Submitted:
21 April 2026
Posted:
22 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods
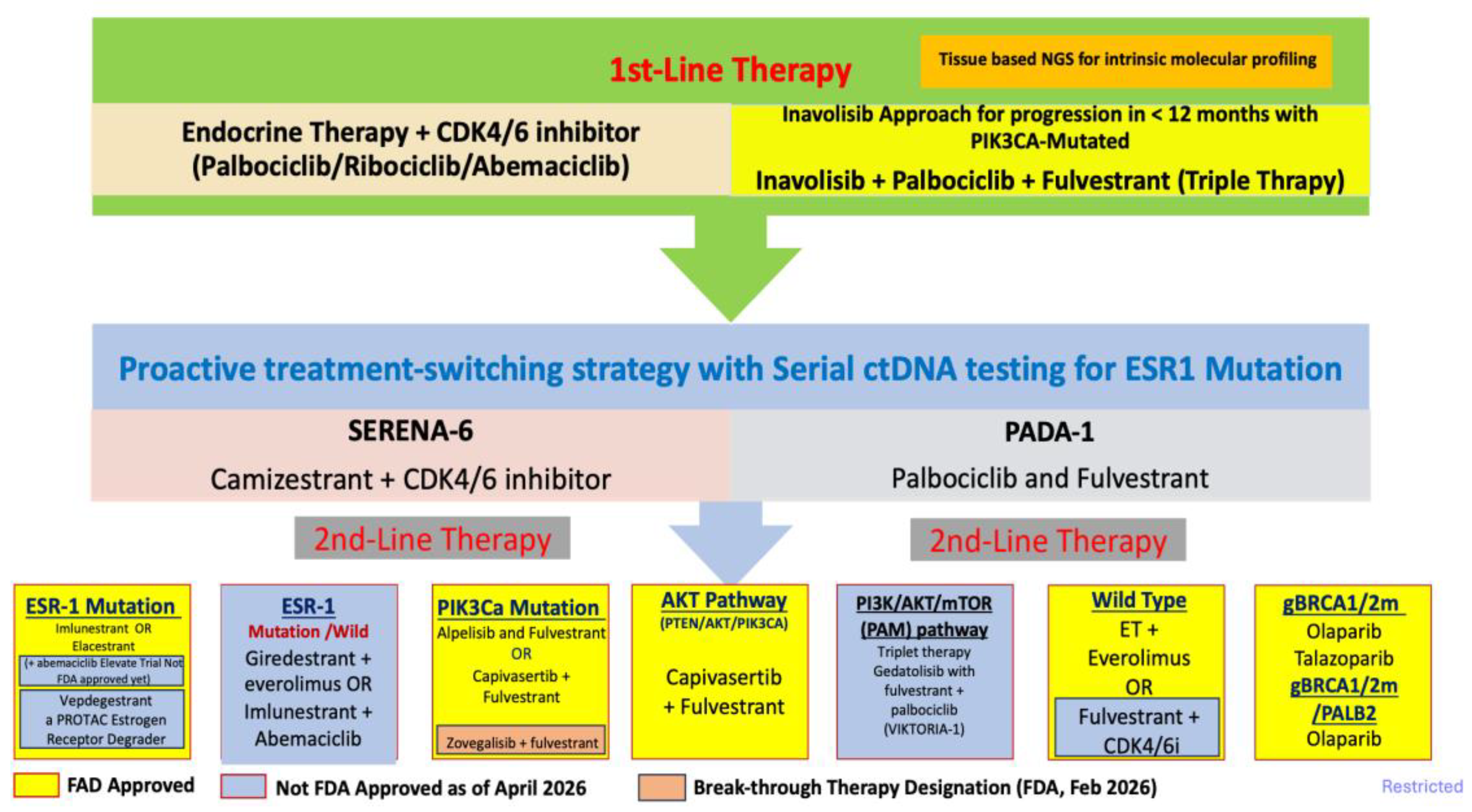
3. Genomic Profiling in Diagnostic Workup
4. Real-World Evidence and Pathway-Directed Therapeutic Strategies
4.1. Alpelisib
4.2. Elacestrant
4.3. Inavolisib
4.4. Capivasertib
4.5. Everolimus
5. Genomic Resistance Architecture and Clinical Decision Frameworks
5.1. Decision Framework for Co-Altered Populations
5.2. Therapeutic Strategies for Patients Lacking Actionable Genomic Alterations in Post-CDK4/6i HR+/HER2− mBC
6. Emerging Molecularly Targeted Therapies
| Trial (NCT) | Phase | Molecular Selection | Intervention vs Control | Primary Endpoint | Key Results | HR (95% CI) | Regulatory Status |
|---|---|---|---|---|---|---|---|
| EMERALD (NCT03778931) | 3 | ESR1-mutated ER+/HER2− mBC post-ET | Elacestrant vs SOC ET | PFS (ESR1-mut) | Median PFS 3.8 vs 1.9 mo; ≥12 mo prior ET+CDK4/6i: 8.6 vs 1.9 mo | 0.55 (0.39–0.77) (Exploratory) | FDA approved (Jan 2023) |
| INAVO120 (NCT04191499) | 3 | PIK3CA-mutated HR+/HER2− LA/mBC | Inavolisib + palbociclib + fulvestrant vs placebo + palbociclib + fulvestrant | PFS | Median PFS 17.2 vs 7.3 mo; OS 34.0 vs 27.0 mo; delayed chemotherapy | PFS: 0.43 (0.32–0.59); OS: 0.67 (0.49–0.91) | FDA approved (Oct 2024) |
| CAPItello-291 (NCT04305496) | 3 | HR+/HER2− advanced BC; AKT pathway-altered subgroup | Capivasertib + fulvestrant vs placebo + fulvestrant | PFS | Overall: 7.2 vs 3.6 mo; Altered: 7.3 vs 3.1 mo | Overall: 0.60 (0.51–0.71); Altered: 0.50 (0.38–0.65) | FDA approved (Nov 2023) |
| BOLERO-2 | 3 | AI-resistant HR+/HER2− BC (pre-CDK4/6 era) | Everolimus + exemestane vs placebo + exemestane | PFS | 7.8 vs 3.2 mo; post-CDK4/6 RWE ~5.3 mo | 0.45 (0.38–0.54) | Established therapy |
| Trial (NCT) | Phase | Therapeutic Class | Population | Intervention | Primary Endpoint | Key Findings | Status |
|---|---|---|---|---|---|---|---|
| VERITAC-2 (NCT05654623) | 3 | PROTAC ER degrader | ESR1-mut HR+/HER2− post-ET + CDK4/6i | Vepdegestrant vs fulvestrant | PFS (ESR1-mut) | 5.0 vs 2.1 mo; HR 0.57–0.58; low AE discontinuation | Regulatory review ongoing |
| EMBER-3 (NCT04975308) | 3 | Oral SERD ± CDK4/6i | HR+/HER2− advanced BC | Imlunestrant vs SOC; Imlunestrant + abemaciclib | PFS | ESR1-mut: 5.5 vs 3.8 mo (HR 0.62); Combo: 9.4 vs 5.5 mo (HR 0.57) | Regulatory status evolving |
| PERSEVERE (NCT04546009) | 3 | Oral SERD | HR+/HER2− post-CDK4/6i | Giredestrant vs physician’s choice ET | PFS | Primary endpoint not met; numerical ESR1-mut benefit | Development strategy ongoing |
| ELEVATE | 2 | SERD-based combinations | HR+/HER2− post-CDK4/6i | Elacestrant + everolimus / abemaciclib | PFS | 8.3 mo (everolimus); 14.3 mo (abemaciclib) | Proof-of-concept |
| ADELA (NCT06382948) | 3 | SERD + mTOR inhibitor | ESR1-mut HR+/HER2− | Elacestrant + everolimus vs monotherapy | PFS | Ongoing | Recruiting |
| INAVO121 | 3 | PI3K inhibitor comparison | PIK3CA-mut HR+/HER2− | Inavolisib vs alpelisib | PFS | Direct comparative efficacy study | Active enrollment |
| ReDiscover (NCT05216432) ReDiscover-2 (NCT06982521) | 1/2 (Ph 3 ongoing) | PIK3CA-mutated HR+/HER2− mBC post-CDK4/6i | Zovegalisib + fulvestrant (Ph 3: vs capivasertib + fulvestrant) | Zovegalisib + fulvestrant (Ph 3: vs capivasertib + fulvestrant) | ReDiscover = Safety, ReDiscover-2 = PFS | Median PFS 11.1 mo (95% CI 7.3–13.0) at recommended Phase 3 dose 400 mg BID with food | Breakthrough Therapy Designation (FDA, Feb 2026); Phase 3 ongoing |
7. Circulating Tumor DNA-Guided Proactive Switching for ESR1 Mutations: Evidence and Implementation
7.1. SERENA-6 Trial
7.2. PADA-1 Trial
| Trial | Strategy | Population | Median PFS (mo) | HR | Key Finding |
|---|---|---|---|---|---|
| SERENA-6 | Camizestrant + CDK4/6i vs AI + CDK4/6i | Emergent ESR1 | 16.0 vs 9.2 | 0.44 | Proactive switch prolongs PFS |
| PADA-1 | Fulvestrant + palbo vs AI + palbo | Rising ESR1 | 11.9 vs 5.7 | 0.61 | Early intervention beneficial |
7.3. Implementation Considerations
8. Limitations
9. Conclusions and Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ADC | antibody–drug conjugate |
| AE | adverse event |
| AI | aromatase inhibitor |
| AKT | protein kinase B |
| ASCO | American Society of Clinical Oncology |
| AURKA | aurora kinase A |
| BC | breast cancer |
| CDK | cyclin-dependent kinase |
| CDK2 | cyclin-dependent kinase 2 |
| CDK4/6 | cyclin-dependent kinase 4 and 6 |
| CDK4/6i | cyclin-dependent kinase 4/6 inhibitor |
| CCNE | cyclin E |
| CI | confidence interval |
| CNS | central nervous system |
| CT | chemotherapy |
