosis through the SIRT1/HMGB1 pathway in heart failure. Chem. Biol. Interact. 2026, 431, 112006. [CrossRef]
- Ma, G.; Li, Y.; Li, X.; Yu, D.; Chao, M.; Li, J.; Guo, J.; Wang, K.; Liu, Z.; Sun, R.; et al. Multi-ion channel nanomedicines targeting zinc transporter 1/ATPase copper transporters disrupt copper/iron homeostasis to enhance tumor immunotherapy. ACS nano 2026, 10.1021/acsnano.6c00358. [CrossRef]
- Xu, L.; Feng, J.; Xu, S.; Jin, M.; Bai, X.; Zhang, H.; Zhu, M.; Lin, S.; Song, J.; Lu, C. GSH-responsive triple-action photosensitizer nanoplatforms orchestrate cuproptosis-ferroptosis synergy to potentiate antitumor PDT efficacy. Journal of nanobiotechnology 2026, 10.1186/s12951-026-04331-3. [CrossRef]
- Lu, Y.; Wu, Y.; Yang, C.; Zhou, Y.; Ren, X.; Li, H.; Huang, H.; Pan, F.; Deng, A.; Lu, Y.; et al. Ferredoxins: master regulators in mitochondrial redox homeostasis and programmed cell death. Redox Biol 2025, 88, 103930. [CrossRef]
- Ding, S.; Hu, D.; Pan, Y.; Wang, J.; Wang, L. Vitisin A inhibits liver fibrosis by promoting Nrf2/HO-1 pathway and inhibiting Cuproptosis. Sci. Rep. 2025, 15, 44186. [CrossRef]
- Zha, Y.; Li, H.; Jiang, S.; Wang, Z.; Liu, Y.; Wei, Z.; Yang, Y. Research on the mechanism of Zuogui Wan in ameliorating oligoasthenozoospermia in rats via modulating cuproptosis-related pathways. J. Ethnopharmacol. 2026, 359, 120990. [CrossRef]
- Xu, F.; Xie, L.; Zhang, Z.; Yang, X.; Xu, X. Nicotine suppresses ferroptosis in colon cancer cells via HMOX1/NF-κB pathway to reduce oxaliplatin sensitivity. Apoptosis 2026, 31, 102. [CrossRef]
- Jin, C.; Li, J.; Han, S.; Bai, C.; Yang, J.; Wang, Z. HELLS inhibits autophagy-dependent ferroptosis in nasopharyngeal carcinoma by modulating the Nrf2/HO-1/GPX4 pathway. Int. J. Mol. Med. 2026, 57. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



