Submitted:
22 April 2026
Posted:
23 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Problem Statement and Scope of this Study
3. Literature Review
3.1. Internal Measures
3.1.1. Direct Injection and Injection Timing
3.1.2. Exhaust Gas Recirculation (EGR)
3.1.3. Miller Cycle
3.2. Passive External Measures
3.2.1. Constructive Measures
3.2.2. Material Development
3.3. Active External Measures
3.3.1. Electric Heating Systems
3.3.2. Fuel Burner
4. Methodology
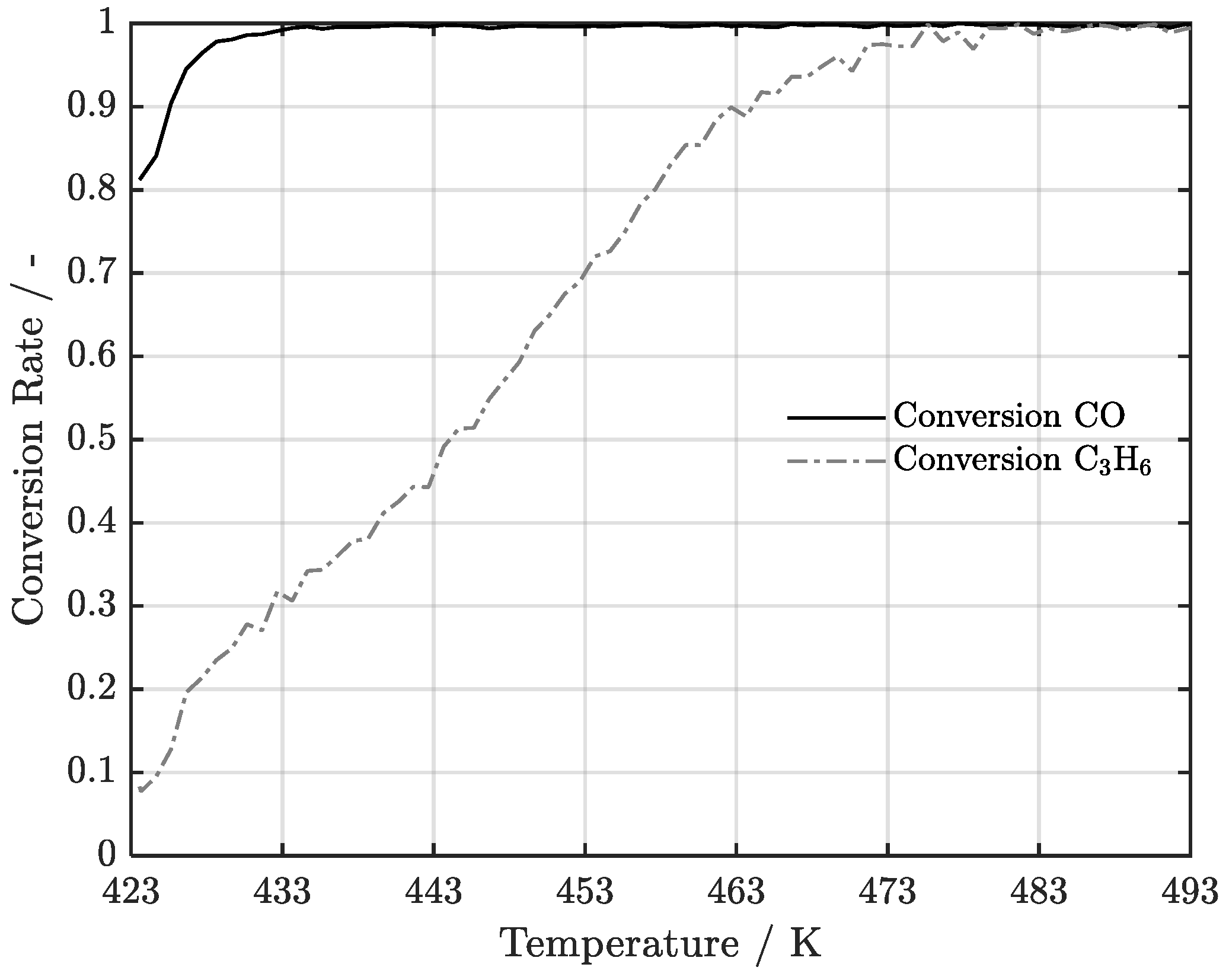
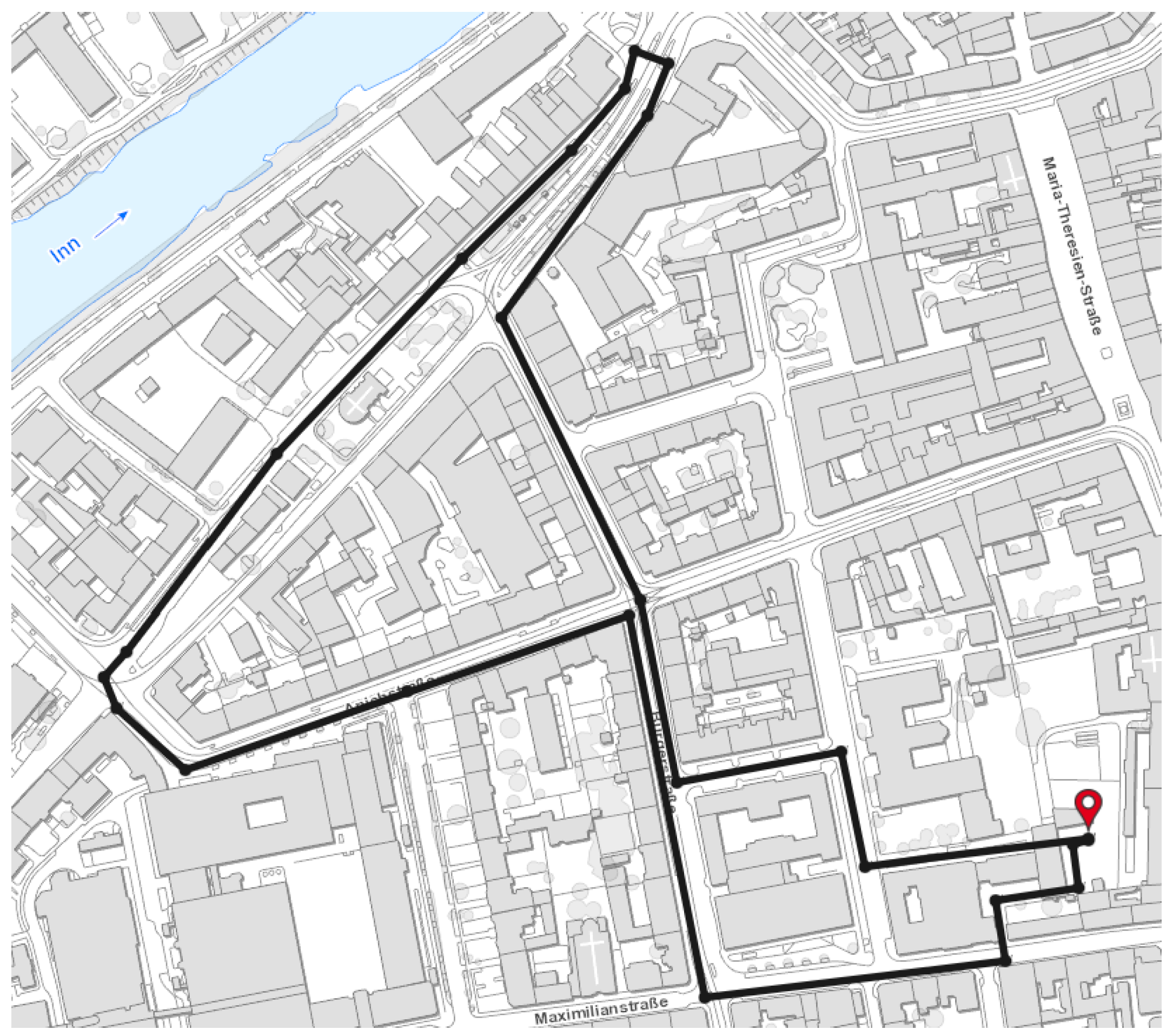
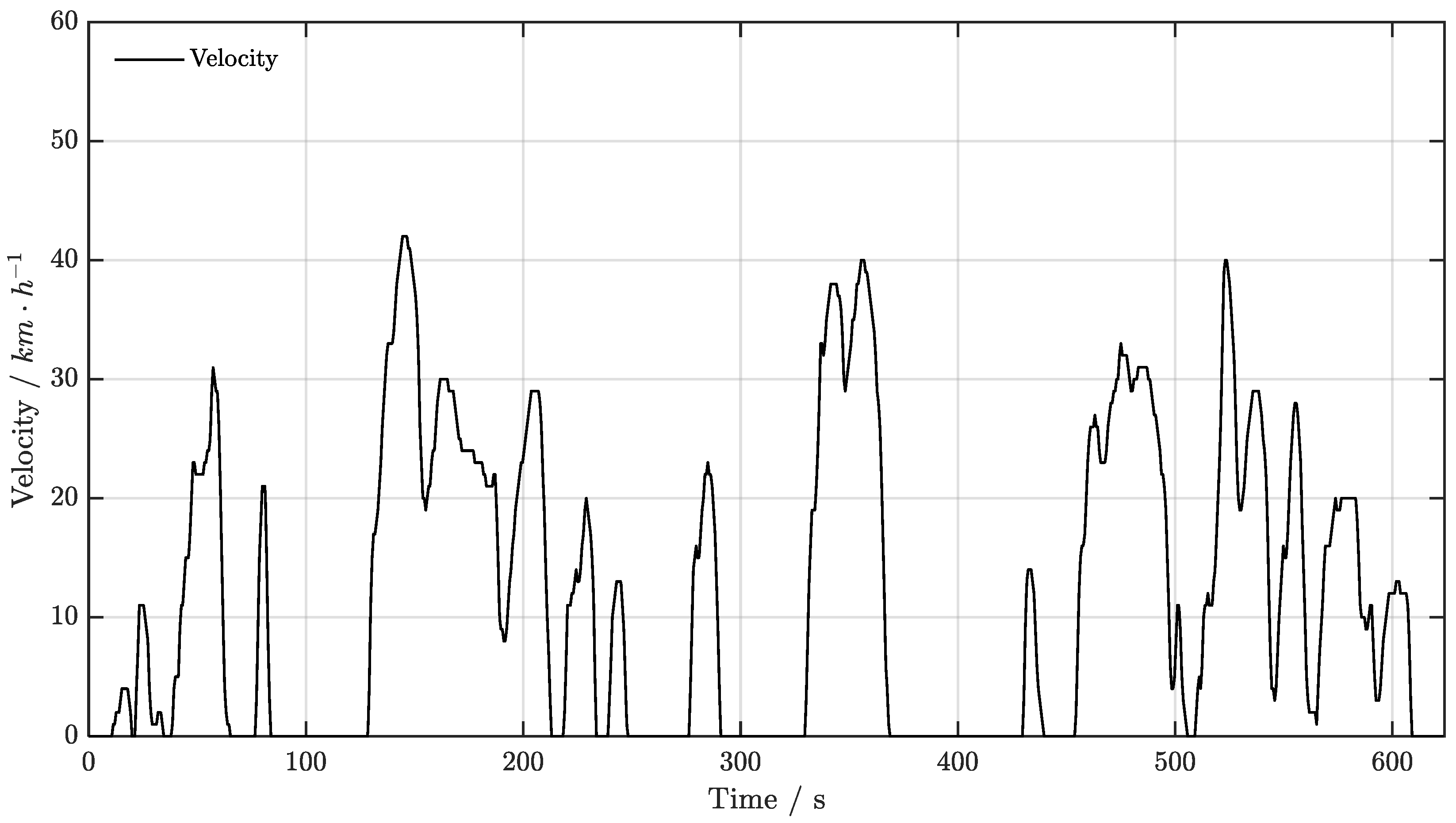
4.1. Experimental Data Acquisition
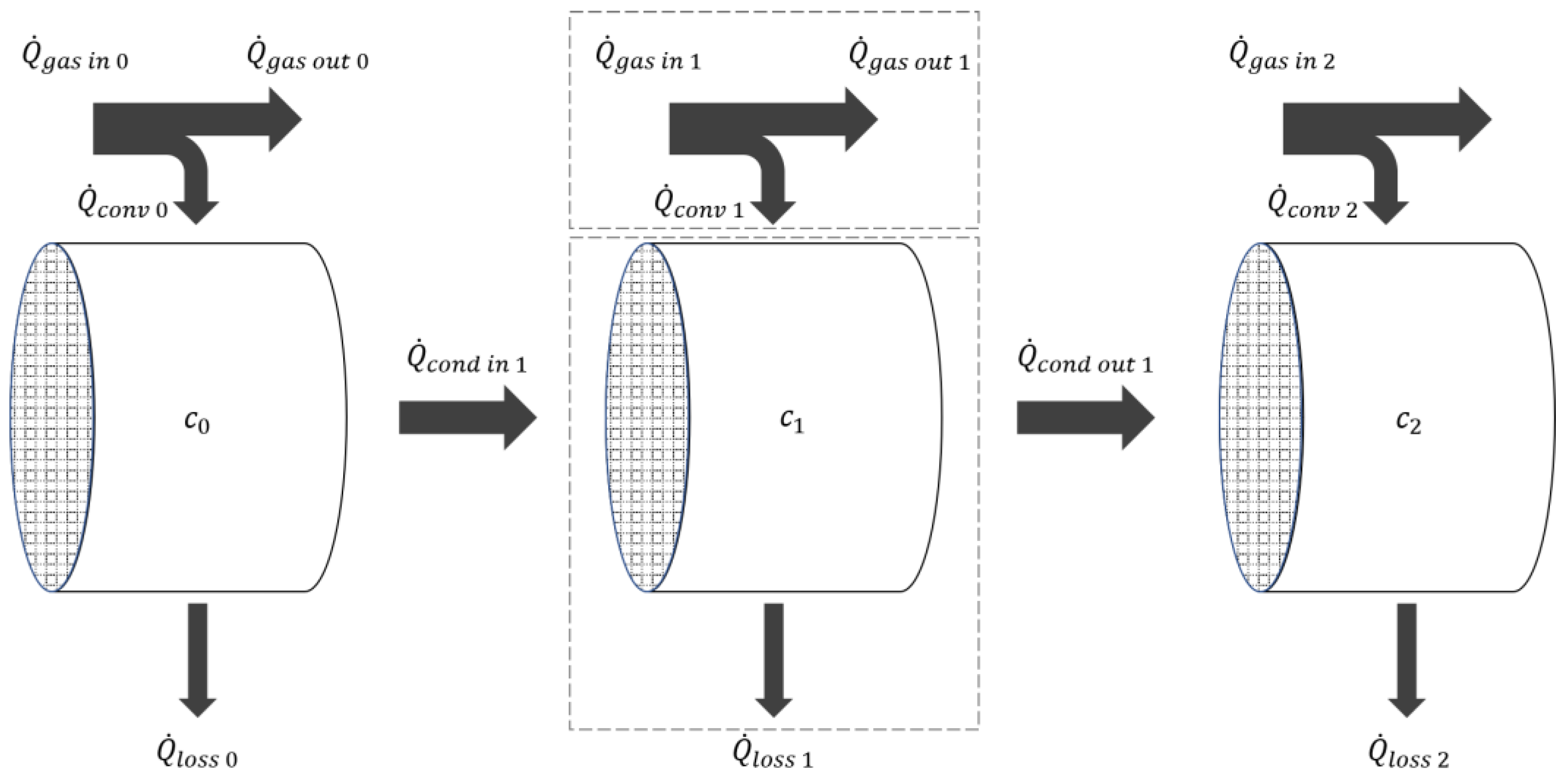
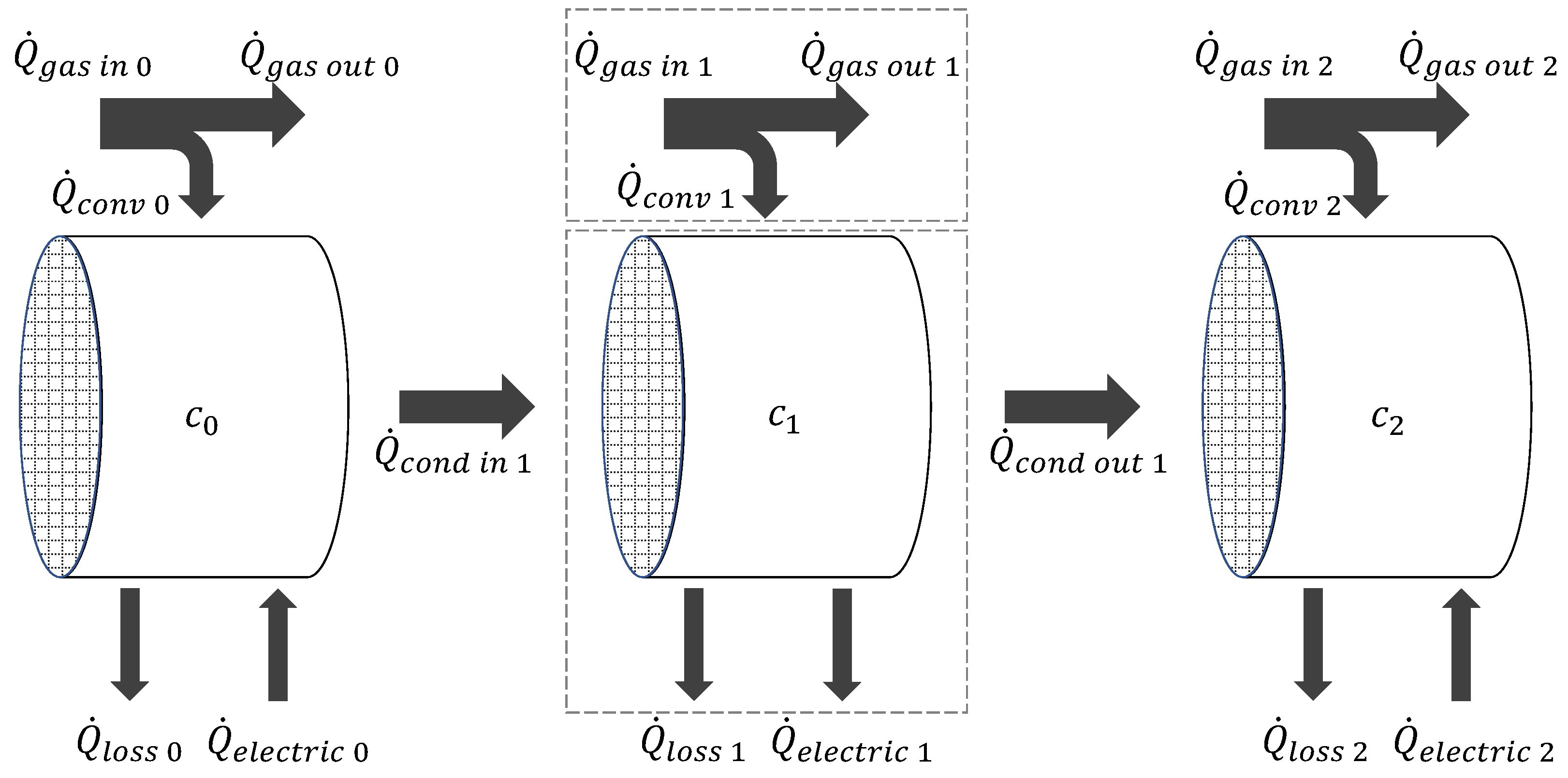
4.2. Simulation Model
- The Converters are placed in a housing, which is designed to offer adiabatic conditions, thereby precluding the possibility of heat loss to the environment.
