Submitted:
21 April 2026
Posted:
22 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials and Equipment
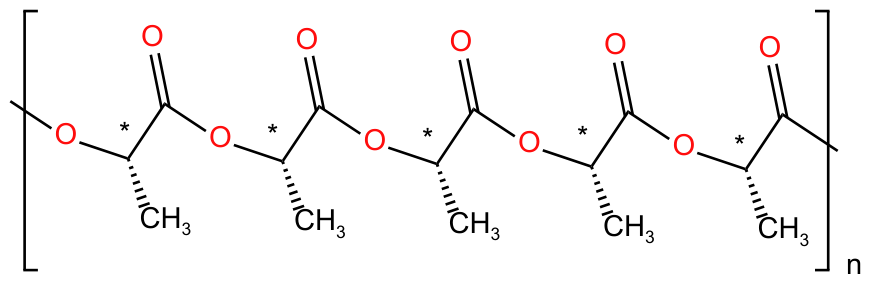
2.2. Preparation of the PLLA-3D or PLLA-40kDa Solutions
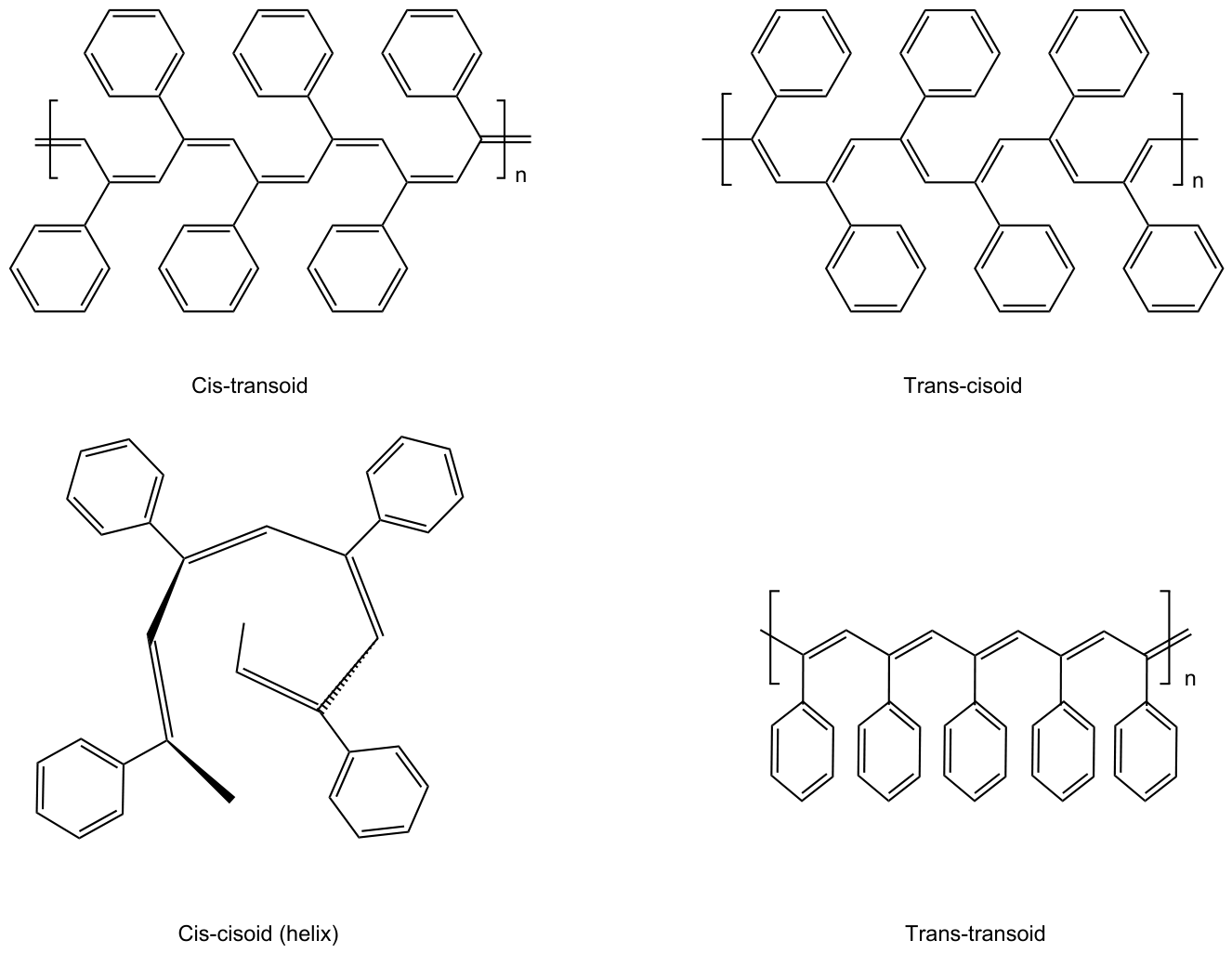
2.3. Synthesis of Polyphenylacetylene (PPA)
2.4. Determination of the Equilibrium Constant Between PLLA-40kDa and PPA
3. Results and Discussion
3.1. Analysis of the ORD Data with the Moffitt-Yang Equation
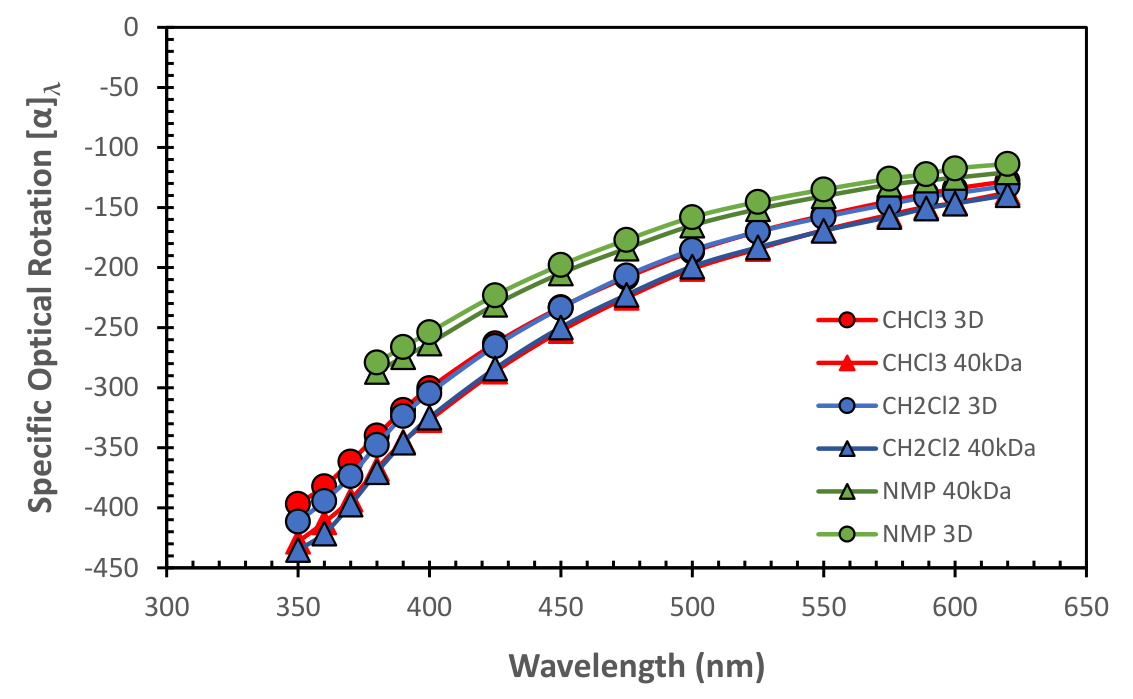
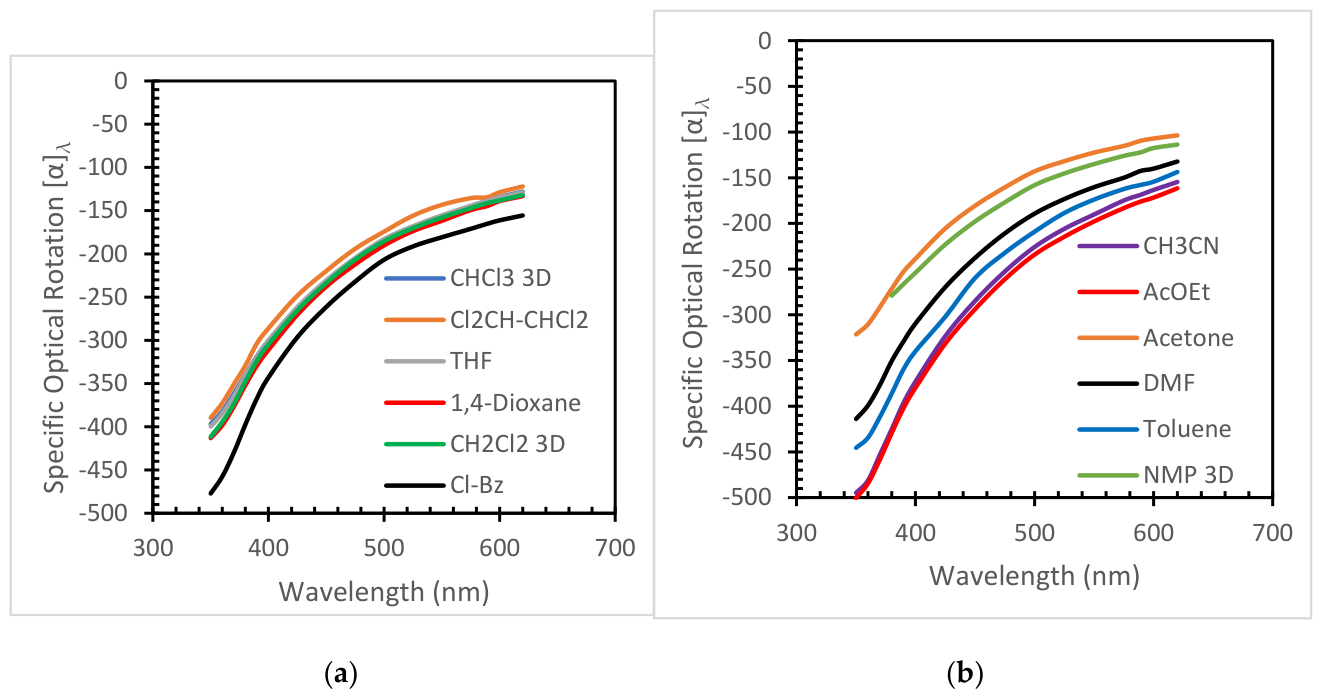
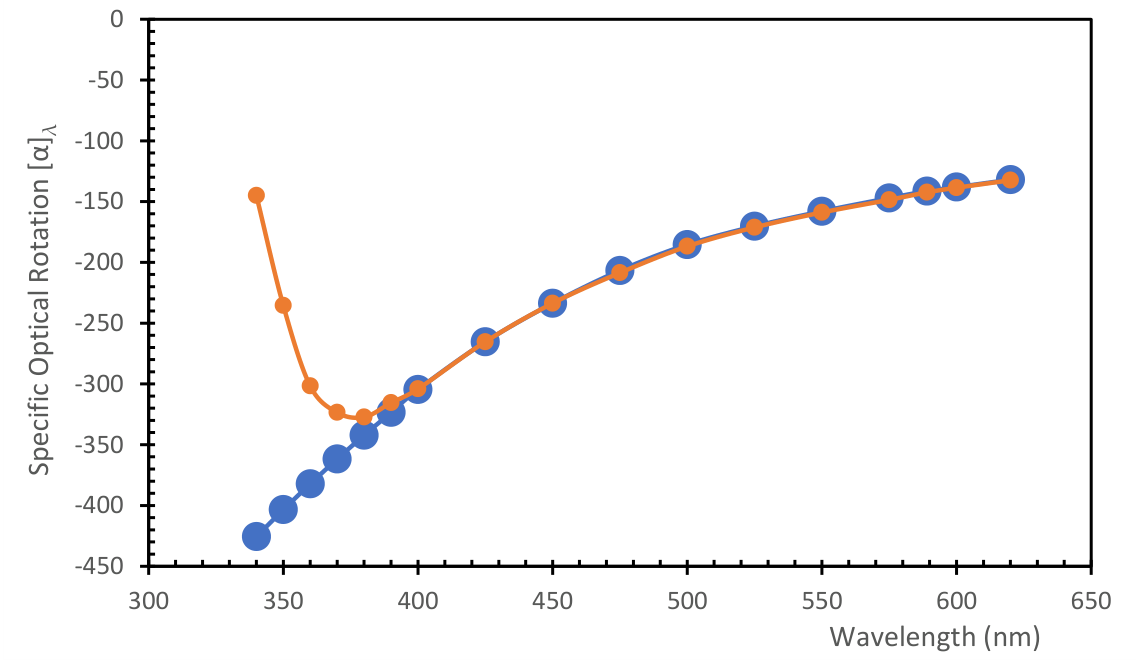
3.1.1. ORD Measurement of PLLA in Selected Solvents
3.1.2. The Moffitt Model and the Moffitt-Yang Equation
3.2.1. The Moffitt-Yang Equation Applied to Previous ORD Data: A Comparison with the Current Data
4.1. On the Interaction Between PLLA-40kDa and PPA in Solution
4.1.1. Expected PLLA-40kDa and PPA Interaction in Solution
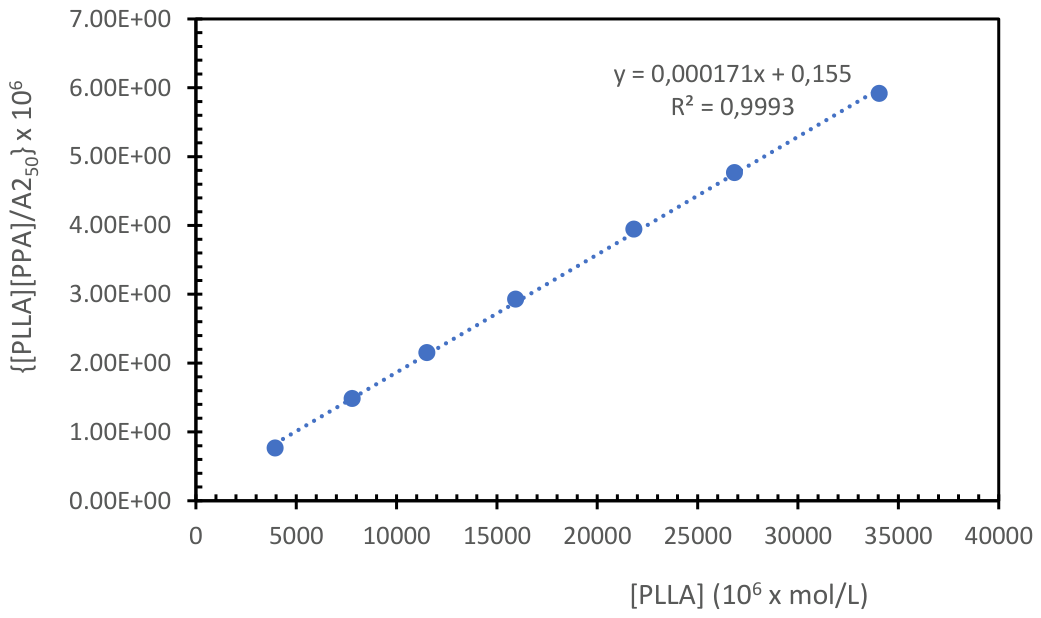
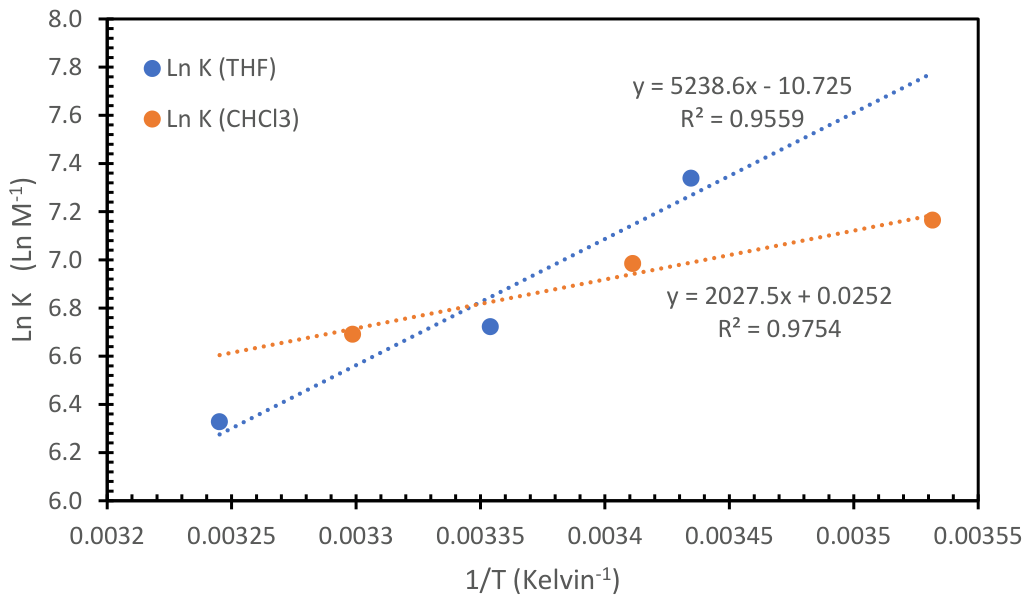
4.1.2. Determination of the Equilibrium Association Constant in the PLLA-40kDa and PPA Interaction in CHCl3 Solution
4.1.3. Determination of the Induced Cotton Effect in PLLA-PPA Complex
4. Conclusions
Funding
Data Availability Statement
Conflicts of Interest
References
- Synthesis, Structure and Properties of Poly (lactic acid); Di Lorenzo, M. L., Androsch, R., Eds.; Springer International Publishing: Cham, Switzerland, 2018; Adv. Polym. Sci.2018; Volume 279, pp. 1–343. [Google Scholar]
- Industrial Applications of Poly (lactic acid); Di Lorenzo, M. L., Androsch, R., Eds.; Springer International Publishing: Cham: Switzerland, Adv. Polym. Sci.; 2018; Volume 282, pp. 1–221. [Google Scholar]
- Rosli, N. A.; Karamanlioglu, M.; Kargarzadeh, H.; Ahmad, I. Comprehensive exploration of natural degradation of poly (lactic acid) blends in various degradation media: A review. Internat. J. Biol. Macromol. 2021, 187, 732–741. [Google Scholar] [CrossRef] [PubMed]
- Tashiro, K.; Wang, H.; Kouno, N.; Koshobu, J.; Watanabe, K. Confirmation of the X-ray-analyzed heterogeneous distribution of the PDLA and PLLA chain stems in the crystal lattice of poly (lactic acid) stereocomplex on the basis of the vibrational circular dichroism IR spectral measurement. Macromol. 2017, 50, 8066–8071. [Google Scholar] [CrossRef]
- Schultz, R.C.; Schwaab, J. Rotationdispersion bei monomeren und polymerem L-(-)-lactid. Macromol. Chem. Phys. 1965, 87, 90–102. [Google Scholar] [CrossRef]