| ctDNA | circulating tumor DNA |
| DCR | disease control rate |
| DNA | deoxyribonucleic acid |
| D/C | discontinuation |
| ddPCR | droplet digital polymerase chain reaction |
| ER | estrogen receptor |
| ERBB2, | Erb-B2 receptor tyrosine kinase 2 |
| ESMO | European Society for Medical Oncology |
| ESR1 | estrogen receptor 1 gene |
| ET | endocrine therapy |
| FDA | Food and Drug Administration |
| FGFR | fibroblast growth factor receptor |
| HR | hazard ratio |
| HR+ | hormone receptor–positive |
| HER2 | human epidermal growth factor receptor 2 |
| HER2− | human epidermal growth factor receptor 2–negative |
| ITT | intention-to-treat |
| mBC | metastatic breast cancer |
| mPFS | median progression-free survival |
| mTOR | mechanistic target of rapamycin |
| NCCN | National Comprehensive Cancer Network |
| NCT | National Clinical Trial identifier |
| NDA | new drug application |
| NGS | next-generation sequencing |
| OS | overall survival |
| PD | progressive disease |
| PDUFA | Prescription Drug User Fee Act |
| PFS | progression-free survival |
| PI3K | phosphoinositide 3-kinase |
| PIK3CA | phosphatidylinositol-4,5-bisphosphate 3-kinase catalytic subunit alpha |
| PRO | patient-reported outcome |
| PROTAC | proteolysis-targeting chimera |
| QoL | quality of life |
| RAS | rat sarcoma viral oncogene |
| RB1 | retinoblastoma 1 gene |
| RECIST | Response Evaluation Criteria in Solid Tumors |
| RWE | real-world evidence |
| SERD | selective estrogen receptor degrader |
| SOC | standard of care |
| TP53 | tumor protein 53 |
| TTNT | time to next treatment |
| VAF | variant allele frequency |
References
- Siegel, R.L.; Giaquinto, A.N. Jemal A: Cancer statistics, 2024. CA Cancer J Clin 2024, 74(1), 12–49. [Google Scholar]
- Harbeck, N.; Brufsky, A.; Rose, C.G.; Korytowsky, B.; Chen, C.; Tantakoun, K.; Jazexhi, E.; Nguyen, D.H.V.; Bartlett, M.; Samjoo, I.A. Real-world effectiveness of CDK4/6i in first-line treatment of HR+/HER2- advanced/metastatic breast cancer: updated systematic review. Front Oncol 2025, 15, 1530391. [Google Scholar] [CrossRef] [PubMed]
- Goyal, R.K.; Nagar, S.P.; Kabir, E.R. e: Progression-free survival and overall survival with CDK 4/6 inhibitors combination therapy versus endocrine monotherapy in hormone receptor-positive, HER2-negative metastatic breast cancer: a systematic literature review and meta-analysis. Breast Cancer 2023, 30(3), 387–402. [Google Scholar]
- Piezzo, M.; Cocco, S.; Caputo, R. e: Targeting cell cycle in breast cancer: CDK4/6 inhibitors. International Journal of Molecular Sciences 2020, 21(18), 6479. [Google Scholar] [CrossRef]
- Wander, S.A.; Cohen, O.; Gong, X.; Johnson, G.N.; Buendia-Buendia, J.E.; Lloyd, M.R.; Kim, D.; Luo, F.; Mao, P.; Helvie, K. The Genomic Landscape of Intrinsic and Acquired Resistance to Cyclin-Dependent Kinase 4/6 Inhibitors in Patients with Hormone Receptor-Positive Metastatic Breast Cancer. Cancer Discov 2020, 10(8), 1174–1193. [Google Scholar] [CrossRef]
- da Silva, J.L.; Oliveira, L.J.C.; de Resende, C.A.A.; Reinert, T.; de Albuquerque, L.Z.; Leite da Silva, L.F.; Mano, M.S. Understanding and overcoming CDK4/6 inhibitor resistance in HR+/HER2- metastatic breast cancer: clinical and molecular perspectives. Ther Adv Med Oncol 2025, 17, 17588359251353623. [Google Scholar] [CrossRef] [PubMed]
- Spoerke, J.M.; Gendreau, S.; Walter, K. e: Heterogeneity and clinical significance of ESR1 mutations in ER-positive metastatic breast cancer patients receiving fulvestrant. Nature Communications 2016, 7, 11579. [Google Scholar] [CrossRef]
- André, F.; Ciruelos, E.; Rubovszky, G. e: Alpelisib for PIK3CA-mutated, hormone receptor-positive advanced breast cancer. New England Journal of Medicine 2019, 380(20), 1929–1940. [Google Scholar] [CrossRef]
- O’Leary, B.; Cutts, R.J.; Liu, Y. e: The genetic landscape and clonal evolution of breast cancer resistance to palbociclib plus fulvestrant in the PALOMA-3 trial. Cancer Discovery 2018, 8(11), 1390–1403. [Google Scholar] [CrossRef]
- Razavi, P.; Chang, M.T.; Xu, G. e: The genomic landscape of endocrine-resistant advanced breast cancers. Cancer Cell 2018, 34(3), 427–438. [Google Scholar] [CrossRef]
- Wander, S.A.; Cohen, O.; Gong, X. e: The genomic landscape of intrinsic and acquired resistance to cyclin-dependent kinase 4/6 inhibitors in patients with hormone receptor-positive metastatic breast cancer. Cancer Discovery 2020, 10(8), 1174–1193. [Google Scholar] [CrossRef]
