Submitted:
21 April 2026
Posted:
22 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
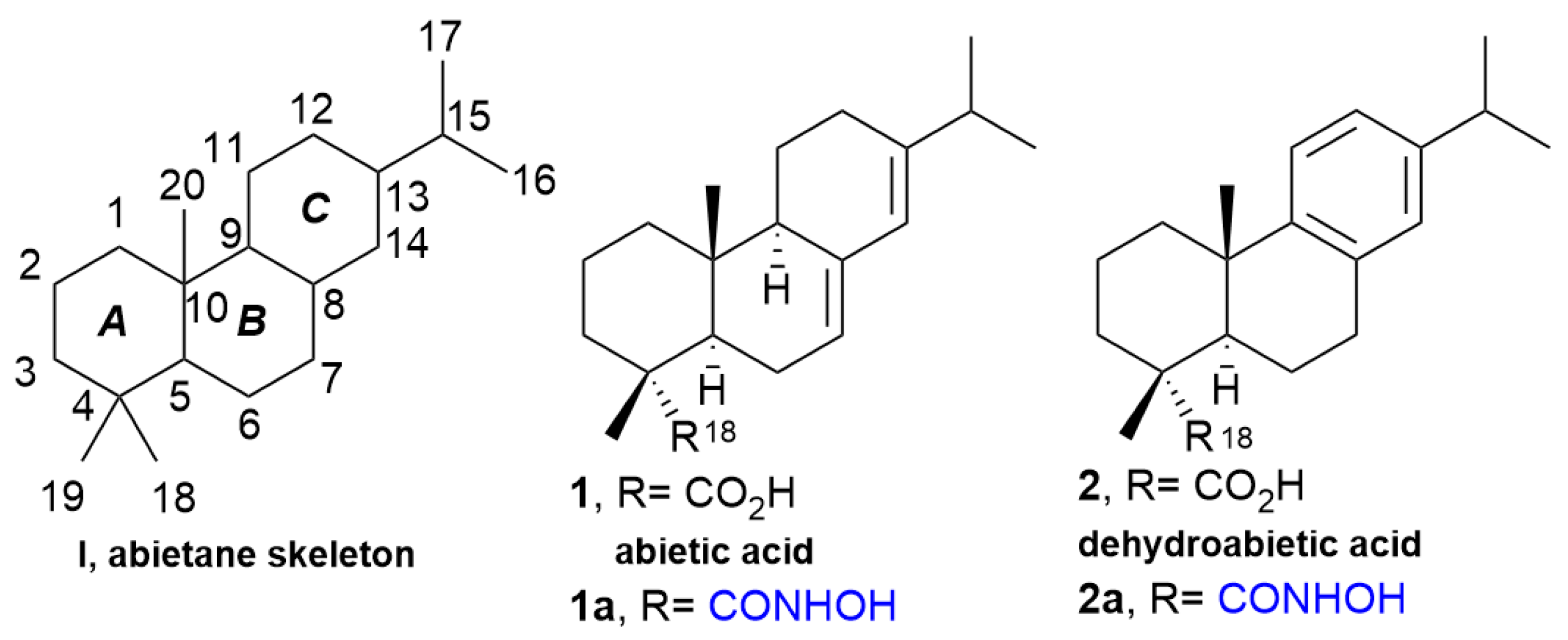
2. Results and Discussion
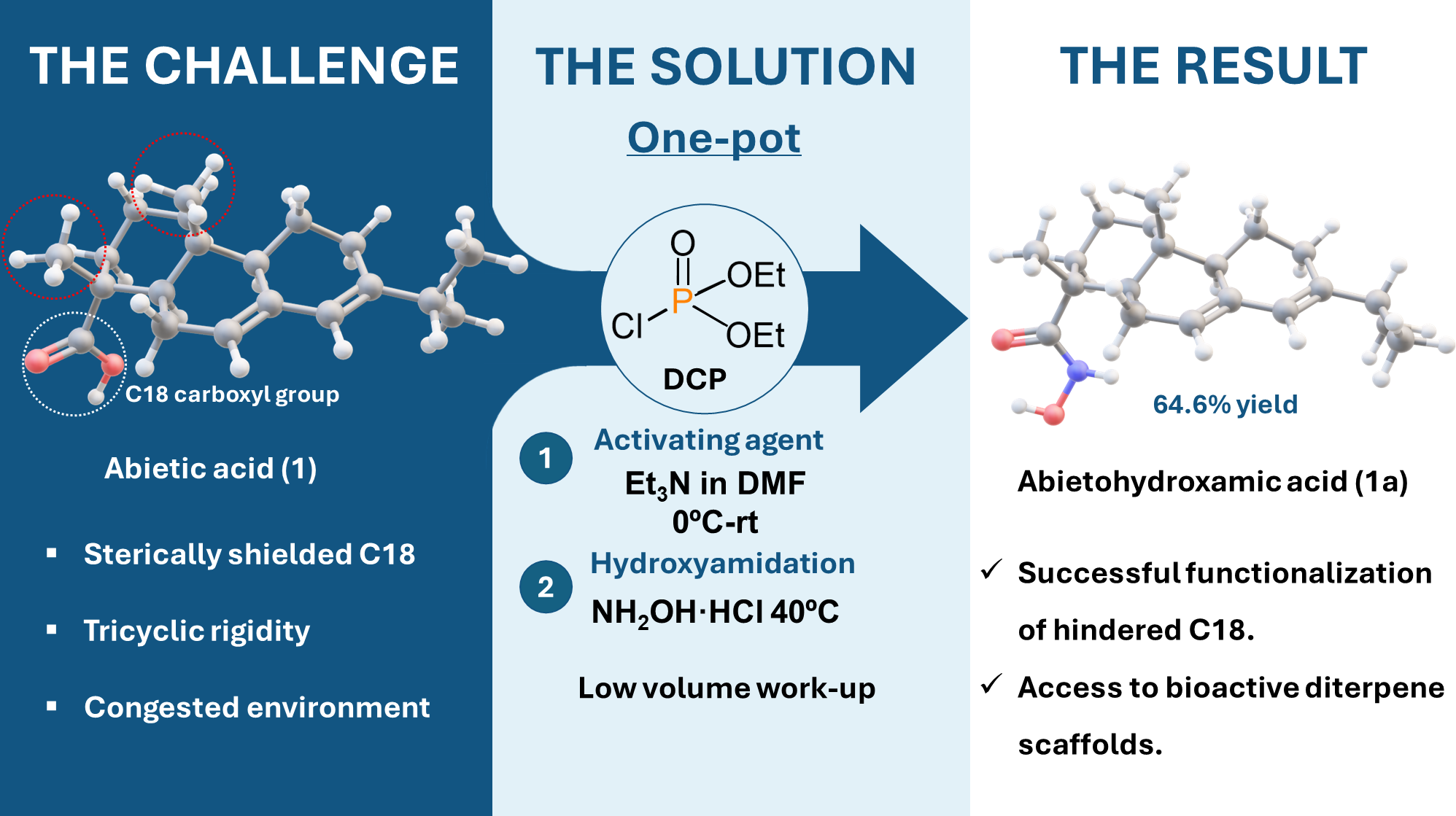
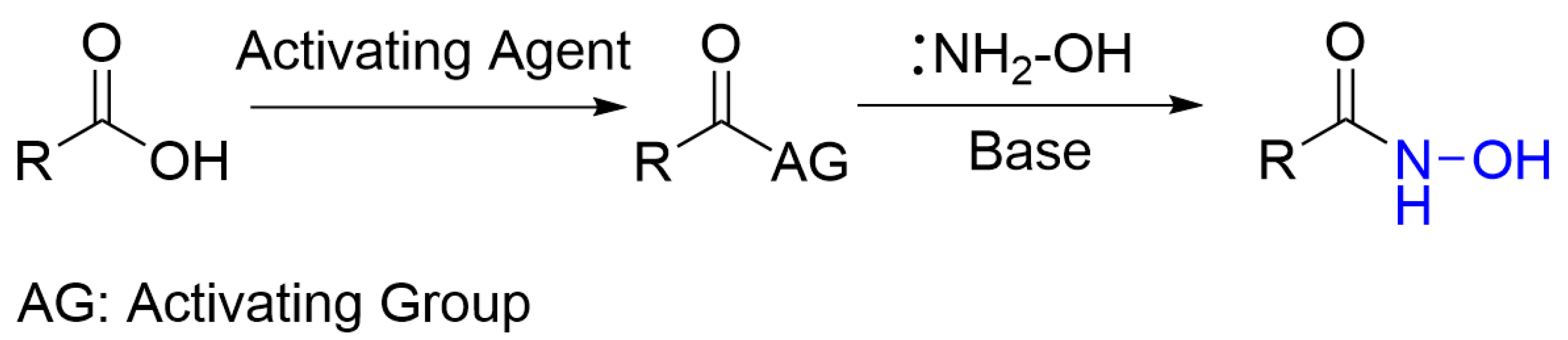
2.1. Screening of Activation Methods and Mechanistic Discovery
2.2. Optimization One-Pot Synthesis
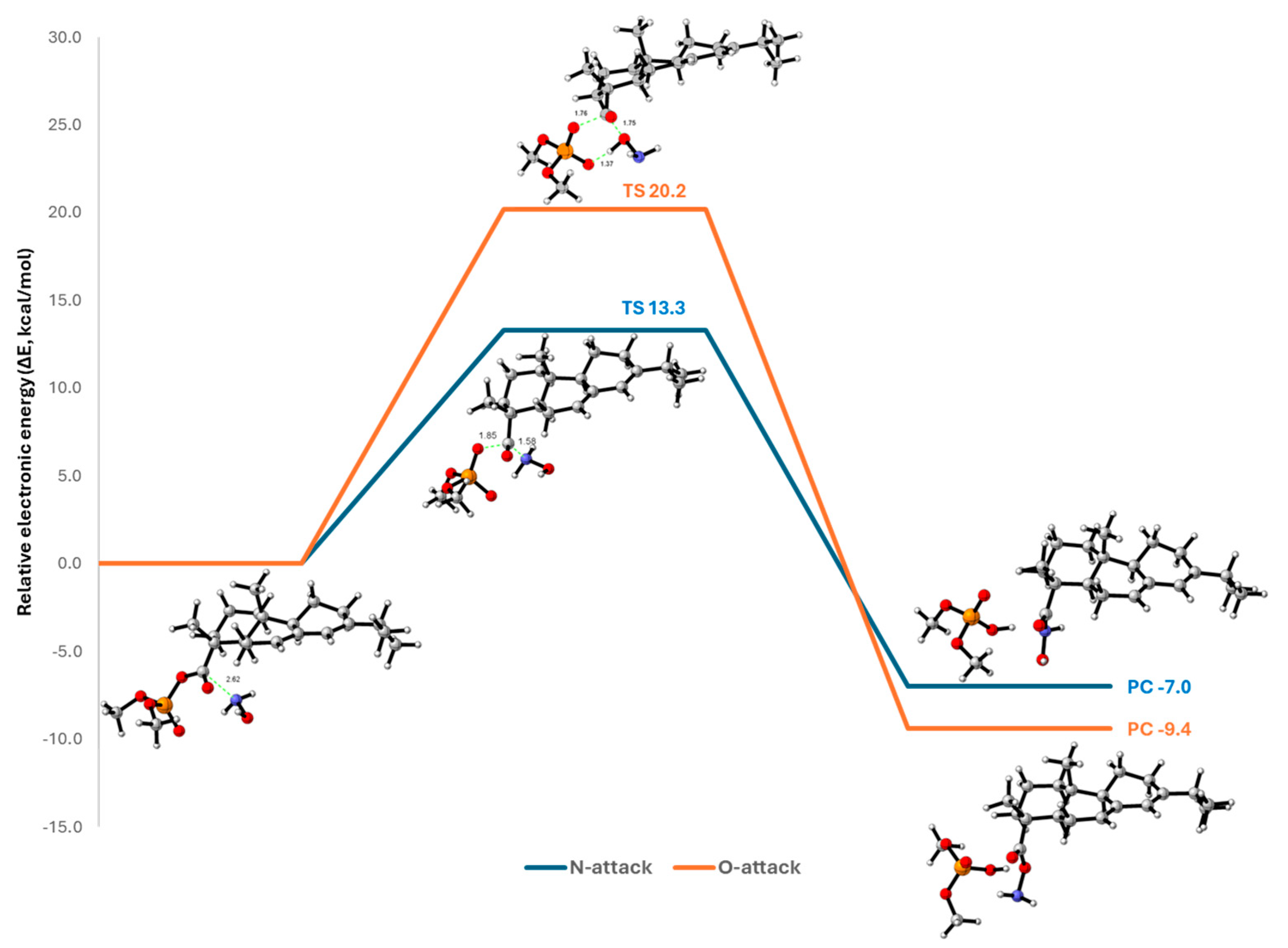
2.3. Mechanistic Rationalization via DFT Studies
3. Materials and Methods
3.1. Materials and Equipment
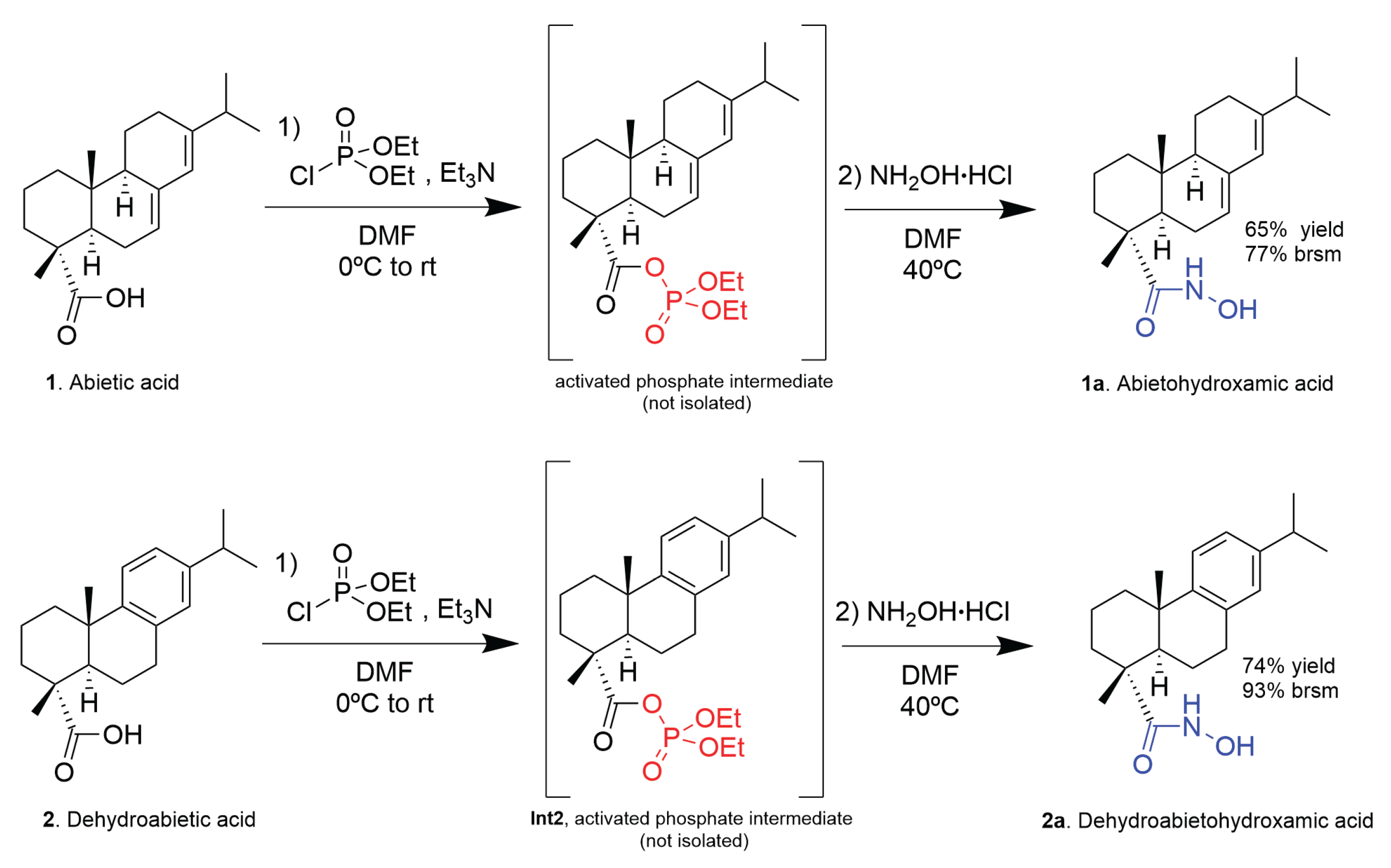
3.2. General Procedure for the Synthesis of N-Hydroxy-abieta-7,13-dien-18-amide (1a, Abietohydroxamic Acid)
3.3. Synthesis of N-Hydroxy-abieta-8,11,13-trien-18-amide (2a, Dehydroabietohydroxamic Acid)
3.4. Isolation and Characterization of the Diethyl Phosphate Mixed Anhydride Intermediate of Dehydroabietic Acid (Int2)
3.5. Computational Methods
