Submitted:
21 April 2026
Posted:
23 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
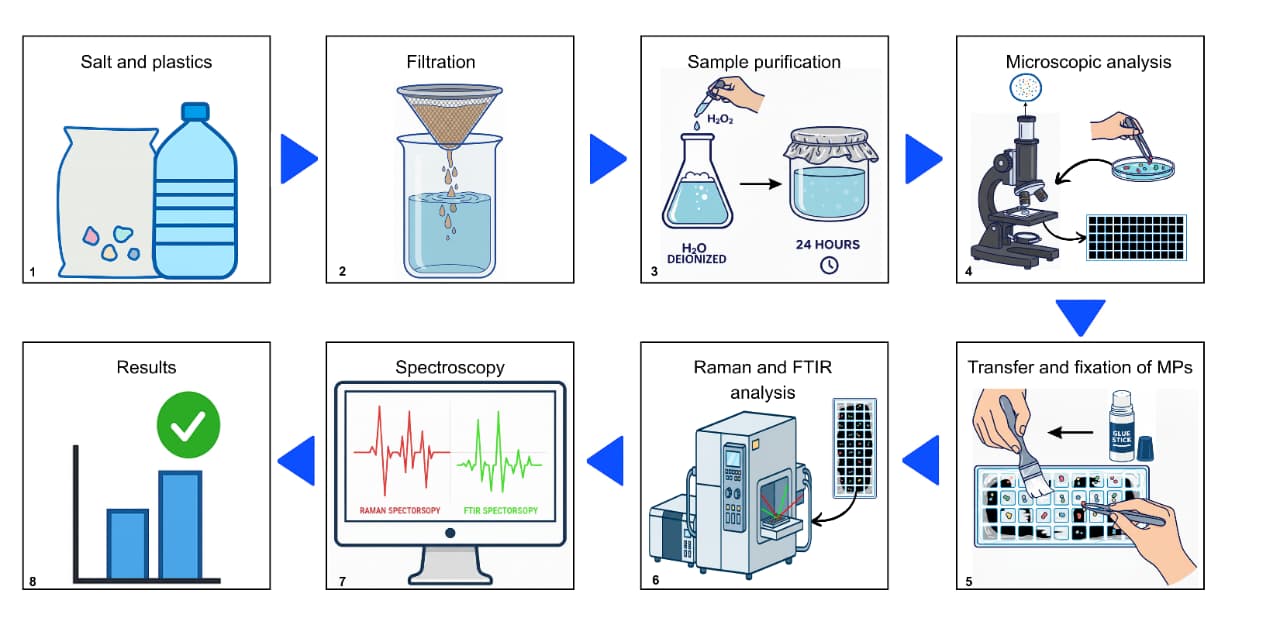
2. Materials and Methods
2.1. Laboratory Screenings
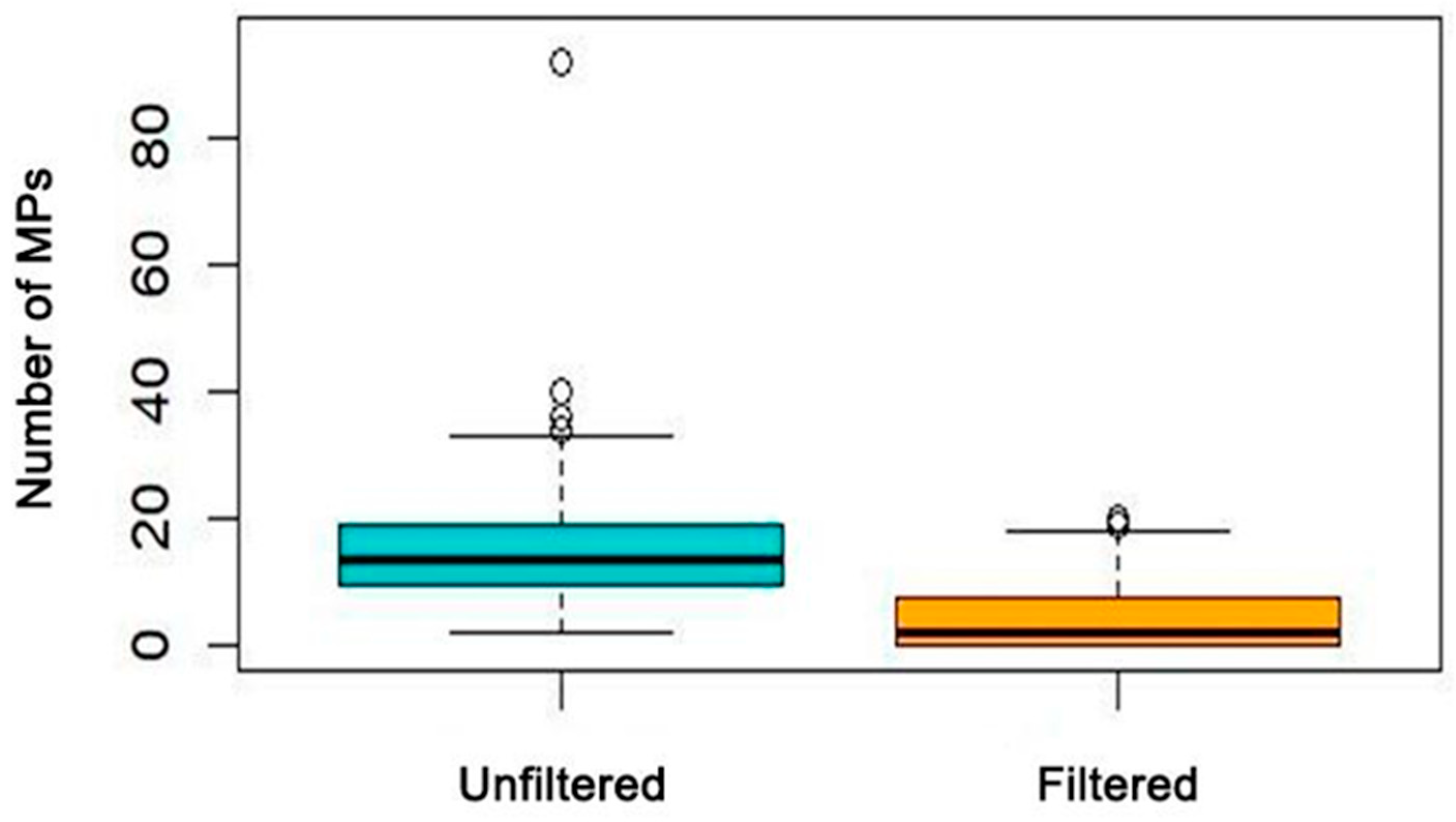
2.2. Screening of Estuary Samples—Unfiltered Saline Solution
2.3. Screening of Reservoir Samples—Filtered Saline Solution
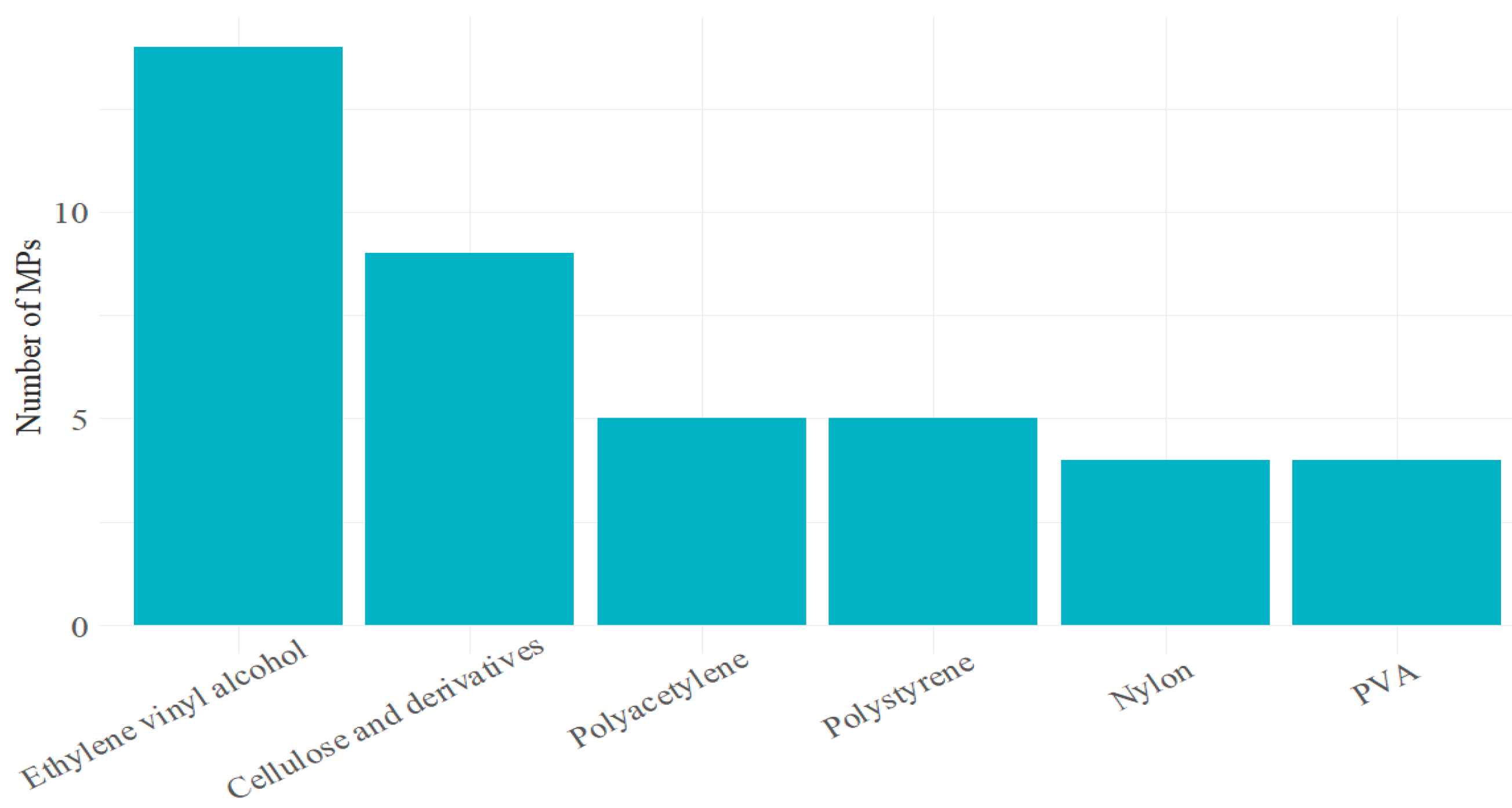
2.4. Identification of the Chemical Composition of Microplastics and Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
- Precision balance
- Drying oven
- Stereoscopic microscope
- Glass beaker (1 L) – (2 units per sample)
- Glass Petri dishes (minimum of 5 units)
- Glass rod
- Filtration membrane (20 µm)
- Aluminum sieve
- Rubber band
- Aluminum container with lid (capacity: 250 mL)
- Wash bottles
- Stainless steel spatula
- Microtubes (1.5/2 mL)
- Sodium chloride (NaCl)
- Distilled water (filtered with 20 µm membrane)
- 70% ethanol
- Aluminum foil
- Personal protective equipment (gloves, mask, cotton lab coat)
- Weigh 250 g of wet sediment.
- Dry in the oven at 60 ± 5 °C for 48 h or until the sample is completely dry.
- Weigh 140 g of NaCl.
- Add the salt to a beaker triple-washed with filtered distilled water.
- Bring the volume to ~1 L with filtered distilled water.