- Apostolidou, K.; Zografos, E.; Papatheodoridi, M.A.; Fiste, O.; Dimopoulos, M.A.; Zagouri, F. Oral SERDs alone or in combination with CDK 4/6 inhibitors in breast cancer: Current perspectives and clinical trials. Breast 2024, 75, 103729. [Google Scholar] [CrossRef]
- Formisano, L.; Lu, Y.; Servetto, A. e: Aberrant FGFR signaling mediates resistance to CDK4/6 inhibitors in ER+ breast cancer. Nature Communications 2019, 10(1), 1373. [Google Scholar] [CrossRef]
- Li, Z.; Zhang, Y.; Zhang, Z. e: The role of CDK4/6 inhibitors in breast cancer treatment: a systematic review and meta-analysis. Oncology Letters 2020, 19(6), 3555–3566. [Google Scholar]
- Lynce, F.; Shajahan-Haq, A.N.; Swain, S.M. CDK4/6 inhibitors in breast cancer therapy: current practice and future opportunities. [CrossRef]
- Turner, N.C.; Oliveira, M.; Howell, S.J. e: Camizestrant + CDK4/6 inhibitor for the treatment of emergent ESR1 mutations during first-line endocrine-based therapy ahead of disease progression in patients with HR+/HER2– advanced breast cancer: phase 3 SERENA-6 trial. Journal of Clinical Oncology 2025, 43, LBA4. [Google Scholar] [CrossRef]
- Chandarlapaty, S.; Chen, D.; He, W. e: Prevalence of ESR1 mutations in cell-free DNA and outcomes in metastatic breast cancer: a secondary analysis of the BOLERO-2 clinical trial. JAMA Oncology 2016, 2(10), 1310–1315. [Google Scholar] [CrossRef] [PubMed]
- Rugo, H.S.; Lerebours, F.; Ciruelos, E. e: Elacestrant in ER-positive, HER2-negative metastatic breast cancer with ESR1-mutated tumors: subgroup analyses from the EMERALD trial by prior duration of CDK4/6i and other clinical and biomarker factors. Clinical Cancer Research 2024, 30(19), 4299–4309. [Google Scholar]
- Baethge, C.; Goldbeck-Wood, S.; Mertens, S. SANRA-a scale for the quality assessment of narrative review articles. Res Integr Peer Rev 2019, 4, 5. [Google Scholar] [CrossRef]
- Rugo, H.S.; Di Palma, J.A.; Cortes, J. e: Real-world outcomes of elacestrant in patients with estrogen receptor-positive, HER2-negative, ESR1-mutant metastatic breast cancer. Clinical Cancer Research 2026, 32(1), 179–187. [Google Scholar] [CrossRef]
- Waks, A.G.; Winer, E.P. Breast cancer treatment: a review. JAMA 2019, 321(3), 288–300. [Google Scholar] [CrossRef]
- André, F.; Ciruelos, E.; Rubovszky, G.; Campone, M.; Loibl, S.; Rugo, H.S.; Iwata, H.; Conte, P.; Mayer, I.A.; Kaufman, B. Alpelisib for PIK3CA-Mutated, Hormone Receptor–Positive Advanced Breast Cancer. New England Journal of Medicine 2019, 380(20), 1929–1940. [Google Scholar] [CrossRef]
- Rugo, H.S.; Lerebours, F.; Ciruelos, E.; Drullinsky, P.; Ruiz-Borrego, M.; Neven, P.; Park, Y.H.; Prat, A.; Bachelot, T.; Juric, D. Alpelisib plus fulvestrant in PIK3CA-mutated, hormone receptor-positive advanced breast cancer after a CDK4/6 inhibitor (BYLieve): one cohort of a phase 2, multicentre, open-label, non-comparative study. The Lancet Oncology 2024, 25(12), e629–e638. [Google Scholar] [CrossRef]
- Rugo, H.S.; et al. Alpelisib plus fulvestrant for PIK3CA-mutated, HR+, HER2− advanced breast cancer after progression on CDK4/6 inhibitor: primary results from EPIK-B5. Abstract RF7-02. Clin Cancer Res. RF7-02. (EPIK-B5 trial). 2026, 32((4_) Suppl. [Google Scholar]
- Turner, N.C.; Oliveira, M.; Howell, S.J.; et al. Capivasertib plus fulvestrant in hormone receptor-positive (HR+) advanced breast cancer (ABC): exploratory ctDNA analyses from the Phase 3 CAPItello-291 trial. Abstract RF7-05 Presented at: San Antonio Breast Cancer Symposium, San Antonio, TX, December 9-12, 2025. [Google Scholar]
- Alaklabi, A.; et al. Real world outcomes with alpelisib in metastatic hormone receptor positive breast cancer: a single institution experience. Front Oncol. 2022, 12, 1012391. [Google Scholar] [CrossRef]
- Sultanbaev, A.K.; Kolyadina, I.V.; et al. Results of a retrospective study on the efficacy and safety of alpelisib in patients with HR+/HER2− metastatic breast cancer in real-world clinical practice. J Clin Oncol. 2024, 42((16_) suppl, e13058. [Google Scholar] [CrossRef]
- Bardia, A.; Aftimos, P.; Bihani, T. e: EMERALD: phase III trial of elacestrant (RAD1901) vs endocrine therapy for previously treated ER+ advanced breast cancer. Future Oncology 2019, 15(28), 3209–3218. [Google Scholar] [CrossRef] [PubMed]
- Bidard, F.C.; Kaklamani, V.G.; Neven, P. e: Elacestrant versus standard endocrine therapy for estrogen receptor-positive, human epidermal growth factor receptor 2-negative advanced breast cancer: results from the randomized phase III EMERALD trial. Journal of Clinical Oncology 2022, 40(26), 3246–3256. [Google Scholar] [CrossRef] [PubMed]
