Submitted:
22 April 2026
Posted:
23 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
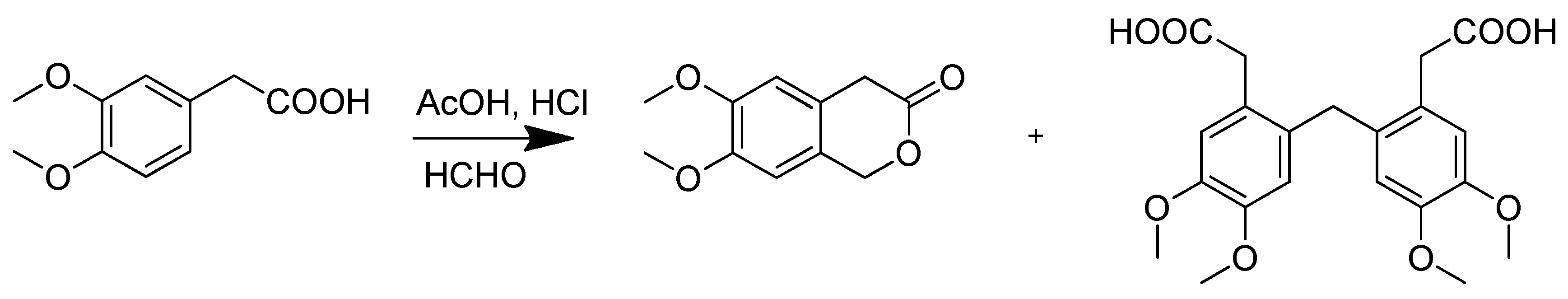
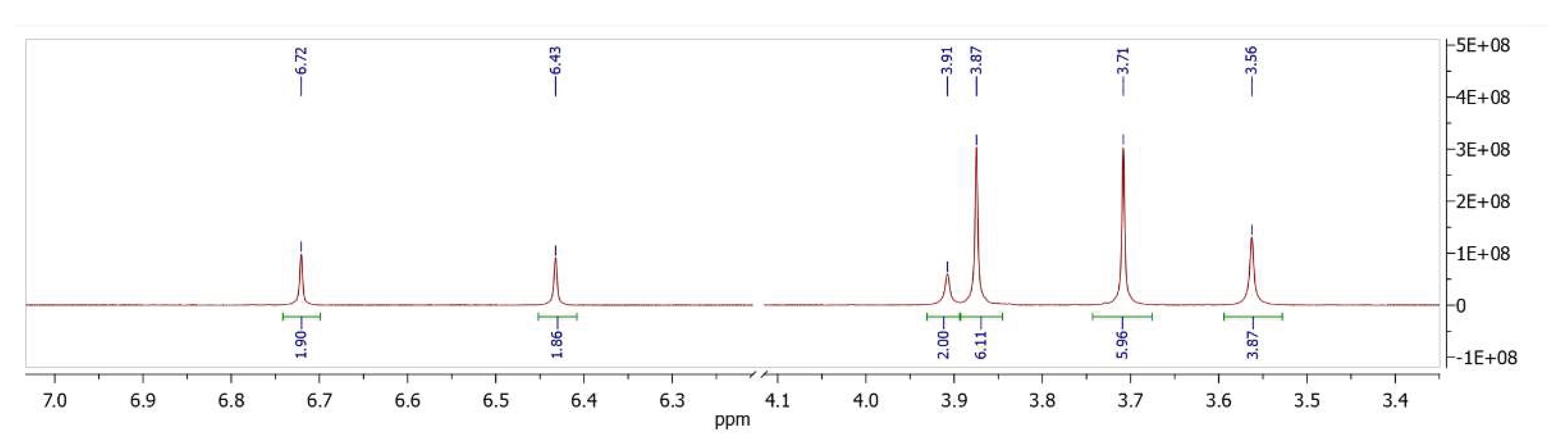
2. Results and Discussion
3. Materials and Methods
3.1. General
3.2. Procedure
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Massol-Frieh, C.; Vantourout, J. Synthesis Side Stories: Capitalizing the Unintended Side-Product. Helv. Chim. Acta 2026, e00001. [Google Scholar] [CrossRef]
- R. van Bommel, M.; de Keijzer, M. Mauveine: The First Synthetic Dye. In Bright Colours from the Past, 1st ed.; Springer: Cham, Switzerland, 2025; pp. 133–143. [Google Scholar]
- Obaida, S.N.B.; Harrity, J.P.A. Base-Mediated Annulation Strategy to Naphthol Boronic Ester Derivatives. J. Org. Chem. 2025, 90(44), 15867–15870. [Google Scholar] [CrossRef] [PubMed]
- Spock, M.; Fettinger, J.C.; Ando, K.; Shaw, J.T. Stereodivergent Synthesis of Complex N-Sulfonimidoyl Lactams via the Castagnoli–Cushman Reaction. Org. Lett. 2025, 27, 1147–1152. [Google Scholar] [CrossRef] [PubMed]
- Saddiqa, A.; Andac, C.A.; Çakmak, O.; Babar, I.; Akhtar, F. Design, synthesis, and structural investigations of novel (S)-amide derivatives as promising ACE inhibitors. Kuwait J. Sci. 2025, 52, 100313. [Google Scholar] [CrossRef]
- Kirad, S.; Deepa, P.R.; Sankaranarayanan, M. Greener alternatives for synthesis of isoquinoline and its derivatives: a comparative review of eco-compatible synthetic routes. RSC Adv. 2025, 15, 30231–30275. [Google Scholar] [CrossRef] [PubMed]
- Stoyanova, S.; Bogdanov, M.G. Rational Design, Synthesis and In Vitro Activity of Diastereomeric Cis-/Trans-3-Substituted-3,4-Dihydroisocoumarin-4-Carboxylic Acids as Potential Carnitine Acetyltransferase Inhibitors. Molecules 2025, 30, 3159. [Google Scholar] [CrossRef] [PubMed]
- Angelov, V.; Stoyanova, S.; Bogdanov, M.G. trans-11-(3,4-Dimethoxyphenyl)-2,3,8,9-tetramethoxy-6-oxo-11,12-dihydro-6H-dibenzo[c,h]chromene-12-carboxylic Acid. Molbank 2024, 2024(4), M1920. [Google Scholar] [CrossRef]