- Goodman, M.; D'Alagni, M. Investigations of poly-S-lactic acid structure in solution. J. Polym. Sci. Part B: Polym. Lett. 1967, 5, 515–521. [Google Scholar] [CrossRef]
- Matsuo, S.; Iwakura, Y. Analysis of optical rotatory dispersion of poly-and oligo (l-lactate) by two-term Drude equation. Macromol. Chem. Phys. 1972, 152, 203–215. [Google Scholar] [CrossRef]
- Hongen, T.; Taniguchi, T.; Nomura, S.; Kadokawa, J. I.; Monde, K. In depth study on solution-state structure of poly (lactic acid) by vibrational circular dichroism. Macromol. 2014, 47, 5313–5319. [Google Scholar] [CrossRef]
- Taniguchi, T.; Hongen, T.; Monde, K. Studying the stereostructures of biomolecules and their analogs by vibrational circular dichroism. Polym. J. 2016, 48, 925–931. [Google Scholar] [CrossRef]
- Taniguchi, T. Analysis of molecular configuration and conformation by (electronic and) vibrational circular dichroism: Theoretical calculation and exciton chirality method. Bull. Chem. Soc. Japan 2017, 90, 1005–1016. [Google Scholar] [CrossRef]
- Wen, T.; Wang, H. F.; Li, M. C.; Ho, R. M. Homochiral evolution in self-assembled chiral polymers and block copolymers. Acc. Chem. Res. 2017, 50, 1011–1021. [Google Scholar] [CrossRef]
- Suzuki, Y.; Watanabe, T.; Kosugi, H.; Ueda, K.; Kikuchi, M.; Narumi, A.; Kawaguchi, S. Dilute solution properties of poly (d, l-lactide) by static light scattering, SAXS, and intrinsic viscosity. Polym. J. 2020, 52, 387–396. [Google Scholar] [CrossRef]
- Suzuki, Y.; Watanabe, T.; Kosugi, H.; Ueda, K.; Kikuchi, M.; Narumi, A.; Kawaguchi, S. Chain conformation of poly(D-lactide) in tetrahydrofuran by static light scattering, small-angle X-ray scattering, and intrinsic viscosity. Macromol. 2020, 53, 1604–1612. [Google Scholar] [CrossRef]
- Cataldo, F. Formation and decomposition of Poly (l-lactic acid) charge-transfer complex with iodine: A new molecular switch. Polym. Degradat.Stabil. 2020, 176, 109155. [Google Scholar] [CrossRef]
- Cataldo, F. On the interaction of C60 fullerene with poly (L-lactic acid) or poly (lactide). Fullerenes Nanot.Carbon Nanostruct. 2020, 28, 621–626. [Google Scholar] [CrossRef]
- Cataldo, F. On the optical activity of poly (l-lactic acid)(PLLA) oligomers and polymer: Detection of multiple cotton effect on thin PLLA solid film loaded with two dyes. Internat. J. Mol. Sci. 2020, 22, 8. [Google Scholar] [CrossRef] [PubMed]
- Cataldo, F. Some aspects concerning the interaction of C70 fullerene with poly (L-lactic acid)(PLLA). Fullerenes Nanot. Carbon Nanostruct. 2021, 29, 608–614. [Google Scholar] [CrossRef]