- Juric, D.; Ciruelos Garcia, E.M.; Loibl, S. e: Inavolisib plus palbociclib and fulvestrant in PIK3CA-mutated, hormone receptor-positive, HER2-negative locally advanced or metastatic breast cancer: updated efficacy and safety results from INAVO120. Annals of Oncology 2024, 35, S339. [Google Scholar]
- Juric, D.; Kalinsky, K.; Turner, N.C. e: Inavolisib or placebo in combination with palbociclib and fulvestrant in patients with PIK3CA-mutated, hormone receptor-positive, HER2-negative locally advanced or metastatic breast cancer: primary results from the phase 3 INAVO120 trial. Journal of Clinical Oncology 2024, 42 (suppl 17), LBA1000. [Google Scholar] [CrossRef]
- Turner, N.C.; et al. INAVO120: Phase III trial final overall survival (OS) analysis of first-line inavolisib/placebo + palbociclib + fulvestrant in patients with PIK3CA-mutated, HR+, HER2– endocrine-resistant advanced breast cancer. J Clin Oncol 2025, 43((16_) suppl, 1003. [Google Scholar] [CrossRef]
- Turner, N.C.; Oliveira, M.; Howell, S.J. e: Capivasertib in hormone receptor-positive advanced breast cancer. New England Journal of Medicine 2023, 388(22), 2058–2070. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, M.; Pominchuk, D.; Nowecki, Z. e: Capivasertib and fulvestrant for patients with aromatase inhibitor-resistant hormone receptor-positive, HER2-negative advanced breast cancer (CAPItello-291): overall survival results from a randomized, double-blind, placebo-controlled, phase 3 trial. Lancet Oncology 2024, 25(11), 1461–1471. [Google Scholar] [CrossRef]
- Schmid, P.; Zaiss, M.; Harper-Wynne, C. e: Capivasertib plus paclitaxel versus placebo plus paclitaxel as first-line therapy for metastatic triple-negative breast cancer: the phase 3 CAPItello-290 trial. Nature Medicine 2024, 30(5), 1408–1416. [Google Scholar]
- Jerusalem, G.; Rorive, A.; Collignon, J. Chemotherapy options for patients with triple-negative breast cancer in the post-anthracycline and taxane era: a systematic review. Current Oncology Reports 2016, 18(4), 24. [Google Scholar]
- Natsuhara, K.H.; Park, L.; Udayachalerm, S.; Mireia, R.; Murphy, B.; Nordstrom, B.; Huppert, L.A. Abstract PS5-05-23: Real-world Treatment Patterns of Capivasertib in Metastatic Breast Cancer in the US. Clinical Cancer Research 2026, 32((4_) Supplement, PS5-05-23-PS05-05-23. [Google Scholar] [CrossRef]
- Yardley, D.A.; Noguchi, S.; Pritchard, K.I. e: Everolimus plus exemestane in postmenopausal patients with HR+ breast cancer: BOLERO-2 final progression-free survival analysis. Advances in Therapy 2013, 30(10), 870–884. [Google Scholar] [CrossRef]
- Lee, J.S.; Yost, S.E.; Li, S.M. e: Real-world clinical outcomes of patients with metastatic breast cancer treated with everolimus plus exemestane after progression on a cyclin-dependent kinase 4/6 inhibitor. Clinical Breast Cancer 2021, 21(6), e675–e682. [Google Scholar]
- Bhave, M.A.; Quintanilha, J.C.F.; Tukachinsky, H.; Li, G.; Scott, T.; Ross, J.S.; Pasquina, L.; Huang, R.S.P.; McArthur, H.; Levy, M.A. Comprehensive genomic profiling of ESR1, PIK3CA, AKT1, and PTEN in HR(+)HER2(-) metastatic breast cancer: prevalence along treatment course and predictive value for endocrine therapy resistance in real-world practice. Breast Cancer Res Treat 2024, 207(3), 599–609. [Google Scholar] [CrossRef]
- Mosele, F.; Remon, J.; Mateo, J. e: Recommendations for the use of next-generation sequencing (NGS) for patients with metastatic cancers: a report from the ESMO Precision Medicine Working Group. Annals of Oncology 2020, 31(11), 1491–1505. [Google Scholar] [CrossRef] [PubMed]
- Nayar, U.; Cohen, O.; Kapstad, C. e: Acquired HER2 mutations in ER+ metastatic breast cancer confer resistance to estrogen receptor-directed therapies. Nature Genetics 2019, 51(2), 207–216. [Google Scholar] [CrossRef]
- Li, Z.; Razavi, P.; Li, Q. e: Loss of the FAT1 tumor suppressor promotes resistance to CDK4/6 inhibitors via the Hippo pathway. Cancer Cell 2018, 34(6), 893–905. [Google Scholar] [CrossRef]
- Turner, N.C.; Liu, Y.; Zhu, Z. e: Cyclin E1 expression and palbociclib efficacy in previously treated hormone receptor-positive metastatic breast cancer. Journal of Clinical Oncology 2019, 37(14), 1169–1178. [Google Scholar] [CrossRef]
- Hanker, A.B.; Sudhan, D.R.; Arteaga, C.L. Overcoming endocrine resistance in breast cancer. Cancer Cell 2020, 37(4), 496–513. [Google Scholar] [CrossRef]
- A Study Evaluating the Efficacy and Safety of Giredestrant Combined With Palbociclib Compared With Letrozole Combined With Palbociclib in Participants With Estrogen Receptor-Positive, HER2-Negative Locally Advanced or Metastatic Breast Cancer (persevERA Breast Cancer). ClinicalTrials.gov Identifier: NCT04546009; accessed; Hoffmann-La Roche, 2020 [updated February 20, 2026; (accessed on 3 March 2026). [Google Scholar]