- The monolith is uniformly considered to be cordierite, with the corresponding material values.
- The air pressure is assumed to be constant at 101,325 Pa.
- The exhaust gas is assumed as ideal gas with a gas constant of Rsp = 287 J kg-1 K-1.
- It is assumed that the cells are homogeneous, meaning that no temperature gradient is present along the cell surface.
- The reaction processes have no impact on the thermal balance.
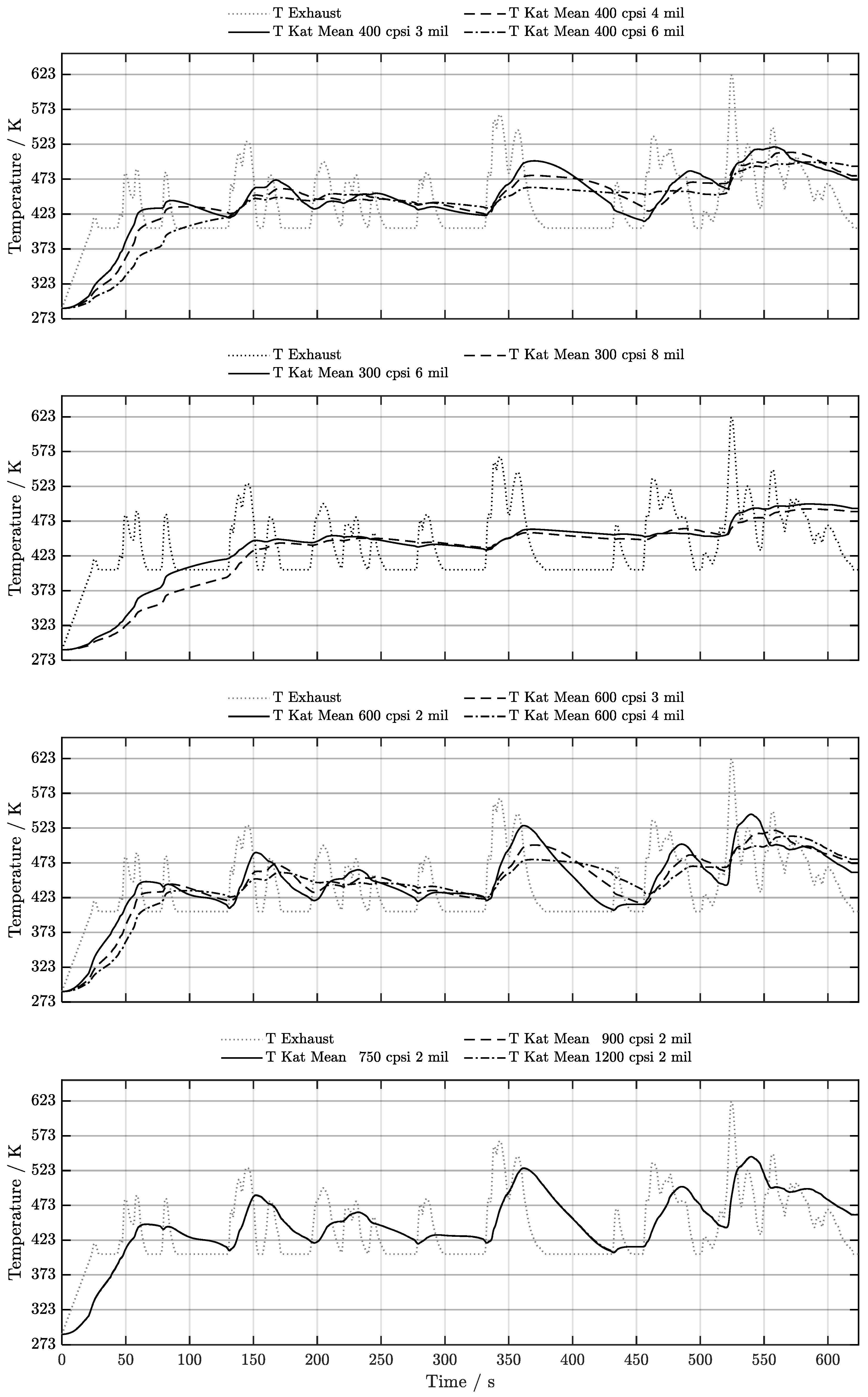
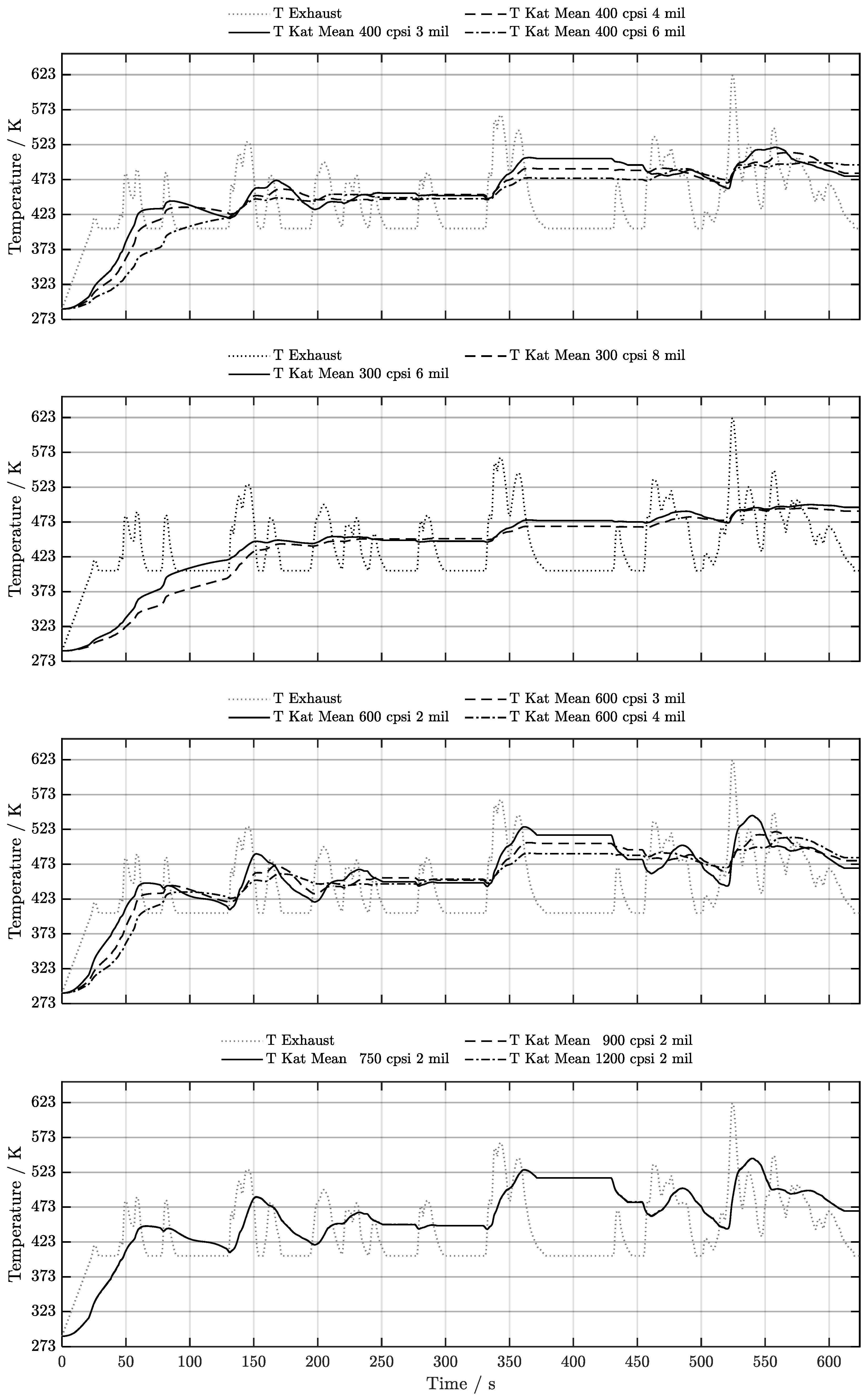
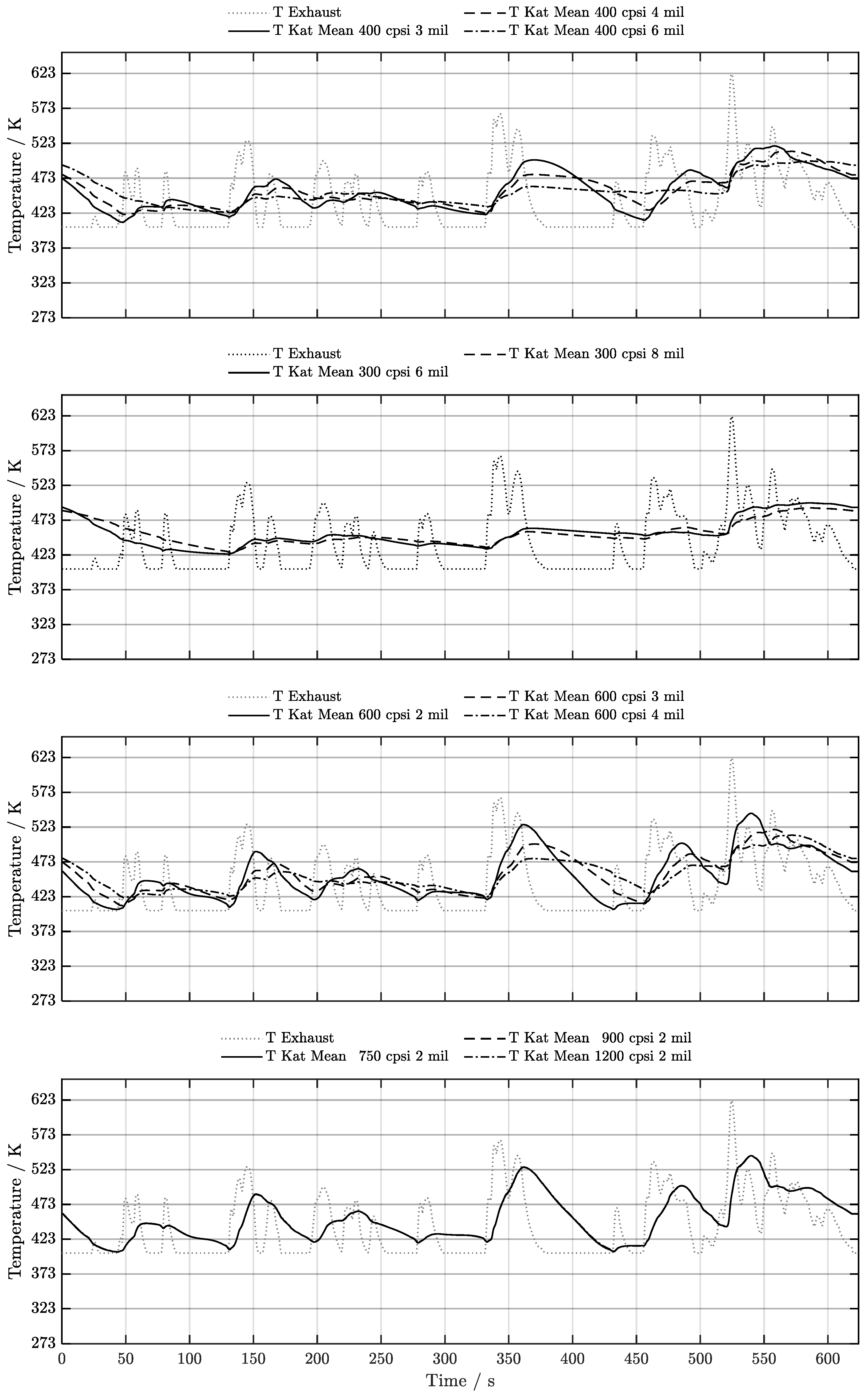
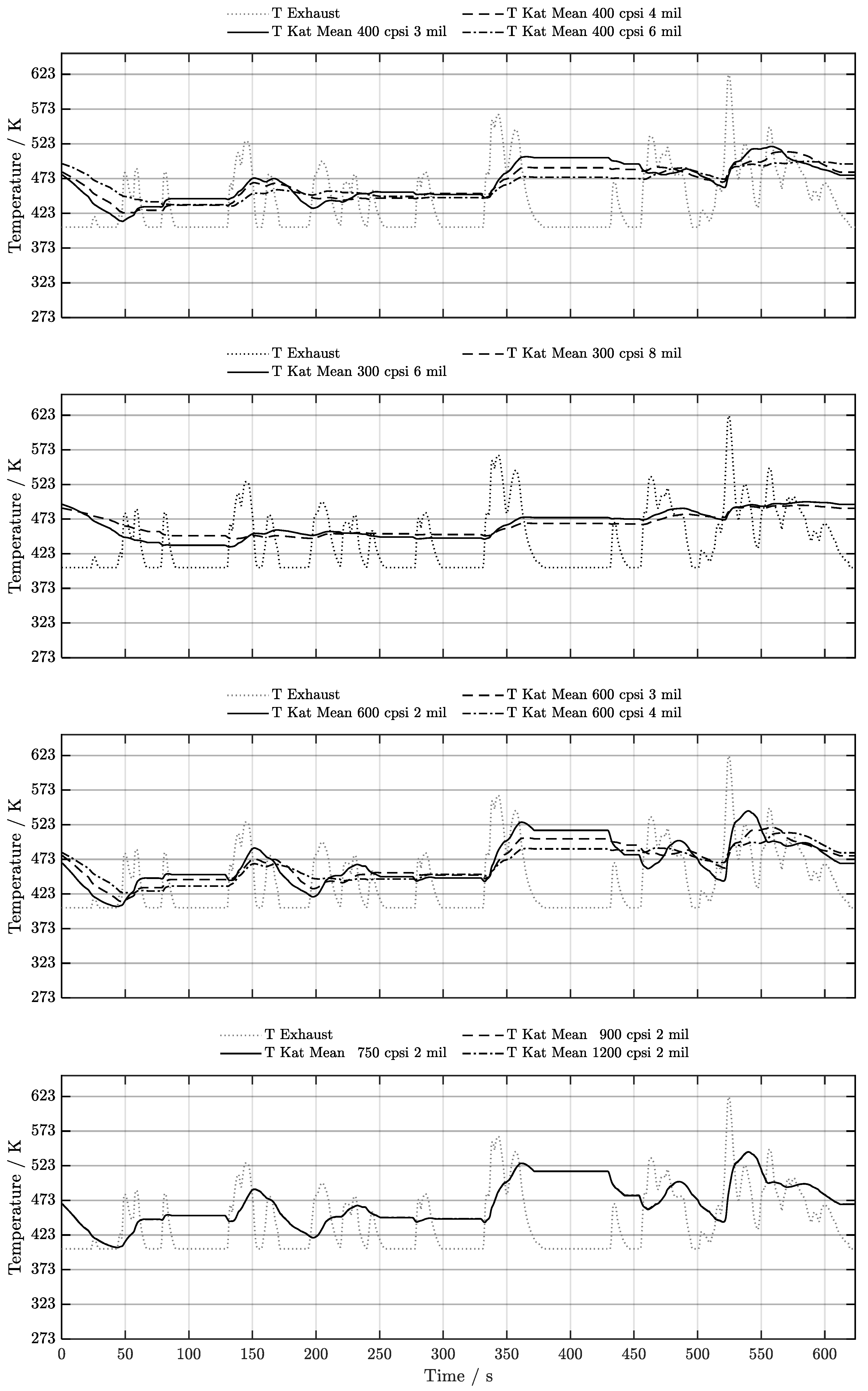
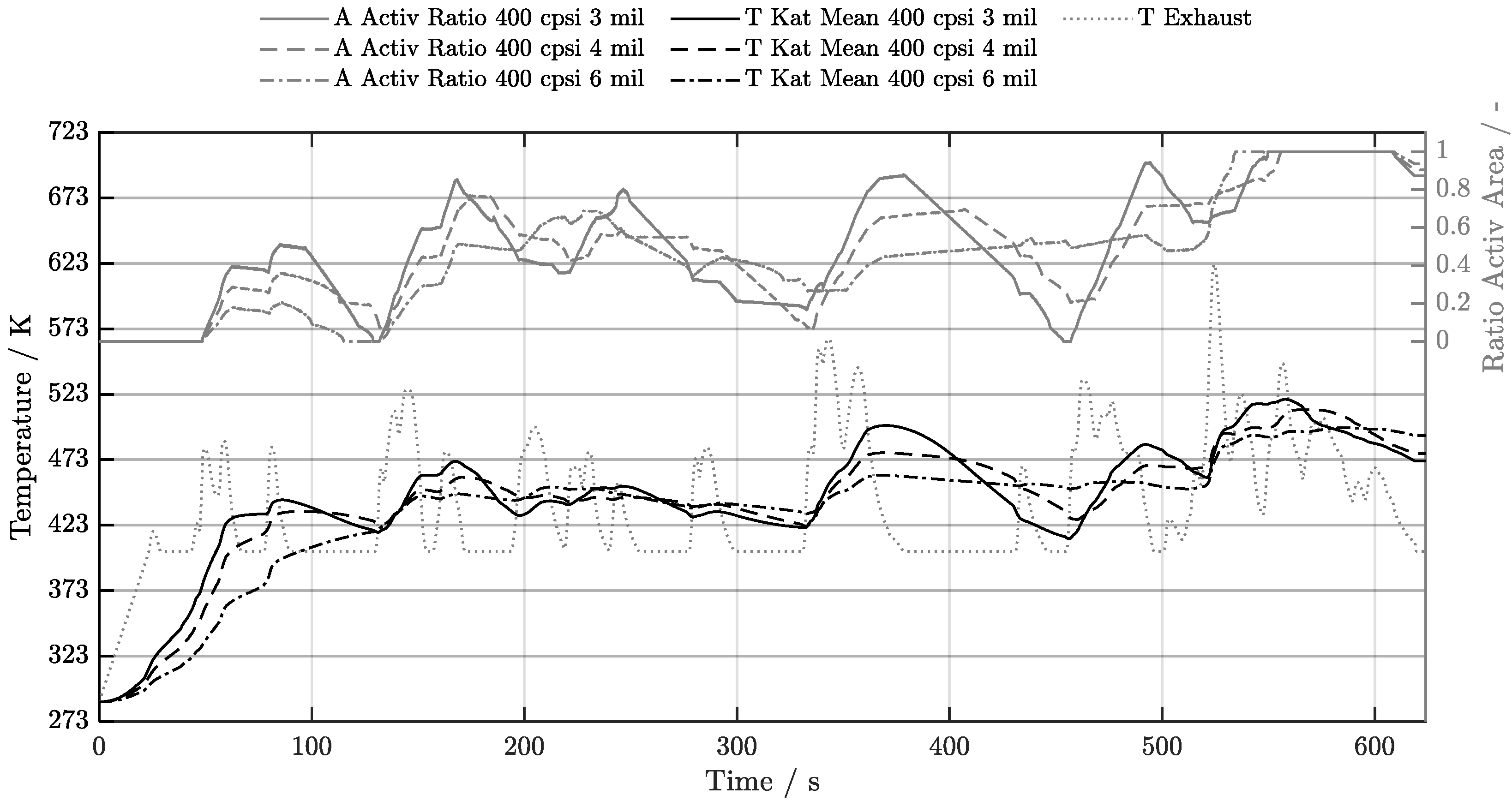
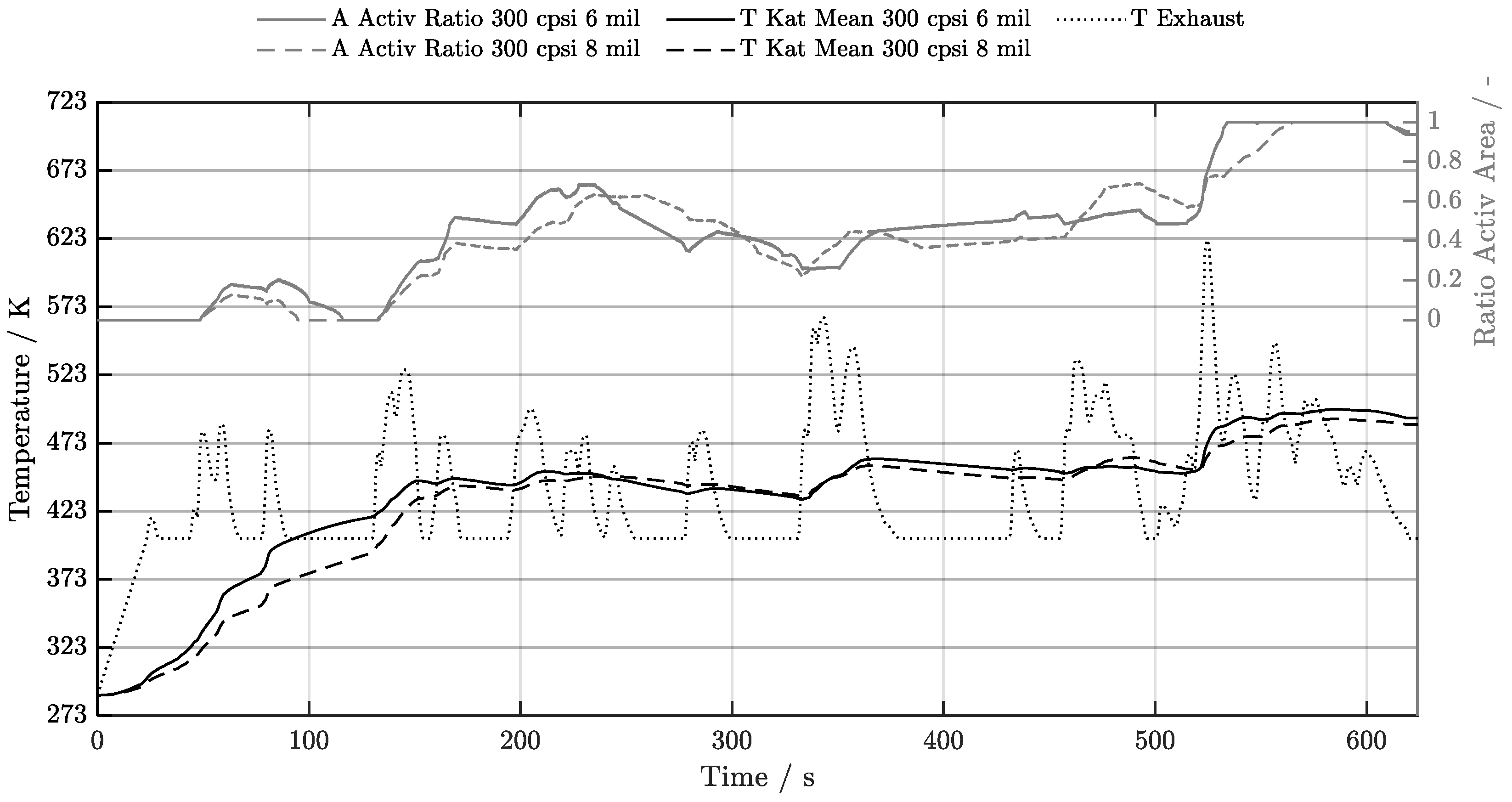
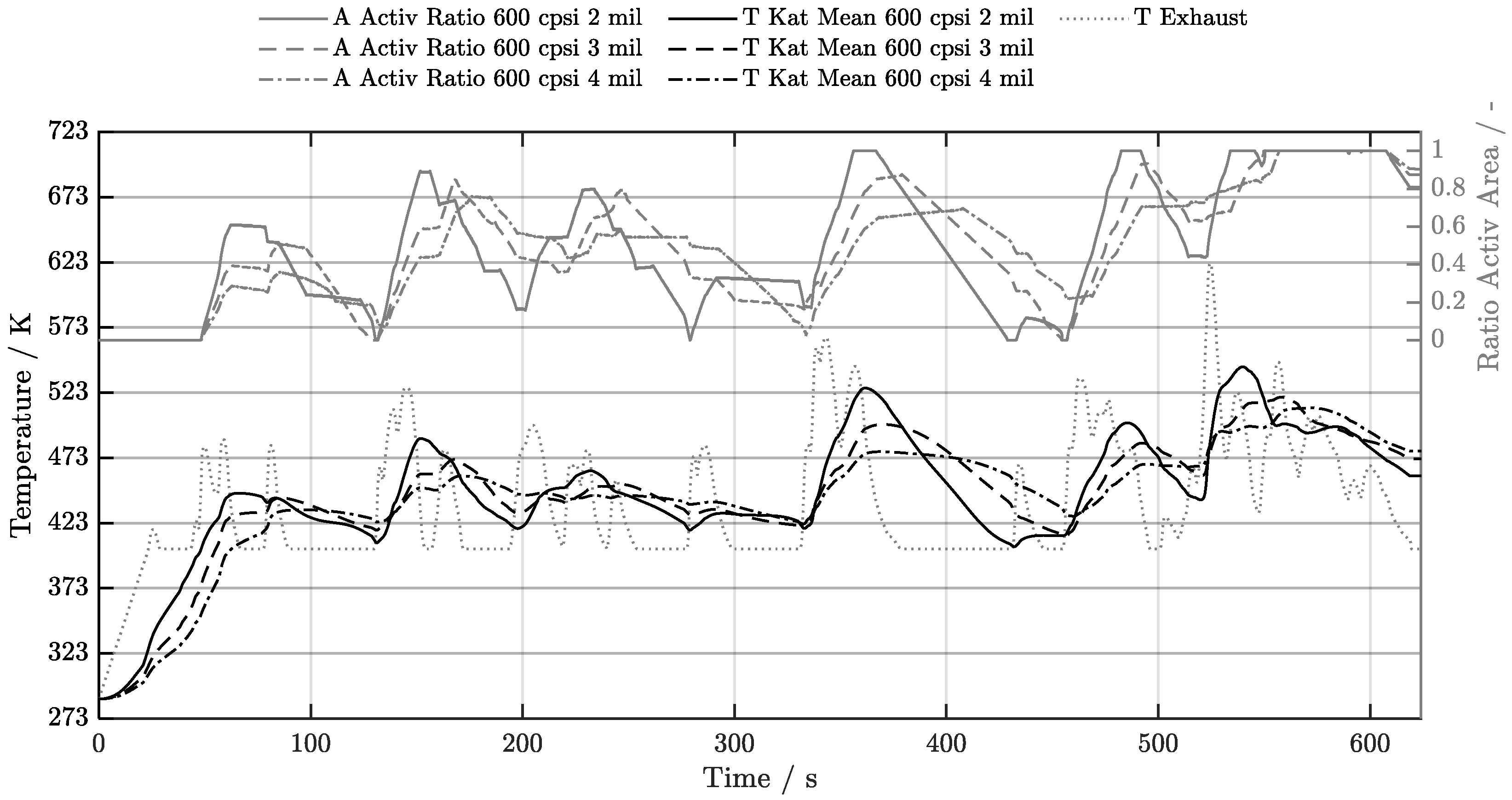
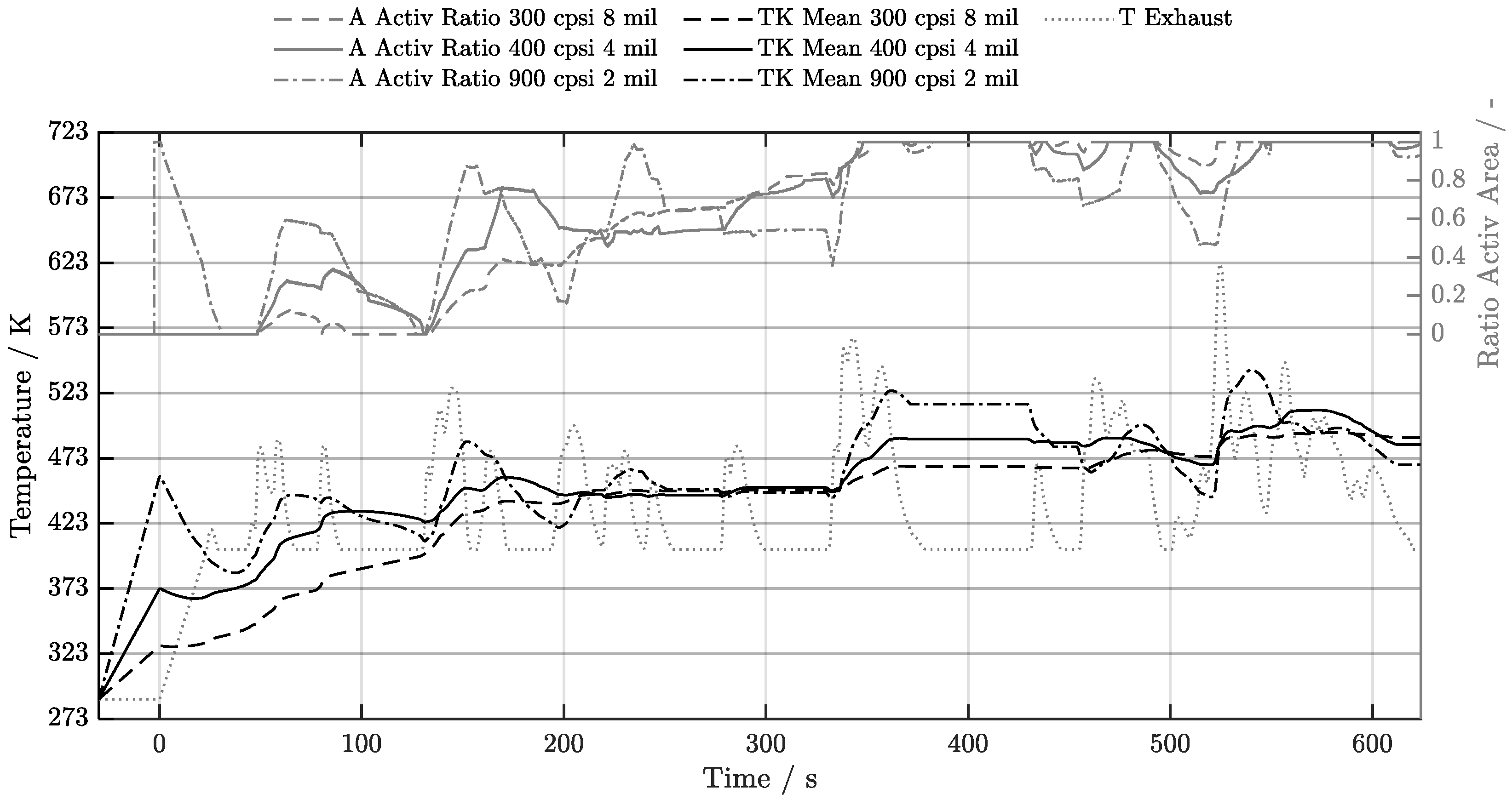
5. Discussion of Results
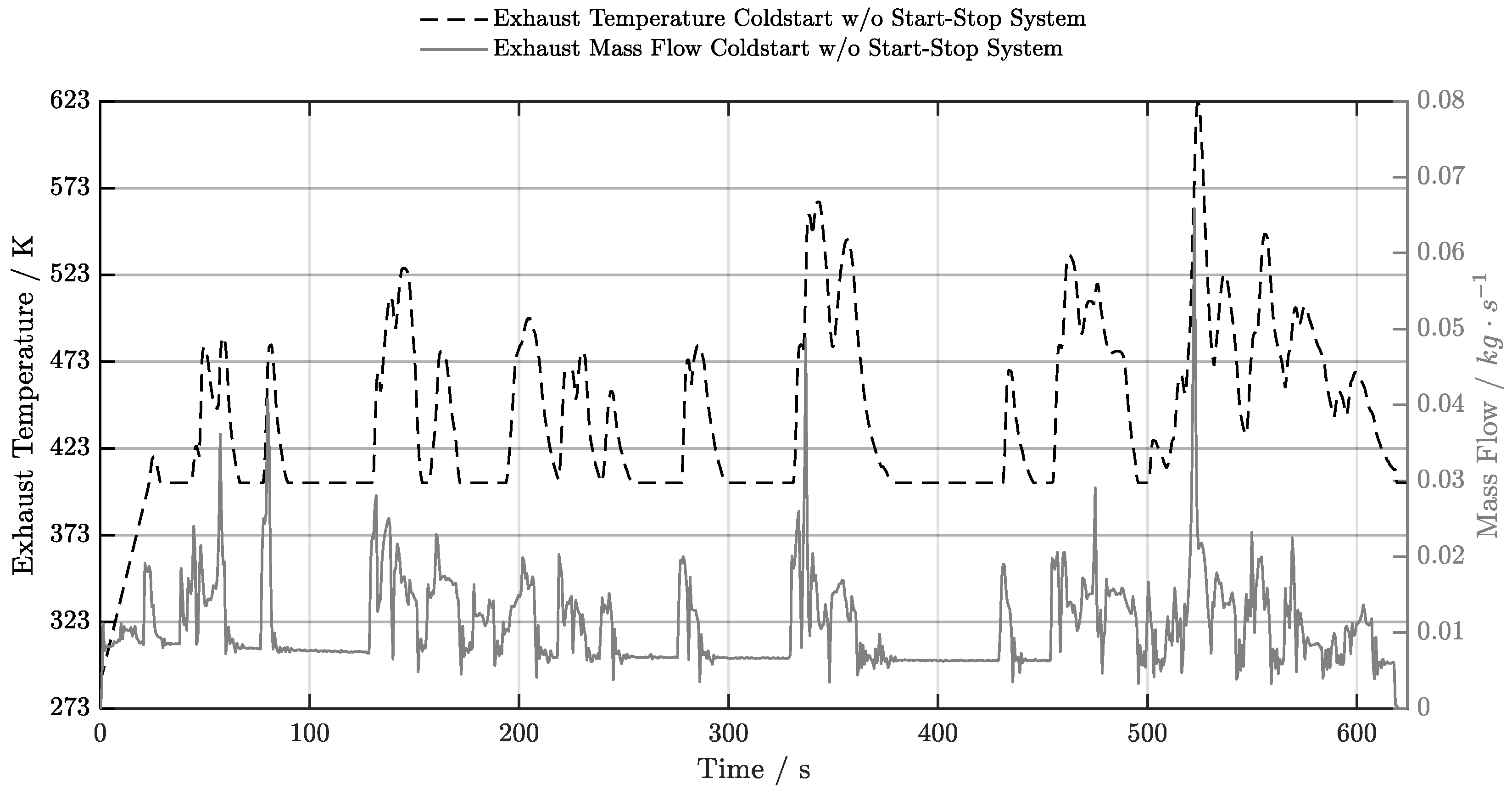
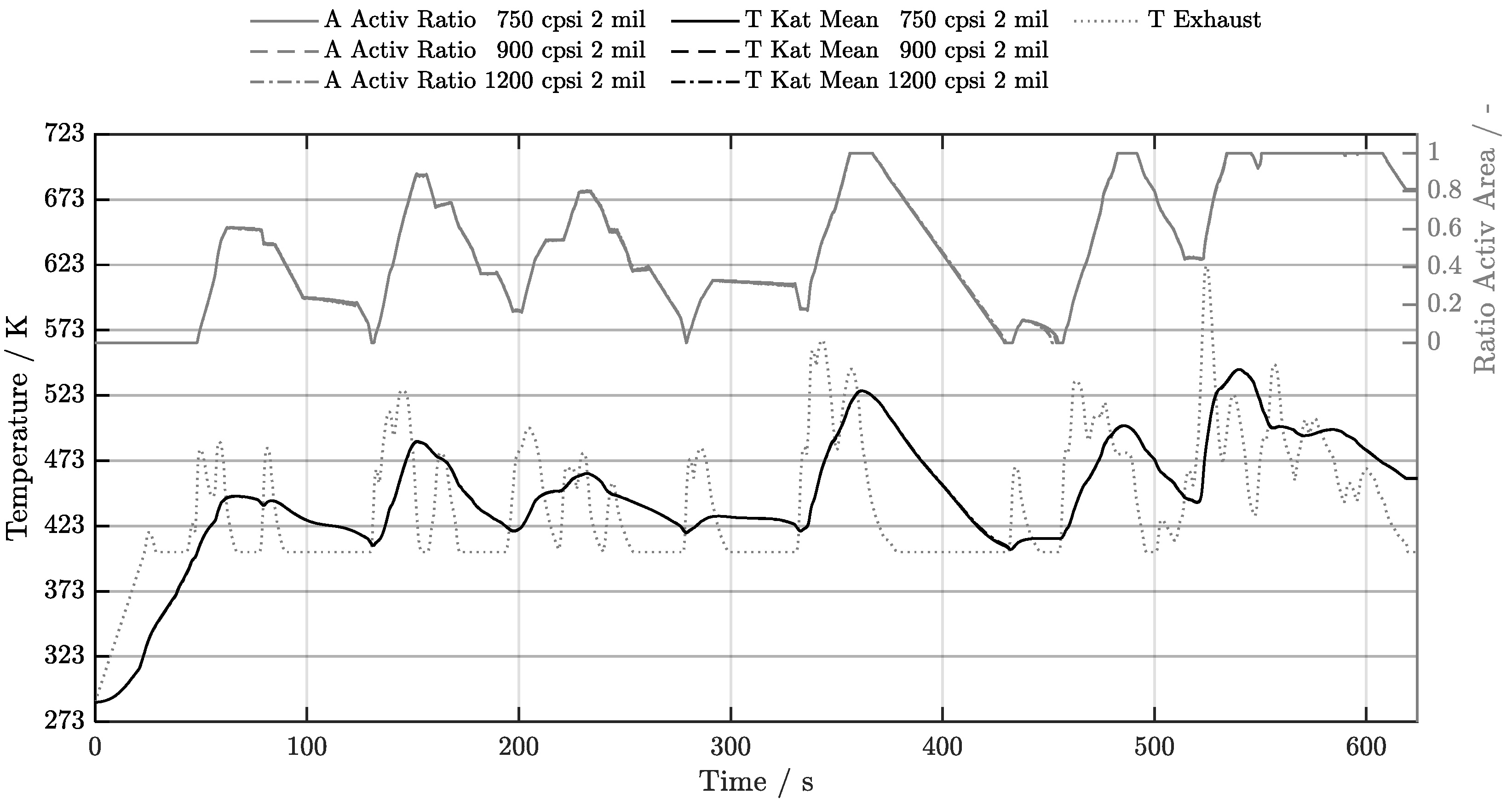
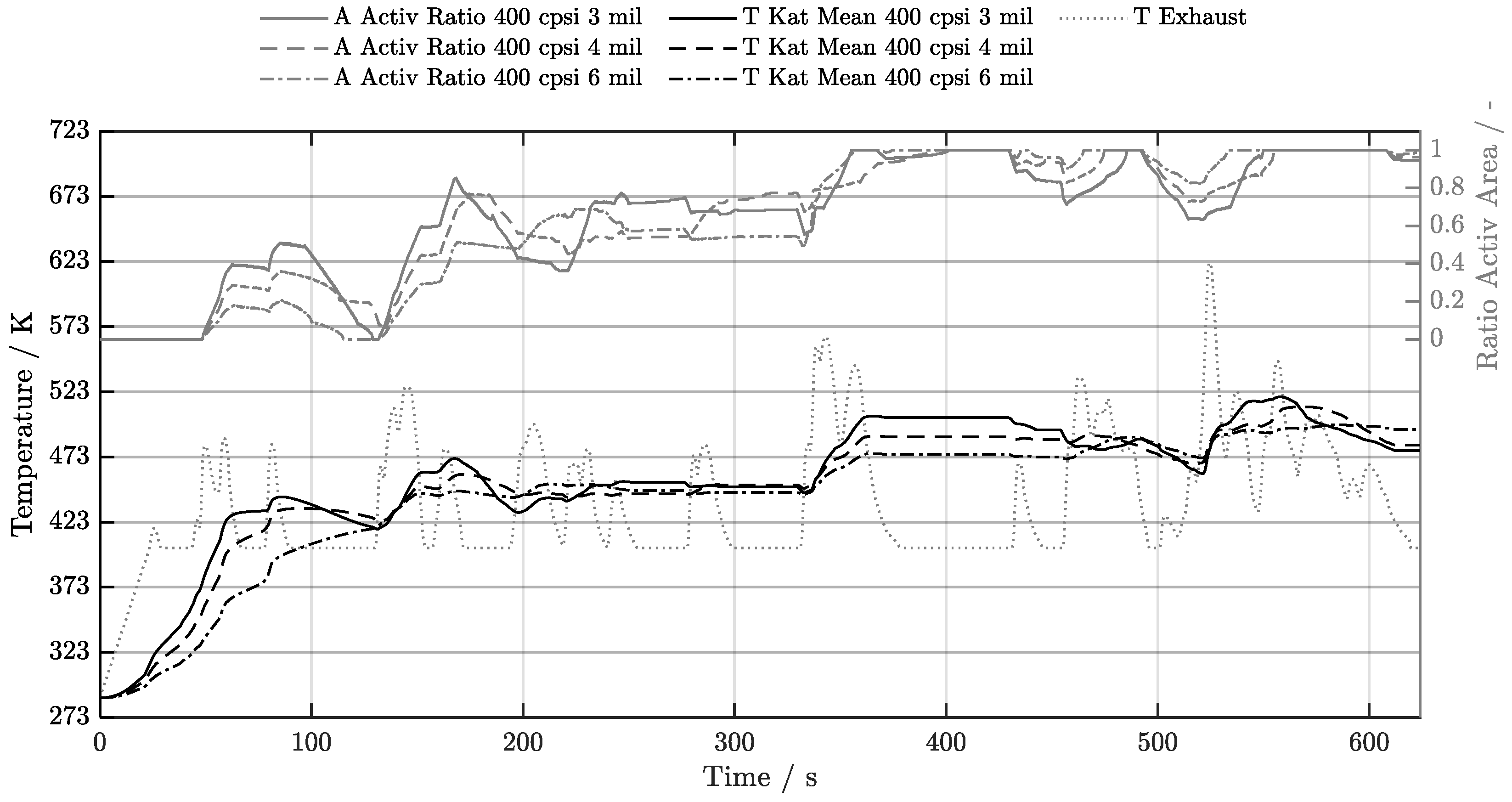
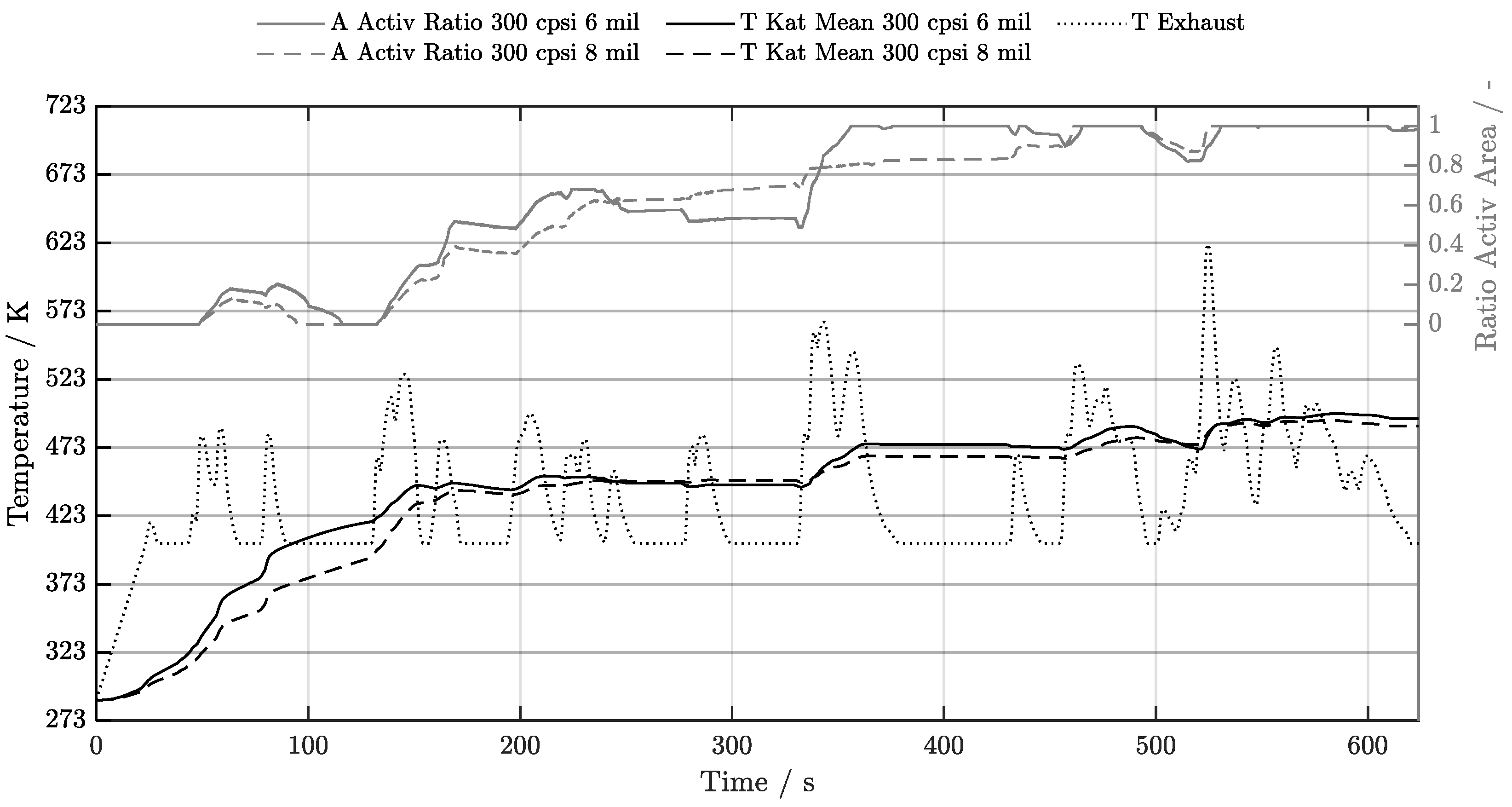
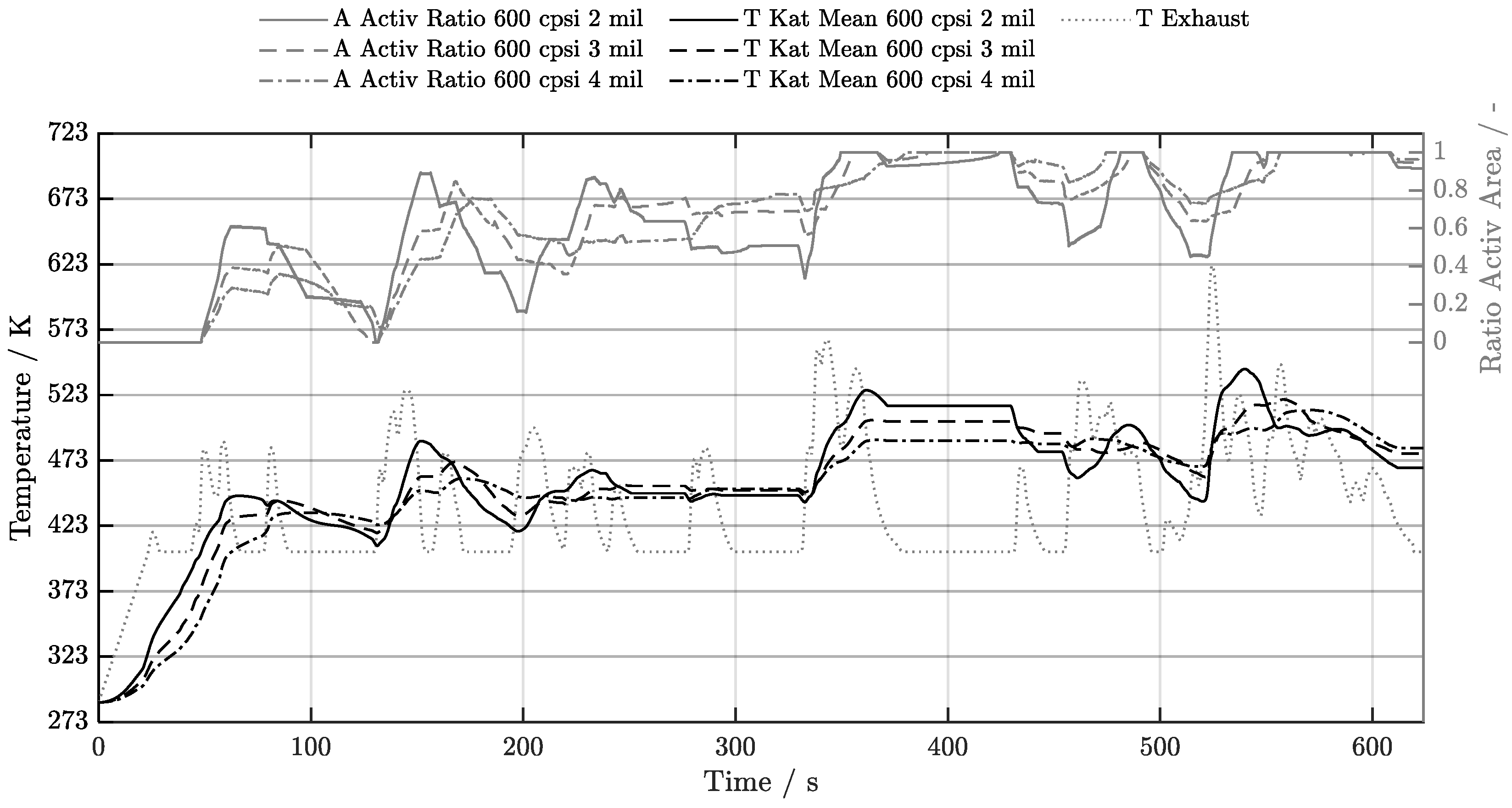
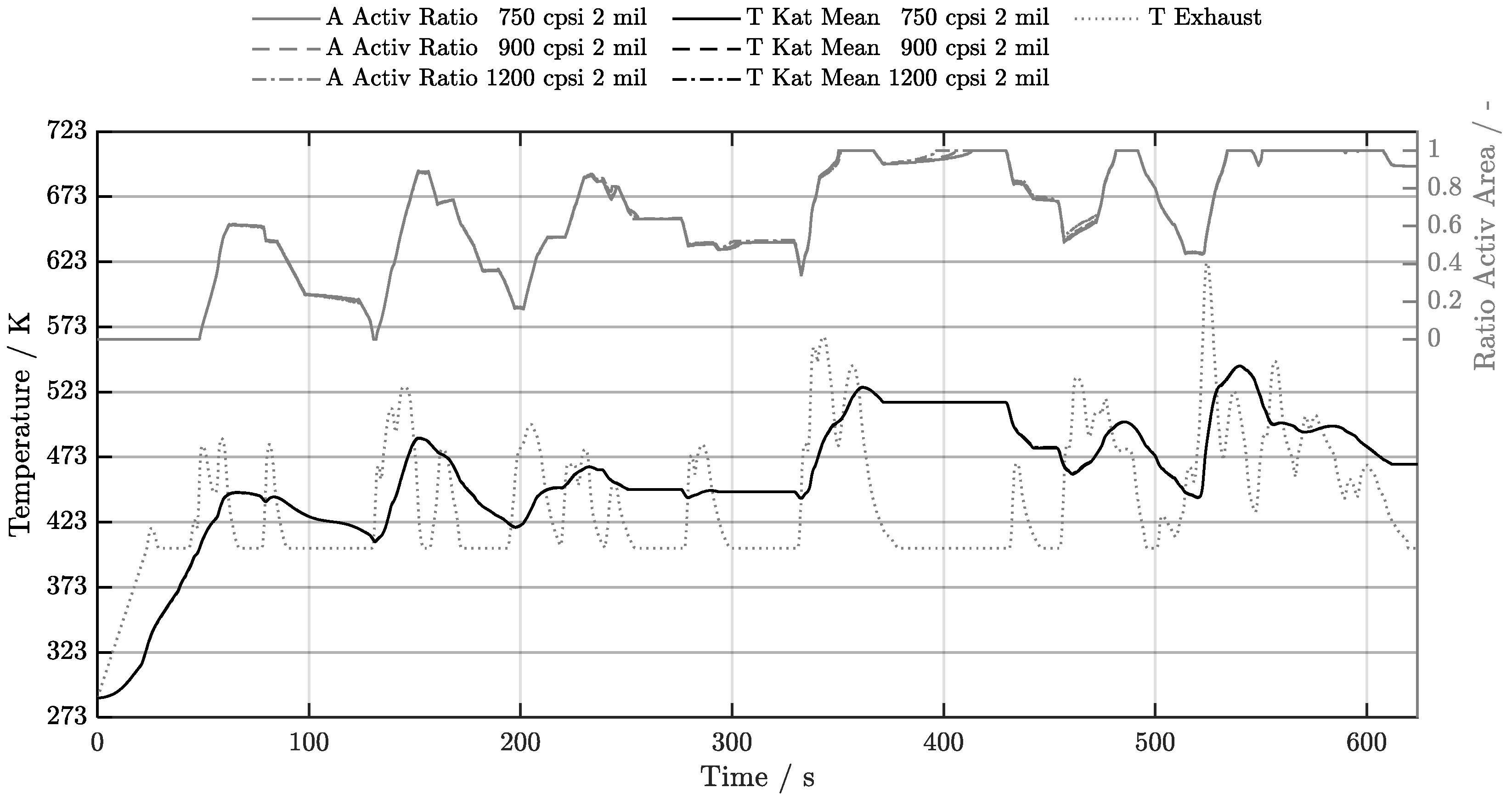
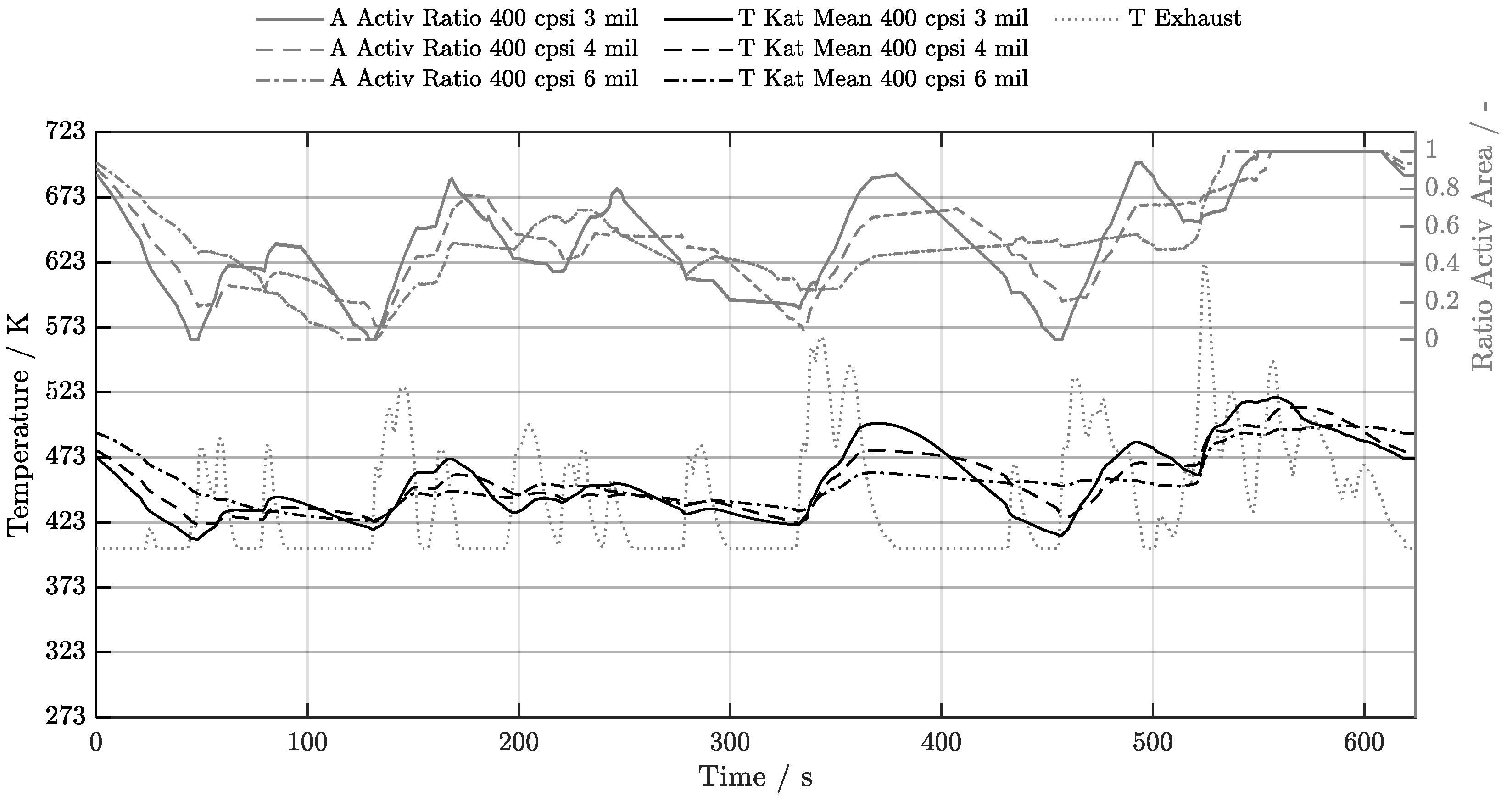
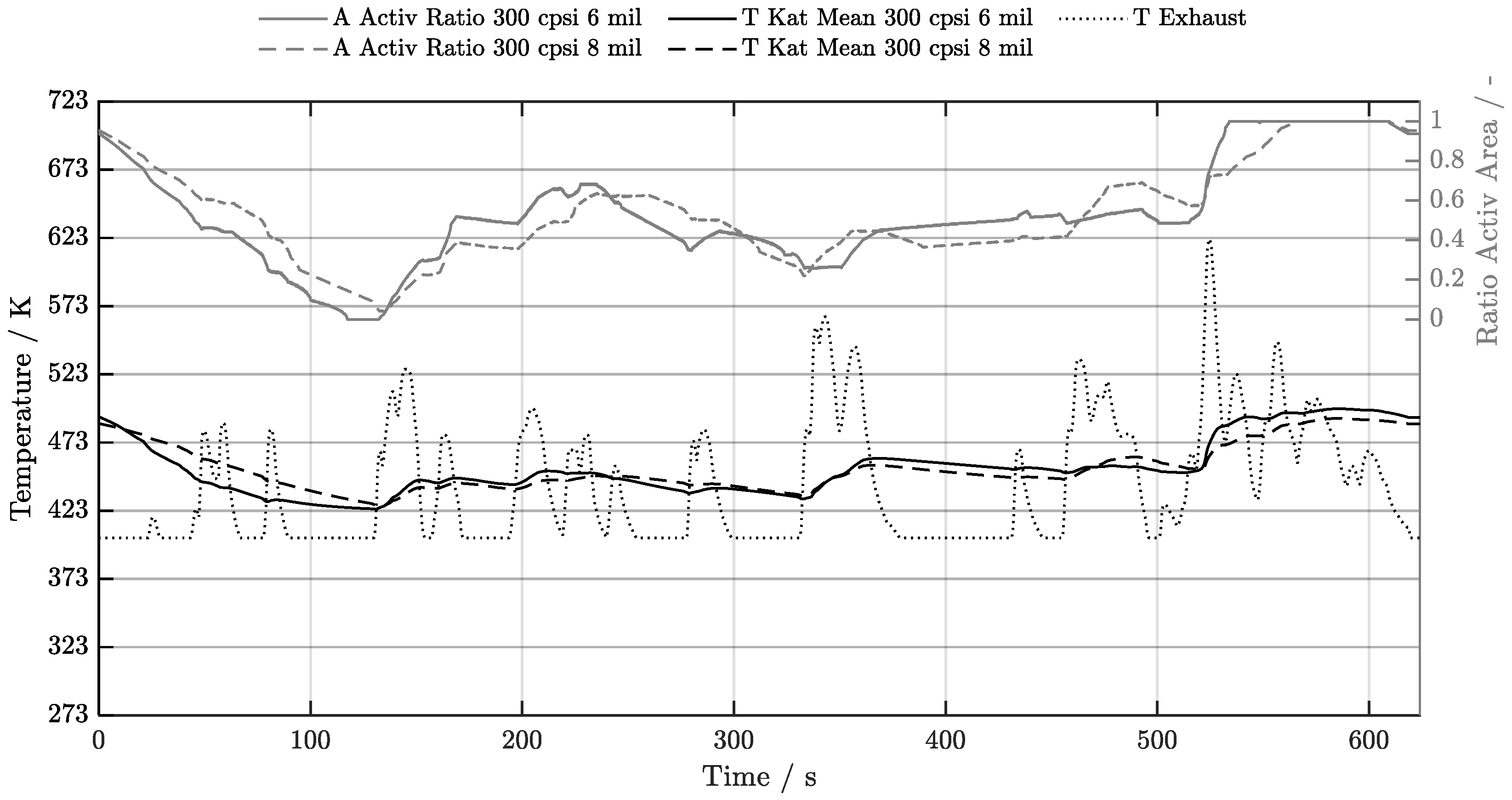
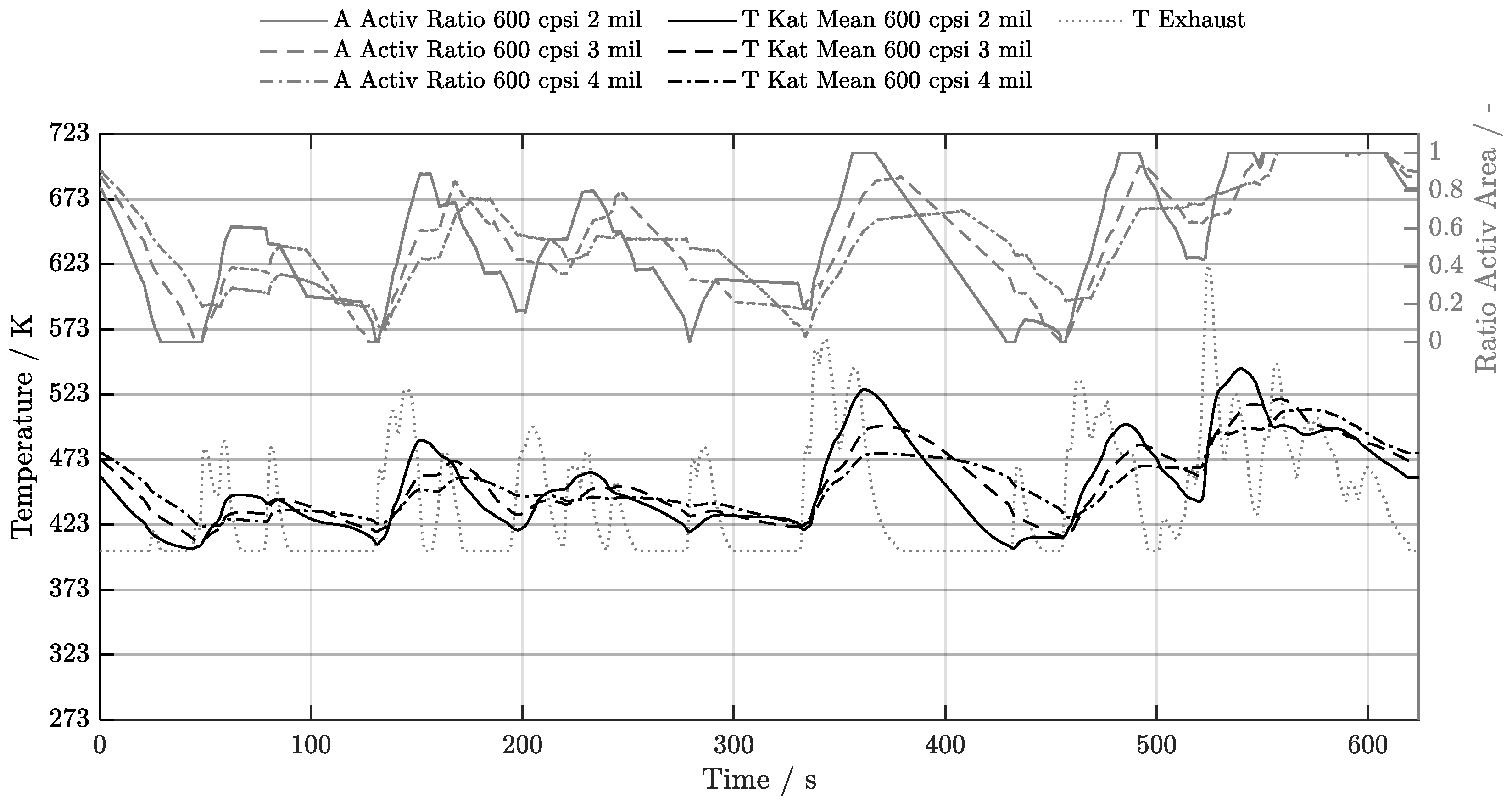
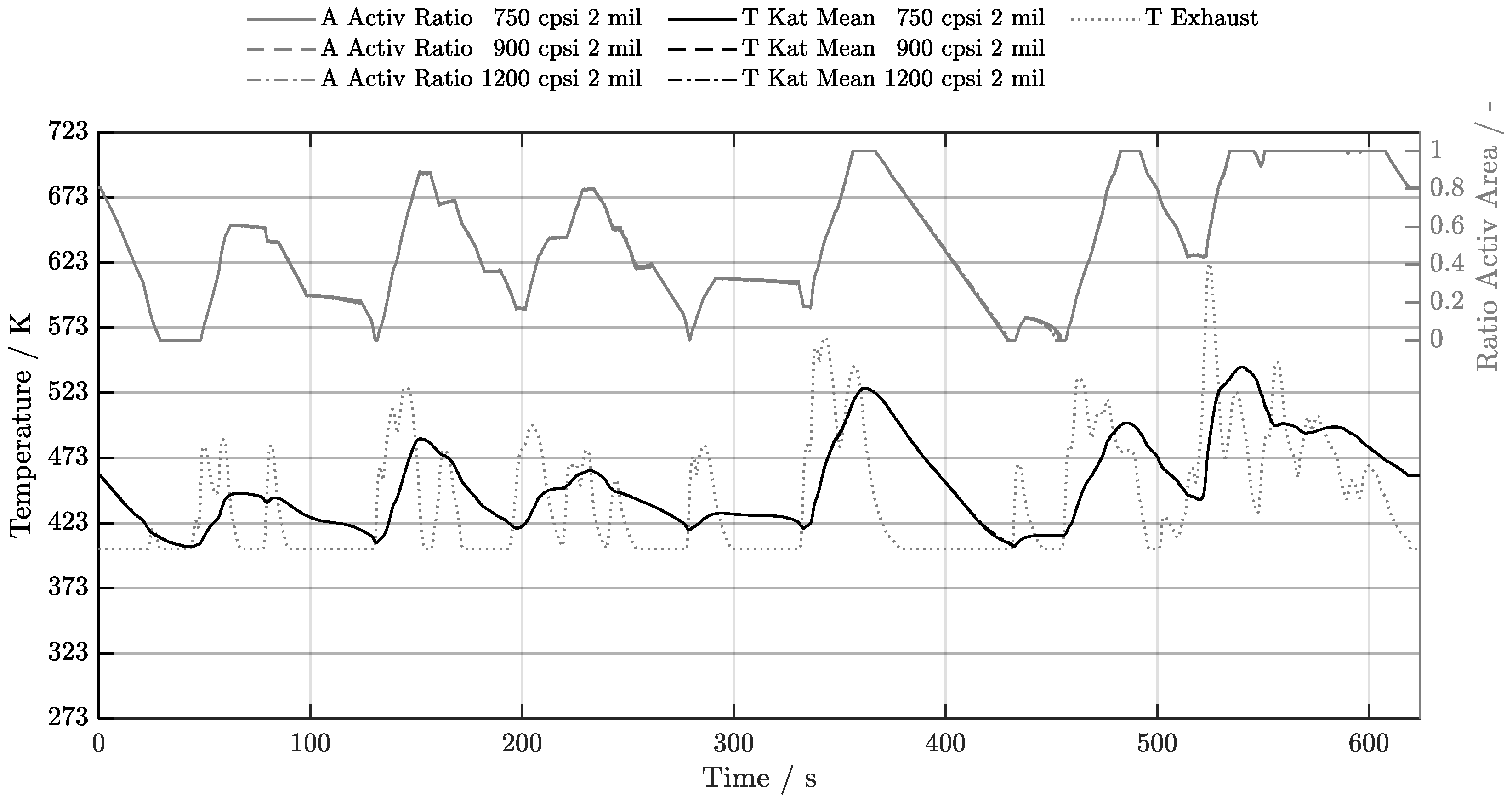
5.1. Cold-Start w/o Start-Stop System
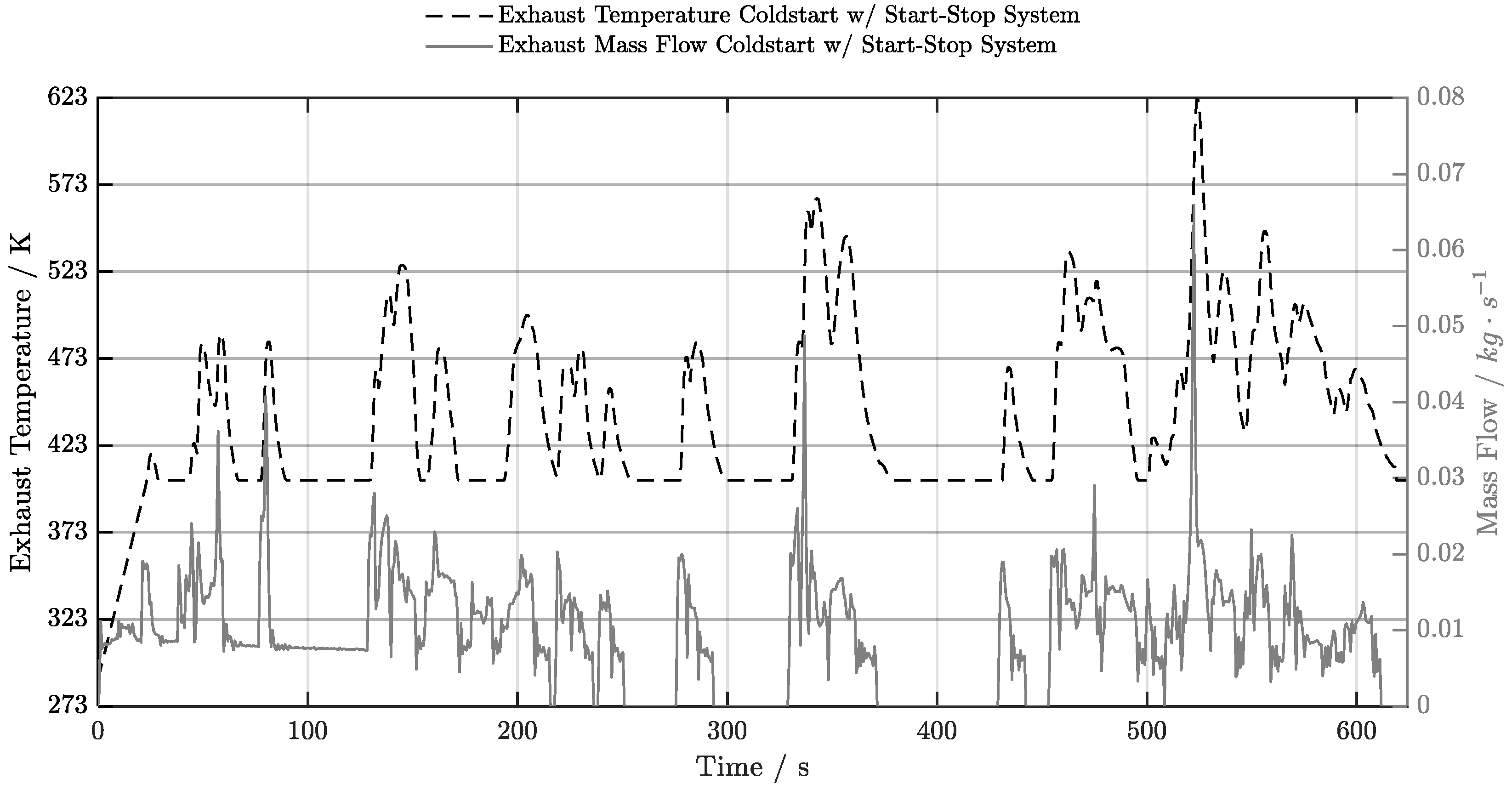
5.2. Cold-Start w/ Start-Stop System
