Submitted:
21 April 2026
Posted:
22 April 2026
You are already at the latest version
Abstract
Antimicrobial resistance (AMR) is one of the most serious threats to global public health, driven in part by extensive antibiotic use in food-producing animals. The poultry industry, a major contributor to global animal protein supply, has depended on antibiotics for growth promotion and disease control, thereby contributing to the emergence and dissemination of AMR zoonotic bacteria. This review synthesizes current evidence on the potential of phytochemicals (PCs), plant-derived bioactive compounds, as sustainable non-antibiotic alternatives for controlling bacterial foodborne pathogens in poultry. Relevant literature including in vitro and in vivo studies assessing PCs against major poultry-associated zoonotic bacteria, including Salmonella enterica, Campylobacter spp., Clostridium perfringens, Listeria monocytogenes, and pathogenic Escherichia coli, is examined. Evidence indicates that PCs exert antimicrobial and anti-virulence effects through mechanisms like bacterial membrane disruption, inhibition of quorum sensing and virulence gene expression, modulation of gut microbiota, and enhancement of host immune responses. In vivo studies demonstrate reductions in pathogen colonization and improvements in gut health and performance metrics in poultry. Despite these promising findings, challenges remain in bioavailability, dose optimization, standardization, and regulatory approval. Overall, PCs represent a promising component of integrated antimicrobial stewardship strategies in poultry production, with significant implications for mitigating zoonotic AMR transmission.
Keywords:
1. Poultry Production at the Crossroads of Zoonotic Disease and Antimicrobial Resistance
1.1. Global Burden of Antimicrobial Resistance and Foodborne Infections
1.2. Antibiotic Dependence in Poultry
1.3. Rationale and Scope of This Review
2. AMR in Poultry: Drivers, Dynamics, and Zoonotic Risk
2.1. Patterns of Antibiotic Use in Poultry Production
2.1.1. Growth Promotion
2.1.2. Disease Prevention and Therapy
2.1.3. Regulatory Shifts
2.2. Selection and Dissemination of Antimicrobial Resistance Genes
2.3. Poultry-to-Human Transmission of AMR Bacteria
2.3.1. Farm-to-Processing Contamination
2.3.2. Retail and Consumer Exposure
2.3.3. Occupational and Environmental Pathways
2.3.4. Public Health Implications
- S. enterica: The most frequently detected MDR pathogen in poultry supply chains is Salmonella, particularly serovars like S. Typhimurium, S. Enteritidis, and S. Kentucky ST198. S. Kentucky ST198 is a particularly concerning clone that produces ESBL and has been linked to severe infections in humans [108,109,110]. The National Antimicrobial Resistance Monitoring System (NARMS) interim analyses emphasize that rising decreased susceptibility among S. Enteritidis is clinically relevant because it may adversely affect fluoroquinolone-treated cases, and NARMS genomic analyses indicate commercial chicken products as a likely source for key strain clusters [111].
- C. jejuni/coli: Campylobacter is the leading cause of bacterial gastroenteritis worldwide, with poultry recognized as the primary reservoir [112,113]. From an AMR perspective, the most consequential public health signal is loss of efficacy of macrolides (erythromycin/azithromycin), which are standard first-line options for severe campylobacteriosis. In Italy, researchers recently detected the erm(N) gene, a marker of erythromycin resistance, for the first time in a food-origin isolate. This gene was located on the chromosome of C. coli isolated from poultry carcasses at slaughterhouses, representing a significant escalation in resistance to a first-line treatment [102].
- Extraintestinal pathogenic E. coli (ExPEC): Poultry products are significant reservoirs for ExPEC, which cause neonatal meningitis, bacteremia, and most human urinary tract infections (UTIs). A comparative study in Canada reported genetic relatedness between E. coli from abattoir animals, particularly chicken, and ExPEC from human urinary tract infections, concluding that chickens were the most probable reservoir among animals sampled [114]. Studies using Caco-2 human epithelial cells have shown that 62.8% of poultry-isolated ExPEC can adhere to human intestinal tissues as effectively as known enteric pathogens, suggesting their high potential for establishing extraintestinal infections after intestinal colonization [115].
2.4. Genomic Evidence of Zoonotic Linkage
3. The Poultry Gut Microbiome as a Reservoir and Amplifier of AMR
4. PCs as Non-Antibiotic Interventions in Poultry Production
4.1. Mechanisms of Action of PCs Relevant to AMR Mitigation
4.1.1. Disruption of Bacterial Cell Membranes
4.1.2. Inhibition of Quorum Sensing
4.1.3. Anti-Biofilm and Anti-Motility Effects
4.1.4. Modulation of Gut Microbiota Composition
4.1.5. Enhancement of Host Immune Responses
4.2. Advantages of PCs over Conventional Antibiotics
5. Effects of Phytochemical Against Major Poultry-Associated Zoonotic Pathogens
5.1. S. enterica
5.2. Campylobacter spp.
5.3. Clostridium Perfringens
5.4. Listeria monocytogenes
5.5. Pathogenic E. coli and Other Emerging Zoonotic Bacteria
| Bacterial species (strain/isolate) | Phytochemical used | Concentration studied | Model details | Other effects | Reference |
|---|---|---|---|---|---|
|
Salmonella enterica serovar Typhimurium ATCC 14028 |
Gallic acid | MIC:3.5 mg/mL; MBC:4.5 mg/mL. | In vitro planktonic susceptibility assay (Broth microdilution in LB broth) | Tested at sublethal levels for membrane integrity/permeability and antivirulence phenotypes; study explicitly addresses antivirulence and antimicrobial effects | [214] |
| Protocatechuic acid | MIC 2.0 mg/mL; MBC 2.0 mg/mL | ||||
| Vanillic acid | MIC 1.5 mg/mL; MBC 2.0 mg/mL | ||||
|
S. Typhimurium ATCC14028 |
Resveratrol | MIC 250 µg/mL. | In vitro MIC (Broth Microdilution); mechanistic assays | Cell wall/membrane structural damage and metabolomics-linked effects | [217] |
| S. Enteritidis PT8 | trans-Cinnamaldehyde | SIC: 0.01% (~0.75 mM) | In vitro transcriptomics at SIC dose | Downregulated genes related to motility, SPI-1 regulation, invasion, transport/outer membrane proteins; Upregulated heat shock genes | [267] |
| Eugenol | SIC: 0.04% (~2.46 mM) |
||||
| S. Enteritidis | Trans-Cinnamaldehyde | Low dose: 0.5% High dose: 0.75% (in-feed) |
In vivo broiler chicken colonization model (n=75/experiment), inoculated day 8 with ~8 log₁₀ CFU/bird, followed through 10 days; cecal enumeration at days 7 and 10 post-inoculation. | ≥3 log₁₀ CFU/g reduction in cecal S. Enteritidis after 10 days infection; SIC exposure led to reduced motility/invasion, downregulation of motility/invasion genes | [268] |
| S. Enteritidis | Eugenol | Low dose: 0.75% High dose: 1% (in-feed) |
≥3 log₁₀ CFU/g reduction in cecal S. Enteritidis after 10 days infection; SIC exposure led to reduced motility/invasion, downregulation of motility/invasion genes; lower body weights vs controls |
||
| S. Enteritidis | Trans-Cinnamaldehyde | 0.75% in-feed for 5 days pre-slaughter | In vivo commercial market-age broilers; gavage challenge on day 30; euthanasia day 31. | Reduced colonization and shedding | [218] |
| S. Enteritidis | Eugenol | 0.1% in-feed for 5 days pre-slaughter | |||
| S. Typhimurium (CVCC541) | Thymol | MIC: 375 µg/mL; MBC 750 µg/mL |
In vitro broth dilution method | Checkerboard assay; combination of thymol and carvacrol showed additive effect | [238] |
| S. Typhimurium (CVCC541) | Carvacrol | MIC: 375 µg/mL; MBC 750 µg/mL |
|||
|
Campylobacter jejuni (S-8, NCTC 81-176) |
Carvacrol | SIC: 0.002% | In vitro Motility assay Adhesion assay LC-MS/MS proteomics |
Reduced motility and adhesion, decreased AI-2 activity, increased acid/bile susceptibility; Reduced expression of proteins linked to motility/adhesion/ metabolism/ respiration. |
[224] |
| C. jejuni | Carvacrol | 120 mg/kg in-feed | In vivo broiler trial: seeder bird model through slaughter age | Reductions in cloacal swab loads and colon counts during early periods; No significant reduction in cecal counts at day 33 | [225] |
| C. jejuni | Carvacrol | 0.25%, 0.5%, 1%, 2% (suspension) | Ex vivo / food model: chicken skin inoculation |
2% wash reduced C. jejuni by ~2.4–4 log₁₀ CFU/sample; emulsion/nanoemulsion not consistently superior to suspension | [228] |
| C. jejuni (five wild-type isolates) | Caprylic acid | In-feed supplementation at 0.35%, 0.7%, 1.4%, 2.8% for the final 72 h of a 15-day trial | In vivo: day-of-hatch broiler chicks (n=60/trial) | 0.7% and 1.4% caprylic acid consistently produced 3-4 log₁₀ reductions in cecal counts vs positive controls | [227] |
| Clostridium perfringens (CVCC2027, CVCC2030) | Thymol | MIC: 375 µg/mL; MBC: 750 µg/mL |
In vitro broth dilution in MHB | Checkerboard assay; combination of thymol and carvacrol showed additive effect | [243] |
| Carvacrol | |||||
| C. perfringens | Tannic acid | 250, 500, 750, 1000 mg/kg diet. | In vivo broiler Necrotic enteritis model | Improvements in anti-inflammatory markers, barrier-associated indicators, and microbiota shifts | [247] |
| Listeria monocytogenes | Resveratrol | MIC: 200 µg/mL | In vitro planktonic MIC + biofilm experiments | Strong biofilm-inhibition even at subinhibitory concentrations | [269] |
| L. monocytogenes ATCC 19115 | Thymol + Cinnamaldehyde |
125 µg/mL thymol + 125 µg/mL cinnamaldehyde | Food model with transcriptomics | Reduced survival and virulence-associated transcriptional activity on meat |
[255] |
| Avian pathogenic E. coli (APEC) | Resveratrol | MIC: 128 µg/mL | In vitro broth dilution, biofilm and motility assays | Biofilm inhibition above 1 µg/mL; structural biofilm effects; biofilm eradication at 32 µg/mL resveratrol + 64 µg/mL florfenicol; highlights synergy with an antibiotic | [261] |
| Methicillin-resistant Staphylococcus aureus (MRSA) | trans-Cinnamaldehyde, Thymol, Carvacrol | In vitro MRSA isolate characterization + compound exposure with virulence gene transcription | Downregulation of key virulence genes; antivirulence plus growth inhibition | [266] | |
| Multidrug-resistant E. coli |
Matrine | MIC: 6.25 mg/mL | In vitro broth microdilution + synergy assays | Checkerboard synergy assay; matrine doses reduced the effective MIC of berberine hydrochloride markedly when combined | [264] |
| Berberine hydrochloride | MIC: 1 mg/mL | ||||
| Matrine + Berberine hydrochloride | 6.25 mg/mL matrine+ 1 mg/mL berberine hydrochloride | In vivo chicken colibacillosis model | Improvements via reduced bacterial load and inflammatory factor modulation |
6. Limitations, Challenges, and Knowledge Gaps
7. Future Perspectives
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AMR | antimicrobial resistance |
| AGPs | antibiotic growth promoters |
| ARGs | antimicrobial resistance genes |
| AHLs | acyl-homoserine lactones |
| AI | autoinducers |
| AI-2 | autoinducer-2 |
| APEC | avian pathogenic Escherichia coli |
| CFU | colony-forming units |
| EPS | extracellular polymeric substances |
| ESBL | extended-spectrum beta-lactamase |
| EU | European Union |
| FDA | Food and Drug Administration |
| FCR | feed conversion ratio |
| FICI | fractional inhibitory concentration index |
| GM | gut microbiota |
| GRAS | generally recognized as safe |
| HGT | horizontal gene transfer |
| IHME | Institute for Health Metrics and Evaluation |
| IgA | immunoglobulin A |
| LA-MRSA | livestock-associated methicillin-resistant Staphylococcus aureus |
| LMICs | low- and middle-income countries |
| MBC | minimum bactericidal concentration |
| MDR | multidrug-resistant |
| MDROs | multidrug-resistant organisms |
| MGEs | mobile genetic elements |
| MIC | minimum inhibitory concentration |
| MPN | most probable number |
| MRSA | methicillin-resistant Staphylococcus aureus |
| NARMS | National Antimicrobial Resistance Monitoring System |
| PCs | phytochemicals |
| QS | quorum sensing |
| QSIs | quorum sensing inhibitors |
| SNPs | single nucleotide polymorphisms |
| STEC | Shiga toxin-producing Escherichia coli |
| UTIs | urinary tract infections |
| VFD | Veterinary Feed Directive |
| VRE | vancomycin-resistant enterococci |
| WEF | World Economic Forum |
| WGS | whole genome sequencing |
| WHO | World Health Organization |
References
- World Economic Forum The Global Risks Report 2026; World Economic Forum: Geneva, Switzerland, 2026.
- Naghavi, M.; Vollset, S.E.; Ikuta, K.S.; Swetschinski, L.R.; Gray, A.P.; Wool, E.E.; Aguilar, G.R.; Mestrovic, T.; Smith, G.; Han, C.; et al. Global Burden of Bacterial Antimicrobial Resistance 1990–2021: A Systematic Analysis with Forecasts to 2050. The Lancet 2024, 404, 1199–1226. [Google Scholar] [CrossRef]
- WHO Global Antimicrobial Resistance and Use Surveillance System (GLASS) Global Antimicrobial Resistance Surveillance Report 2025; World Health Organization: Geneva, 2025.
- Economou, V.; Gousia, P. Agriculture and Food Animals as a Source of Antimicrobial-Resistant Bacteria. Infect Drug Resist 2015, 8, 49–61. [Google Scholar] [CrossRef] [PubMed]
- Antimicrobial Resistance. Available online: https://www.who.int/news-room/fact-sheets/detail/antimicrobial-resistance (accessed on 18 March 2026).