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data and sample Availability Statement
Acknowledgments
Conflicts of Interest
References
- Talapatra, S.K.; Talapatra, B. Diterpenoids (C20). In Chemistry of Plant Natural Products; Springer Berlin Heidelberg: Berlin, Heidelberg, 2015; pp. 469–510. [CrossRef]
- Sargazifar, Z.; Ghorbanian Charami, D.; Esmaeilzadeh Kashi, M.; Asili, J.; Shakeri, A. Abietane-Type Diterpenoids: Insights into Structural Diversity and Therapeutic Potential. Chemistry & Biodiversity 2024, 21, e202400808. [CrossRef]
- González, M.A. Aromatic Abietane Diterpenoids: Total Syntheses and Synthetic Studies. Tetrahedron 2015, 71, 1883–1908. [CrossRef]
- Antoniuk, O.; Maranha, A.; Salvador, J.A.R.; Empadinhas, N.; Moreira, V.M. Bi- and Tricyclic Diterpenoids: Landmarks from a Decade (2013–2023) in Search of Leads against Infectious Diseases. Nat. Prod. Rep. 2024, 41, 1858–1894. [CrossRef]
- Hamulić, D.; Stadler, M.; Hering, S.; Padrón, J.M.; Bassett, R.; Rivas, F.; Loza-Mejía, M.A.; Dea-Ayuela, M.A.; González-Cardenete, M.A. Synthesis and Biological Studies of (+)-Liquiditerpenoic Acid A (Abietopinoic Acid) and Representative Analogues: SAR Studies. J. Nat. Prod. 2019, 82, 823–831. [CrossRef]
- Citarella, A.; Moi, D.; Pinzi, L.; Bonanni, D.; Rastelli, G. Hydroxamic Acid Derivatives: From Synthetic Strategies to Medicinal Chemistry Applications. ACS Omega 2021, 6, 21843–21849. [CrossRef]
- Al Shaer, D.; Al Musaimi, O.; De La Torre, B.G.; Albericio, F. Hydroxamate Siderophores: Natural Occurrence, Chemical Synthesis, Iron Binding Affinity and Use as Trojan Horses against Pathogens. Eur. J. Med. Chem. 2020, 208, 112791. [CrossRef]