- Cataldo, F.; Barzaga, R.; García-Hernández, D. A.; Manchado, A.; Di Sarcina, I.; Cemmi, A. High energy radiation induced degradation of poly (lactic acid)(PLLA) film: radiation chemical yield, kinetics, mechanism and radioracemization evidences. J. Radioanal. Nucl. Chem. 2025, 334, 2875–2885. [Google Scholar] [CrossRef]
- Marubayashi, H.; Asai, S.; Sumita, M. Complex crystal formation of poly (l-lactide) with solvent molecules. Macromol. 2012, 45, 1384–1397. [Google Scholar] [CrossRef]
- Tanaka, T.; Tsutsui, A.; Tanaka, K.; Yamamoto, K.; Kadokawa, J. I. Evaluation of stability of amylose inclusion complexes depending on guest polymers and their application to supramolecular polymeric materials. Biomolecules 2017, 7, 28. [Google Scholar] [CrossRef]
- Orio, S.; Yamamoto, K.; Kadokawa, J. I. Preparation and material application of amylose-polymer inclusion complexes by enzymatic polymerization approach. Polymers 2017, 9, 729. [Google Scholar] [CrossRef] [PubMed]
- Kowalewska, A.; Nowacka, M. Supramolecular interactions in hybrid polylactide blends—the structures, mechanisms and properties. Molecules 2020, 25, 3351. [Google Scholar] [CrossRef] [PubMed]
- Świerczyńska, M.; Kudzin, M. H.; Chruściel, J. J. Poly (lactide)-based materials modified with biomolecules: a review. Materials 2024, 17, 5184. [Google Scholar] [CrossRef]
- Jang, Y.; Jang, J.; Kim, B. Y.; Song, Y. S.; Lee, D. Y. Effect of gelatin content on degradation behavior of PLLA/gelatin hybrid membranes. Tissue Engin. Regenerat. Medicine 2024, 21, 557–569. [Google Scholar] [CrossRef]
- Lu, H.; Oh, H. H.; Kawazoe, N.; Yamagishi, K.; Chen, G. PLLA–collagen and PLLA–gelatin hybrid scaffolds with funnel-like porous structure for skin tissue engineering. Sci. Technol. Adv. Mater. 2012, 13, 064210. [Google Scholar] [CrossRef]
- Chen, S.; Hao, Y.; Cui, W.; Chang, J.; Zhou, Y. Biodegradable electrospun PLLA/chitosan membrane as guided tissue regeneration membrane for treating periodontitis. J. Mater. Sci. 2013, 48, 6567–6577. [Google Scholar] [CrossRef]
- Li, Y.; Zhao, L.; Han, C.; Xiao, L.; Yu, Y.; Zhou, G.; Xu, M. Effect of the molecular weight of poly (vinyl acetate) on the polymorphism and thermomechanical properties of poly (L-lactic acid)/poly (D-lactic acid) blends. J. Therm. Anal. Calorim. 2022, 147, 3171–3184. [Google Scholar] [CrossRef]