- Murray, C.J.L.; Ikuta, K.S.; Sharara, F.; Swetschinski, L.; Aguilar, G.R.; Gray, A.; Han, C.; Bisignano, C.; Rao, P.; Wool, E.; et al. Global Burden of Bacterial Antimicrobial Resistance in 2019: A Systematic Analysis. The Lancet 2022, 399, 629–655. [Google Scholar] [CrossRef]
- Kirchhelle, C. Pharming Animals: A Global History of Antibiotics in Food Production (1935–2017). Palgrave Commun 2018, 4, 96. [Google Scholar] [CrossRef]
- Butaye, P.; Devriese, L.A.; Haesebrouck, F. Antimicrobial Growth Promoters Used in Animal Feed: Effects of Less Well Known Antibiotics on Gram-Positive Bacteria. Clinical Microbiology Reviews 2003, 16, 175–188. [Google Scholar] [CrossRef]
- El-Fateh, M.; Bilal, M.; Zhao, X. Effect of Antibiotic Growth Promoters (AGPs) on Feed Conversion Ratio (FCR) of Broiler Chickens: A Meta-Analysis. Poult Sci 2024, 103, 104472. [Google Scholar] [CrossRef]
- Novel Growth Promoters Replacing AGPs in Poultry Feed. Available online: https://www.feedstrategy.com/animal-nutrition/poultry/article/15438597/novel-growth-promoters-replacing-agps-in-poultry-feed (accessed on 14 March 2026).
- Nutrition, E.W. Beyond AGPs: Controlling necrotic enteritis through gut health optimization. EW Nutrition 2020.
- Sana, S.S.; Atuahene, D.; Nagy, V.; Shaikh, A.M.; Knop, R. The Rising Threat of Antibiotic Resistance in Poultry: Veterinary and One Health Perspectives. Vet Sci 2025, 12, 1059. [Google Scholar] [CrossRef] [PubMed]
- Wegener, H.C.; Aarestrup, F.M.; Jensen, L.B.; Hammerum, A.M.; Bager, F. Use of Antimicrobial Growth Promoters in Food Animals and Enterococcus Faecium Resistance to Therapeutic Antimicrobial Drugs in Europe -. Number 3—June 1999 - Emerging Infectious Diseases Journal - CDC Volume 5. [CrossRef] [PubMed]
- Wegener, H.C. Historical Yearly Usage of Glycopeptides for Animals and Humans: The American-European Paradox Revisited. Antimicrobial Agents and Chemotherapy 1998, 42, 3049–3049. [Google Scholar] [CrossRef]
- Sharma, G.; Dey, T.K.; Hazarika, R.A.; Shome, B.R.; Shome, R.; Singh, V.P.; Deka, R.P.; Grace, D.; Lindahl, J.F. Knowledge and Practices Related to Antibiotics among Poultry Producers and Veterinarians in Two Indian States. One Health 2024, 18, 100700. [Google Scholar] [CrossRef]
- Abreu, R.; Semedo-Lemsaddek, T.; Cunha, E.; Tavares, L.; Oliveira, M. Antimicrobial Drug Resistance in Poultry Production: Current Status and Innovative Strategies for Bacterial Control. Microorganisms 2023, 11, 953. [Google Scholar] [CrossRef]
- Nelson, J.M.; Chiller, T.M.; Powers, J.H.; Angulo, F.J. Fluoroquinolone-Resistant Campylobacter Species and the Withdrawal of Fluoroquinolones from Use in Poultry: A Public Health Success Story. Clin Infect Dis 2007, 44, 977–980. [Google Scholar] [CrossRef]
- CDC - October 6, 2011 - Epi - Salmonella Altona / Johannesburg Infections Linked to Chicks / Ducklings. Available online: https://archive.cdc.gov/www_cdc_gov/salmonella/2011/chicks-ducklings-10-6-2011.html (accessed on 14 March 2026).
- Punchihewage-Don, A.J.; Hawkins, J.; Adnan, A.M.; Hashem, F.; Parveen, S. The Outbreaks and Prevalence of Antimicrobial Resistant Salmonella in Poultry in the United States: An Overview. Heliyon 2022, 8, e11571. [Google Scholar] [CrossRef] [PubMed]
- 2018 Salmonella Infections Linked to Raw Chicken Products | Multistate Outbreak of Salmonella Infections Linked to Raw Chicken Products | October 2018 | Salmonella | CDC. Available online: https://archive.cdc.gov/www_cdc_gov/salmonella/infantis-10-18/index.html (accessed on 14 March 2026).
- O’Neill, L.; Manzanilla, E.G.; Ekhlas, D.; Leonard, F.C. Antimicrobial Resistance in Commensal Escherichia Coli of the Porcine Gastrointestinal Tract. Antibiotics (Basel) 2023, 12, 1616. [Google Scholar] [CrossRef]
- Lambrecht, E.; Van Coillie, E.; Van Meervenne, E.; Boon, N.; Heyndrickx, M.; Van de Wiele, T. Commensal E. Coli Rapidly Transfer Antibiotic Resistance Genes to Human Intestinal Microbiota in the Mucosal Simulator of the Human Intestinal Microbial Ecosystem (M-SHIME). Int J Food Microbiol 2019, 311, 108357. [Google Scholar] [CrossRef]
- Rivera-Gomis, J.; Marín, P.; Martínez-Conesa, C.; Otal, J.; Jordán, M.J.; Escudero, E.; Cubero, M.J. Antimicrobial Resistance of Campylobacter Jejuni, Escherichia Coli and Enterococcus Faecalis Commensal Isolates from Laying Hen Farms in Spain. Animals 2021, 11, 1284. [Google Scholar] [CrossRef]
- Marshall, B.M.; Ochieng, D.J.; Levy, S.B. Commensals: Underappreciated Reservoir of Antibiotic Resistance. Microbe Magazine 2009, 4, 231–238. [Google Scholar] [CrossRef]
- de Kraker, M.E.A.; Wolkewitz, M.; Davey, P.G.; Koller, W.; Berger, J.; Nagler, J.; Icket, C.; Kalenic, S.; Horvatic, J.; Seifert, H.; et al. Burden of Antimicrobial Resistance in European Hospitals: Excess Mortality and Length of Hospital Stay Associated with Bloodstream Infections Due to Escherichia Coli Resistant to Third-Generation Cephalosporins. J Antimicrob Chemother 2011, 66, 398–407. [Google Scholar] [CrossRef] [PubMed]
- Dimuccio, M.M.; Conforti, V.; Celentano, F.E.; Circella, E.; Salvaggiulo, A.; Bozzo, G.; Corrente, M. Regulation of Antibiotic Use in Livestock: European and International Strategies to Prevent and Control Antimicrobial Resistance and Ensure Animal Welfare. Antibiotics (Basel) 2026, 15, 67. [Google Scholar] [CrossRef] [PubMed]
- Mshana, S.E.; Sindato, C.; Matee, M.I.; Mboera, L.E.G. Antimicrobial Use and Resistance in Agriculture and Food Production Systems in Africa: A Systematic Review. Antibiotics 2021, 10, 976. [Google Scholar] [CrossRef] [PubMed]
- The Economic Costs of Withdrawing Antimicrobial Growth Promoters in the Livestock Sector. In One Health Trust.
- Tilahun, H.E.; Ofa, D.A. Antimicrobial-Resistant Escherichia Coli and Salmonella in Poultry Production and Spread and Effect in the One Health Framework. Poultry Science 2026, 106752. [Google Scholar] [CrossRef]
- Lima Barbieri, N.; Nielsen, D.W.; Wannemuehler, Y.; Cavender, T.; Hussein, A.; Yan, S.-G.; Nolan, L.K.; Logue, C.M. Mcr-1 Identified in Avian Pathogenic Escherichia Coli (APEC). PLoS One 2017, 12, e0172997. [Google Scholar] [CrossRef] [PubMed]
- Jones-Dias, D.; Manageiro, V.; Caniça, M. Influence of Agricultural Practice on Mobile Bla Genes: IncI1-Bearing CTX-M, SHV, CMY and TEM in Escherichia Coli from Intensive Farming Soils. Environ Microbiol 2016, 18, 260–272. [Google Scholar] [CrossRef]
- Lv, D.; Duan, R.; Fan, R.; Mu, H.; Liang, J.; Xiao, M.; He, Z.; Qin, S.; Yang, J.; Jing, H.; et al. blaNDM and Mcr-1 to Mcr-5 Gene Distribution Characteristics in Gut Specimens from Different Regions of China. Antibiotics (Basel) 2021, 10, 233. [Google Scholar] [CrossRef] [PubMed]
- Oladeinde, A.; Abdo, Z.; Press, M.O.; Cook, K.; Cox, N.A.; Zwirzitz, B.; Woyda, R.; Lakin, S.M.; Thomas, J.C.; Looft, T.; et al. Horizontal Gene Transfer Is the Main Driver of Antimicrobial Resistance in Broiler Chicks Infected with Salmonella Enterica Serovar Heidelberg. mSystems 2021, 6, e00729-21. [Google Scholar] [CrossRef]
- Yao, X.; Doi, Y.; Zeng, L.; Lv, L.; Liu, J.-H. Carbapenem-Resistant and Colistin-Resistant Escherichia Coli Co-Producing NDM-9 and MCR-1. The Lancet Infectious Diseases 2016, 16, 288–289. [Google Scholar] [CrossRef]
- Raised Without Antibiotics: Lessons from Voluntary Labeling of Antibiotic Use Practices in The Broiler Industry - Bowman - 2016 - American Journal of Agricultural Economics - Wiley Online Library. Available online: https://onlinelibrary.wiley.com/doi/10.1093/ajae/aaw008 (accessed on 13 March 2026).
- Gemechu, F.G. Embracing Nutritional Qualities, Biological Activities and Technological Properties of Coffee Byproducts in Functional Food Formulation. Trends in Food Science & Technology 2020, 104, 235–261. [Google Scholar] [CrossRef]
- Tsuchiya, H. Membrane Interactions of Phytochemicals as Their Molecular Mechanism Applicable to the Discovery of Drug Leads from Plants. Molecules 2015, 20, 18923–18966. [Google Scholar] [CrossRef] [PubMed]
- Papaneophytou, C. Phytochemical Quorum-Sensing Inhibitors Against Bacterial Pathogens: Mechanisms of Action and Translational Challenges. Curr Issues Mol Biol 2026, 48, 214. [Google Scholar] [CrossRef]
- Lillehoj, H.; Liu, Y.; Calsamiglia, S.; Fernandez-Miyakawa, M.E.; Chi, F.; Cravens, R.L.; Oh, S.; Gay, C.G. Phytochemicals as Antibiotic Alternatives to Promote Growth and Enhance Host Health. Veterinary Research 2018, 49, 76. [Google Scholar] [CrossRef]
- Suganya, T.; Packiavathy, I.A.S.V.; Aseervatham, G.S.B.; Carmona, A.; Rashmi, V.; Mariappan, S.; Devi, N.R.; Ananth, D.A. Tackling Multiple-Drug-Resistant Bacteria With Conventional and Complex Phytochemicals. Front Cell Infect Microbiol 2022, 12, 883839. [Google Scholar] [CrossRef] [PubMed]
- Mcleod, A.; Thieme, O.; Mack, S.D. Structural Changes in the Poultry Sector: Will There Be Smallholder Poultry Development in 2030? World’s Poultry Science Journal 2009, 65, 191–200. [Google Scholar] [CrossRef]
- Farkas, Z.; Strang, O.; Zentai, A.; Csorba, S.; Farkas, M.; Bittsánszky, A.; Tóth, A.; Süth, M.; Jóźwiak, Á. Scoping Review of Factors Affecting Antimicrobial Use and the Spread of Antimicrobial Resistance in the Poultry Production Chain. Veterinary Sciences 2025, 12. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.; Bose, P.; Rahman, M.; Muktaruzzaman, M.; Sultana, P.; Ahamed, T.; Khatun, M. A Review of Antimicrobial Usage Practice in Livestock and Poultry Production and Its Consequences on Human and Animal Health. J Adv Vet Anim Res 2024, 11, 675. [Google Scholar] [CrossRef] [PubMed]
- Lawal, J.R.; Jajere, S.M.; Geidam, Y.A.; Wakil, Y. Antibiotic Residues in Edible Poultry Tissues and Products in Nigeria: A Potential Public Health Hazard. International Journal of Animal and Veterinary Advances 2015, 7, 55–61. [Google Scholar] [CrossRef]
- Abou-Jaoudeh, C.; Andary, J.; Abou-Khalil, R. Antibiotic Residues in Poultry Products and Bacterial Resistance: A Review in Developing Countries. Journal of Infection and Public Health 2024, 17, 102592. [Google Scholar] [CrossRef]
- Hedman, H.D.; Vasco, K.A.; Zhang, L. A Review of Antimicrobial Resistance in Poultry Farming within Low-Resource Settings. Animals (Basel) 2020, 10, 1264. [Google Scholar] [CrossRef]
- Farkas, Z.; Strang, O.; Zentai, A.; Csorba, S.; Farkas, M.; Bittsánszky, A.; Tóth, A.; Süth, M.; Jóźwiak, Á. Scoping Review of Factors Affecting Antimicrobial Use and the Spread of Antimicrobial Resistance in the Poultry Production Chain. Vet Sci 2025, 12, 881. [Google Scholar] [CrossRef]
- Hogs, Antibiotics, and the Industrial Environments of Postwar Agriculture. In Industrializing Organisms; Routledge, 2004.
- Fernández Miyakawa, M.E.; Casanova, N.A.; Kogut, M.H. How Did Antibiotic Growth Promoters Increase Growth and Feed Efficiency in Poultry? Poultry Science 2024, 103, 103278. [Google Scholar] [CrossRef]
- Jacobs, R.L.; Elam, J.F.; Anderson, G.W.; Gee, L.L.; Fowler, J.; Couch, J.R. Further Evidence as to the Possible Mechanism Involved in the Growth-Promoting Responses Obtained from Antibiotics. The Journal of Nutrition 1953, 51, 507–513. [Google Scholar] [CrossRef]
- Graham, J.P.; Boland, J.J.; Silbergeld, E. Growth Promoting Antibiotics in Food Animal Production: An Economic Analysis. Public Health Rep 2007, 122, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Izah, S.C.; Nurmahanova, A.; Ogwu, M.C.; Toktarbay, Z.; Umirbayeva, Z.; Ussen, K.; Koibasova, L.; Nazarbekova, S.; Tynybekov, B.; Guo, Z. Public Health Risks Associated with Antibiotic Residues in Poultry Food Products. Journal of Agriculture and Food Research 2025, 21, 101815. [Google Scholar] [CrossRef]
- Baptiste, K.E.; Kyvsgaard, N.C. Do Antimicrobial Mass Medications Work? A Systematic Review and Meta-Analysis of Randomised Clinical Trials Investigating Antimicrobial Prophylaxis or Metaphylaxis against Naturally Occurring Bovine Respiratory Disease. Pathog Dis 2017, 75, ftx083. [Google Scholar] [CrossRef]
- Adhikari, J.; Thapaliya, S.; Munakarmi, R.; Acharya, P.; Basnet, H.B.; Paudyal, N. Drivers and Patterns of Antibiotic Use in Small to Medium-Sized Chicken Farms in Selected Districts of Nepal. Front Vet Sci 2025, 12, 1570822. [Google Scholar] [CrossRef]
- Sawadogo, A.; Kagambèga, A.; Moodley, A.; Ouedraogo, A.A.; Barro, N.; Dione, M. Knowledge, Attitudes, and Practices Related to Antibiotic Use and Antibiotic Resistance among Poultry Farmers in Urban and Peri-Urban Areas of Ouagadougou, Burkina Faso. Antibiotics (Basel) 2023, 12, 133. [Google Scholar] [CrossRef]
- U.S. Food and Drug Administration Guidance for Industry #263: Recommendations for Sponsors of Medically Important Antimicrobial Drugs Approved for Use in Animals to Voluntarily Bring Under Veterinary Oversight All Products That Continue to Be Available Over-the-Counter; U.S. Food and Drug Administration, 2023.