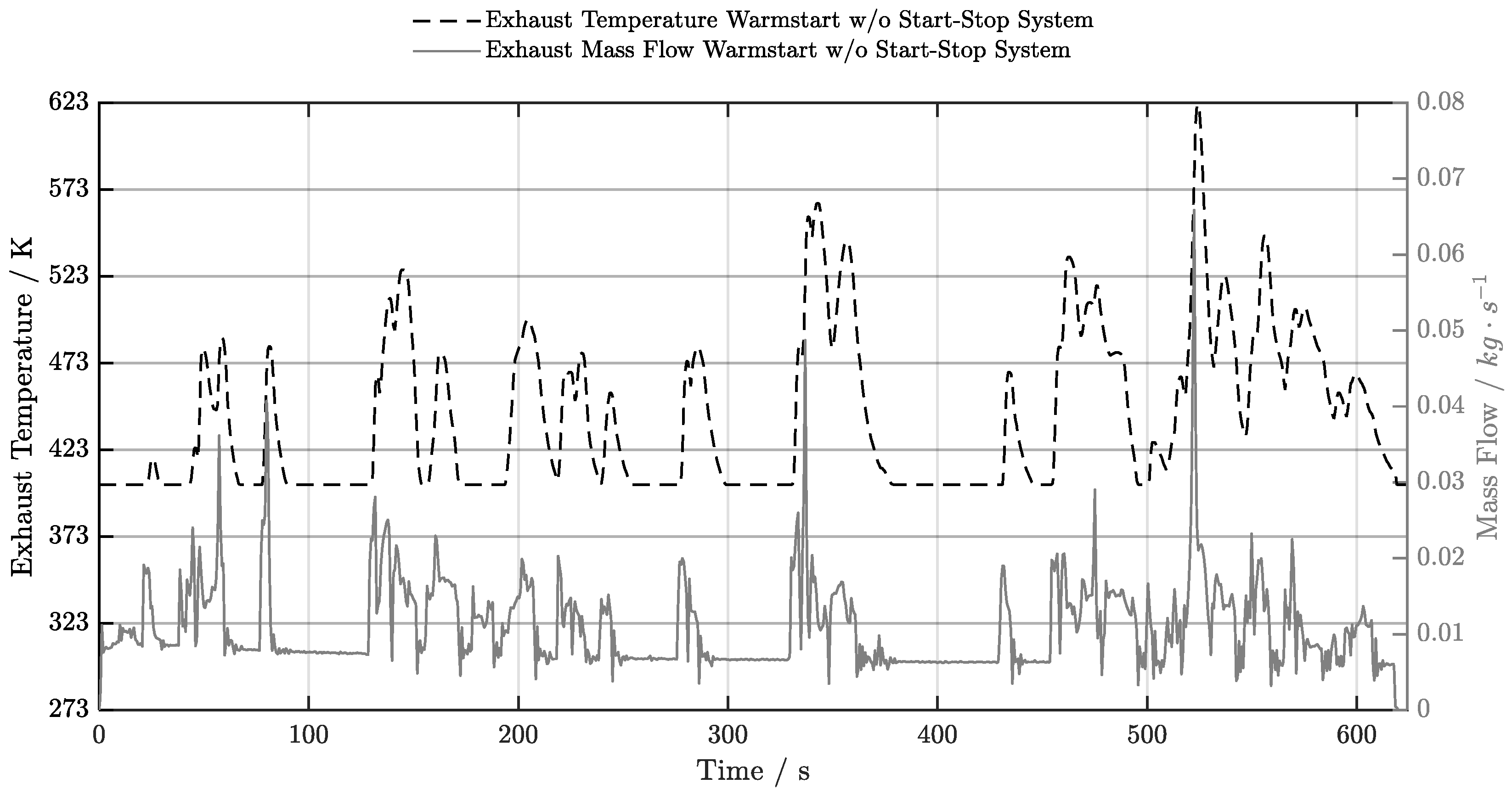
5.3. Warm-Start w/o Start-Stop System
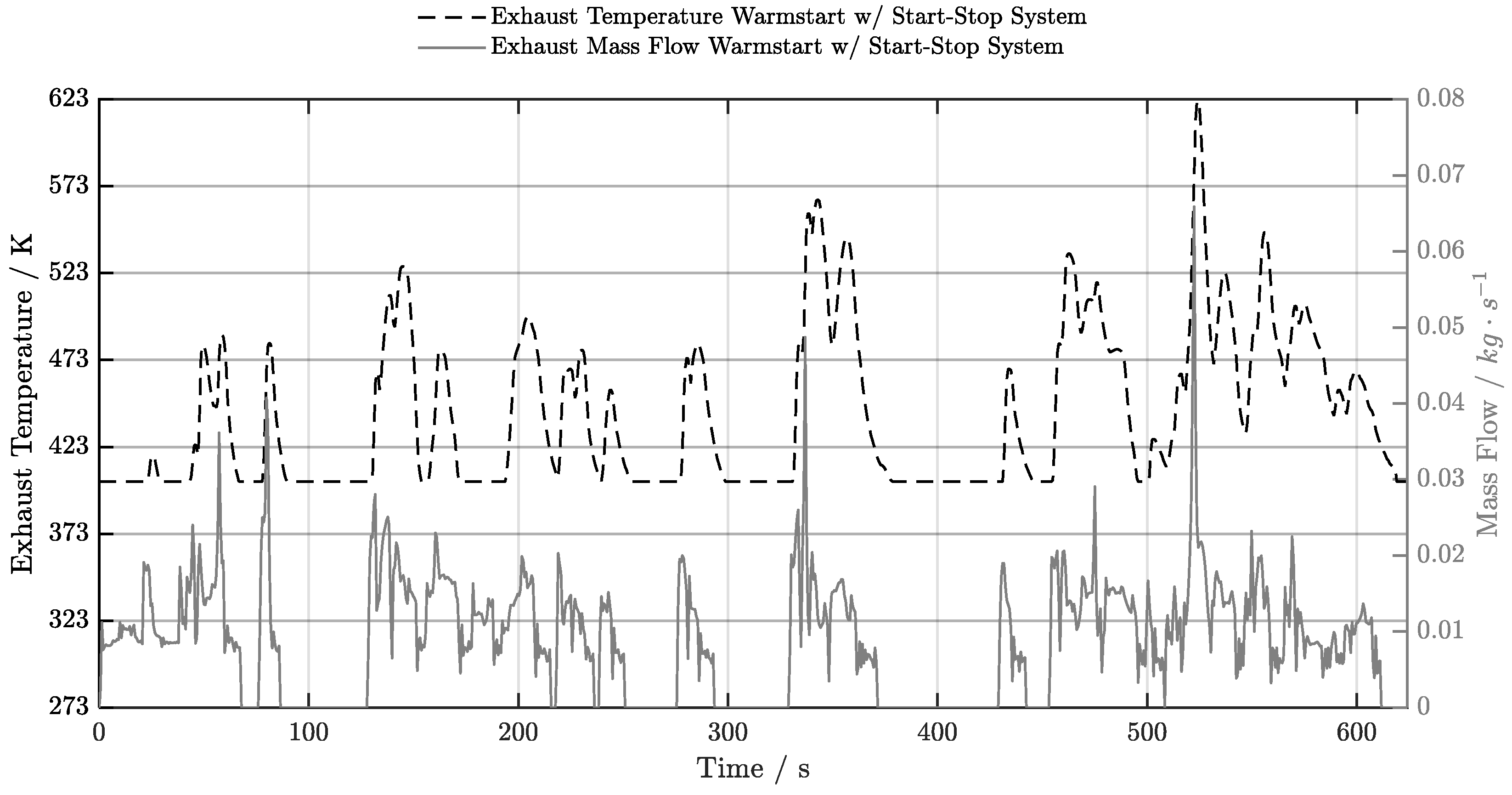
5.4. Warm-Start w/ Start-Stop System
5.5. Electrical Preheating
6. Conclusion
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| Latin Letters | ||
| area | m2 | |
| isobaric heat capacity | J∙kg-1∙K-1 | |
| diameter | m | |
| length | m | |
| mass | kg | |
| gravimetric flow rate | kg∙s-1 | |
| Nusselt number | - | |
| pressure | Pa | |
| Peclet number | - | |
| Prandtl number | - | |
| heat | J | |
| heat flux | W | |
| specific gas constant | J∙kg-1∙K-1 | |
| Reynolds number | - | |
| temperature | K | |
| time | s | |
| velocity | m∙s-1 | |
| uniformity index | - | |
| volume | m3 | |
| volumetric flow rate | m3∙s-1 | |
| Greek Letters | ||
| heat transfer coefficient | W∙m-2∙K-1 | |
| difference | - | |
| heat conductivity | W∙m-1∙K-1 | |
| dynamic viscosity | kg∙m-1∙s-1 | |
| density | kg∙m-3 | |
| Subscripts | ||
| average | ||
| air | air | |
| catalytic converter | ||
| cell | ||
| conductive | ||
| convective | ||
| exhaust | ||
| fuel gas | ||
| exhaust gas | ||
| hydraulic | ||
| inflowing | ||
| internal | ||
| logarithmic | ||
| heat loss | ||
| light off | ||
| mean value | ||
| monolith | ||
| open | ||
| outflowing | ||
| ratio active area to internal area | ||