- Stir the solution with a glass rod for 5 minutes or until fully saturated.
- Wash the sieve, rubber band, and filtration membrane 3× with filtered distilled water.
- Cover the top of the sieve with the membrane and secure it with the rubber band.
- Fit the filtration system over a new and clean beaker.
- Filter the saline solution.
- Weigh 100 g of dry sediment and transfer it to the beaker containing the filtered saline solution.
- Homogenize the mixture for 5 minutes with a glass rod.
- Cover the beaker with aluminum foil and allow it to rest for 4 h for decantation.
- Filter the supernatant using a 20 µm mesh, avoiding disturbance of the settled sediment.
- Clean the bench with 70% ethanol.
- Place three Petri dishes beside the stereoscopic microscope, filling the bottom with filtered distilled water (blank).
- Rinse the 20 µm mesh with filtered distilled water, transferring the material to a new Petri dish.
- Analyze the plates under the stereoscopic microscope and identify possible microplastics.
- Record morphology and predominant color of each microplastic particle.
- Transfer the microplastics to labeled microtubes (1.5–2 mL) containing 70% ethanol.
References
- Montagner, C.C.; Dias, M.A.; Paiva, E.M.; Vidal, C. Microplásticos: ocorrência ambiental e desafios analíticos. Quim Nova 2021, 44, 1328–52. [Google Scholar] [CrossRef]
- Geyer, R.; Jambeck, J.R.; Law, K.L. Production, use, and fate of all plastics ever made. Sci Adv. 2017, 3(7), e1700782. [Google Scholar] [CrossRef]
- Pappis, T.; Kapusta, S.C.; Ojeda, T. Metodologia de extração de microplásticos associados a sedimentos de ambientes de água doce. Eng Sanit Ambient. 2021, 26, 749–56. [Google Scholar] [CrossRef]
- Browne, M.A.; Galloway, T.; Thompson, R. Microplastic-an emerging contaminant of potential concern? Integr Environ Assess Manag 2007, 3(4). [Google Scholar] [CrossRef] [PubMed]
- Thompson, R.C.; Olsen, Y.; Mitchell, R.P.; Davis, A.; Rowland, S.J.; John, A.W.; et al. Lost at sea: where is all the plastic? Science 2004, 304(5672), 838. [Google Scholar] [CrossRef]
- Vendel, A.L.; Bessa, F.; Alves, V.E.; Amorim, A.L.; Patrício, J.; Palma, A.R. Widespread microplastic ingestion by fish assemblages in tropical estuaries subjected to anthropogenic pressures. Mar Pollut Bull. Available at. 2017, 117(1-2), 448–55. [Google Scholar] [CrossRef] [PubMed]
- Hooper, D.U.; Chapin, F.S., III; Ewel, J.J.; Hector, A.; Inchausti, P.; Lavorel, S.; et al. Effects of biodiversity on ecosystem functioning: a consensus of current knowledge. Ecol Monogr. 2005, 75. [Google Scholar] [CrossRef]
- Falasco, C.F. Avaliação da presença de partículas insolúveis e microplásticos em sais de origem marinha do Brasil [tese]; Universidade de São Paulo: São Paulo, 2019. [Google Scholar]
- Rakib, M.R.; Al Nahian, S.; Newton, S.; Parvin, A.; Eke, W.; Komola, R.; et al. Microplastics pollution in salt pans from the Maheshkhali Channel, Bangladesh. Sci Rep. 2021, 11(1), 23187. [Google Scholar] [CrossRef]
- Imhof, H.K.; Ivleva, N.P.; Schmid, J.; Niessner, R.; Laforsch, C. Contamination of beach sediments of a subalpine lake with microplastic particles. Curr Biol. 2013, 23, R867–8. [Google Scholar] [CrossRef]