- Ekakoro, J.E.; Caldwell, M.; Strand, E.B.; Okafor, C.C. Drivers, Alternatives, Knowledge, and Perceptions towards Antimicrobial Use among Tennessee Beef Cattle Producers: A Qualitative Study. BMC Vet Res 2019, 15, 16. [Google Scholar] [CrossRef]
- Ben Ali, M.; Chtioui, B.; Bouchrit, H.; Laamiri, H.; Attia El Hili, H. Antibiotic Use in Poultry Farming: A Cross-Sectional Study of Veterinary Practices in Tunisia. Front Antibiot 2025, 4, 1646766. [Google Scholar] [CrossRef]
- Regulation (EU) 2019/6 of the European Parliament and of the Council of 11 December 2018 on Veterinary Medicinal Products and Repealing Directive 2001/82/EC (Text with EEA Relevance); 2018; Vol. 004.
- European Commission Farm to Fork Strategy: For a Fair, Healthy and Environmentally-Friendly Food System; European Commission: Brussels, Belgium, 2020.
- Sneeringer, S. The U.S. and EU Animal Pharmaceutical Industries in the Age of Antibiotic Resistance.
- Researchers Find Evidence of Banned Antibiotics in Poultry Products | Johns Hopkins | Bloomberg School of Public Health. Available online: https://publichealth.jhu.edu/2012/feather_meal_clf (accessed on 13 March 2026).
- Otaigbe, I.I.; Elikwu, C.J. Drivers of Inappropriate Antibiotic Use in Low- and Middle-Income Countries. JAC Antimicrob Resist 2023, 5, dlad062. [Google Scholar] [CrossRef] [PubMed]
- The Economic Costs of Withdrawing Antimicrobial Growth Promoters from the Livestock Sector; OECD Food, Agriculture and Fisheries Papers; 2015; Vol. 78.
- Van Boeckel, T.P.; Brower, C.; Gilbert, M.; Grenfell, B.T.; Levin, S.A.; Robinson, T.P.; Teillant, A.; Laxminarayan, R. Global Trends in Antimicrobial Use in Food Animals. Proc Natl Acad Sci U S A 2015, 112, 5649–5654. [Google Scholar] [CrossRef] [PubMed]
- Algarni, S.; Ricke, S.C.; Foley, S.L.; Han, J. The Dynamics of the Antimicrobial Resistance Mobilome of Salmonella Enterica and Related Enteric Bacteria. Front. Microbiol. 2022, 13. [Google Scholar] [CrossRef]
- Frost, L.S.; Leplae, R.; Summers, A.O.; Toussaint, A. Mobile Genetic Elements: The Agents of Open Source Evolution. Nat Rev Microbiol 2005, 3, 722–732. [Google Scholar] [CrossRef]
- Chen, Y.; Liu, Y.; Zhao, C.; Ma, J.; Guo, J. Antibiotic Resistance Gene Pollution in Poultry Farming Environments and Approaches for Mitigation: A System Review. Poult Sci 2025, 104, 104858. [Google Scholar] [CrossRef]
- Liu, K.; Wang, M.; Zhang, Y.; Fang, C.; Zhang, R.; Fang, L.; Sun, J.; Liu, Y.; Liao, X. Distribution of Antibiotic Resistance Genes and Their Pathogen Hosts in Duck Farm Environments in South-East Coastal China. Appl Microbiol Biotechnol 2024, 108, 136. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Li, Y.; Zhang, L.; Wu, Z.; Huang, Y.; Yan, H.; Zhong, J.; Wang, L.-J.; Abdullah, H.M.; Wang, H.H. Antibiotic Administration Routes and Oral Exposure to Antibiotic Resistant Bacteria as Key Drivers for Gut Microbiota Disruption and Resistome in Poultry. Front Microbiol 2020, 11, 1319. [Google Scholar] [CrossRef] [PubMed]
- Partridge, S.R.; Kwong, S.M.; Firth, N.; Jensen, S.O. Mobile Genetic Elements Associated with Antimicrobial Resistance. Clin Microbiol Rev 2018, 31, e00088-17. [Google Scholar] [CrossRef] [PubMed]
- Johansson, M.H.K.; Aarestrup, F.M.; Petersen, T.N. Importance of Mobile Genetic Elements for Dissemination of Antimicrobial Resistance in Metagenomic Sewage Samples across the World. PLoS One 2023, 18, e0293169. [Google Scholar] [CrossRef]
- Shang, D.; Zhao, H.; Xu, X.; Arunachalam, K.; Chang, J.; Bai, L.; Shi, C. Conjugative IncHI2 Plasmid Harboring Novel Class 1 Integron Mediated Dissemination of Multidrug Resistance Genes in Salmonella Typhimurium. Food Control 2021, 122, 107810. [Google Scholar] [CrossRef]
- Chen, W.; Fang, T.; Zhou, X.; Zhang, D.; Shi, X.; Shi, C. IncHI2 Plasmids Are Predominant in Antibiotic-Resistant Salmonella Isolates. Front. Microbiol. 2016, 7. [Google Scholar] [CrossRef]
- Foley, S.L.; Kaldhone, P.R.; Ricke, S.C.; Han, J. Incompatibility Group I1 (IncI1) Plasmids: Their Genetics, Biology, and Public Health Relevance. Microbiol Mol Biol Rev 2021, 85, e00031-20. [Google Scholar] [CrossRef]
- Chouchani, C.; El Salabi, A.; Marrakchi, R.; Ferchichi, L.; Walsh, T.R. Characterization of IncA/C Conjugative Plasmid Harboring Bla TEM-52 and Bla CTX-M-15 Extended-Spectrum β-Lactamases in Clinical Isolates of Escherichia Coli in Tunisia. Eur J Clin Microbiol Infect Dis 2012, 31, 1081–1087. [Google Scholar] [CrossRef]
- Cloeckaert, A.; Praud, K.; Doublet, B.; Bertini, A.; Carattoli, A.; Butaye, P.; Imberechts, H.; Bertrand, S.; Collard, J.-M.; Arlet, G.; et al. Dissemination of an Extended-Spectrum-Beta-Lactamase blaTEM-52 Gene-Carrying IncI1 Plasmid in Various Salmonella Enterica Serovars Isolated from Poultry and Humans in Belgium and France between 2001 and 2005. Antimicrob Agents Chemother 2007, 51, 1872–1875. [Google Scholar] [CrossRef]
- Liu, Y.-Y.; Wang, Y.; Walsh, T.R.; Yi, L.-X.; Zhang, R.; Spencer, J.; Doi, Y.; Tian, G.; Dong, B.; Huang, X.; et al. Emergence of Plasmid-Mediated Colistin Resistance Mechanism MCR-1 in Animals and Human Beings in China: A Microbiological and Molecular Biological Study. Lancet Infect Dis 2016, 16, 161–168. [Google Scholar] [CrossRef]
- Huang, J.; Deng, S.; Ren, J.; Tu, J.; Ye, M.; Wang, M. Characterization of a blaNDM-1-Harboring Plasmid from a Salmonella Enterica Clinical Isolate in China. Mol Med Rep 2017, 16, 1087–1092. [Google Scholar] [CrossRef] [PubMed]
- Racewicz, P.; Majewski, M.; Biesiada, H.; Nowaczewski, S.; Wilczyński, J.; Wystalska, D.; Kubiak, M.; Pszczoła, M.; Madeja, Z.E. Prevalence and Characterisation of Antimicrobial Resistance Genes and Class 1 and 2 Integrons in Multiresistant Escherichia Coli Isolated from Poultry Production. Sci Rep 2022, 12, 6062. [Google Scholar] [CrossRef]
- Lee, M.D.; Sanchez, S.; Zimmer, M.; Idris, U.; Berrang, M.E.; McDermott, P.F. Class 1 Integron-Associated Tobramycin-Gentamicin Resistance in Campylobacter Jejuni Isolated from the Broiler Chicken House Environment. Antimicrob Agents Chemother 2002, 46, 3660–3664. [Google Scholar] [CrossRef]
- Rao, S.; Linke, L.; Doster, E.; Hyatt, D.; Burgess, B.A.; Magnuson, R.; Pabilonia, K.L.; Morley, P.S. Genomic Diversity of Class I Integrons from Antimicrobial Resistant Strains of Salmonella Typhimurium Isolated from Livestock, Poultry and Humans. PLoS One 2020, 15, e0243477. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, C.; Lee, M.D.; Sanchez, S.; Hudson, C.; Phillips, B.; Register, B.; Grady, M.; Liebert, C.; Summers, A.O.; White, D.G.; et al. Incidence of Class 1 and 2 Integrases in Clinical and Commensal Bacteria from Livestock, Companion Animals, and Exotics. Antimicrob Agents Chemother 2001, 45, 723–726. [Google Scholar] [CrossRef] [PubMed]
- Hsu, C.-H.; Li, C.; Harrison, L.; Zhao, S. Genomic Structure of Class 1 and 2 Integrons in Non-Typhoidal Salmonella Isolated from Food Animals and Related Meat Products in the USA. J Antimicrob Chemother 2025, 80, 2844–2853. [Google Scholar] [CrossRef]
- Zhao, H.; Chen, W.; Xu, X.; Zhou, X.; Shi, C. Transmissible ST3-IncHI2 Plasmids Are Predominant Carriers of Diverse Complex IS26-Class 1 Integron Arrangements in Multidrug-Resistant Salmonella. Front. Microbiol. 2018, 9. [Google Scholar] [CrossRef]
- Yang, J.; Tong, C.; Xiao, D.; Xie, L.; Zhao, R.; Huo, Z.; Tang, Z.; Hao, J.; Zeng, Z.; Xiong, W. Metagenomic Insights into Chicken Gut Antibiotic Resistomes and Microbiomes. Microbiology Spectrum 2022, 10, e01907-21. [Google Scholar] [CrossRef]
- Gargiullo, L.; Del Chierico, F.; D’Argenio, P.; Putignani, L. Gut Microbiota Modulation for Multidrug-Resistant Organism Decolonization: Present and Future Perspectives. Front Microbiol 2019, 10, 1704. [Google Scholar] [CrossRef]
- Salyers, A.A.; Gupta, A.; Wang, Y. Human Intestinal Bacteria as Reservoirs for Antibiotic Resistance Genes. Trends in Microbiology 2004, 12, 412–416. [Google Scholar] [CrossRef]
- Elbehiry, A.; Marzouk, E. From Farm to Fork: Antimicrobial-Resistant Bacterial Pathogens in Livestock Production and the Food Chain. Vet Sci 2025, 12, 862. [Google Scholar] [CrossRef] [PubMed]
- P, R.; Srijith, L.; G, K.; V.S, R.; Kopula, S.; Paul, A.; Perumal, K. Genomic Analysis of ST117, 155, 1011, 167, 744, and 17391 in Poultry-Associated Multidrug Resistant Escherichia Coli Isolates from India. Scientific Reports 2026, 16. [Google Scholar] [CrossRef] [PubMed]
- Al-Khalaifah, H.; Rahman, M.H.; Al-Surrayai, T.; Al-Dhumair, A.; Al-Hasan, M. A One-Health Perspective of Antimicrobial Resistance (AMR): Human, Animals and Environmental Health. Life 2025, 15. [Google Scholar] [CrossRef] [PubMed]
- Gymoese, P.; Kiil, K.; Torpdahl, M.; Østerlund, M.T.; Sørensen, G.; Olsen, J.E.; Nielsen, E.M.; Litrup, E. WGS Based Study of the Population Structure of Salmonella Enterica Serovar Infantis. BMC Genomics 2019, 20, 870. [Google Scholar] [CrossRef]
- Kumar, R.; Adeyemi, N.O.; Chattaraj, S.; Alloun, W.; Thamarsha, A.K.A.N.W.M.R.K.; Anđelković, S.; Mitra, D.; Gautam, P. Antimicrobial Resistance in Salmonella: One Health Perspective on Global Food Safety Challenges. Sci One Health 2025, 4, 100117. [Google Scholar] [CrossRef]
- Dunn, L.L.; Sharma, V.; Chapin, T.K.; Friedrich, L.M.; Larson, C.C.; Rodrigues, C.; Jay-Russell, M.; Schneider, K.R.; Danyluk, M.D. The Prevalence and Concentration of Salmonella Enterica in Poultry Litter in the Southern United States. PLOS ONE 2022, 17, e0268231. [Google Scholar] [CrossRef]
- Roberts, B.N.; Bailey, R.H.; McLaughlin, M.R.; Miles, D.M.; Brooks, J.P. Spatial and Temporal Analysis of Microbial Populations in Production Broiler House Litter in the Southeastern United States1. Journal of Applied Poultry Research 2013, 22, 759–770. [Google Scholar] [CrossRef]
- The Role of Biofilms as a Reservoir for Foodborne Pathogens in Irrigation Systems | National Agricultural Library. Available online: https://www.nal.usda.gov/research-tools/food-safety-research-projects/role-biofilms-reservoir-foodborne-pathogens-irrigation (accessed on 13 March 2026).