- Li, Y.; Liu, L.; Shi, Y.; Xiang, F.; Huang, T.; Wang, Y.; Zhou, Z. Morphology, rheological, crystallization behavior, and mechanical properties of poly (l-lactide)/ethylene-co-vinyl acetate blends with different VA contents. J. Appl. Polym. Sci. 2011, 121, 2688–2698. [Google Scholar] [CrossRef]
- Le, K. P.; Lehman, R.; Remmert, J.; Vanness, K.; Ward, P. M. L.; Idol, J. D. Multiphase blends from poly (L-lactide) and poly(methyl mathacrylate). J. Biomater. Sci. Polym. Ed. 2006, 17, 121–137. [Google Scholar] [CrossRef] [PubMed]
- Samuel, C.; Cayuela, J.; Barakat, I.; Müller, A. J.; Raquez, J. M.; Dubois, P. Stereocomplexation of polylactide enhanced by poly(methyl methacrylate): improved processability and thermomechanical properties of stereocomplexable polylactide-based materials. ACS Appl. Mater. Interf. 2013, 5, 11797–11807. [Google Scholar] [CrossRef] [PubMed]
- Samuel, C.; Raquez, J. M.; Dubois, P. PLLA/PMMA blends: A shear-induced miscibility with tunable morphologies and properties? Polymer 2013, 54, 3931–3939. [Google Scholar] [CrossRef]
- Lim, M. P. A.; Lee, W. L.; Widjaja, E.; Loo, S. C. J. One-step fabrication of core–shell structured alginate–PLGA/PLLA microparticles as a novel drug delivery system for water soluble drugs. Biomater. Sci. 2013, 1, 486–493. [Google Scholar] [CrossRef]
- Liang, J.; Deng, J. Chiral particles consisting of helical polylactide and helical substituted polyacetylene: preparation and synergistic effects in enantio-differentiating release. Macromol. 2018, 51, 4003–4011. [Google Scholar] [CrossRef]
- Djerassi, C. Optical Rotatory Dispersion. Applications to Organic Chemistry; McGraw-Hill Book Co. Inc.: New York, 1960; Volume Chapter 17. [Google Scholar]
- Jirgensons, B. Optical Activity of Proteins and Other Macromolecules, 2nd Ed. ed; Chapman & Hall Ltd.: London, UK, 1973; pp. 16–17. [Google Scholar]
- Schultz, R. Addition compounds and complexes with polymers and models. Pure Appl. Chem. 1974, 38, 227–247. [Google Scholar] [CrossRef]
- Schultz, R. Modification of chiral properties due to the interaction of polymers and small molecules or ions. In Optically Active Polymers; Selegny, E., Ed.; Reidel: Dordrecht, The Netherlands, 1979; pp. 267–290. [Google Scholar]
- Hatano, M. Induced circular dichroism in biopolymer-dye systems. Adv. Polym. Sci. 1986, 77, 1–136. [Google Scholar]
- Furlani, A.; Napoletano, C.; Russo, M. V.; Feast, W. J. Stereoregular polyphenylacetylene. Polym. Bull. 1986, 16, 311–317. [Google Scholar] [CrossRef]