| Definitions/Abbreviations | ||
| 1D | one-dimensional | |
| BlmSchV | Bundes-Immissionsschutzverordnung | |
| C3H6 | propene | |
| CO | carbon monoxide | |
| CO2 | carbon dioxide | |
| cpsi | cells per square inch | |
| CxHy | hydrocarbons | |
| DOC | diesel oxidation catalyst | |
| EGR | exhaust gas recirculation | |
| EURO | European emissions standard | |
| FAQA | Federal Air Quality Act | |
| GPS | Global Positioning System | |
| H2O | water | |
| HC | hydrocarbons | |
| ICE | internal combustion engines | |
| moDiag | motor diagnostic | |
| NO | nitrogen monoxide | |
| NOx | nitrogen oxides | |
| O2 | oxygen | |
| OFA | open frontal area | |
| Pd | palladium | |
| PM | particulate matter | |
| Pt | platinum | |
| RDE | real driving emissions | |
| Rh | rhodium | |
| SCR | selective catalytic reduction | |
| SynAir | synthetic air | |
| TIER | US emission standard | |
| TWC | three-way catalytic converter | |
| USA | United States of America | |
| WLTP | Worldwide Harmonized Light Vehicles Test Procedure | |
Appendix A
References
- European Commission. Proposal for a Regulation of the European Parliament and of the Council on the Type-Approval of Motor Vehicles and Engines […] (Euro 7). COM(2022) 586 final, Brussels, Belgium, 10 November 2022. [Google Scholar]