- Maes, S.; Vackier, T.; Nguyen Huu, S.; Heyndrickx, M.; Steenackers, H.; Sampers, I.; Raes, K.; Verplaetse, A.; De Reu, K. Occurrence and Characterisation of Biofilms in Drinking Water Systems of Broiler Houses. BMC Microbiol 2019, 19, 77. [Google Scholar] [CrossRef]
- Katsande, P.; Davies, A.R.; Owusu-Nyantakyi, C.; Gufe, C.; Musari, S.; Majuru, C.S.; Machakwa, J. Genetic Diversity and Antimicrobial Resistance Profiles of Salmonella Enterica in the Broiler Supply Chain in Harare, Zimbabwe: Tracking Transmission from Farm to Table. Microb Genom 2025, 11, 001550. [Google Scholar] [CrossRef]
- Al-Hadidi, S.H.; Al mana, H.; Almoghrabi, S.Z.; El-Obeid, T.; AlAli, W.Q.; Eltai, N.O. Retail Chicken Carcasses as a Reservoir of Multidrug-Resistant Salmonella. Microb Drug Resist 2022, 28, 824–831. [Google Scholar] [CrossRef]
- Dimaapi, L.K.A.; Dela Cruz, A.L.G.; Francisco, R.A.D.; Noble, R.G.D.; Sabangan, H.E.G.; Gavino-Lacuna, A.R.; Lota, M.M.M. Antimicrobial Resistance Profile of Escherichia Coli Isolated from Raw Chicken Meat in a Selected Wet Market in Manila City, Philippines. Acta Med Philipp 2025, 59, 107–122. [Google Scholar] [CrossRef] [PubMed]
- Fearnley, C.; Rodgers, J. A Survey of Antimicrobial Resistant (AMR) E. Coli, Campylobacter and Salmonella on Chicken and Turkey Meat on Retail Sale in the UK (2022); Food Standards Agency, 2023.
- García-Fernández, A.; Artuso, I.; Marotta, F.; Di Romualdo, R.; Arena, S.; De Marchis, M.L.; Pitti, M.; Primavilla, S.; Napoleoni, M.; Aschbacher, R.; et al. WGS-Based Surveillance for Campylobacter Spp. in Human Infections and Chicken Meat Production in Italy (2023). BMC Microbiol 2025, 25, 696. [Google Scholar] [CrossRef] [PubMed]
- Njoga, E.O.; Chah, K.F.; Oguttu, J.W. Drivers of Zoonotic Campylobacter Species Transmission in Slaughterhouse Settings: Insights from Nigeria for Global One Health Improvement. One Health 2026, 22, 101371. [Google Scholar] [CrossRef]
- Klous, G.; Huss, A.; Heederik, D.J.J.; Coutinho, R.A. Human–Livestock Contacts and Their Relationship to Transmission of Zoonotic Pathogens, a Systematic Review of Literature. One Health 2016, 2, 65–76. [Google Scholar] [CrossRef] [PubMed]
- Khairullah, A.R.; Widodo, A.; Riwu, K.H.P.; Yanestria, S.M.; Moses, I.B.; Effendi, M.H.; Fauzia, K.A.; Fauziah, I.; Hasib, A.; Kusala, M.K.J.; et al. Spread of Livestock-Associated Methicillin-Resistant Staphylococcus Aureus in Poultry and Its Risks to Public Health: A Comprehensive Review. Open Vet J 2024, 14, 2116–2128. [Google Scholar] [CrossRef] [PubMed]
- Dierikx, C.; van der Goot, J.; Fabri, T.; van Essen-Zandbergen, A.; Smith, H.; Mevius, D. Extended-Spectrum-β-Lactamase- and AmpC-β-Lactamase-Producing Escherichia Coli in Dutch Broilers and Broiler Farmers. J Antimicrob Chemother 2013, 68, 60–67. [Google Scholar] [CrossRef]
- Aminudin, M.H.; Amalina, F.; Ab Hamid, M.R.; Sulaiman, S.; Azfa, N.; Razak, A.S.A. Environmental and Public Health Risks of Antibiotic Resistance Gene Pollution in Poultry Systems: Sustainability Impact, Transmission Pathways, and Mitigation Strategies. The Microbe 2026, 10, 100658. [Google Scholar] [CrossRef]
- Mashe, T.; Thilliez, G.; Chaibva, B.V.; Leekitcharoenphon, P.; Bawn, M.; Nyanzunda, M.; Robertson, V.; Tarupiwa, A.; Al-Khanaq, H.; Baker, D.; et al. Highly Drug Resistant Clone of Salmonella Kentucky ST198 in Clinical Infections and Poultry in Zimbabwe. NPJ Antimicrob Resist 2023, 1, 6. [Google Scholar] [CrossRef]
- Le Hello, S.; Hendriksen, R.S.; Doublet, B.; Fisher, I.; Nielsen, E.M.; Whichard, J.M.; Bouchrif, B.; Fashae, K.; Granier, S.A.; Jourdan-Da Silva, N.; et al. International Spread of an Epidemic Population of Salmonella Enterica Serotype Kentucky ST198 Resistant to Ciprofloxacin. J Infect Dis 2011, 204, 675–684. [Google Scholar] [CrossRef]
- Liu, B.; Meng, C.; Wang, Z.; Li, Q.; Xu, C.; Kang, X.; Chen, L.; Wang, F.; Jiao, X.; Pan, Z. Prevalence and Transmission of Extensively Drug-Resistant Salmonella Enterica Serovar Kentucky ST198 Based on Whole-Genome Sequence in an Intensive Laying Hen Farm in Jiangsu, China. Poult Sci 2024, 103, 103608. [Google Scholar] [CrossRef]
- Medicine, C. for V. NARMS Interim Data Updates. In FDA; 2026. [Google Scholar]
- Greene, G.; Koolman, L.; Whyte, P.; Lynch, H.; Coffey, A.; Lucey, B.; Egan, J.; O’Connor, L.; Bolton, D. An in Vitro Investigation of the Survival and/or Growth of Campylobacter Jejuni in Broiler Digestate from Different Feed Types. Lett Appl Microbiol 2021, 72, 36–40. [Google Scholar] [CrossRef]
- Gottapu, C.; Edison, L.K.; Butcher, G.D.; Kariyawasam, S. Preharvest Control of Campylobacter Colonization in Chickens, with a Special Emphasis on Vaccination Strategies. Microorganisms 2025, 13. [Google Scholar] [CrossRef]
- Bergeron, C.R.; Prussing, C.; Boerlin, P.; Daignault, D.; Dutil, L.; Reid-Smith, R.J.; Zhanel, G.G.; Manges, A.R. Chicken as Reservoir for Extraintestinal Pathogenic Escherichia Coli in Humans, Canada. Emerg Infect Dis 2012, 18, 415–421. [Google Scholar] [CrossRef] [PubMed]
- Guragain, M.; Bagi, L.; Liu, Y.; Bosilevac, J.M. Characterization of Extraintestinal Pathogenic Escherichia Coli from Human Clinical and Poultry Samples. Microorganisms 2025, 13. [Google Scholar] [CrossRef] [PubMed]
- Conforti, S.; du Plessis, L.; Bagutti, C.; Becker, J.; Brugger, S.D.; Cusini, A.; Egli, A.; Gaia, V.; Greub, G.; Guler, C.; et al. From Clinics to Sewers: Leveraging Environmental Surveillance and Whole Genome Sequencing to Inform Transmission of ESBL-Escherichia Coli in Switzerland 2025, 2025.09.12.25335617.
- P, R.; Srijith, L.; G, K.; V.S, R.; Sridharan, K.S.; Paul, A.; Perumal, K. Genomic Analysis of ST117, 155, 1011, 167, 744, and 17391 in Poultry-Associated Multidrug Resistant Escherichia Coli Isolates from India. Sci Rep 2026, 16, 7438. [Google Scholar] [CrossRef]
- Feng, Y.; Wang, Y.; Zhu, B.; Gao, G.F.; Guo, Y.; Hu, Y. Metagenome-Assembled Genomes and Gene Catalog from the Chicken Gut Microbiome Aid in Deciphering Antibiotic Resistomes. Commun Biol 2021, 4, 1305. [Google Scholar] [CrossRef]
- Błażejewska, A.; Zalewska, M.; Grudniak, A.; Popowska, M. A Comprehensive Study of the Microbiome, Resistome, and Physical and Chemical Characteristics of Chicken Waste from Intensive Farms. Biomolecules 2022, 12, 1132. [Google Scholar] [CrossRef]
- Mazhar, S.H.; Li, X.; Rashid, A.; Su, J.; Xu, J.; Brejnrod, A.D.; Su, J.-Q.; Wu, Y.; Zhu, Y.-G.; Zhou, S.G.; et al. Co-Selection of Antibiotic Resistance Genes, and Mobile Genetic Elements in the Presence of Heavy Metals in Poultry Farm Environments. Science of The Total Environment 2021, 755, 142702. [Google Scholar] [CrossRef]
- Huber, L.; Agunos, A.; Gow, S.P.; Carson, C.A.; Boeckel, T.P.V. Reduction in Antimicrobial Use and Resistance to Salmonella, Campylobacter, and Escherichia Coli in Broiler Chickens, Canada, 2013–2019 -. Number 9—September 2021 - Emerging Infectious Diseases Journal - CDC Volume 27. [CrossRef]
- Sackey, B.A.; Mensah, P.; Collison, E.; Sakyi-Dawson, E. Campylobacter, Salmonella, Shigella and Escherichia Coli in Live and Dressed Poultry from Metropolitan Accra. Int J Food Microbiol 2001, 71, 21–28. [Google Scholar] [CrossRef]
- Ahmed, Z.S.; Hashad, M.E.; Atef, Y.; Badr, H.; Elhariri, M.; Kadry, M. Public Health Threat of Antimicrobial Resistance and Virulence Genes in Escherichia Coli from Human-Chicken Transmission in Egypt. Sci Rep 2025, 15, 12627. [Google Scholar] [CrossRef]
- Szoke, Z.; Fauszt, P.; Mikolas, M.; David, P.; Szilagyi-Tolnai, E.; Pesti-Asboth, G.; Homoki, J.R.; Kovacs-Forgacs, I.; Gal, F.; Stundl, L.; et al. Comprehensive Analysis of Antimicrobial Resistance Dynamics among Broiler and Duck Intensive Production Systems. Sci Rep 2025, 15, 4673. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Ling, W. Health Benefits and Future Research of Phytochemicals: A Literature Review. The Journal of Nutrition 2025, 155, 87–101. [Google Scholar] [CrossRef]
- Qassadi, F.I.; Zhu, Z.; Monaghan, T.M. Plant-Derived Products with Therapeutic Potential against Gastrointestinal Bacteria. Pathogens 2023, 12, 333. [Google Scholar] [CrossRef]
- Yang, C.; Chowdhury, M. A.K.; Hou, Y.; Gong, J. Phytogenic Compounds as Alternatives to In-Feed Antibiotics: Potentials and Challenges in Application. Pathogens 2015, 4, 137–156. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.H. Potential Synergy of Phytochemicals in Cancer Prevention: Mechanism of Action. The Journal of Nutrition 2004, 134, 3479S–3485S. [Google Scholar] [CrossRef] [PubMed]
- Hurdle, J.G.; O’Neill, A.J.; Chopra, I.; Lee, R.E. Targeting Bacterial Membrane Function: An Underexploited Mechanism for Treating Persistent Infections. Nat Rev Microbiol 2011, 9, 62–75. [Google Scholar] [CrossRef]
- Pérez-Flores, J.G.; García-Curiel, L.; Pérez-Escalante, E.; Contreras-López, E.; Aguilar-Lira, G.Y.; Ángel-Jijón, C.; González-Olivares, L.G.; Baena-Santillán, E.S.; Ocampo-Salinas, I.O.; Guerrero-Solano, J.A.; et al. Plant Antimicrobial Compounds and Their Mechanisms of Action on Spoilage and Pathogenic Bacteria: A Bibliometric Study and Literature Review. Applied Sciences 2025, 15. [Google Scholar] [CrossRef]
- Valdez-Lara, A.G.; Jaramillo-Granada, ÁM.; Ortega-Zambrano, D.; García-Marquez, E.; García-Fajardo, J.A.; Mercado-Uribe, H.; Ruiz-Suárez, J.C. Disruption of Biological Membranes by Hydrophobic Molecules: A Way to Inhibit Bacterial Growth. Front Microbiol 2025, 15, 1478519. [Google Scholar] [CrossRef]
- Li, K.; Guan, G.; Zhu, J.; Wu, H.; Sun, Q. Antibacterial Activity and Mechanism of a Laccase-Catalyzed Chitosan–Gallic Acid Derivative against Escherichia Coli and Staphylococcus Aureus. Food Control 2019, 96, 234–243. [Google Scholar] [CrossRef]
- Tang, Y.; Li, F.; Gu, D.; Wang, W.; Huang, J.; Jiao, X. Antimicrobial Effect and the Mechanism of Diallyl Trisulfide against Campylobacter Jejuni. Antibiotics (Basel) 2021, 10, 246. [Google Scholar] [CrossRef] [PubMed]