- Furlani, A.; Napoletano, C.; Russo, M. V.; Camus, A.; Marsich, N. The influence of the ligands on the catalytic activity of a series of Rh(I) complexes in reactions with phenylacetylene: Synthesis of stereoregular poly(phenyl)acetylene. J. Polym. Sci.Part A: Polym. Chem. 1989, 27, 75–86. [Google Scholar] [CrossRef]
- Cataldo, F. A spectroscopic study of polyacetylene prepared by using Rh (I) catalysts. Polymer 1994, 35, 5235–5240. [Google Scholar] [CrossRef]
- Yoshida, Y.; Mawatari, Y.; Motoshige, A.; Motodshige, R.; Hiraoki, T.; Tabata, M. Helix oscillation of polyacetylene esters detected by dynamic 1H NMR, IR, and UV-vis methods in solution. Polym. Chem. 2013, 4, 2982–2988. [Google Scholar] [CrossRef]
- Motoshige, R.; Mawatari, Y.; Motoshige, A.; Yoshida, Y.; Sasaki, T.; Yoshimizu, H.; Suzuki, T.; Tsujita, Y.; Tabata, M. Mutual conversion between stretched and contracted helices accompanied by a drastic change in color and spatial structure of poly (phenylacetylene) prepared with a [Rh(nbd)Cl]2-amine catalyst. J. Polym. Sci. Part A Polym. Chem. 2014, 52, 752–759. [Google Scholar] [CrossRef]
- Tabata, M.; Mawatari, Y. Emerging π-conjugated stretched and contracted helices and their mutual conversions of substituted polyacetylenes prepared using an organo-rhodium catalyst. Polym. Rev. 2017, 57, 65–88. [Google Scholar] [CrossRef]
- Akagi, K.; Mori, T. Helical polyacetylene—origins and synthesis. Chem. Record 2008, 8, 395–406. [Google Scholar] [CrossRef] [PubMed]
- Yashima, E.; Maeda, K.; Iida, H.; Furusho, Y.; Nagai, K. Helical polymers: synthesis, structures, and functions. Chem. Rev. 2009, 109, 6102–6211. [Google Scholar] [CrossRef]
- Masuda, T. Substituted polyacetylenes: Synthesis, properties, and functions. Polymer Reviews 2017, 57, 1–14. [Google Scholar] [CrossRef]
- Pisetsky, W.; Müller, T. J. Recent advances in the synthesis of substituted polyacetylenes. Catalysts 2025, 15, 50. [Google Scholar] [CrossRef]
- Maeda, K.; Yashima, E. Helical polyacetylenes induced via noncovalent chiral interactions and their applications as chiral materials. Top. Curr. Chem. 2017, 375, 72. [Google Scholar] [CrossRef]
- Cataldo, F.; Ursini, O.; Angelini, G. Synthesis and study of the thermal and chiro-optical properties of polyacetylenes with bulky side groups: Poly (1-ethynyl-4-biphenyl), Poly (1-ethynyl-4-phenoxybenzene) and Poly (1-ethynyl-4-pentylbenzene). J. Macromol. Sci. Part A: Pure Appl. Chem. 2009, 46, 860–869. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, X.; Huang, C. B.; Hu, L.; Wang, X. Q.; Wang, W.; Yang, H. B. Inducing and switching the handedness of polyacetylenes with topologically chiral [2] catenane pendants. Angew. Chemie Int. Ed. 2024, 63, e202408271. [Google Scholar] [CrossRef]