- Stanetty, C.; Czollner, L.; Koller, I.; Shah, P.; Gaware, R.; Cunha, T.D.; Odermatt, A.; Jordis, U.; Kosma, P.; Claßen-Houben, D. Synthesis of Novel 3-Amino and 29-Hydroxamic Acid Derivatives of Glycyrrhetinic Acid as Selective 11β-Hydroxysteroid Dehydrogenase 2 Inhibitors. Bioorg. Med. Chem. 2010, 18, 7522–7541. [CrossRef]
- Wiemann, J.; Heller, L.; Perl, V.; Kluge, R.; Ströhl, D.; Csuk, R. Betulinic Acid Derived Hydroxamates and Betulin Derived Carbamates Are Interesting Scaffolds for the Synthesis of Novel Cytotoxic Compounds. Eur. J. Med. Chem. 2015, 106, 194–210. [CrossRef]
- Wiemann, J.; Heller, L.; Csuk, R. Targeting Cancer Cells with Oleanolic and Ursolic Acid Derived Hydroxamates. Bioorg. Med. Chem. Lett. 2016, 26, 907–909. [CrossRef]
- Bardyshev, I.I. Diterpenoid Carboxylic Acid Anhydrides of the Abietane, Pimarane, and Isopimarane Series. Russ. J. Org. Chem. 1999, 35, 41–55.