- Tyagi, P.; Singh, M.; Kumari, H.; Kumari, A.; Mukhopadhyay, K. Bactericidal Activity of Curcumin I Is Associated with Damaging of Bacterial Membrane. PLoS One 2015, 10, e0121313. [Google Scholar] [CrossRef]
- Khameneh, B.; Eskin, N.A.M.; Iranshahy, M.; Fazly Bazzaz, B.S. Phytochemicals: A Promising Weapon in the Arsenal against Antibiotic-Resistant Bacteria. Antibiotics (Basel) 2021, 10, 1044. [Google Scholar] [CrossRef]
- Guay, I.; Boulanger, S.; Isabelle, C.; Brouillette, E.; Chagnon, F.; Bouarab, K.; Marsault, E.; Malouin, F. Tomatidine and Analog FC04-100 Possess Bactericidal Activities against Listeria, Bacillus and Staphylococcus Spp. BMC Pharmacol Toxicol 2018, 19, 7. [Google Scholar] [CrossRef]
- Lamontagne Boulet, M.; Isabelle, C.; Guay, I.; Brouillette, E.; Langlois, J.-P.; Jacques, P.-É; Rodrigue, S.; Brzezinski, R.; Beauregard, P.B.; Bouarab, K.; et al. Tomatidine Is a Lead Antibiotic Molecule That Targets Staphylococcus Aureus ATP Synthase Subunit C. Antimicrobial Agents and Chemotherapy 2018, 62. [Google Scholar] [CrossRef]
- Wu, H.-Z.; Fei, H.-J.; Zhao, Y.; Liu, X.; Huang, Y.; Wu, S. Antibacterial mechanism of sulforaphane on Escherichia coli. Sichuan Da Xue Xue Bao Yi Xue Ban 2012, 43, 386–390. [Google Scholar] [PubMed]
- Gaurav, A.; Bakht, P.; Saini, M.; Pandey, S.; Pathania, R. Role of Bacterial Efflux Pumps in Antibiotic Resistance, Virulence, and Strategies to Discover Novel Efflux Pump Inhibitors. Microbiology (Reading) 2023, 169, 001333. [Google Scholar] [CrossRef]
- Kumawat, M.; Nabi, B.; Daswani, M.; Viquar, I.; Pal, N.; Sharma, P.; Tiwari, S.; Sarma, D.K.; Shubham, S.; Kumar, M. Role of Bacterial Efflux Pump Proteins in Antibiotic Resistance across Microbial Species. Microbial Pathogenesis 2023, 181, 106182. [Google Scholar] [CrossRef]
- Siriyong, T.; Srimanote, P.; Chusri, S.; Yingyongnarongkul, B.-E.; Suaisom, C.; Tipmanee, V.; Voravuthikunchai, S.P. Conessine as a Novel Inhibitor of Multidrug Efflux Pump Systems in Pseudomonas Aeruginosa. BMC Complement Altern Med 2017, 17, 405. [Google Scholar] [CrossRef]
- Kristiansen, M.M.; Leandro, C.; Ordway, D.; Martins, M.; Viveiros, M.; Pacheco, T.; Molnar, J.; Kristiansen, J.E.; Amaral, L. Thioridazine Reduces Resistance of Methicillin-Resistant Staphylococcus Aureus by Inhibiting a Reserpine-Sensitive Efflux Pump. In Vivo 2006, 20, 361–366. [Google Scholar]
- Zhang, L.; Tian, X.; Sun, L.; Mi, K.; Wang, R.; Gong, F.; Huang, L. Bacterial Efflux Pump Inhibitors Reduce Antibiotic Resistance. Pharmaceutics 2024, 16, 170. [Google Scholar] [CrossRef]
- Vasil, M.L. DNA Microarrays in Analysis of Quorum Sensing: Strengths and Limitations. J Bacteriol 2003, 185, 2061–2065. [Google Scholar] [CrossRef] [PubMed]
- Deep, A.; Chaudhary, U.; Gupta, V. Quorum Sensing and Bacterial Pathogenicity: From Molecules to Disease. J Lab Physicians 2011, 3, 4–11. [Google Scholar] [CrossRef] [PubMed]
- Rutherford, S.T.; Bassler, B.L. Bacterial Quorum Sensing: Its Role in Virulence and Possibilities for Its Control. Cold Spring Harb Perspect Med 2012, 2, a012427. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Yu, Z.; Ding, T. Quorum-Sensing Regulation of Antimicrobial Resistance in Bacteria. Microorganisms 2020, 8, 425. [Google Scholar] [CrossRef]
- Naga, N.G.; El-Badan, D.E.; Ghanem, K.M.; Shaaban, M.I. It Is the Time for Quorum Sensing Inhibition as Alternative Strategy of Antimicrobial Therapy. Cell Commun Signal 2023, 21, 133. [Google Scholar] [CrossRef] [PubMed]
- Case, R.J.; Labbate, M.; Kjelleberg, S. AHL-Driven Quorum-Sensing Circuits: Their Frequency and Function among the Proteobacteria. ISME J 2008, 2, 345–349. [Google Scholar] [CrossRef]
- Galloway, W.R.J.D.; Hodgkinson, J.T.; Bowden, S.D.; Welch, M.; Spring, D.R. Quorum Sensing in Gram-Negative Bacteria: Small-Molecule Modulation of AHL and AI-2 Quorum Sensing Pathways. Chem Rev 2011, 111, 28–67. [Google Scholar] [CrossRef]
- Burt, S.A.; Ojo-Fakunle, V.T.A.; Woertman, J.; Veldhuizen, E.J.A. The Natural Antimicrobial Carvacrol Inhibits Quorum Sensing in Chromobacterium Violaceum and Reduces Bacterial Biofilm Formation at Sub-Lethal Concentrations. PLOS ONE 2014, 9, e93414. [Google Scholar] [CrossRef]
- Corral-Lugo, A.; Daddaoua, A.; Ortega, A.; Espinosa-Urgel, M.; Krell, T. So Different and Still so Similar: The Plant Compound Rosmarinic Acid Mimics Bacterial Homoserine Lactone Quorum Sensing Signals. Communicative & Integrative Biology 2016, 9, e1156832. [Google Scholar] [CrossRef]
- Ren, Y.; Zhu, R.; You, X.; Li, D.; Guo, M.; Fei, B.; Liu, Y.; Yang, X.; Liu, X.; Li, Y. Quercetin: A Promising Virulence Inhibitor of Pseudomonas Aeruginosa LasB in Vitro. Appl Microbiol Biotechnol 2024, 108, 57. [Google Scholar] [CrossRef]
- Sturme, M.H.J.; Kleerebezem, M.; Nakayama, J.; Akkermans, A.D.L.; Vaugha, E.E.; de Vos, W.M. Cell to Cell Communication by Autoinducing Peptides in Gram-Positive Bacteria. Antonie Van Leeuwenhoek 2002, 81, 233–243. [Google Scholar] [CrossRef] [PubMed]
- Alqahtani, M.; Almukainzi, M.; Alghoribi, M.F.; El-Mahdy, A.M. Antivirulence Effects of Trans-Resveratrol and Curcumin on Methicillin-Resistant Staphylococcus Aureus (MRSA) from Saudi Arabia. Life 2024, 14. [Google Scholar] [CrossRef] [PubMed]
- Qin, N.; Tan, X.; Jiao, Y.; Liu, L.; Zhao, W.; Yang, S.; Jia, A. RNA-Seq-Based Transcriptome Analysis of Methicillin-Resistant Staphylococcus Aureus Biofilm Inhibition by Ursolic Acid and Resveratrol. Sci Rep 2014, 4, 5467. [Google Scholar] [CrossRef]
- Khaleghian, M.; Sahrayi, H.; Hafezi, Y.; Mirshafeeyan, M.; Moghaddam, Z.S.; Farasati Far, B.; Noorbazargan, H.; Mirzaie, A.; Ren, Q. In Silico Design and Mechanistic Study of Niosome-Encapsulated Curcumin against Multidrug-Resistant Staphylococcus Aureus Biofilms. Front. Microbiol. 2023, 14. [Google Scholar] [CrossRef] [PubMed]
- Nakagawa, S.; Hillebrand, G.G.; Nunez, G. Rosmarinus Officinalis L. (Rosemary) Extracts Containing Carnosic Acid and Carnosol Are Potent Quorum Sensing Inhibitors of Staphylococcus Aureus Virulence. Antibiotics (Basel) 2020, 9, 149. [Google Scholar] [CrossRef]
- Federle, M.J.; Bassler, B.L. Interspecies Communication in Bacteria. J Clin Invest 2003, 112, 1291–1299. [Google Scholar] [CrossRef]
- Sarigiannis, Y.; Papaneophytou, C. Targeting Bacterial Communication: Evaluating Phytochemicals as LuxS Inhibitors to Disrupt Quorum Sensing. Macromol 2024, 4, 753–771. [Google Scholar] [CrossRef]
- Morgaan, H.A.; Omar, H.M.G.; Zakaria, A.S.; Mohamed, N.M. Repurposing Carvacrol, Cinnamaldehyde, and Eugenol as Potential Anti-Quorum Sensing Agents against Uropathogenic Escherichia Coli Isolates in Alexandria, Egypt. BMC Microbiol 2023, 23, 300. [Google Scholar] [CrossRef]
- Flemming, H.-C.; Wingender, J. The Biofilm Matrix. Nat Rev Microbiol 2010, 8, 623–633. [Google Scholar] [CrossRef] [PubMed]
- Ceri, H.; Olson, M.E.; Stremick, C.; Read, R.R.; Morck, D.; Buret, A. The Calgary Biofilm Device: New Technology for Rapid Determination of Antibiotic Susceptibilities of Bacterial Biofilms. J Clin Microbiol 1999, 37, 1771–1776. [Google Scholar] [CrossRef] [PubMed]
- Mattick, J.S. Type IV Pili and Twitching Motility. Annual Review of Microbiology 2002, 56, 289–314. [Google Scholar] [CrossRef]
- Wei, G.; Palalay, J.-J.S.; Sanfilippo, J.E.; Yang, J.Q. Flagellum-Driven Motility Enhances Pseudomonas Aeruginosa Biofilm Formation by Altering Cell Orientation. Appl Environ Microbiol 91 e00821-25. [CrossRef]
- Pratt, L.A.; Kolter, R. Genetic Analysis of Escherichia Coli Biofilm Formation: Roles of Flagella, Motility, Chemotaxis and Type I Pili. Molecular Microbiology 1998, 30, 285–293. [Google Scholar] [CrossRef]
- Monte, J.; Abreu, A.C.; Borges, A.; Simões, L.C.; Simões, M. Antimicrobial Activity of Selected Phytochemicals against Escherichia Coli and Staphylococcus Aureus and Their Biofilms. Pathogens 2014, 3, 473–498. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-H.; Kim, Y.-G.; Ryu, S.Y.; Cho, M.H.; Lee, J. Ginkgolic Acids and Ginkgo Biloba Extract Inhibit Escherichia Coli O157:H7 and Staphylococcus Aureus Biofilm Formation. Int J Food Microbiol 2014, 174, 47–55. [Google Scholar] [CrossRef]
- Lee, J.-H.; Regmi, S.C.; Kim, J.-A.; Cho, M.H.; Yun, H.; Lee, C.-S.; Lee, J. Apple Flavonoid Phloretin Inhibits Escherichia Coli O157:H7 Biofilm Formation and Ameliorates Colon Inflammation in Rats ▿. Infect Immun 2011, 79, 4819–4827. [Google Scholar] [CrossRef]
- Joseph, J.; Boby, S.; Muyyarikkandy, M.S. Phytochemicals: A Promising Strategy to Combat Biofilm-Associated Antimicrobial Resistance. In Exploring Bacterial Biofilms; IntechOpen, 2025; ISBN 978-0-85466-946-2. [Google Scholar]
- Qian, W.; Fu, Y.; Liu, M.; Zhang, J.; Wang, W.; Li, J.; Zeng, Q.; Wang, T.; Li, Y. Mechanisms of Action of Luteolin Against Single- and Dual-Species of Escherichia Coli and Enterobacter Cloacae and Its Antibiofilm Activities. Appl Biochem Biotechnol 2021, 193, 1397–1414. [Google Scholar] [CrossRef]
- Ouyang, J.; Sun, F.; Feng, W.; Sun, Y.; Qiu, X.; Xiong, L.; Liu, Y.; Chen, Y. Quercetin Is an Effective Inhibitor of Quorum Sensing, Biofilm Formation and Virulence Factors in Pseudomonas Aeruginosa. J Appl Microbiol 2016, 120, 966–974. [Google Scholar] [CrossRef] [PubMed]
- Karnwal, A.; Jassim, A.Y.; Mohammed, A.A.; Sharma, V.; Al-Tawaha, A.R.M.S.; Sivanesan, I. Nanotechnology for Healthcare: Plant-Derived Nanoparticles in Disease Treatment and Regenerative Medicine. Pharmaceuticals (Basel) 2024, 17, 1711. [Google Scholar] [CrossRef] [PubMed]
- Meesaragandla, B.; Hayet, S.; Fine, T.; Janke, U.; Chai, L.; Delcea, M. Inhibitory Effect of Epigallocatechin Gallate-Silver Nanoparticles and Their Lysozyme Bioconjugates on Biofilm Formation and Cytotoxicity. ACS Appl Bio Mater 2022, 5, 4213–4221. [Google Scholar] [CrossRef]
- Guinane, C.M.; Cotter, P.D. Role of the Gut Microbiota in Health and Chronic Gastrointestinal Disease: Understanding a Hidden Metabolic Organ. Therap Adv Gastroenterol 2013, 6, 295–308. [Google Scholar] [CrossRef]
- Effect on the Host Metabolism. In The Microbiota in Gastrointestinal Pathophysiology; Academic Press, 2017; pp. 249–253.