- U.S. Environmental Protection Agency. Control of Air Pollution From Motor Vehicles: Tier 3 Motor Vehicle Emission and Fuel Standards. Fed. Regist. 2014, 79(57), 16122–16247. [Google Scholar]
- U.S. Congress. Federal Air Quality Act of 1967, Public Law 90–148; 81 Stat. 485. 2 November 1967. Available online: https://www.govinfo.gov/content/pkg/STATUTE-81/pdf/STATUTE-81-Pg485.pdf (accessed on 2025.05.05).
- Bundesministerium des Innern. Erste Verordnung zur Durchführung des Bundes-Immissionsschutzgesetzes (1. BImSchV). Bundesgesetzblatt Teil I 1974, 2121. Available online: https://www.bgbl.de/xaver/bgbl/start.xav?start=//*%5B@attr_id='bgbl174103.pdf'%5D (accessed on 2025.05.05).
- Peer, J.; Backes, F.; Sauerland, H.; Härtl, M.; Wachtmeister, G. Development of a High Turbulence, Low Particle Number, High Injection Pressure Gasoline Direct Injection Combustion System. SAE Int. J. Engines 2016, 9, 2301–2311. [Google Scholar] [CrossRef]
- Ming, Z.; Graham, T.R.; Gary, H.J. Diesel engine exhaust gas recirculation—a review on advanced and novel concepts. Energy Convers. Manag. 2004, 45, 883–900. [Google Scholar] [CrossRef]
- Bin, C.; Li, Z.; Jinlin, H.; Qing, Z. A combination of electric supercharger and Miller Cycle in a gasoline engine to improve thermal efficiency without performance degradation. Case Stud. Therm. Eng. 2019, 14, 100429. [Google Scholar] [CrossRef]