- Ganeshpurkar, A.; Kumar, D.; Singh, S.K. Strategies for the Synthesis of Hydroxamic Acids. Curr. Org. Synth. 2018, 15, 154–165. [CrossRef]
- Alam, M.A. Methods for Hydroxamic Acid Synthesis. Curr. Org. Chem. 2019, 23, 978–993. [CrossRef]
- Mendoza-Hernández, W.E.; Zaragozá, R.J.; González-Cardenete, M.A. Study and Development on the Hydroxamation of Natural Resinic Acids: Synthesis and Computational Studies. In Proceedings of the ECSOC 2025; MDPI, November 12 2025; p. 81. [CrossRef]
- Ech-Chahad, A.; Minassi, A.; Berton, L.; Appendino, G. An Expeditious Hydroxyamidation of Carboxylic Acids. Tetrahedron Lett. 2005, 46, 5113–5115. [CrossRef]
- Harusawa, S.; Shioiri, T. Diethyl Phosphorocyanidate (DEPC): A Versatile Reagent for Organic Synthesis. Tetrahedron 2016, 72, 8125–8200. [CrossRef]
- Halbrook, N.J.; Lawrence, R.V. The Isolation of Dehydroabietic Acid from Disproportionated Rosin. J. Org. Chem. 1966, 31, 4246–4247. [CrossRef]
