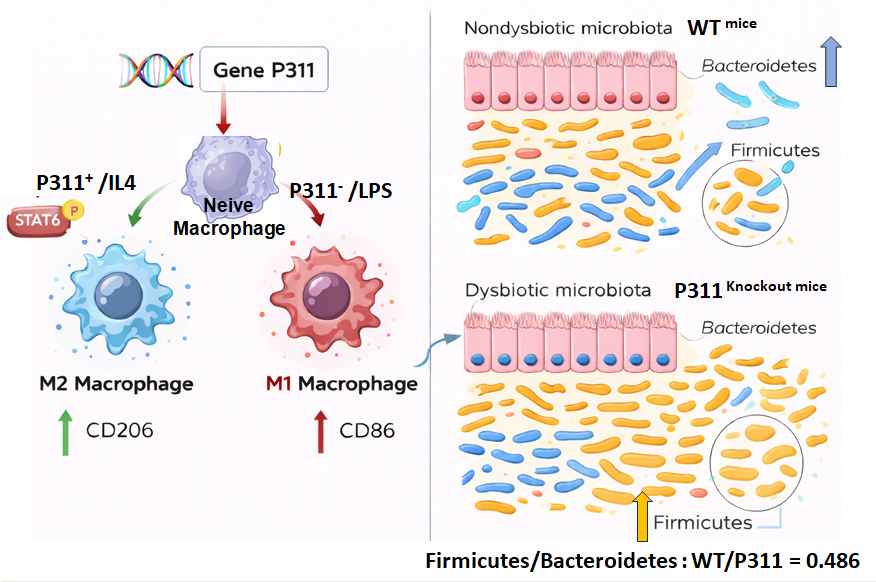
Adipose tissue is a heterogenous organ with newly identified endocrine function, consisting of immune cells along with main populations of fat storing adipocytes and adipose precursor cells. Dysregulated immune cell function and infiltration cause low grade chronic inflammation in adipose tissue that leads to various metabolic disorders like obesity, insulin resistance, type2 diabetes and cardiovascular disease. Our research showed the role of P311 protein in adipogenesis, amoeboid migration, vascular wall homeostasis, and blood pressure regulation. Studies from our laboratory and other labs showed the potential involvement of P311 in wound healing and predicted immune function. Here we studied the role of P311 on inflammation mainly focusing on macrophage phenotypes and functions as macrophages are predominant immune cells in adipose tissue that switch the inflammatory micro-environment between pro- and anti-inflammatory conditions. For the first time, we show the expression of P311 in macrophages implicating its role in inflammation directly. Further, P311 expression in macrophages induced anti-inflammatory phenotype (M2 macrophages) through phosphorylation of STAT6 of canonical JAK/STAT signaling pathway.
The human gastrointestinal (GI) system harbors high populations of both healthier and pathogenic microbial communities that provide immunity, inflammation, nutrients, and GI tract epithelial homeostasis. Given the roles of P311 in macrophage mediated inflammation and metabolic diseases, we verified whether lack of P311 in P311 knockout mice has any effects on GI microbiome compared to wildtype mice. Our studies demonstrate that lack of P311 led to changes in the intestinal microbial strains. Together, current studies implicate a larger role of P311 connecting inflammation and microbiome with obesity.