- Marsh, P.; Acke, F.; Konieczny, R.; Brück, R.; Hirth, P. Application Guideline to Define Catalyst Layout for Maximum Catalytic Efficiency. SAE 2001 World Congress, Detroit, MI, USA, 5–8 March 2001; SAE International, 2001. [Google Scholar]
- Steiner, T.; Neurauter, D.; Moewius, P.; Pfeifer, C.; Schallhart, V.; Moeltner, L. Heat-Up Performance of Catalyst Carriers—A Parameter Study and Thermodynamic Analysis. Energies 2021, 14, 964. [Google Scholar] [CrossRef]
- Brück, R. Neue Katalysatorträger-Innovation für RDE- und SULEV-30-Grenzwerte. MTZ 2016, 78, 18–27. [Google Scholar] [CrossRef]
- Harth, G. Intelligente Katalysatorentechnologien für zukünftige Emissionsanforderungen. In 32. Internationales Wiener Motorensymposium; Lenz, H.-P., Ed.; VDI Verlag GmbH: Düsseldorf, Germany, 2011; pp. 246–257. ISBN 978-3-18-373512-9. [Google Scholar]
- Möltner, L.; Schallhart, V.; Berthold, H. Silikatkeramischer Kompositwerkstoff und Anwendungen davon. 2021. [Google Scholar]
- Nieken, U.; Eigenberger, G.; Tuttlies, U.S.; Bernnat, J. Katalytischer Brenner für ein wärmeintegriertes Motorabgasreinigungssystem; Universitätsbibliothek der Universität Stuttgart: Stuttgart, Germany, 2009. [Google Scholar]