- Ikai, T.; Okuda, S.; Tsuzuki, A.; Suzuki, N.; Yashima, E. Switchable and self-learning enantioseparation based on ultrafast helix induction and memory of a helical polyacetylene. J. Amer. Chem. Soc. 2026. [Google Scholar] [CrossRef]
- Zhang, C.; Wang, H.; Su, G.; Li, R.; Shen, X.; Zhang, S.; Geng, Q.; Liu, F.; Otsuka, I.; Satoh, T.; Kakuchi, T. Synthesis and conformation effects of poly (phenylacetylene) s having chiral and racemic polylactide pendants. Polym. Internat. 2012, 61, 1158–1162. [Google Scholar] [CrossRef]
- Gao, X.; Wang, J.; Yang, K.; Zhao, B.; Deng, J. Regulating the helical chirality of racemic polyacetylene by chiral polylactide for realizing full-color and white circularly polarized luminescence. Chem. Mater. 2022, 34, 6116–6128. [Google Scholar] [CrossRef]
- Orellana Barrasa, J.; Ferrández-Montero, A.; Ferrari, B.; Pastor, J. Y. Characterisation and modelling of PLA filaments and evolution with time. Polymers 2021, 13, 2899. [Google Scholar] [CrossRef] [PubMed]
- Báez, J. E.; Marcos-Fernández, Á; Galindo-Iranzo, P. Exploring the effect of alkyl end group on poly (L-lactide) oligo-esters. Synthesis and characterization. J. Polym. Res. 2011, 18, 1137–1146. [Google Scholar] [CrossRef]
- Tsuji, H.; Sugimoto, S. Long terminal linear alkyl group as internal crystallization accelerating moiety of poly (l-lactide). Polymer 2014, 55, 4786–4798. [Google Scholar] [CrossRef]
- Garcia-Hernandez, D. A.; Cataldo, F.; Manchado, A. Charge-transfer interaction between C60 fullerene and alkylnaphthalenes. Fullerenes Nanot.Carbon Nanostruct. 2017, 25, 223–229. [Google Scholar] [CrossRef]
- García-Hernández, D. A.; Cataldo, F.; Manchado, A. C70 Fullerene charge-transfer interaction with alkylnaphthalenes, pinenes, and a diene. Fullerenes Nanot.Carbon Nanostruct 2017, 25, 505–511. [Google Scholar] [CrossRef]
- Scott, R. L. Some comments on the Benesi-Hildebrand equation. Recueil Trav. Chim. Pays-Bas 1956, 75, 787–789. [Google Scholar] [CrossRef]
- Moffitt, W. Optical rotatory dispersion of helical polymers. J. Chem. Phys. 1956, 25, 467–478. [Google Scholar] [CrossRef]
- Moffitt, W.; Fitts, D. D.; Kirkwood, J. G. Critique of the theory of optical activity of helical polymers. Proc. Nation. Acad. Sci. 1957, 43, 723–730. [Google Scholar] [CrossRef]