- Jacobson, A. Alpelisib Plus Fulvestrant or Letrozole Demonstrates Sustained Benefits Across Subgroups of Patients with PIK3CA-Mutated HR+/HER2- Advanced Breast Cancer. Oncologist 2022, 27 (Suppl 1), S13–S14. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Rugo, H.S.; Lerebours, F.; Ciruelos, E. e: Alpelisib plus fulvestrant in PIK3CA-mutated, hormone receptor-positive advanced breast cancer after a CDK4/6 inhibitor (BYLieve): one cohort of a phase 2, multicentre, open-label, non-comparative study. Lancet Oncology 2021, 22(4), 489–498. [Google Scholar] [CrossRef] [PubMed]
- Piccart, M.; Hortobagyi, G.N.; Campone, M.; Pritchard, K.I.; Lebrun, F.; Ito, Y.; Noguchi, S.; Perez, A.; Rugo, H.S.; Deleu, I. Everolimus plus exemestane for hormone-receptor-positive, human epidermal growth factor receptor-2-negative advanced breast cancer: overall survival results from BOLERO-2†. Ann Oncol 2014, 25(12), 2357–2362. [Google Scholar] [CrossRef]
- Mo, H.; Renna, C.E.; Moore, H.C.F.; Abraham, J.; Kruse, M.L.; Montero, A.J.; LeGrand, S.B.; Wang, L.; Budd, G.T. Real-World Outcomes of Everolimus and Exemestane for the Treatment of Metastatic Hormone Receptor-Positive Breast Cancer in Patients Previously Treated With CDK4/6 Inhibitors. Clin Breast Cancer 2022, 22(2), 143–148. [Google Scholar] [CrossRef] [PubMed]
- Kalinsky, K.; Accordino, M.K.; Chiuzan, C.; Mundi, P.S.; Sakach, E.; Sathe, C.; Ahn, H.; Trivedi, M.S.; Novik, Y.; Tiersten, A. Randomized Phase II Trial of Endocrine Therapy With or Without Ribociclib After Progression on Cyclin-Dependent Kinase 4/6 Inhibition in Hormone Receptor-Positive, Human Epidermal Growth Factor Receptor 2-Negative Metastatic Breast Cancer: MAINTAIN Trial. J Clin Oncol 2023, 41(24), 4004–4013. [Google Scholar] [CrossRef]
- Kalinsky, K.; Bianchini, G.; Hamilton, E.; Graff, S.L.; Park, K.H.; Jeselsohn, R.; Demirci, U.; Martin, M.; Layman, R.M.; Hurvitz, S.A. Abemaciclib Plus Fulvestrant in Advanced Breast Cancer After Progression on CDK4/6 Inhibition: Results From the Phase III postMONARCH Trial. J Clin Oncol 2025, 43(9), 1101–1112. [Google Scholar] [CrossRef]
- Singareeka Raghavendra, A.; Damodaran, S.; Barcenas, C.H.; Fuqua, S.A.; Layman, R.M. Tripathy D: Personalizing therapies over the course of hormone receptor-positive/HER2-negative metastatic breast cancer. CA: A Cancer Journal for Clinicians 2026, 76(1), e70055. [Google Scholar] [PubMed]
- Cutts, R.J.; Ijaz, H.; Huang, X. e: Patterns of genomic instability and clonal evolution in breast cancer from AURORA, the Breast International Group Molecular Screening Initiative. Journal of Clinical Oncology 2023, 41((16) suppl, 1009. [Google Scholar]
- Damodaran, S.; Plourde, P.V.; Moore, H.C.F. e: Vepdegestrant versus fulvestrant in patients with oestrogen receptor-positive, HER2-negative, locally advanced or metastatic breast cancer (VERITAC-2): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncology 2024, 25(11), 1441–1454. [Google Scholar]
- Hamilton, E.P.; Schott, A.F.; Nanda, R. e: ARV-471, a PROTAC estrogen receptor degrader, combined with palbociclib in advanced ER+/HER2− breast cancer: phase 1b cohort of a phase 1/2 study. Journal of Clinical Oncology 2024, 42((16) suppl, 1050. [Google Scholar]
- Food, U.; Administration, D. FDA grants accelerated approval to erdafitinib for metastatic urothelial carcinoma. FDA. updated 12 Apr 2019) 2019. Available online: https://www.
- Jhaveri, K.; Shapiro, G.I.; Krop, I.E. e: The oral selective estrogen receptor degrader imlunestrant versus investigator choice of endocrine monotherapy in patients with ER+, HER2− advanced breast cancer following progression on endocrine therapy: primary results from EMBER-3. Journal of Clinical Oncology 2024, 42, LBA1000. [Google Scholar]
- Jhaveri, K.L.; Neven, P.; Casalnuovo, M.L.; Kim, S.-B.; Tokunaga, E.; Aftimos, P.; Saura, C.; O’Shaughnessy, J.; Harbeck, N.; Carey, L.A. Imlunestrant with or without Abemaciclib in Advanced Breast Cancer. New England Journal of Medicine 2025, 392(12), 1189–1202. [Google Scholar] [CrossRef]
- A Study Evaluating the Efficacy and Safety of Giredestrant Combined With Palbociclib Compared With Letrozole Combined With Palbociclib in Participants With Estrogen Receptor-Positive, HER2-Negative Locally Advanced or Metastatic Breast Cancer (persevERA Breast Cancer). 20 February 2026. Available online: https://ClinicalTrials.gov.