- Hudec, J.; Šarkan, B.; Cződörová, R. Examination of the results of the vehicles technical inspections in relation to the average age of vehicles in selected EU states. Transportation Research Procedia 55. 2–9. [CrossRef]
- Landeshauptstadt Innsbruck, City-Map Innsbruck. Available online: https://city-map.innsbruck.gv.at/stadtplan/client_workspace/FatalError.jsp;jsessionid=D5D860C04CCB6987F2D5BF6FF01D6775?synergis_session=02d2ef88-5742-4502-bf69-7e581dc57f08&tsp=1746781930310 (accessed on 2025.09.09).
- Moeltner, L.; Hohensinner, M.; Schallhart, V. Experimental and Numerical Analysis of Low Temperature NOx Conversion in Urban Busses. SAE 2016 Commercial Vehicle Engineering Congress, 2016; SAE International. [Google Scholar]
- Moore, W.J. Physikalische Chemie; De Gruyter: Berlin, Germany, 1986; ISBN 978-3-11-010979-5. [Google Scholar]
- Kleiber, M.; Joh, R. VDI-Wärmeatlas; Verein Deutscher Ingenieure (VDI-GVC): Berlin, Germany, 2006; ISBN 978-3-540-25504-8. [Google Scholar]
- Wishart, J.; Shirk, M.; Gray, T.; Fengler, N. Quantifying the Effects of Idle-Stop Systems on Fuel Economy in Light-Duty Passenger Vehicles. SAE 2012 World Congress & Exhibition, 2012; SAE International. [Google Scholar]
- Statistisches Bundesamt. Erwärbstätigkeit – Berufspendler. Available online: https://www.destatis.de/DE/Themen/Arbeit/Arbeitsmarkt/Erwerbstaetigkeit/Tabellen/pendler1.html (accessed on 2025.09.15).