- Frias, J.P.; Otero, V.; Sobral, P. Evidence of microplastics in samples of zooplankton from Portuguese coastal waters. Mar Environ Res. 2014, 95, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.K.; Hong, S.H.; Jang, M.; Kang, J.H.; Kwon, O.Y.; Han, G.M.; et al. Large accumulation of micro-sized synthetic polymer particles in the sea surface microlayer. Environ Sci Technol. 2014, 48, 9014–21. [Google Scholar] [CrossRef]
- Enders, K.; Lenz, R.; Stedmon, C.A.; Nielsen, T.G. Abundance, size and polymer composition of marine microplastics ≥10 μm in the Atlantic Ocean and their modelled vertical distribution. Mar Pollut Bull. 2015, 100, 70–81. [Google Scholar] [CrossRef]
- Everall, N.J.; Chalmer, J.M.; Griffiths, P.R. Vibrational spectroscopy of polymers: principles and practice; Wiley-VCH: Weinheim, 2007. [Google Scholar]
- Käppler, A.; Fischer, D.; Oberbeckmann, S.; Schernewski, G.; Labrenz, M.; Eichhorn, K.J.; et al. Analysis of environmental microplastics by vibrational microspectroscopy: FTIR, Raman or both? Anal Bioanal Chem. 2016, 408, 8377–91. [Google Scholar] [CrossRef]
- Paiva, B.O.; Lima, A.C.; Silva, C.P.; Souza, T.C.; Vendel, A.L. How to control the airborne contamination in laboratory analyses of microplastics? Braz Arch Biol Technol. 2022, 65. [Google Scholar] [CrossRef]
- Koelmans, A.A.; Redondo-Hasselerharm, P.E.; Nor, N.H.; de Ruijter, V.N.; Mintenig, S.M.; Kooi, M. Risk assessment of microplastic particles. Nat Rev Mater. Available from. 2022, 7(2), 138–52. [Google Scholar] [CrossRef]
- Pereira, C. Impacto por microplásticos no sedimento em sistemas aquáticos da Paraíba [relatório PIBIC]; UEPB: Campina Grande, 2023. [Google Scholar]
- Yang, D.; Shi, H.; Li, L.; Li, J.; Jabeen, K.; Kolandhasamy, P. Microplastic pollution in table salts from China. Environ Sci Technol. 2015, 49(22), 13622–7. [Google Scholar] [CrossRef]
- Ha, D.T. Microplastic contamination in commercial sea salt of Vietnam. Vietnam J Sci Technol. 2021, 59(3), 333–44. [Google Scholar]
- Makhdoumi, P.; Hossini, H.; Pirsaheb, M. Microplastic pollution in table salt and sugar: occurrence, qualification and quantification and risk assessment. J Food Compos Anal. 2023, 119, 105261. [Google Scholar] [CrossRef]
- Rochman, C.M.; Tahir, A.; Williams, S.L.; Baxa, D.V.; Lam, R.; Miller, J.T.; et al. Anthropogenic debris in seafood: plastic debris and fibers from textiles in fish and bivalves sold for human consumption. Sci Rep. 2015, 5(1), 1–10. [Google Scholar] [CrossRef] [PubMed]
- da Silva Batista, K.A.; Amado, E.M. Impactos da poluição marinha por microplásticos em anêmonas do mar: estado da arte e perspectivas futuras. In Editora Licuri; 2023; pp. 104–17. [Google Scholar] [CrossRef]
- Alves, V.E.N.; Figueiredo, G.M. Microplastic in the sediments of a highly eutrophic tropical estuary. Mar Pollut. Bull. 2019, 146, 326–35. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Chae, K.J. A systematic protocol of microplastics analysis from their identification to quantification in water environment: A comprehensive review. J Hazard Mater. 2020. [Google Scholar] [CrossRef] [PubMed]
- Abourich, M.; Bellamy, A.; Ajji, A.; Claveau-Mallet, D. Washing machine filters to mitigate microplastics release: Citizen science study to estimate microfibers capture potential and assess their social acceptability. Environ Chall. 2024, 16, 100984. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