- Basu, S.; Rosas Diaz, A.N.; Narvaez-Paliza, J.M.; Curtin, C.; Kurella, S.; Urias, M.; More Verde, L.; Patel, J.M.; Asnani, A. Selective Estrogen Receptor Degraders Induce Bradycardia by Modulating Nuclear Estrogen Signaling. JACC Basic Transl Sci 2026, 11(2), 101454. [Google Scholar] [CrossRef]
- Brooks, L.; Dolton, M.; Langenhorst, J.; Yoshida, K.; Lien, Y.T.K.; Malhi, V.; Li, C.; Perez-Moreno, P.; Bond, J.; Chen, Y.C. Concentration QTc analysis of giredestrant: Overcoming QT/heart rate confounding in the presence of drug-induced heart rate changes. Clin Transl Sci 2023, 16(5), 823–834. [Google Scholar] [CrossRef]
- A Phase III Randomized, Double-Blind, Placebo-Controlled, Multicenter Study Evaluating the Efficacy and Safety of GDC-9545 Combined With Palbociclib Compared With Letrozole Combined With Palbociclib in Patients With Estrogen Receptor-Positive, HER2-Negative Locally Advanced or Metastatic Breast Cancer. NCT04546009; In. 2020.
- Turner, N.C.; et al. persevERA Breast Cancer (BC): Phase III study evaluating the efficacy and safety of giredestrant (GDC-9545) + palbociclib compared with letrozole + palbociclib for patients with ER-positive, HER2-negative locally advanced or metastatic BC. J Clin Oncol. 2021, 39((15_) suppl, TPS1103. [Google Scholar] [CrossRef]
- Hurvitz, S.; Pistilli, B.; Kaklamani, V.; Layman, R.M.; Cortés, J.; Park, K.H.; Cristofanilli, M.; Sullivan, B.; Moser, E.C.; Gorbatchevsky, I. Abstract CT258: A randomized, open-label, phase 3 study of fulvestrant and CDK4/6 inhibitors with or without gedatolisib as first-line treatment in patients with HR+/HER2- advanced breast cancer (VIKTORIA-2). Cancer Research 2025, 85((8_) Supplement_2. [Google Scholar] [CrossRef]
- Hurvitz, S.A.; Layman, R.M.; Curigliano, G.; André, F.; Cristofanilli, M.; Kim, S.B.; Rodriguez, J.L.M.; Nadal, J.C.; Kim, G.M.; Emile, G. LBA17 Gedatolisib (geda) + fulvestrant ± palbociclib (palbo) vs fulvestrant in patients (pts) with HR+/ HER2-/PIK3CA wild-type (WT) advanced breast cancer (ABC): First results from VIKTORIA-1. Annals of Oncology 2025, 36, S1562–S1563. [Google Scholar] [CrossRef]
- Rugo, H.; Tolaney, S.M.; Chan, N.; Borges, G.; Yerushalmi, R.; Sharifi, M.; McHayleh, W.; Beck, T.; Vidula, N.; Hamilton, E. Abstract RF7-01: Elacestrant in combination with everolimus or abemaciclib in patients with ER+/HER2- locally advanced or metastatic breast cancer (mBC): phase 2 results from ELEVATE, an open-label, umbrella study. Clinical Cancer Research 2026, 32((4_) Supplement, RF7-01-RF07-01. [Google Scholar] [CrossRef]
- ClinicalTrials.gov ID NCT06382948 details a Phase 3 study on Elacestrant plus Everolimus for ER+/HER2- advanced breast cancer, active but not recruiting. 2025.
- Juric, D.; Kalinsky, K.; Im, S.-A.; Ciruelos, E.; Bianchini, G.; Barrios, C.H.; Jacot, W.; Schmid, P.; Loi, S.; Rugo, H.S. INAVO121: Phase III study of inavolisib (INAVO) + fulvestrant (FUL) vs. alpelisib (ALP) + FUL in patients (pts) with hormone receptor-positive, HER2-negative (HR+, HER2–), PIK3CA-mutated (mut) locally advanced or metastatic breast cancer (LA/mBC). Journal of Clinical Oncology 2024, 42, TPS1136–TPS1136. [Google Scholar] [CrossRef]
- Therapeutics, Relay. Zovegalisib granted Breakthrough Therapy designation by the FDA for PIK3CA-mutated HR-positive, HER2-negative advanced breast cancer [Internet]. 3 Feb 2026. Available online: https://ir.relaytx.com/news-releases/news-release-details/relay-therapeutics-announces-zovegalisib-granted-breakthrough.
- Varkaris, A.; Italiano, A.; Sammons, S.; Felip Falgas, E.; Curigliano, G.; et al. Dose optimization of zovegalisib, a novel PI3Kα inhibitor, in patients (pts) with PIK3CA-mutant (m) HR+/HER2− advanced breast cancer (BC): Results from the first-in-human (FIH) study to support the recommended phase III dose [abstract 90O]. ESMO Open. 2026, 11 (Suppl 2), 106232. Available online: https://oncologypro.esmo.org/congress-resources/esmo-targeted-anticancer-therapies-congress-2026?presentation=dose_optimization_of_zovegalisib__a_novel_pi3ka_in. [CrossRef]
- ClinicalTrials.gov; National Library of Medicine (US). Identifier NCT06982521, A Phase 3 Open-Label Randomized Study Assessing the Efficacy and Safety of RLY-2608 + Fulvestrant Versus Capivasertib + Fulvestrant as Treatment for PIK3CA-mutant Hormone Receptor Positive, Human Epidermal Growth Factor Receptor 2 Negative (HR+/HER2-) Locally Advanced or Metastatic Breast Cancer Following Recurrence or Progression On or After Treatment With a CDK4/6 Inhibitor. Bethesda (MD), 21 May 2025. Available online: https://clinicaltrials.gov/study/NCT06982521.