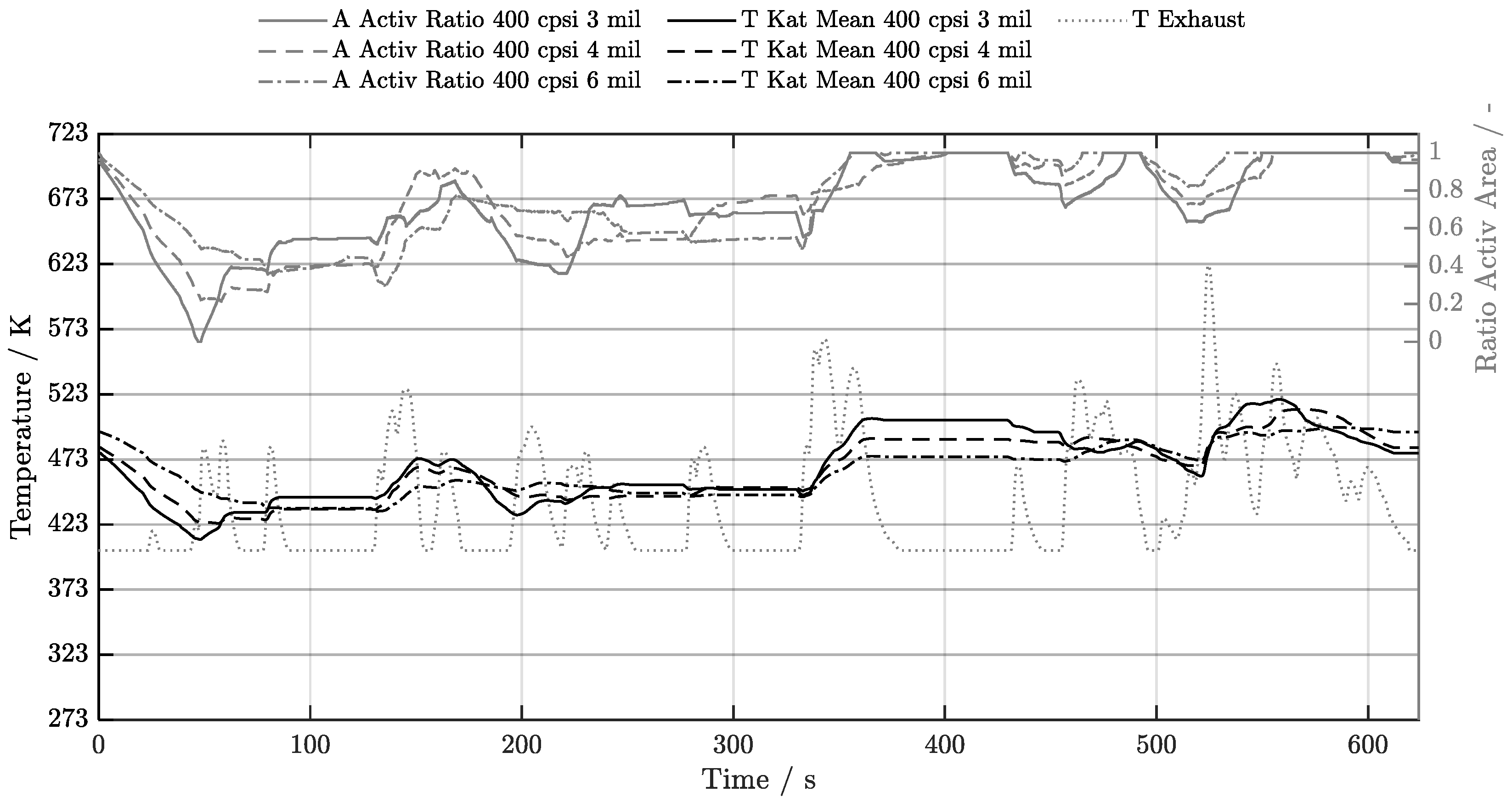
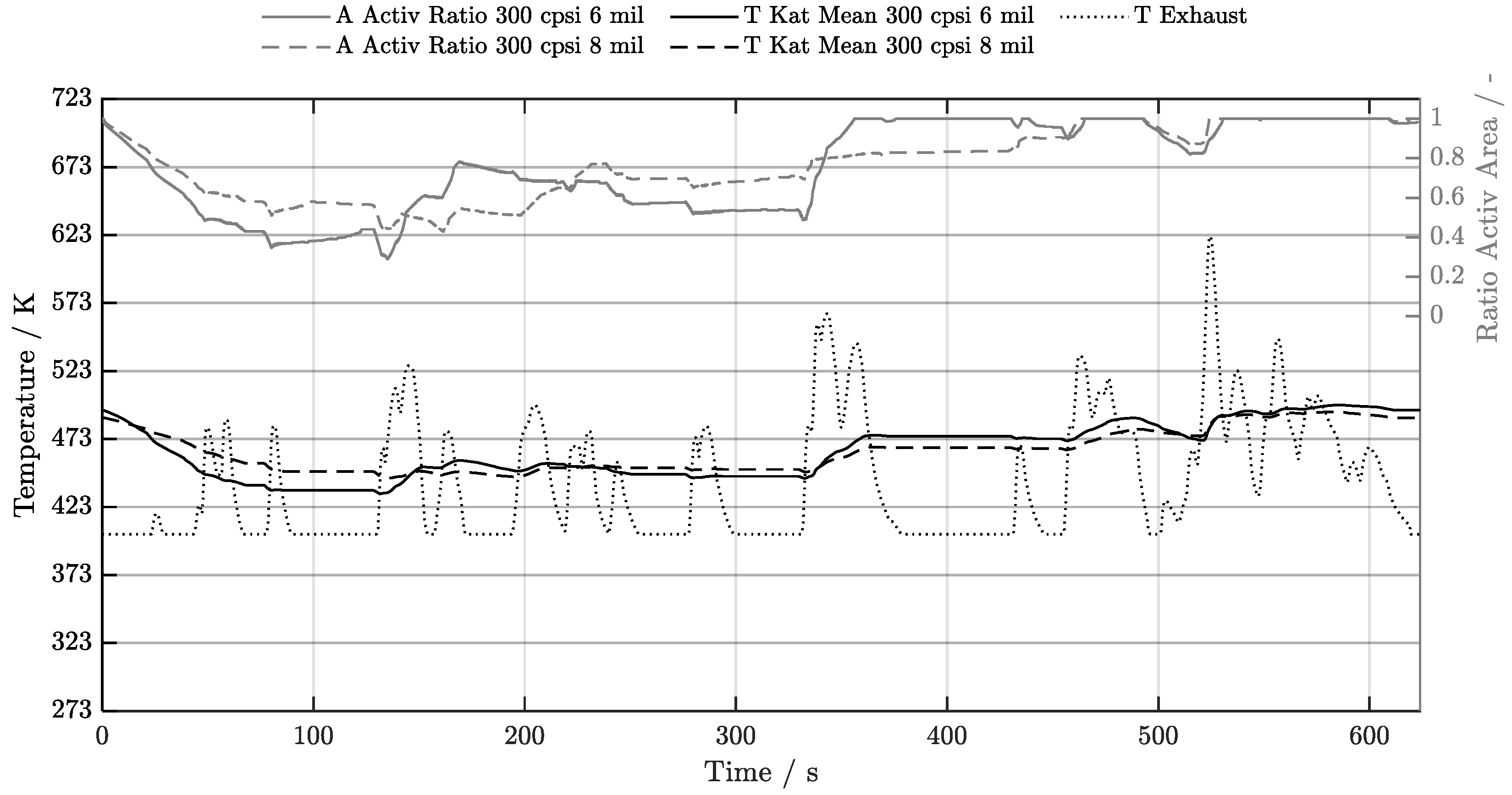
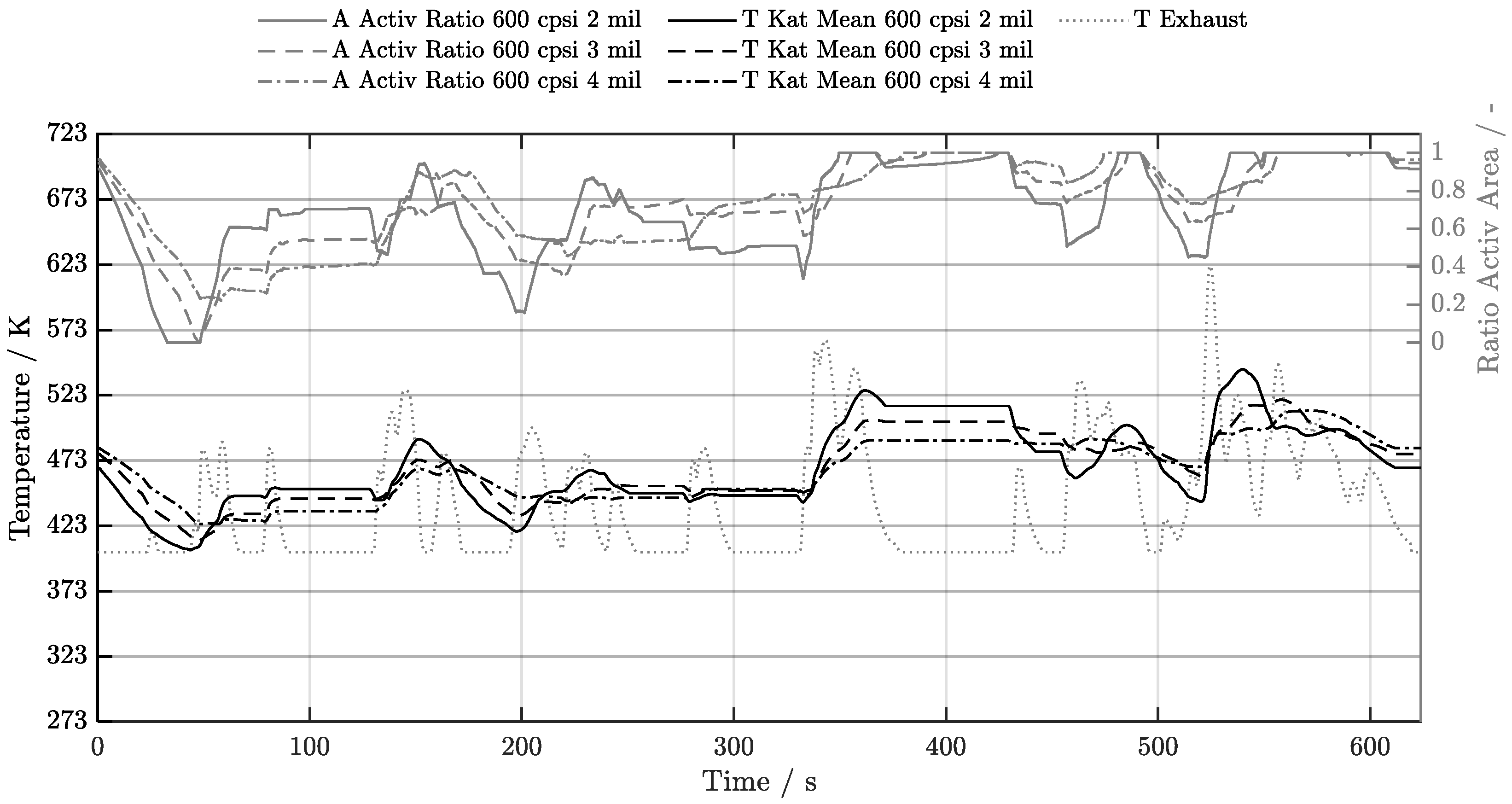
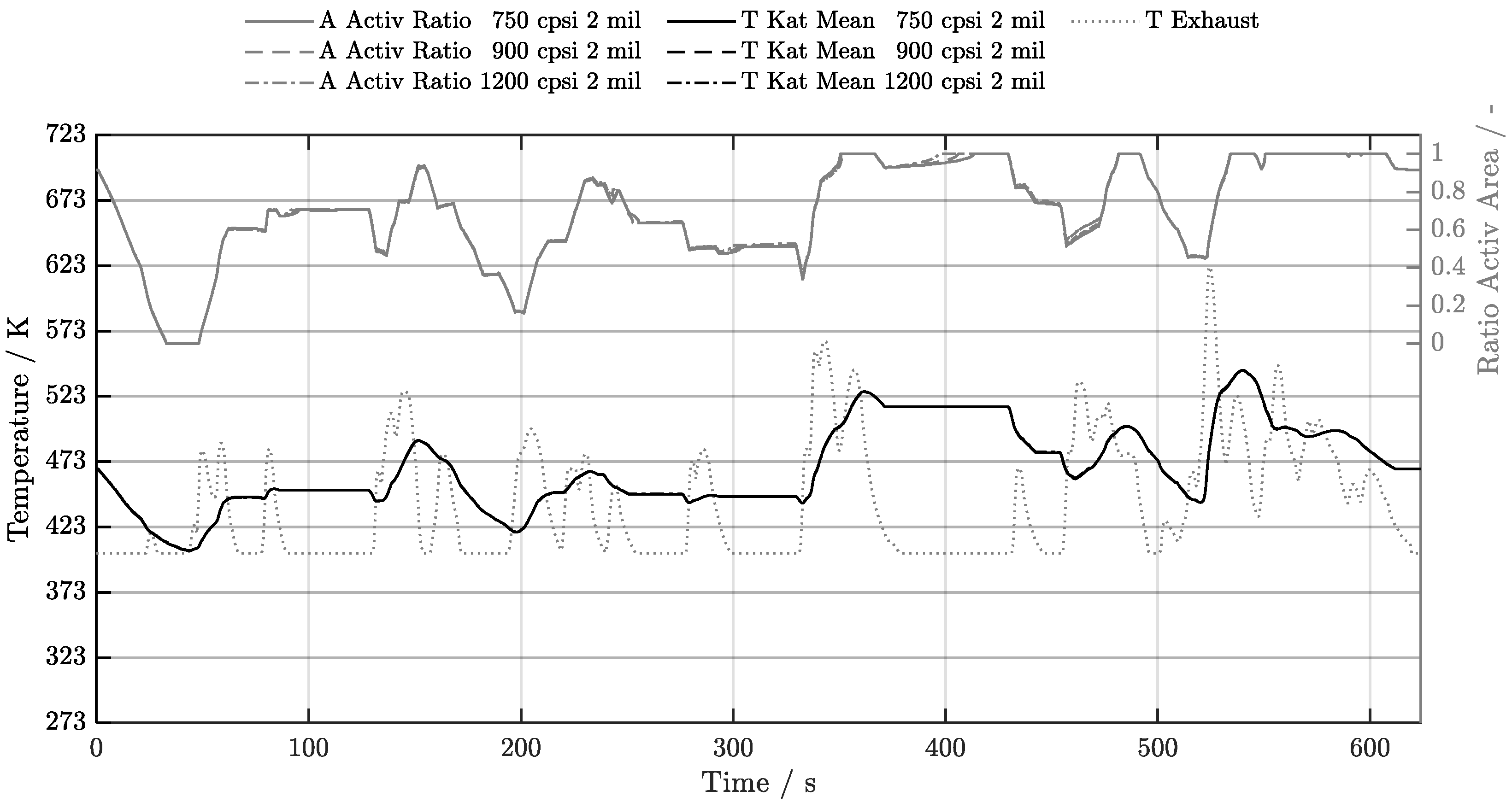
| Geometry | 300 cpsi | 400 cpsi | 600 cpsi | 750 cpsi | 900 cpsi | 1200 cpsi |
|---|---|---|---|---|---|---|
| 2 mil | - | - | x | x | x | x |
| 3 mil | - | x | x | - | - | - |
| 4 mil | - | x | x | - | - | - |
| 6 mil | x | x | - | - | - | - |
| 8 mil | x | - | - | - | - | - |
| Manufacturer | Skoda |
|---|---|
| Model | Octavia |
| Year of manufacture | 2015 |
| Emission standard | EURO V |
| Number of cylinders | 4 |
| Operating principle | Four-Stroke-Diesel |
| Engine displacement | 1598 ccm³ |
| Vehicle weight | 1381 kg |
| Power output | 77 kW (@4000 rpm) |
| Cell density | Wall thickness | Aint | Length | Length | Diamter |
|---|---|---|---|---|---|
| / cpsi | / mil | / m² | / in | / m | / m |
| 300 | 6 | 7.169 | 7.113 | 0.181 | 0.144 |
| 300 | 8 | 7.169 | 7.399 | 0.188 | 0.144 |
| 400 | 3 | 7.169 | 5.873 | 0.149 | 0.144 |
| 400 | 4 | 7.169 | 6.000 | 0.152 | 0.144 |
| 400 | 6 | 7.169 | 6.273 | 0.159 | 0.144 |
| 600 | 2 | 7.169 | 4.740 | 0.120 | 0.144 |
| 600 | 3 | 7.169 | 4.865 | 0.124 | 0.144 |
| 600 | 4 | 7.169 | 4.997 | 0.127 | 0.144 |
| 750 | 2 | 7.169 | 4.265 | 0.108 | 0.144 |
| 900 | 2 | 7.196 | 3.915 | 0.099 | 0.144 |
| 1200 | 2 | 7.196 | 3.424 | 0.087 | 0.144 |
| Weight | 300 cpsi | 400 cpsi | 600 cpsi | 750 cpsi | 900 cpsi | 1200 cpsi |
|---|---|---|---|---|---|---|
| 2 mil | - | - | 0.392 kg | 0.393 kg | 0.395 kg | 0.397 kg |
| 3 mil | - | 0.592 kg | 0.596 kg | - | - | - |
| 4 mil | - | 0.798 kg | 0.806 kg | - | - | - |
| 6 mil | 1.214 kg | 1.225 kg | - | - | - | - |
| 8 mil | 1.653 kg | - | - | - | - | - |
| α-value / W m-1 K-1 |
300 cpsi | 400 cpsi | 600 cpsi | 750 cpsi | 900 cpsi | 1200 cpsi |
|---|---|---|---|---|---|---|
| 2 mil | - | - | 151.8 | 170.7 | 188.0 | 219.2 |
| 3 mil | - | 125.4 | 155.8 | - | - | - |
| 4 mil | - | 128.1 | 160.0 | - | - | - |
| 6 mil | 114.0 | 134.0 | - | - | - | - |
| 8 mil | 118.5 | - | - | - | - | - |
| Material | Cordierit | Hybrid Ceramic |
|---|---|---|
| Density | 2100 kg m-3 | 2670 kg m-3 |
| Heat Capacity | 1000 J kg-1 K-1 | 805 J kg-1 K-1 |
| Thermal Conductivity | 2.0 W m-1 K-1 | 4.7 W m-1 K-1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