- de Vos, W.M.; Tilg, H.; Hul, M.V.; Cani, P.D. Gut Microbiome and Health: Mechanistic Insights. Gut 2022, 71, 1020–1032. [Google Scholar] [CrossRef] [PubMed]
- Martel, J.; Ojcius, D.M.; Ko, Y.-F.; Young, J.D. Phytochemicals as Prebiotics and Biological Stress Inducers. Trends in Biochemical Sciences 2020, 45, 462–471. [Google Scholar] [CrossRef]
- Kan, J.; Wu, F.; Wang, F.; Zheng, J.; Cheng, J.; Li, Y.; Yang, Y.; Du, J. Phytonutrients: Sources, Bioavailability, Interaction with Gut Microbiota, and Their Impacts on Human Health. Front Nutr 2022, 9, 960309. [Google Scholar] [CrossRef]
- Clifford, M.N. Diet-Derived Phenols in Plasma and Tissues and Their Implications for Health. Planta Med 2004, 70, 1103–1114. [Google Scholar] [CrossRef]
- Clavel, T.; Fallani, M.; Lepage, P.; Levenez, F.; Mathey, J.; Rochet, V.; Sérézat, M.; Sutren, M.; Henderson, G.; Bennetau-Pelissero, C.; et al. Isoflavones and Functional Foods Alter the Dominant Intestinal Microbiota in Postmenopausal Women1. The Journal of Nutrition 2005, 135, 2786–2792. [Google Scholar] [CrossRef] [PubMed]
- Schröder, C.; Matthies, A.; Engst, W.; Blaut, M.; Braune, A. Identification and Expression of Genes Involved in the Conversion of Daidzein and Genistein by the Equol-Forming Bacterium Slackia Isoflavoniconvertens. Appl Environ Microbiol 2013, 79, 3494–3502. [Google Scholar] [CrossRef]
- Parkar, S.G.; Stevenson, D.E.; Skinner, M.A. The Potential Influence of Fruit Polyphenols on Colonic Microflora and Human Gut Health. Int J Food Microbiol 2008, 124, 295–298. [Google Scholar] [CrossRef]
- Lin, R.; Piao, M.; Song, Y. Dietary Quercetin Increases Colonic Microbial Diversity and Attenuates Colitis Severity in Citrobacter Rodentium-Infected Mice. Front Microbiol 2019, 10, 1092. [Google Scholar] [CrossRef]
- Larrosa, M.; González-Sarrías, A.; Yáñez-Gascón, M.J.; Selma, M.V.; Azorín-Ortuño, M.; Toti, S.; Tomás-Barberán, F.; Dolara, P.; Espín, J.C. Anti-Inflammatory Properties of a Pomegranate Extract and Its Metabolite Urolithin-A in a Colitis Rat Model and the Effect of Colon Inflammation on Phenolic Metabolism. J Nutr Biochem 2010, 21, 717–725. [Google Scholar] [CrossRef]
- Hidalgo, M.; Oruna-Concha, M.J.; Kolida, S.; Walton, G.E.; Kallithraka, S.; Spencer, J.P.E.; Gibson, G.R.; de Pascual-Teresa, S. Metabolism of Anthocyanins by Human Gut Microflora and Their Influence on Gut Bacterial Growth. J. Agric. Food Chem. 2012, 60, 3882–3890. [Google Scholar] [CrossRef] [PubMed]
- Faria, A.; Fernandes, I.; Norberto, S.; Mateus, N.; Calhau, C. Interplay between Anthocyanins and Gut Microbiota. J. Agric. Food Chem. 2014, 62, 6898–6902. [Google Scholar] [CrossRef]
- Dongre, D.S.; Saha, U.B.; Saroj, S.D. Exploring the Role of Gut Microbiota in Antibiotic Resistance and Prevention. Ann Med 57 2478317. [CrossRef] [PubMed]
- Oliver, A.; Xue, Z.; Villanueva, Y.T.; Durbin-Johnson, B.; Alkan, Z.; Taft, D.H.; Liu, J.; Korf, I.; Laugero, K.D.; Stephensen, C.B.; et al. Association of Diet and Antimicrobial Resistance in Healthy U.S. Adults. mBio 2022, 13, e0010122. [Google Scholar] [CrossRef]
- Wang, X.; Qi, Y.; Zheng, H. Dietary Polyphenol, Gut Microbiota, and Health Benefits. Antioxidants (Basel) 2022, 11, 1212. [Google Scholar] [CrossRef]
- Poles, J.; Karhu, E.; McGill, M.; McDaniel, H.R.; Lewis, J.E. The Effects of Twenty-Four Nutrients and Phytonutrients on Immune System Function and Inflammation: A Narrative Review. J Clin Transl Res 2021, 7, 333–376. [Google Scholar] [PubMed]
- Behl, T.; Kumar, K.; Brisc, C.; Rus, M.; Nistor-Cseppento, D.C.; Bustea, C.; Aron, R.A.C.; Pantis, C.; Zengin, G.; Sehgal, A.; et al. Exploring the Multifocal Role of Phytochemicals as Immunomodulators. Biomedicine & Pharmacotherapy 2021, 133, 110959. [Google Scholar] [CrossRef]
- Hui, J.; Li, L.; Li, R.; Wu, M.; Yang, Y.; Wang, J.; Fan, Y.; Zheng, X. Effects of Supplementation with β-Carotene on the Growth Performance and Intestinal Mucosal Barriers in Layer-Type Cockerels. Animal Science Journal 2020, 91, e13344. [Google Scholar] [CrossRef]
- Mantis, N.J.; Rol, N.; Corthésy, B. Secretory IgA’s Complex Roles in Immunity and Mucosal Homeostasis in the Gut. Mucosal Immunol 2011, 4, 603–611. [Google Scholar] [CrossRef]
- Hong, E.-H.; Song, J.H.; Kang, K.B.; Sung, S.H.; Ko, H.-J.; Yang, H. Anti-Influenza Activity of Betulinic Acid from Zizyphus Jujuba on Influenza A/PR/8 Virus. Biomol Ther (Seoul) 2015, 23, 345–349. [Google Scholar] [CrossRef]
- Guo, N.; Liu, Z.; Yan, Z.; Liu, Z.; Hao, K.; Liu, C.; Wang, J. Subinhibitory Concentrations of Honokiol Reduce α-Hemolysin (Hla) Secretion by Staphylococcus Aureus and the Hla-Induced Inflammatory Response by Inactivating the NLRP3 Inflammasome. Emerg Microbes Infect 2019, 8, 707–716. [Google Scholar] [CrossRef]
- Xu, T.; Kuang, T.; Du, H.; Li, Q.; Feng, T.; Zhang, Y.; Fan, G. Magnoflorine: A Review of Its Pharmacology, Pharmacokinetics and Toxicity. Pharmacological Research 2020, 152, 104632. [Google Scholar] [CrossRef]
- Muscolo, A.; Mariateresa, O.; Giulio, T.; Mariateresa, R. Oxidative Stress: The Role of Antioxidant Phytochemicals in the Prevention and Treatment of Diseases. Int J Mol Sci 2024, 25, 3264. [Google Scholar] [CrossRef]
- Long, D.R.; Mead, J.; Hendricks, J.M.; Hardy, M.E.; Voyich, J.M. 18β-Glycyrrhetinic Acid Inhibits Methicillin-Resistant Staphylococcus Aureus Survival and Attenuates Virulence Gene Expression. Antimicrob Agents Chemother 2013, 57, 241–247. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.T.; Lin, W.C.; Yu, B.; Lee, T.T. Antioxidant Capacity of Phytochemicals and Their Potential Effects on Oxidative Status in Animals — A Review. Asian-Australas J Anim Sci 2017, 30, 299–308. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.M.; Lee, T.T. Immunomodulatory Effects of Phytogenics in Chickens and Pigs — A Review. Asian-Australas J Anim Sci 2018, 31, 617–627. [Google Scholar] [CrossRef]
- Alarabei, A.A.; Abd Aziz, N.A.L.; AB Razak, N.I.; Abas, R.; Bahari, H.; Abdullah, M.A.; Hussain, M.K.; Abdul Majid, A.M.S.; Basir, R. Immunomodulating Phytochemicals: An Insight Into Their Potential Use in Cytokine Storm Situations. Adv Pharm Bull 2024, 14, 105–119. [Google Scholar] [CrossRef] [PubMed]
- Mahasneh, Z.M.H.; Abuajamieh, M.; Abedal-Majed, M.A.; Al-Qaisi, M.; Abdelqader, A.; Al-Fataftah, A.-R.A. Effects of Medical Plants on Alleviating the Effects of Heat Stress on Chickens. Poult Sci 2024, 103, 103391. [Google Scholar] [CrossRef]
- Muscolo, A.; Mariateresa, O.; Giulio, T.; Mariateresa, R. Oxidative Stress: The Role of Antioxidant Phytochemicals in the Prevention and Treatment of Diseases. Int J Mol Sci 2024, 25, 3264. [Google Scholar] [CrossRef] [PubMed]
- Darmawan, A.; Hermana, W.; Suci, D.M.; Mutia, R.; Sumiati; Jayanegara, A.; Ozturk, E. Dietary Phytogenic Extracts Favorably Influence Productivity, Egg Quality, Blood Constituents, Antioxidant and Immunological Parameters of Laying Hens: A Meta-Analysis. Animals 2022, 12, 2278. [Google Scholar] [CrossRef] [PubMed]
- Dong, S.; Li, L.; Hao, F.; Fang, Z.; Zhong, R.; Wu, J.; Fang, X. Improving Quality of Poultry and Its Meat Products with Probiotics, Prebiotics, and Phytoextracts. Poultry Science 2024, 103, 103287. [Google Scholar] [CrossRef]
- Abuoghaba, A.A.-K.; Ragab, M.A.; Shazly, S.A.; Kokoszyński, D.; Saleh, M. Impact of Treating Hatching Eggs with Curcumin after Exposure to Thermal Stress on Embryonic Development, Hatchability, Physiological Body Reactions, and Hormonal Profiles of Dokki-4 Chickens. Animals (Basel) 2021, 11, 3220. [Google Scholar] [CrossRef]
- Ling, C.; Chen, X.; Lin, W.; Geng, Z. Green Tea Powder Inclusion Promoted Hatchability through Increased Yolk Antioxidant Activity. Poult Sci 2022, 101, 101804. [Google Scholar] [CrossRef]
- Poslinski, H.; Hatley, M.; Tramell, J.; Song, B.-H. Harnessing Phytochemicals in Sustainable and Green Agriculture. Journal of Agriculture and Food Research 2025, 19, 101633. [Google Scholar] [CrossRef]
- Hoffmann, S.; White, A.E.; McQueen, R.B.; Ahn, J.-W.; Gunn-Sandell, L.B.; Scallan Walter, E.J. Economic Burden of Foodborne Illnesses Acquired in the United States. Foodborne Pathog Dis 2024. [Google Scholar] [CrossRef]
- Chlebicz, A.; Śliżewska, K. Campylobacteriosis, Salmonellosis, Yersiniosis, and Listeriosis as Zoonotic Foodborne Diseases: A Review. Int J Environ Res Public Health 2018, 15, 863. [Google Scholar] [CrossRef]
- Punchihewage-Don, A.J.; Hawkins, J.; Adnan, A.M.; Hashem, F.; Parveen, S. The Outbreaks and Prevalence of Antimicrobial Resistant Salmonella in Poultry in the United States: An Overview. Heliyon 2022, 8, e11571. [Google Scholar] [CrossRef] [PubMed]
- Seyedtaghiya, M.H.; Fasaei, B.N.; Peighambari, S.M. Antimicrobial and Antibiofilm Effects of Satureja Hortensis Essential Oil against Escherichia Coli and Salmonella Isolated from Poultry. Iran J Microbiol 2021, 13, 74–80. [Google Scholar] [CrossRef] [PubMed]
- Alvarado-Martinez, Z.; Bravo, P.; Kennedy, N.-F.; Krishna, M.; Hussain, S.; Young, A.C.; Biswas, D. Antimicrobial and Antivirulence Impacts of Phenolics on Salmonella Enterica Serovar Typhimurium. Antibiotics 2020, 9. [Google Scholar] [CrossRef]
- Marathe, S.A.; Balakrishnan, A.; Negi, V.D.; Sakorey, D.; Chandra, N.; Chakravortty, D. Curcumin Reduces the Motility of Salmonella Enterica Serovar Typhimurium by Binding to the Flagella, Thereby Leading to Flagellar Fragility and Shedding. Journal of Bacteriology 2016, 198, 1798–1811. [Google Scholar] [CrossRef]
- Dahl, T.A.; McGowan, W.M.; Shand, M.A.; Srinivasan, V.S. Photokilling of Bacteria by the Natural Dye Curcumin. Arch Microbiol 1989, 151, 183–185. [Google Scholar] [CrossRef]
- Wang, N.; Ning, C.; Zhao, Z.; Yang, C.; Ren, H.; Chen, L.; Yu, Q.; Zhang, G. Antibacterial Mechanism Analysis of Resveratrol against Salmonella Typhimurium via Metabolomics. Appl Microbiol Biotechnol 2024, 108, 512. [Google Scholar] [CrossRef]
- Kollanoor-Johny, A.; Upadhyay, A.; Baskaran, S.A.; Upadhyaya, I.; Mooyottu, S.; Mishra, N.; Darre, M.J.; Khan, M.I.; Donoghue, A.M.; Donoghue, D.J.; et al. Effect of Therapeutic Supplementation of the Plant Compounds Trans-Cinnamaldehyde and Eugenol on Salmonella Enterica Serovar Enteritidis Colonization in Market-Age Broiler Chickens1 2. Journal of Applied Poultry Research 2012, 21, 816–822. [Google Scholar] [CrossRef]
- CDC About Campylobacter Infection. Available online: https://www.cdc.gov/campylobacter/about/index.html (accessed on 9 March 2026).
- Hoffmann, S.; White, A.E.; McQueen, R.B.; Ahn, J.-W.; Gunn-Sandell, L.B.; Scallan Walter, E.J. Economic Burden of Foodborne Illnesses Acquired in the United States. Foodborne Pathogens and Disease 2025, 22, 4–14. [Google Scholar] [CrossRef]
- Gharbi, M.; Béjaoui, A.; Hamrouni, S.; Arfaoui, A.; Maaroufi, A. Persistence of Campylobacter Spp. in Poultry Flocks after Disinfection, Virulence, and Antimicrobial Resistance Traits of Recovered Isolates. Antibiotics 2023, 12. [Google Scholar] [CrossRef]
- García-Fernández, A.; Dionisi, A.M.; Arena, S.; Iglesias-Torrens, Y.; Carattoli, A.; Luzzi, I. Human Campylobacteriosis in Italy: Emergence of Multi-Drug Resistance to Ciprofloxacin, Tetracycline, and Erythromycin. Front. Microbiol. 2018, 9. [Google Scholar] [CrossRef]
- Alfredson, D.A.; Korolik, V. Antibiotic Resistance and Resistance Mechanisms in Campylobacter Jejuni and Campylobacter Coli. FEMS Microbiol Lett 2007, 277, 123–132. [Google Scholar] [CrossRef]
- Wagle, B.R.; Donoghue, A.M.; Shrestha, S.; Upadhyaya, I.; Arsi, K.; Gupta, A.; Liyanage, R.; Rath, N.C.; Donoghue, D.J.; Upadhyay, A. Carvacrol Attenuates Campylobacter Jejuni Colonization Factors and Proteome Critical for Persistence in the Chicken Gut. Poult Sci 2020, 99, 4566–4577. [Google Scholar] [CrossRef]
- Szott, V.; Reichelt, B.; Alter, T.; Friese, A.; Roesler, U. In Vivo Efficacy of Carvacrol on Campylobacter Jejuni Prevalence in Broiler Chickens during an Entire Fattening Period. Eur J Microbiol Immunol (Bp) 2020, 10, 131–138. [Google Scholar] [CrossRef]
- Allaoua, M.; Bonnafé, E.; Etienne, P.; Noirot, V.; Gabarrou, J.; Castinel, A.; Pascal, G.; Darbot, V.; Treilhou, M.; Combes, S. A Carvacrol-based Product Reduces Campylobacter Jejuni Load and Alters Microbiota Composition in the Caeca of Chickens. J Appl Microbiol 2022, 132, 4501–4516. [Google Scholar] [CrossRef]
- de los Santos, F.S.; Donoghue, A.M.; Venkitanarayanan, K.; Reyes-Herrera, I.; Metcalf, J.H.; Dirain, M.L.; Aguiar, V.F.; Blore, P.J.; Donoghue, D.J. Therapeutic Supplementation of Caprylic Acid in Feed Reduces Campylobacter Jejuni Colonization in Broiler Chicks. Appl Environ Microbiol 2008, 74, 4564–4566. [Google Scholar] [CrossRef]
- Shrestha, S.; Wagle, B.R.; Upadhyay, A.; Arsi, K.; Donoghue, D.J.; Donoghue, A.M. Carvacrol Antimicrobial Wash Treatments Reduce Campylobacter Jejuni and Aerobic Bacteria on Broiler Chicken Skin. Poultry Science 2019, 98, 4073–4083. [Google Scholar] [CrossRef]
- Abd El-Hack, M.E.; El-Saadony, M.T.; Elbestawy, A.R.; El-Shall, N.A.; Saad, A.M.; Salem, H.M.; El-Tahan, A.M.; Khafaga, A.F.; Taha, A.E.; AbuQamar, S.F.; et al. Necrotic Enteritis in Broiler Chickens: Disease Characteristics and Prevention Using Organic Antibiotic Alternatives – a Comprehensive Review. Poult Sci 2021, 101, 101590. [Google Scholar] [CrossRef]
- Van Immerseel, F.; De Buck, J.; Pasmans, F.; Huyghebaert, G.; Haesebrouck, F.; Ducatelle, R. Clostridium Perfringens in Poultry: An Emerging Threat for Animal and Public Health. Avian Pathol 2004, 33, 537–549. [Google Scholar] [CrossRef]
- la Mora, Z.V.; Macías-Rodríguez, M.E.; Arratia-Quijada, J.; Gonzalez-Torres, Y.S.; Nuño, K.; Villarruel-López, A. Clostridium Perfringens as Foodborne Pathogen in Broiler Production: Pathophysiology and Potential Strategies for Controlling Necrotic Enteritis. Animals 2020, 10, 1718. [Google Scholar] [CrossRef]
- CDC Clostridium Perfringens Root Causes. Available online: https://www.cdc.gov/restaurant-food-safety/php/practices/clostridium-perfringens-root-causes.html (accessed on 16 March 2026).