- Liu, S.-J.; Chen, Z.-H.; Chen, J.-Y.; Ni, S.-F.; Zhang, Y.-C.; Shi, F. Rational Design of Axially Chiral Styrene-Based Organocatalysts and Their Application in Catalytic Asymmetric (2+4) Cyclizations. Angew. Chem. Int. Ed. Angew. Chem. 2022, 134, e202112226.. 2022, 61, e202112226. [Google Scholar] [CrossRef] [PubMed]
- Kazantsev, A.; Bakulina, O.; Dar’in, D.; Kantin, G.; Bunev, A.; Krasavin, M. Unexpected Ring Contraction of Homophthalic Anhydrides under Diazo Transfer Conditions. Org. Lett. 2022, 24, 4762–4765. [Google Scholar] [CrossRef] [PubMed]
- Bayles, T.; Guillou, C. Trifluoroethanol Promoted Castagnoli–Cushman Cycloadditions of Imines with Homophthalic Anhydride. Molecules 2022, 27, 844. [Google Scholar] [CrossRef] [PubMed]
- Miliovsky, M.; Svinyarov, I.; Prokopova, E.; Batovska, D.; Stoyanov, S.; Bogdanov, M.G. Synthesis and Antioxidant Activity of Polyhydroxylated trans-Restricted 2-Arylcinnamic Acids. Molecules 2015, 20, 2555–2575. [Google Scholar] [CrossRef] [PubMed]
- Svinyarov, I.; Bogdanov, M.G. One-pot synthesis and radical scavenging activity of novel polyhydroxylated 3-arylcoumarins. Eur. J. Med. Chem. 2014, 78, 198–206. [Google Scholar] [CrossRef] [PubMed]
- Weimar, C.; Angerer, S.; Wiegrebe, W. Methoxy- and acetoxy-8-oxoberbines - synthesis, antitumor activity, and interaction with DNA. Arch. Pharm. Pharm. Med. Chem. 1991, 324, 509–518. [Google Scholar] [CrossRef] [PubMed]
- Saeed, A. Oxa-Pictet–Spengler reaction in water. Synthesis of some (±)-1-aryl-6,7-dimethoxyisochromans. Chin. Chem. Lett. 2010, 21, 261–264. [Google Scholar] [CrossRef]
- Krohn, K.; Cludius-Brandt, S. 2-(Trimethylsilyl)-1,3-dithiane 1-Oxide as a Convenient Reagent for the Transformation of Aldehydes and Ketones into Homologous Carboxylic Acids. Synth. 2010, 15, 2616–2620. [Google Scholar] [CrossRef]
- Wang, H.; Li, L.; Bai, X.-F.; Shang, J.-Y.; Yang, K.-F.; Xu, L.-W. Efficient Palladium-Catalyzed C-O Hydrogenolysis of Benzylic Alcohols and Aromatic Ketones with Polymethylhydrosiloxane. Adv. Synth. Catal. 2013, 355, 341–347. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