- McLachlan, A. D. Macromolecules Optical rotation in helical polymers. Proc. Royal Soc. London Ser. A. Mathem. Phys. Sci. 1967, 297, 141–149. [Google Scholar] [CrossRef]
- Stern, E.S.; Timmons, C. Gillam and Stern’s Introduction to Electronic Absorption Spectroscopy in Organic Chemistry, 3rd Ed. ed; Edward Arnold Publishers Ltd: London, UK, 1970; p. 54. [Google Scholar]
- McClellan, A.L. Tables of Experimental Dipole Moments; Freeman Co.: San Francisco, USA, 1963. [Google Scholar]
- Maryott, A.; Smith, E.R. Tables of Dielectric Constants. US Dept. of Commerce; National Bureau of Standards, 10 August 1951; Volume Circular 514. [Google Scholar]
- Mohamed, M. H.; Wilson, L. D.; Headley, J. V.; Peru, K. M. Thermodynamic properties of inclusion complexes between β-cyclodextrin and naphthenic acid fraction components. Energy & Fuels 2015, 29, 3591–3600. [Google Scholar] [CrossRef]
- Preuss, M. D.; Jansen, S. A.; Vantomme, G.; Meijer, E. W. Chirality and Supramolecular Copolymerizations–The Elusive Role of Subtle Solvation Effects. Israel J. Chem. 2021, 61, 622–628. [Google Scholar] [CrossRef]
- Aparicio, F.; Sánchez, L. Thermodynamics of the helical, supramolecular polymerization of linear self-asembling molecules: influence of hydrogen bonds and π stacking. Chem. Eur. J. 2013, 19(32). [Google Scholar] [CrossRef] [PubMed]
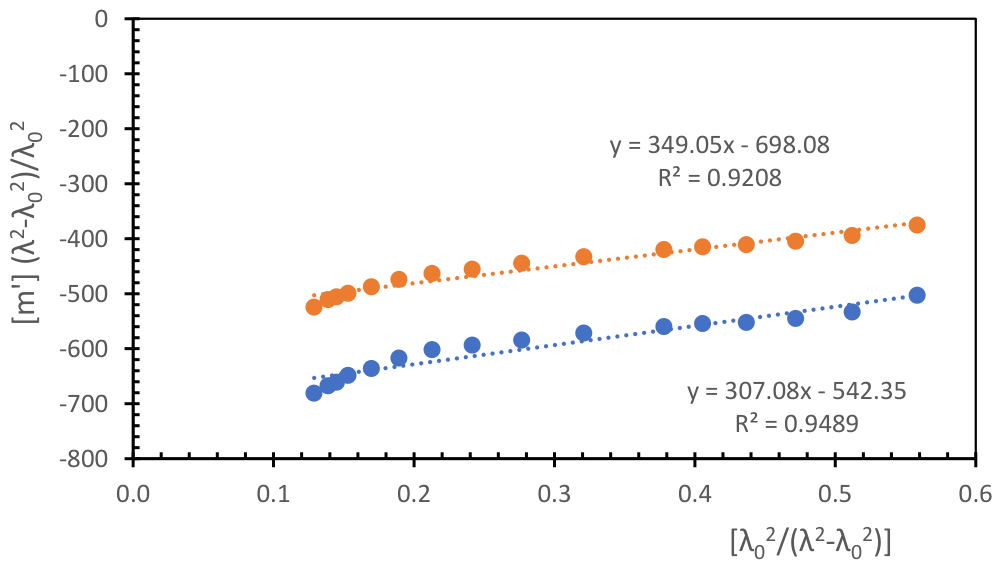
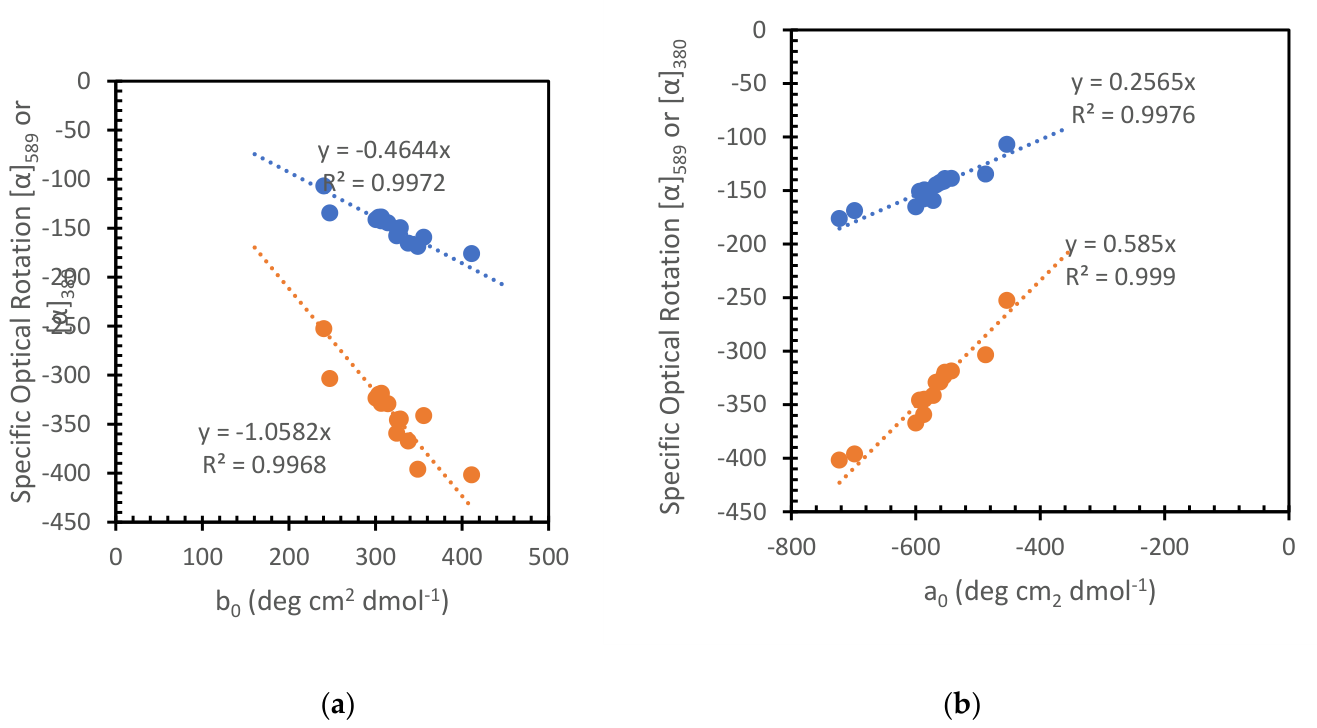
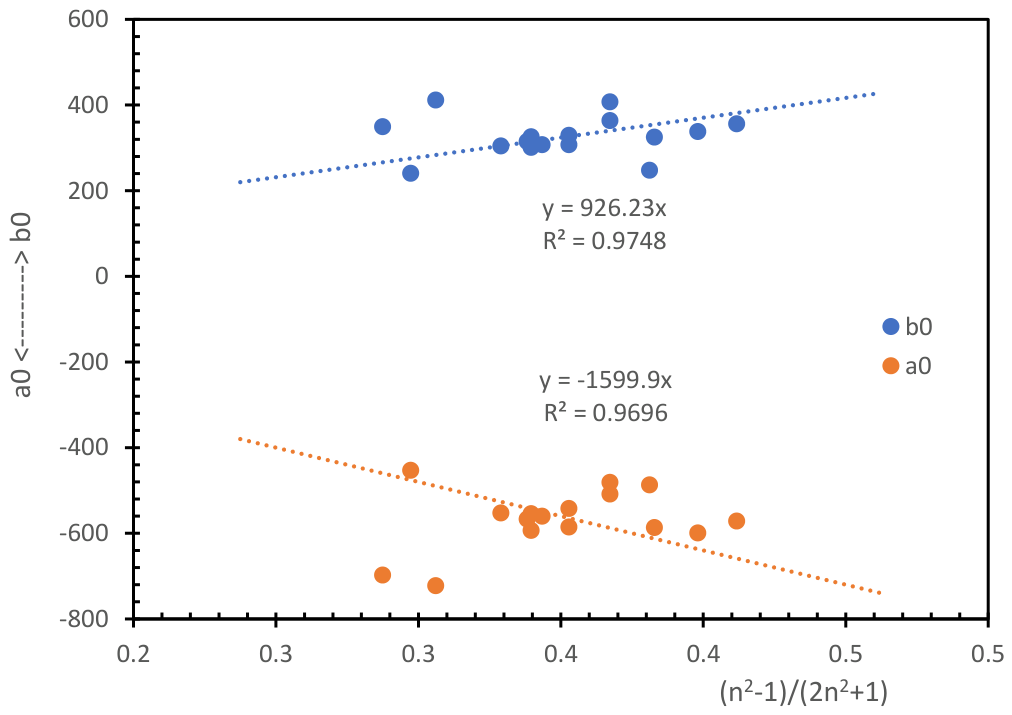
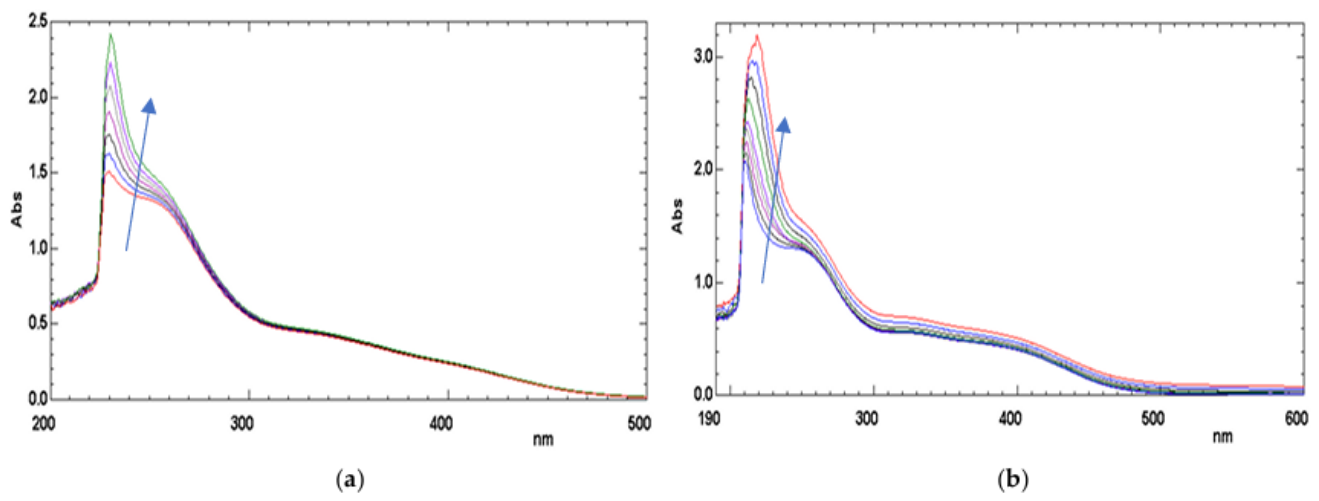
| SOLVENT and SOLUTE |
SOLVENT POLARITY (ε)/(μ) (*) |
Polarizability (**) |
b0 (Equation (4)) |
a0 (Equation (4)) |
[a]589 | [a]380 | % (w/V) (***) |
| Ethyl Acetate (****) [PLLA-3D] | 6.02 / 2.05 | 0.3061 | 411.1 | -722.7 | -176.1 | -401.8 | 0.135 |
| Acetonitrile [PLLA-3D] | 37.5 /3.39 | 0.2875 | 349.0 | -698.1 | -168.7 | -396.1 | 0.484 |
| o-Dichlorobenzene (†) [PLLA-3D] | 9.93/2.56 | 0.4118 | 345.8 | -571.7 | -159.3 | -341.4 | 0.246 |
| Chlorobenzene [PLLA-3D] | 5.71/1.75 | 0.3981 | 337.9 | -599.8 | -165.3 | -367.2 | 0.246 |
| Chloroform [PLLA-40kDa] | 4.81/1.10 | 0.3529 | 328.9 | -585.8 | -149.5 | -344.9 | 0.670 |
| Dichloromethane [PLLA-40kDa] | 9.08/1.85 | 0.3396 | 325.5 | -593.6 | -150.7 | -345.8 | 0.450 |
| Toluene (****)[PLLA-3D] | 2.38/0.36 | 0.3929 | 324.7 | -587.0 | -157.9 | -359.3 | 0.450 |
| Dioxane [PLLA-3D] | 2.21/0.0 | 0.3382 | 314.6 | -567.1 | -144.7 | -329.2 | 0.484 |
| Chloroform [PLLA-3D] | 4.81/1.1 | 0.3529 | 307.1 | -542.3 | -138.7 | -318.6 | 0.670 |