- Casewell, M.; Friis, C.; Marco, E.; McMullin, P.; Phillips, I. The European Ban on Growth-Promoting Antibiotics and Emerging Consequences for Human and Animal Health. J Antimicrob Chemother 2003, 52, 159–161. [Google Scholar] [CrossRef]
- Friedlein, U.; Dorn-In, S.; Schwaiger, K. Antimicrobial Effects of Plant Extracts against Clostridium Perfringens with Respect to Food-Relevant Influencing Factors. Journal of Food Protection 2021, 84, 1809–1818. [Google Scholar] [CrossRef]
- Alfattani, S.Q.; Banawas, S.S.; Sarker, M.R. Inhibitory Effects of Natural Products on Germination, Outgrowth, and Vegetative Growth of Clostridium Perfringens Spores in Laboratory Medium and Chicken Meat. Microorganisms 2025, 13. [Google Scholar] [CrossRef]
- Liu, Y.; Zhu, J.; Qu, S.; Shen, J.; Zhu, K. Plant-Derived Xanthones against Clostridial Enteric Infections. Antibiotics 2023, 12. [Google Scholar] [CrossRef]
- Ghiselli, F.; Giovagnoni, G.; Felici, M.; Tugnoli, B.; Piva, A.; Grilli, E. A Mixture of Organic Acids and Thymol Protects Primary Chicken Intestinal Epithelial Cells from Clostridium Perfringens Infection in Vitro. Poult Sci 2022, 101, 102101. [Google Scholar] [CrossRef]
- Du, E.; Gan, L.; Li, Z.; Wang, W.; Liu, D.; Guo, Y. In Vitro Antibacterial Activity of Thymol and Carvacrol and Their Effects on Broiler Chickens Challenged with Clostridium Perfringens. J Anim Sci Biotechnol 2015, 6, 58. [Google Scholar] [CrossRef]
- Juneja, V.K.; Friedman, M. Carvacrol, Cinnamaldehyde, Oregano Oil, and Thymol Inhibit Clostridium Perfringens Spore Germination and Outgrowth in Ground Turkey during Chilling. J Food Prot 2007, 70, 218–222. [Google Scholar] [CrossRef]
- Yin, D.; Du, E.; Yuan, J.; Gao, J.; Wang, Y.; Aggrey, S.E.; Guo, Y. Supplemental Thymol and Carvacrol Increases Ileum Lactobacillus Population and Reduces Effect of Necrotic Enteritis Caused by Clostridium Perfringes in Chickens. Sci Rep 2017, 7, 7334. [Google Scholar] [CrossRef]
- Jerzsele, A.; Szeker, K.; Csizinszky, R.; Gere, E.; Jakab, C.; Mallo, J.J.; Galfi, P. Efficacy of Protected Sodium Butyrate, a Protected Blend of Essential Oils, Their Combination, and Bacillus Amyloliquefaciens Spore Suspension against Artificially Induced Necrotic Enteritis in Broilers. Poultry Science 2012, 91, 837–843. [Google Scholar] [CrossRef]
- Dahiya, J.P.; Wilkie, D.C.; Van Kessel, A.G.; Drew, M.D. Potential Strategies for Controlling Necrotic Enteritis in Broiler Chickens in Post-Antibiotic Era. Animal Feed Science and Technology 2006, 129, 60–88. [Google Scholar] [CrossRef]
- Du, E.; Wang, W.; Gan, L.; Li, Z.; Guo, S.; Guo, Y. Effects of Thymol and Carvacrol Supplementation on Intestinal Integrity and Immune Responses of Broiler Chickens Challenged with Clostridium Perfringens. J Anim Sci Biotechnol 2016, 7, 19. [Google Scholar] [CrossRef]
- Santhiravel, S.; Bekhit, A.E.-D.A.; Mendis, E.; Jacobs, J.L.; Dunshea, F.R.; Rajapakse, N.; Ponnampalam, E.N. The Impact of Plant Phytochemicals on the Gut Microbiota of Humans for a Balanced Life. Int J Mol Sci 2022, 23, 8124. [Google Scholar] [CrossRef] [PubMed]
- Nantapo, C.W.T.; Marume, U. Strategic Technologies to Improve Phytogenic Feed Additive Efficacy in Pigs and Poultry. Animal Nutrition 2025, 23, 286–303. [Google Scholar] [CrossRef]
- Abdelli, N.; Pérez, J.F.; Vilarrasa, E.; Cabeza Luna, I.; Melo-Duran, D.; D’Angelo, M.; Solà-Oriol, D. Targeted-Release Organic Acids and Essential Oils Improve Performance and Digestive Function in Broilers under a Necrotic Enteritis Challenge. Animals (Basel) 2020, 10, 259. [Google Scholar] [CrossRef]
- Xu, H.; Zhang, X.; Li, P.; Luo, Y.; Fu, J.; Gong, L.; Lv, Z.; Guo, Y. Effects of Tannic Acid Supplementation on the Intestinal Health, Immunity, and Antioxidant Function of Broilers Challenged with Necrotic Enteritis. Antioxidants (Basel) 2023, 12, 1476. [Google Scholar] [CrossRef]
- Osek, J.; Lachtara, B.; Wieczorek, K. Listeria Monocytogenes – How This Pathogen Survives in Food-Production Environments? Front Microbiol 2022, 13, 866462. [Google Scholar] [CrossRef]
- Rørvik, L.M.; Aase, B.; Alvestad, T.; Caugant, D.A. Molecular Epidemiological Survey of Listeria Monocytogenes in Broilers and Poultry Products. J. Appl. Microbiol. 2003, 94, 633–640. [Google Scholar] [CrossRef]
- Jamshidi, A.; Zeinali, T. Significance and Characteristics of Listeria Monocytogenes in Poultry Products. International Journal of Food Science 2019, 2019, 7835253. [Google Scholar] [CrossRef]
- CDC About Listeria Infection. Available online: https://www.cdc.gov/listeria/about/index.html (accessed on 16 March 2026).
- Panera Martínez, S.; Capita, R.; Pedriza-González, Á; Díez-Moura, M.; Riesco-Peláez, F.; Alonso-Calleja, C. Occurrence, Antibiotic Resistance and Biofilm-Forming Ability of Listeria Monocytogenes in Chicken Carcasses and Cuts. Foods 2024, 13, 3822. [Google Scholar] [CrossRef]
- Morshdy, A.E.M.A.; Al-Mogbel, M.S.; Mohamed, M.E.M.; Elabbasy, M.T.; Elshafee, A.K.; Hussein, M.A. Bioactivity of Essential Oils for Mitigation of Listeria Monocytogenes Isolated from Fresh Retail Chicken Meat. Foods 2021, 10, 3006. [Google Scholar] [CrossRef]
- Osaili, T.M.; Hasan, F.; Dhanasekaran, D.K.; Obaid, R.S.; Al-Nabulsi, A.A.; Ayyash, M.; Karam, L.; Savvaidis, I.N.; Holley, R. Effect of Active Essential Oils Added to Chicken Tawook on the Behaviour of Listeria Monocytogenes, Salmonella Spp. and Escherichia Coli O157:H7 during Storage. International Journal of Food Microbiology 2021, 337, 108947. [Google Scholar] [CrossRef]
- Liang, S.; Hu, X.; Wang, R.; Fang, M.; Yu, Y.; Xiao, X. The Combination of Thymol and Cinnamaldehyde Reduces the Survival and Virulence of Listeria Monocytogenes on Autoclaved Chicken Breast. J Appl Microbiol 2022, 132, 3937–3950. [Google Scholar] [CrossRef]
- Liu, Y.; Wu, L.; Han, J.; Dong, P.; Luo, X.; Zhang, Y.; Zhu, L. Inhibition of Biofilm Formation and Related Gene Expression of Listeria Monocytogenes in Response to Four Natural Antimicrobial Compounds and Sodium Hypochlorite. Front Microbiol 2021, 11, 617473. [Google Scholar] [CrossRef] [PubMed]
- Roy, P.K.; Song, M.G.; Park, S.Y. The Inhibitory Effect of Quercetin on Biofilm Formation of Listeria Monocytogenes Mixed Culture and Repression of Virulence. Antioxidants (Basel) 2022, 11, 1733. [Google Scholar] [CrossRef] [PubMed]
- Risalvato, J.; Sewid, A.H.; Eda, S.; Gerhold, R.W.; Wu, J.J. Strategic Detection of Escherichia Coli in the Poultry Industry: Food Safety Challenges, One Health Approaches, and Advances in Biosensor Technologies. Biosensors 2025, 15, 419. [Google Scholar] [CrossRef] [PubMed]
- Nawaz, S.; Wang, Z.; Zhang, Y.; Jia, Y.; Jiang, W.; Chen, Z.; Yin, H.; Huang, C.; Han, X. Avian Pathogenic Escherichia Coli (APEC): Current Insights and Future Challenges. Poult Sci 2024, 103, 104359. [Google Scholar] [CrossRef]
- Casalino, G.; Dinardo, F.R.; D’Amico, F.; Bozzo, G.; Bove, A.; Camarda, A.; Lombardi, R.; Dimuccio, M.M.; Circella, E. Antimicrobial Efficacy of Cinnamon Essential Oil against Avian Pathogenic Escherichia Coli from Poultry. Animals (Basel) 2023, 13, 2639. [Google Scholar] [CrossRef]
- Ruan, X.; Deng, X.; Tan, M.; Wang, Y.; Hu, J.; Sun, Y.; Yu, C.; Zhang, M.; Jiang, N.; Jiang, R. Effect of Resveratrol on the Biofilm Formation and Physiological Properties of Avian Pathogenic Escherichia Coli. J Proteomics 2021, 249, 104357. [Google Scholar] [CrossRef]
- Pham, V.H.; Abbas, W.; Huang, J.; Guo, F.; Zhang, K.; Kong, L.; Zhen, W.; Guo, Y.; Wang, Z. Dietary Coated Essential Oil and Organic Acid Mixture Supplementation Improves Health of Broilers Infected with Avian Pathogenic Escherichiacoli. Anim Nutr 2022, 12, 245–262. [Google Scholar] [CrossRef]
- Qu, Q.; Liu, M.; Hu, Y.; Huang, G.; Xuan, Z.; Lun, J.; Chen, X.; Lv, W.; Guo, S. Modulatory Effects of Polyherbal Mixture on the Immuno-Antioxidant Capacity and Intestinal Health of Chicks Infected with Escherichia Coli O78. Poult Sci 2025, 104, 105156. [Google Scholar] [CrossRef]
- Meng, J.; Wang, W.; Ding, J.; Gu, B.; Zhou, F.; Wu, D.; Fu, X.; Qiao, M.; Liu, J. The Synergy Effect of Matrine and Berberine Hydrochloride on Treating Colibacillosis Caused by an Avian Highly Pathogenic Multidrug-Resistant Escherichia Coli. Poult Sci 2024, 103, 104151. [Google Scholar] [CrossRef]
- Li, Y.; Ge, X. Role of Berberine as a Potential Efflux Pump Inhibitor against MdfA from Escherichia Coli: In Vitro and In Silico Studies. Microbiol Spectr 2023, 11, e0332422. [Google Scholar] [CrossRef]
- Abdel-Raheem, S.M.; Abd El-Hamid, M.I.; Ibrahim, D.; El-Malt, R.M.S.; El-Ghareeb, W.R.; Ismail, H.A.; Al-Sultan, S.I.; Meligy, A.M.A.; ELTarabili, R.M. Future Scope of Plant-Derived Bioactive Compounds in the Management of Methicillin-Resistant Staphylococcus Aureus: In Vitro Antimicrobial and Antivirulence Prospects to Combat MRSA. Microbial Pathogenesis 2023, 183, 106301. [Google Scholar] [CrossRef]
- Kollanoor Johny, A.; Frye, J.G.; Donoghue, A.; Donoghue, D.J.; Porwollik, S.; McClelland, M.; Venkitanarayanan, K. Gene Expression Response of Salmonella Enterica Serotype Enteritidis Phage Type 8 to Subinhibitory Concentrations of the Plant-Derived Compounds Trans-Cinnamaldehyde and Eugenol. Front Microbiol 2017, 8, 1828. [Google Scholar] [CrossRef]
- Kollanoor-Johny, A.; Mattson, T.; Baskaran, S.A.; Amalaradjou, M.A.; Babapoor, S.; March, B.; Valipe, S.; Darre, M.; Hoagland, T.; Schreiber, D.; et al. Reduction of Salmonella Enterica Serovar Enteritidis Colonization in 20-Day-Old Broiler Chickens by the Plant-Derived Compounds Trans-Cinnamaldehyde and Eugenol. Appl Environ Microbiol 2012, 78, 2981–2987. [Google Scholar] [CrossRef]
- Ferreira, S.; Domingues, F. The Antimicrobial Action of Resveratrol against Listeria Monocytogenes in Food-Based Models and Its Antibiofilm Properties. J Sci Food Agric 2016, 96, 4531–4535. [Google Scholar] [CrossRef] [PubMed]
- Valenzuela-Grijalva, N.V.; Pinelli-Saavedra, A.; Muhlia-Almazan, A.; Domínguez-Díaz, D.; González-Ríos, H. Dietary Inclusion Effects of Phytochemicals as Growth Promoters in Animal Production. J Anim Sci Technol 2017, 59, 8. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Salas, P.; Morales-Soto, A.; Segura-Carretero, A.; Fernández-Gutiérrez, A. Phenolic-Compound-Extraction Systems for Fruit and Vegetable Samples. Molecules 2010, 15, 8813–8826. [Google Scholar] [CrossRef]
- Grashorn, M. Use of Phytobiotics in Broiler Nutrition– an Alternative to Infeed Antibiotics? J. Anim. Feed Sci. 2010, 19, 338–347. [Google Scholar] [CrossRef]
- Aqil, F.; Munagala, R.; Jeyabalan, J.; Vadhanam, M.V. Bioavailability of Phytochemicals and Its Enhancement by Drug Delivery Systems. Cancer Lett 2013, 334, 133–141. [Google Scholar] [CrossRef] [PubMed]
- Bubonja-Šonje, M.; Knežević, S.; Abram, M. Challenges to Antimicrobial Susceptibility Testing of Plant-Derived Polyphenolic Compounds. Arh Hig Rada Toksikol 2020, 71, 300–311. [Google Scholar] [CrossRef] [PubMed]
- Obianwuna, U.E.; Chang, X.; Oleforuh-Okoleh, V.U.; Onu, P.N.; Zhang, H.; Qiu, K.; Wu, S. Phytobiotics in Poultry: Revolutionizing Broiler Chicken Nutrition with Plant-Derived Gut Health Enhancers. Journal of Animal Science and Biotechnology 2024, 15, 169. [Google Scholar] [CrossRef] [PubMed]
- Al-Sultan, S.I.; Gameel, A.A. Histopathological Changes in the Livers of Broiler Chicken Supplemented with Turmeric (Curcuma Longa). Available online: https://scialert.net/abstract/?doi=ijps.2004.333.336 (accessed on 26 August 2024).