| Entry | Substrate | Reaction conditions | Results |
|---|---|---|---|
| 1 | 1 | PPAA, Et3N, NH2OH· HCl in MeCN rt | SM recovered |
| 2 | 2 | PPAA, Et3N, NH2OH· HCl in MeCN rt | 20.1% yield of 2a |
| 3 | 1 | DEPC, Et3N, NH2OH/Et3N in THF rt-40 °C | 13.7% yield of 1a |
| 4 | 2 | DEPC, Et3N, NH2OH/Et3N in THF rt | Phosphate intermediate isolated (Int2) |
| 5 | Int2 | NH2OH· HCl, Et3N in DMF | Confirmed formation of 2a |
| 6 | 1 | 1) DCP, Et3N 0 °C-rt 2) NH2OH· HCl 40 °C in DMF one pot |
17.3% yield of 1a |
| 7 | 2 | 1) DCP, Et3N 0 °C-rt 2) NH2OH· HCl rt in DMF one pot |
34.2% yield of 2a |
| 81 | 1 | 1) DCP, Et3N 0 °C-rt 2) NH2OH· HCl 40 °C in DMF one pot |
64.6% yield of 1a |
| 91 | 2 | 1)DCP, Et3N 0 °C-rt 2)NH2OH· HCl 40 °C in DMF one pot |
73.8% yield of 2a |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