| N,N'-dimethylformamide [PLLA-3D] | 36.7/3.85 | 0.3435 | 306.9 | -560.7 | -142.6 | -328.8 | 0.463 |
| Tetrahydrofuran [PLLA-3D) | 8.20/1.63 | 0.3290 | 303.9 | -552.8 | -138.9 | -319.7 | 0.495 |
| Dichloromethane [PLLA-3D] | 9.08/1.85 | 0.3396 | 300.7 | -554.6 | -141.4 | -323.5 | 0.423 |
| 1,1',2,2'-Tetrachloroethane [PLLA-3D] | 8.2/1.85 | 0.3812 | 247.4 | -487.5 | -134.6 | -303.5 | 0.413 |
| Acetone (****) [PLLA-3D] | 20.7/2.86 | 0.2973 | 240.4 | -453.4 | -106.9 | -252.6 | 0.467 |
| N-Methyl-pyrrolidone (‡) [PLLA-40kDa] | 32.3/4.09 | 0.3673 | 407.0 | -508.8 | -127.9 | -274.7 | 0.261 |
| N-Methyl-pyrrolidone (‡) [PLLA-3D] | 32.3/4.09 | 0.3673 | 363.2 | -481.3 | -122.2 | -266.2 | 0.468 |
| SOLVENT (‡) |
b0 (Equation (4)) |
a0 (Equation (4)) |
λ0 (nm) (*) | Spectral Range (nm) (**) | % (w/V) (***) | Reference |
| Acetonitrile PLLA-3D | 349.0 | -698.1 | 209.5 | 350-620 | 0.484 | This Work |
| Acetonitrile | 277.3 | -572.9 | 209.5 | 280-400 | n.r. (#) | [6] |
| Chloroform PLLA-40kDa | 328.9 | -585.8 | 209.5 | 350-620 | 0.67 | This Work |
| Chloroform | 317.3 | -634.4 | 201.0 | 365-578 | 0.72 | [5] |
| Chloroform PLLA-3D | 307.1 | -542.3 | 209.5 | 350-620 | 0.67 | This Work |
| Chloroform | 280.9 | -713.2 | 209.5 | 280-400 | n.r. (#) | [6] |
| Dichloromethane PLLA-40kDa | 325.5 | -593.6 | 209.5 | 350-620 | 0.45 | This Work |
| Dichloromethane | 309.9 | -654.8 | 201.0 | 365-578 | 0.72 | [5] |
| Dichloromethane PLLA-3D | 300.7 | -554.6 | 209.5 | 350-620 | 0.42 | This Work |
| m-Cresol | 305.7 | -984.6 | 201.0 | 365-578 | 0.72 | [5] |
| Trifluoroethanol | 298.8 | -899.9 | 209.5 | 280-400 | n.r. (#) | [6] |
| 1,1,2-Trichloroethane | 297.3 | -574.3 | 201.0 | 365-578 | 0.72 | [5] |
| Dichloroacetic acid | 279.7 | -680.9 | 201.0 | 365-578 | 0.72 | [5] |
| Trifluoroacetic acid | 273.9 | -901.8 | 209.5 | 260-400 | n.r. (#) | [6] |
| Sulfuric acid | 199.6 | -585.3 | 201.0 | 365-578 | 0.72 | [5] |
| Solvent | T(°C) | Λ (nm) | K (M-1) |
| Tetrahydrofuran | 18°C | 247/315/400 | 1539 |
| Tetrahydrofuran | 25°C | 247/315/400 | 831 |
| Tetrahydrofuran | 35°C | 247/315/400 | 560 |
| Chlorofom | 10°C | 240/250 | 1293 |
| Chlorofom | 20°C | 240/250 | 1080 |
| Chlorofom | 30°C | 240/250 | 805 |
| Dichloromethane | 16°C | 250 | 1103 |
| Solvent | ΔH°(kJ/mol) | ΔS°(J mol-1 K-1) | ΔG°(kJ/mol) |
| Tetrahydrofuran | -43.6 | -89.17 | -17.0 |
| Chloroform | -16.9 | +0.210 | -16.8 |
| Dichloromethane | n.d | n.d. | -16.8 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