- Roper, T.J. Olfaction in Birds. In Advances in the Study of Behavior; Slater, P.J.B., Rosenblat, J.S., Snowden, C.T., Roper, T.J., Eds.; Academic Press, 1999; Vol. 28, pp. 247–332. [Google Scholar]
- Luiken, R.E.C.; Van Gompel, L.; Munk, P.; Sarrazin, S.; Joosten, P.; Dorado-García, A.; Borup Hansen, R.; Knudsen, B.E.; Bossers, A.; Wagenaar, J.A.; et al. Associations between Antimicrobial Use and the Faecal Resistome on Broiler Farms from Nine European Countries. J Antimicrob Chemother 2019, 74, 2596–2604. [Google Scholar] [CrossRef]
- Gaonkar, P.P.; Santana-Pereira, A.L.R.; Golden, R.; Lambert, A.; Higgins, C.; Adhikari, Y.; Bailey, M.; Macklin, K.; Huber, L. Microbiome and Resistome Dynamics in Different Stages of Commercial Broiler Production with Restricted Antimicrobial Use. BMC Microbiol 2026, 26, 126. [Google Scholar] [CrossRef]
- Li, W.; Li, Y.; Jia, Y.; Sun, H.; Zhang, C.; Hu, G.; Yuan, L. Genomic Characteristics of Mcr-1 and blaCTX-M-Type in a Single Multidrug-Resistant Escherichia Coli ST93 from Chicken in China. Poult Sci 2021, 100, 101074. [Google Scholar] [CrossRef]
- Khalifa, H.O.; Elbediwi, M.; Mohammed, T.; Abdalla, A.; Mohamed, M.-Y.I.; Lakshmi, G.B.; Habib, I. Molecular Characterization of Mcr-1.1-Harboring Multidrug-Resistant Escherichia Coli Isolates from Chicken in the United Arab Emirates: Implications for One Health Surveillance. Front. Vet. Sci. 2026, 12. [Google Scholar] [CrossRef]
- Huang, J.; Guo, F.; Abbas, W.; Hu, Z.; Liu, L.; Qiao, J.; Bi, R.; Xu, T.; Zhang, K.; Huang, J.; et al. Effects of Microencapsulated Essential Oils and Organic Acids Preparation on Growth Performance, Slaughter Characteristics, Nutrient Digestibility and Intestinal Microenvironment of Broiler Chickens. Poultry Science 2024, 103, 103655. [Google Scholar] [CrossRef]
- Ibrahim, D.; Abdelfattah-Hassan, A.; Badawi, M.; Ismail, T.A.; Bendary, M.M.; Abdelaziz, A.M.; Mosbah, R.A.; Mohamed, D.I.; Arisha, A.H.; El-Hamid, M.I.A. Thymol Nanoemulsion Promoted Broiler Chicken’s Growth, Gastrointestinal Barrier and Bacterial Community and Conferred Protection against Salmonella Typhimurium. Sci Rep 2021, 11, 7742. [Google Scholar] [CrossRef] [PubMed]
- Bendary, M.M.; Ibrahim, D.; Mosbah, R.A.; Mosallam, F.; Hegazy, W.A.H.; Awad, N.F.S.; Alshareef, W.A.; Alomar, S.Y.; Zaitone, S.A.; Abd El-Hamid, M.I. Thymol Nanoemulsion: A New Therapeutic Option for Extensively Drug Resistant Foodborne Pathogens. Antibiotics 2021, 10, 25. [Google Scholar] [CrossRef]
- Press, M.O.; Wiser, A.H.; Kronenberg, Z.N.; Langford, K.W.; Shakya, M.; Lo, C.-C.; Mueller, K.A.; Sullivan, S.T.; Chain, P.S.G.; Liachko, I. Hi-C Deconvolution of a Human Gut Microbiome Yields High-Quality Draft Genomes and Reveals Plasmid-Genome Interactions 2017. 198713.
- Stalder, T.; Press, M.O.; Sullivan, S.; Liachko, I.; Top, E.M. Linking the Resistome and Plasmidome to the Microbiome. ISME J 2019, 13, 2437–2446. [Google Scholar] [CrossRef] [PubMed]
- Castañeda-Barba, S.; Ridenhour, B.J.; Top, E.M.; Stalder, T. Detection of Rare Plasmid Hosts Using a Targeted Hi-C Approach. ISME Commun 2025, 5, ycae161. [Google Scholar] [CrossRef] [PubMed]
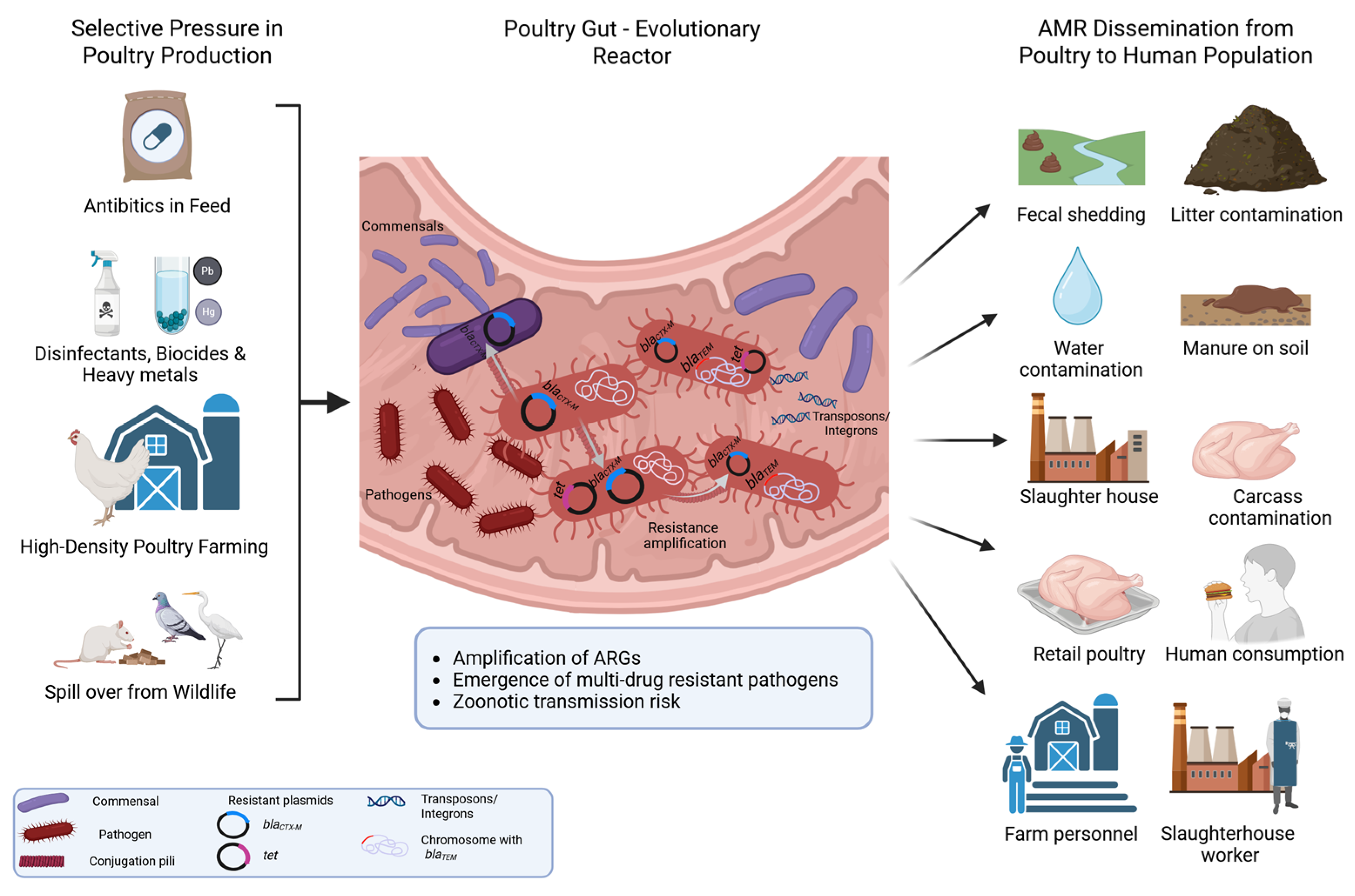
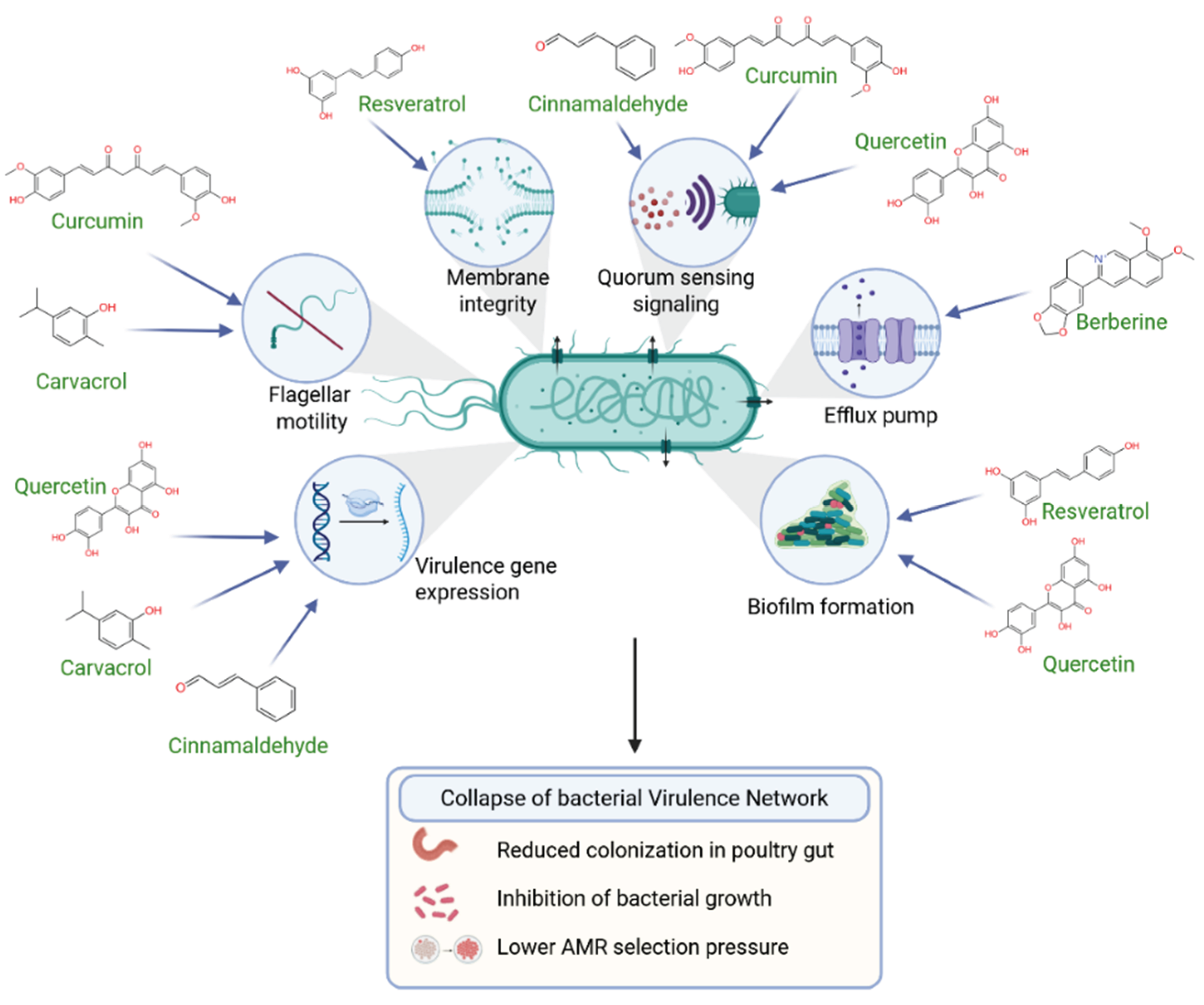
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