- Lloyd, M.R.; Wander, S.A.; Hamilton, E.; Razavi, P.; Bardia, A. Next-generation selective estrogen receptor degraders and other novel endocrine therapies for management of metastatic hormone receptor-positive breast cancer: current and emerging role. Ther Adv Med Oncol 2022, 14, 17588359221113694. [Google Scholar] [CrossRef]
- Turner, N.C.; Oliveira, M.; Howell, S.J. e: Camizestrant, a next-generation oral SERD, versus continued aromatase inhibitor, for treatment of ER-positive, HER2-negative, advanced breast cancer patients with acquired ESR1 mutation on first-line aromatase inhibitor-based therapy ahead of disease progression: primary results from the randomised, phase 3 SERENA-6 study. New England Journal of Medicine 2025, 393(6), 569–580. [Google Scholar]
- Bidard, F.C.; Callens, C.; Dalenc, F. e: Circulating tumor DNA analysis in the randomized PADA-1 trial: clinical implications for hormone receptor-positive metastatic breast cancer. Cancer Discovery 2023, 13(10), 2128–2143. [Google Scholar]
- Lux, M.P.; Becnel, W.E.; Loibl, S. e: Health-related quality of life results from SERENA-6. Annals of Oncology 2025, 36, S1234. [Google Scholar]
- Bertrand, F.; Rochigneux, P.; Vicier, C. e: Circulating tumour DNA-guided switch from aromatase inhibitor plus palbociclib to fulvestrant plus palbociclib in women with oestrogen receptor-positive, HER2-negative advanced breast cancer developing an ESR1 mutation (PADA-1): a randomised, open-label, phase 3 trial. Lancet Oncology 2022, 23(11), 1367–1377. [Google Scholar]
- Bidard, F.; Hardy-Bessard, A.; Dalenc, F.; et al. Switch to fulvestrant and palbociclib versus no switch in advanced breast cancer with rising ESR1 mutation during aromatase inhibitor and palbociclib therapy (PADA-1): a randomised, open-label, multicentre, phase 3 trial. The Lancet Oncology 2022, 23, 1367–1377. [Google Scholar] [CrossRef]
- Keup, C.; Storbeck, M.; Hauch, S. e: Multimodal targeted deep sequencing of circulating tumor DNA in breast cancer patients. Genome Medicine 2021, 13(1), 4. [Google Scholar]
- Tolaney, S.M.; Garrett-Mayer, E.; White, J. e: Updated standardized definitions for efficacy end points in adjuvant breast cancer clinical trials: STEEP version 2.0. Journal of Clinical Oncology 2021, 39(24), 2720–2731. [Google Scholar] [CrossRef]
- Collin, S.M.; Hillman, S.; Shah, C.H.; Tank, R.; Bhave, M.; Traina, T.A. Real-World Treatment Patterns and Outcomes for Patients With Metastatic Triple-Negative Breast Cancer in the United States: An Observational Study. JCO Oncology Practice 2025, OP-25-00822. [Google Scholar] [CrossRef]
- Gennari, A.; André, F.; Barrios, C.H. e: ESMO Clinical Practice Guideline for the diagnosis, staging and treatment of patients with metastatic breast cancer. Annals of Oncology 2021, 32(12), 1475–1495. [Google Scholar] [CrossRef]
- Kamli, H.; Khan, N.U. Emerging Role of ctDNA Fragmentomics and Epigenetic Signatures in the Early Detection, Minimal Residual Disease Assessment, and Precision Monitoring of Renal Cell Carcinoma. Journal of Cellular and Molecular Medicine 2026, 30(3), e71019. [Google Scholar] [CrossRef] [PubMed]
- Turner, S.; Chia, S.; Kanakamedala, H.; Hsu, W.C.; Park, J.; Chandiwana, D.; Ridolfi, A.; Yu, C.L.; Zarate, J.P.; Rugo, H.S. Effectiveness of Alpelisib + Fulvestrant Compared with Real-World Standard Treatment Among Patients with HR+, HER2-, PIK3CA-Mutated Breast Cancer. Oncologist 2021, 26(7), e1133–e1142. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Turner, Nicholas C.; et al. INAVO120: Phase III trial final overall survival (OS) analysis of first-line inavolisib (INAVO)/placebo (PBO) + palbociclib (PALBO) + fulvestrant (FULV) in patients (pts) with PIK3CA-mutated, hormone receptor-positive (HR+), HER2-negative (HER2–), endocrine-resistant advanced breast cancer (aBC). J Clin Oncol 2025, 43, 1003–1003. [Google Scholar] [CrossRef]
- Agostinetto, Elisa; et al. Clinico-molecular characteristics associated with outcomes in breast cancer patients treated with CDK4/6 inhibitors: Results from the AURORA Molecular Screening Initiative. J Clin Oncol 2023, 41, 1019–1019. [Google Scholar] [CrossRef]
- Hamilton, E.P.; et al. Vepdegestrant versus fulvestrant in ER-positive/HER2-negative advanced breast cancer: Results of the global, randomized, phase 3 VERITAC-2 study. J Clin Oncol 2025, 43, LBA1000. [Google Scholar] [CrossRef]
| Therapeutic Class | Agent | Molecular Selection | Pivotal Trial(s) | Median PFS (months) | OS Signal | Regulatory Status (as of early 2026) | Clinical Positioning / Notes |
|---|---|---|---|---|---|---|---|
| Oral SERD | Elacestrant | ESR1-mutated | EMERALD | 3.8 (overall); 8.6 (≥12 mo prior CDK4/6i) | No statistically significant OS benefit yet | FDA approved (2023) | Preferred in ESR1-mut endocrine-sensitive disease; best in longer prior CDK4/6i exposure |
| AKT inhibitor | Capivasertib | AKT1/PIK3CA/PTEN altered | CAPItello-291 | 7.2 (overall); 7.3 (altered) | Not mature / no clear OS benefit yet | FDA approved | Pathway-altered (AKT/PI3K/PTEN) dominant tumors; post-CDK4/6i option |
| PI3Kα inhibitor (triplet) | Inavolisib (triplet: + palbociclib + fulvestrant) | PIK3CA-mutated | INAVO120 | 15.0 | Yes (HR 0.67; mature OS benefit) | FDA approved (2024) | PIK3CA-mut endocrine-resistant tumors; triplet strategy with clear OS benefit |
