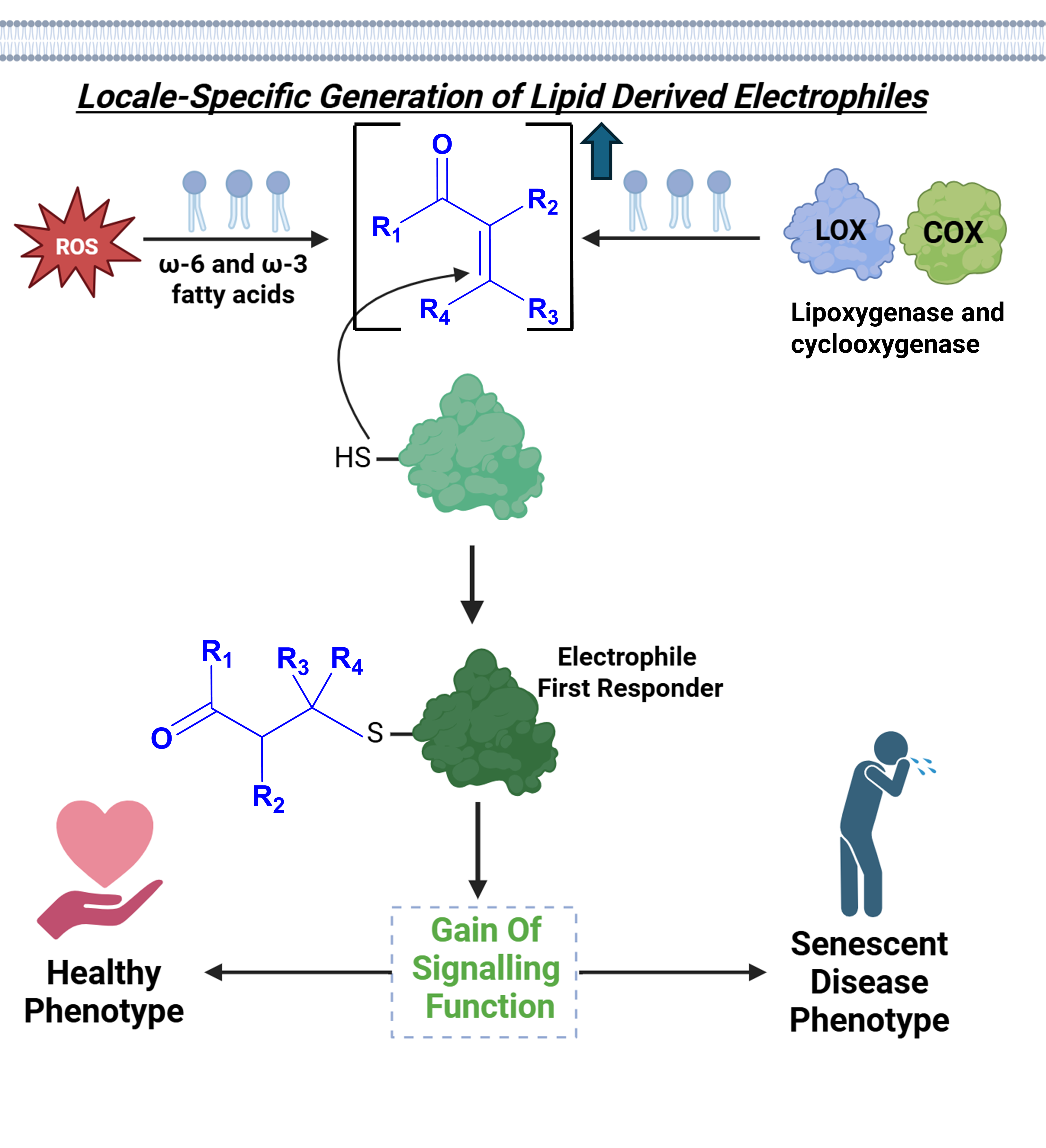
Cellular stress signaling conveys vital messages to the cell’s machinery to respond dynamically to internal and external environmental cues. One prevailing hypothesis for such signaling is to bring about crucial downstream functional changes in the cell’s ability to withstand intra- and extra-cellular stress, as exemplified by the NRF2-ARE pathway. Reactive electrophilic metabolites (REMs) are often generated from membrane lipids or respiratory metabolites in inflammatory and stress signaling contexts. REMs harbour an innate ability to irreversibly bind protein-cysteines, and form DNA and RNA adducts. Our work has led us to propose a new hypothesis regarding the role of locale-specific REM build-up in stress signalling; whereby they can label both the majority stable protein residents in a given locale, as well as minority guests transiently existing within a subcellular compartment, at a kinetically significant rate to trigger a “gain of function” signalling cascade. Much like the NRF2 signalling pathway, such downstream signalling messages could assist the cell’s ability to survive against microenvironment-specific stress and adapt on-demand. As REMs accumulate in several disease-specific cells, especially age-related disorders such as cancer and neurodegeneration, understanding the functional signal propagation mechanisms shaped by specific REMs engaging with specific biomolecular targets will prove vital for future therapeutic interventions with enhanced precision and context.