| mTOR inhibitor | Everolimus (+ exemestane) | None required | BOLERO-2 | 7.8 (pre-CDK4/6i era); ~3.8–5.4 in RWE post-CDK4/6i | No OS benefit | Approved (earlier line) | Later-line endocrine-based strategy; reduced efficacy post-CDK4/6i in real-world data |
| PI3K inhibitor | Alpelisib (+ fulvestrant) | PIK3CA-mutated | SOLAR-1 | 11.0 | No OS (numeric 7.9 mo improvement, not stat sig) | Approved | Earlier PI3K option post-CDK4/6i in PIK3CA-mut; established but hyperglycemia common |
| Endocrine therapy (backbone) | Fulvestrant | ER+ | Various (e.g., PALOMA-3 reference) | Limited monotherapy activity post-CDK4/6i | N/A | Approved | Backbone; limited single-agent efficacy post-CDK4/6i; used in combinations |
| PARP inhibitor | Olaparib | Germline BRCA1/2 mutated | OlympiAD / others | N/A (PARP context) | OS benefit in gBRCA population | Approved | Germline BRCA-mutated population; post-CDK4/6i if applicable |
| PARP inhibitor | Talazoparib | Germline BRCA1/2 mutated/PALB2 mutated | EMBRACA | N/A (PARP context) | OS benefit in gBRCA population | Approved | Germline BRCA-mutated population; post-CDK4/6i if applicable |
| Oral SERD | Camizestrant | ESR1-mutated | SERENA-6 (proactive switch) | N/A (investigational) | N/A | Phase III / investigational | Investigational; proactive ctDNA-guided in ESR1-mut |
| Oral SERD / PROTAC ER degrader | Imlunestrant | ESR1-mutated | EMBER-3 | N/A (investigational) | N/A | Phase III / region-dependent | Investigational; emerging oral SERD |
| PROTAC ER degrader | Vepdegestrant | ESR1-mutated | VERITAC-2 | N/A (investigational) | N/A | Regulatory review / emerging | Emerging; PROTAC-based for ESR1-mut |
| Oral SERD | Giredestrant | ESR1-mutated | persevERA (negative primary) | N/A (investigational) | N/A | Phase III (primary endpoint negative) | Investigational; limited promise based on trial results |
| PI3K/mTOR inhibitor | Gedatolisib | PIK3CA-WT / PI3K pathway | VIKTORIA-1 | N/A (investigational) | N/A | Phase III | Investigational; for PIK3CA wild-type pathway-driven tumors |
| Pan-mutant selective PI3K inhibitor | Zovegalisi (+fulvestrant) | PIK3CA-mutated | ReDiscover (Ph 1/2) ReDiscover-2(Ph3 ongoing) | 11.1 | Not mature | Breakthrough Therapy Designation (FDA, Feb 2026); Ph 3 ongoing | Mutant-selective (allosteric, pan-mutant) PI3Kα inhibitor. |
| Study/Agent | Study Type | Population (n) | Key Subgroups | Median TTNT/PFS (95% CI) |
Median OS (95% CI) |
Key Findings |
|---|---|---|---|---|---|---|
| Elacestrant RWE (2023-2026 cohorts) | Multicenter retrospective | ESR1-mutated HR+/HER2− mBC (n~300-750) | Overall 1-2 prior ET lines No prior fulvestrant ≥12mo prior CDK4/6i ESR1+PIK3CA co-mut |
Overall: 7.9 mo (7.1-9.8) 1-2 prior: 8.2-10.8 mo No fulv: 12.9 mo ≥12mo: 8.4 mo Co-mut: 6.3 mo |
Not reported | RWE exceeds trial PFS Retained benefit in co-mutated Low discontinuation |
| Everolimus + ET Post-CDK4/6i RWE | Multicenter retrospective | HR+/HER2− mBC with prior CDK4/6i (n~200-400) | Post-CDK4/6i CDK4/6i-naïve |
Post-CDK4/6i: 5.3 mo (4.6-7.1) Naïve: 6.7 mo (5.8-7.6) P=0.046 |
Post-CDK4/6i: 21.8 mo (18.5-25.5) Naïve: 27.3 mo (23.2-30.2) P=0.01 |
Attenuated vs pre-CDK4/6i era Toxicity limits use (20-30% D/C) |
| Capivasertib RWE (emerging 2025) | Early real-world cohorts | Pathway-altered HR+/HER2− mBC post-CDK4/6i | AKT/PIK3CA/PTEN-altered | 5-7 mo (limited data) | Not reported | Modest benefit Manageable toxicity with dose modifications |
| Study | Study Type | Population (n) | Key Findings | Resistance Mechanisms Identified | Clinical Implications |
|---|---|---|---|---|---|
| Wander et al. (Cancer Discovery 2020) | Whole-exome sequencing | CDK4/6i-exposed tumors (n=59) | 8 distinct resistance mechanisms in 66% of cases 29.3% harbor ≥2 concurrent drivers (polyclonal) |
RB1 loss (9.8%) AKT1 mutations (9.8%) RAS pathway (9.8%) AURKA amp CCNE2 amp ERBB2 mutations FGFR2 alterations ER loss |
Multi-targeted strategies needed Single-pathway approaches inadequate in 30% due to polyclonal resistance |
| PALOMA-3 ctDNA Analysis | Serial liquid biopsy | Palbociclib-treated patients | Acquired RB1 mutations during treatment ESR1 mutations emerge on therapy |
RB1 mutations (~5%) ESR1 mutations (increasing VAF) |
Direct CDK4/6i target inactivation Serial monitoring detects resistance early |
| BYLieve ctDNA Substudy | Prospective ctDNA profiling | PIK3CA-mutated mBC post-CDK4/6i | ctDNA fraction strongly prognostic Low ctDNA (<10%) predicts superior PFS |
High ctDNA burden associated with worse outcomes | Low ctDNA: PFS 16.7 mo High ctDNA: PFS 5.4 mo HR 0.31 ctDNA quantification is prognostic biomarker |
| AURORA Molecular Screening Program | Large-scale genomic profiling | HR+/HER2− mBC | TP53 and ESR1 mutations independently predict worse outcomes | TP53 mutations: HR 1.59 for PFS ESR1 mutations: HR 3.10 for PFS |
Prognostic stratification TP53-mutant may benefit from alternative strategies |
| Multiple ctDNA Cohorts (FGFR Analysis) | Retrospective ctDNA profiling | Post-CDK4/6i populations | FGFR amplifications highly prevalent in resistant disease | FGFR1/2 amplifications (15-41% by ctDNA) | Potentially actionable target FGFR inhibitors under investigation |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




