Submitted:
21 April 2026
Posted:
22 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design and Ethics
2.2. Patient Inclusion and Exclusion Criteria
2.3. Melatonin Sampling Protocol
2.4. Microbiome Analysis
2.5. Neuroimaging and Cognitive Assessment
2.6. Statistical Analysis
3. Results
3.1. Participant Characteristics
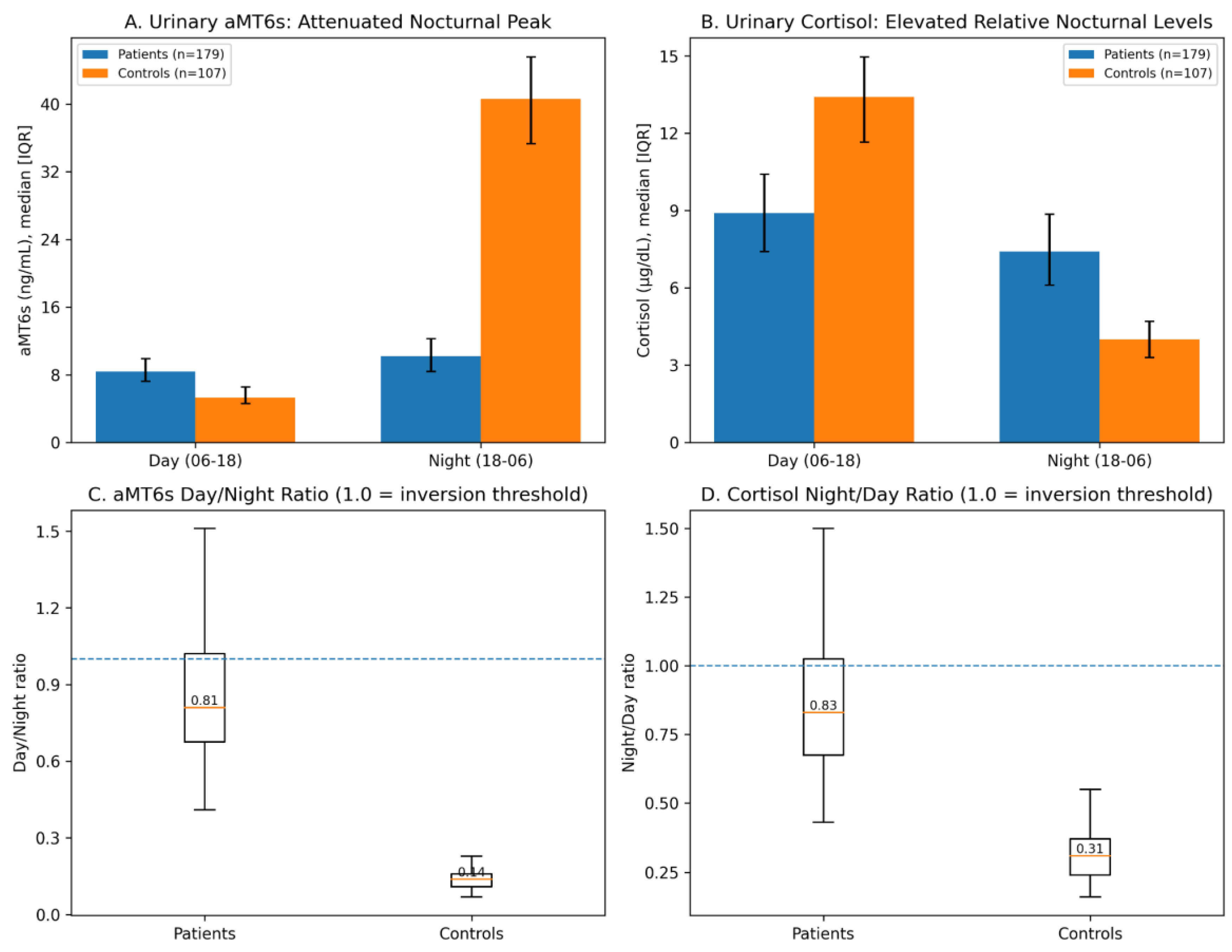
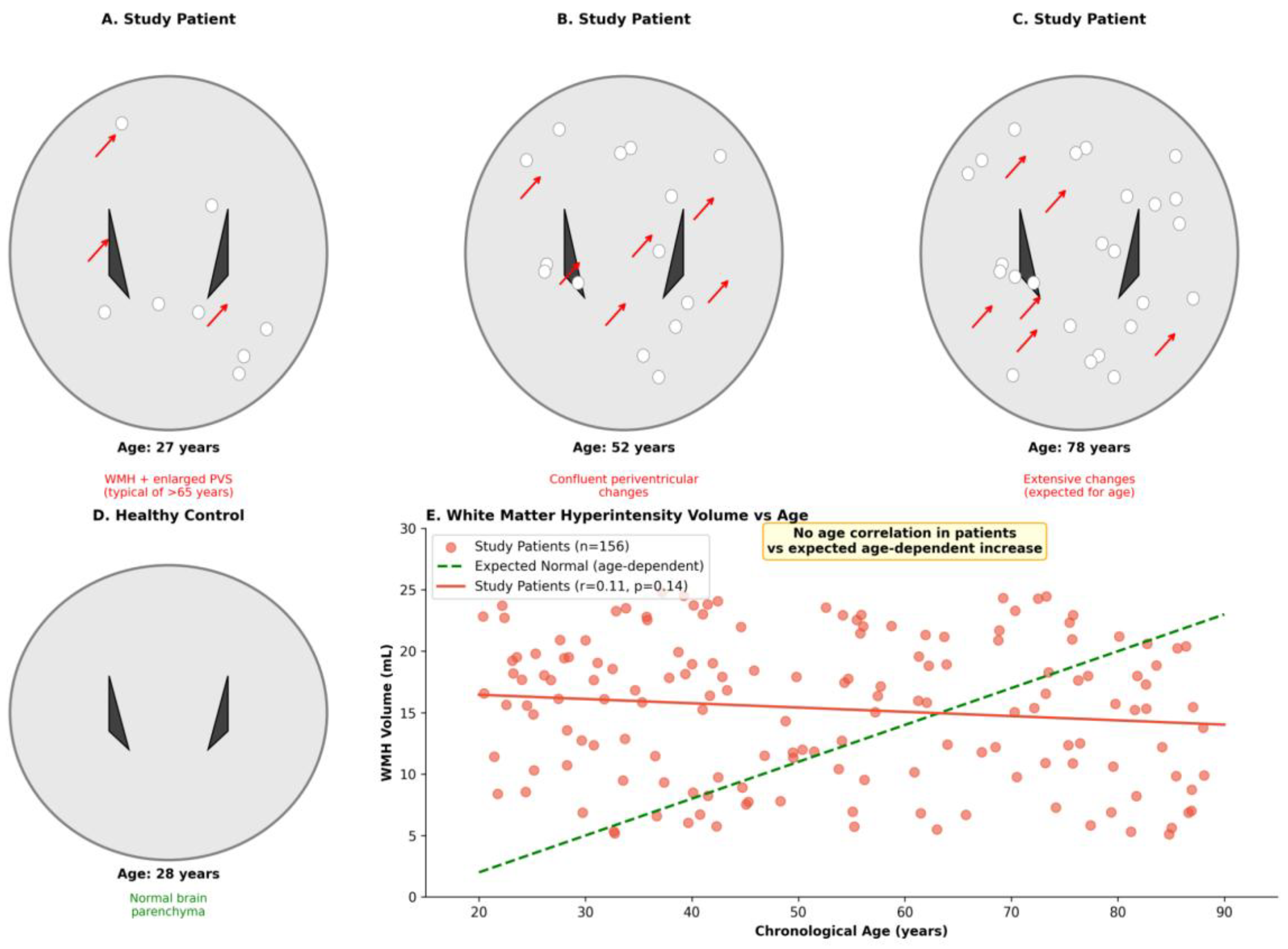
3.2. Circadian Endocrine Findings—Melatonin Amplitude Inversion
| Marker | Patients (median [IQR]) | Controls (median [IQR]) | p-value |
|---|---|---|---|
| Urinary aMT6s (24 h), ng/mL | 19.2 (14.0–27.1) | 46.6 (39.0–52.0) | <0.0001 |
| Urinary aMT6s (day 06:00–18:00), ng/mL | 8.4 (7.2–9.9) | 5.3 (4.6–6.5) | <0.0001 |
| Urinary aMT6s (night 18:00–06:00), ng/mL | 10.2 (8.4–12.2) | 40.6 (35.3–45.5) | <0.0001 |
| Urinary aMT6s Day/Night ratio | 0.81 (0.68–1.02) | 0.14 (0.11–0.16) | <0.0001 |
| Urinary cortisol (day 06:00–18:00), µg/dL | 8.9 (7.4–10.4) | 13.4 (11.6–14.9) | <0.0001 |
| Urinary cortisol (night 18:00–06:00), µg/dL | 7.4 (6.1–8.9) | 4.0 (3.3–4.7) | <0.0001 |
| Urinary cortisol Night/Day ratio | 0.83 (0.68–1.02) | 0.31 (0.24–0.37) | <0.0001 |
| Plasma melatonin (day 14:00–16:00), pg/mL | 6.1 (5.0–7.3) | 2.8 (1.9–3.7) | <0.0001 |
| Plasma melatonin (night 02:00–04:00), pg/mL | 12.7 (10.1–15.2) | 54.4 (47.2–62.5) | <0.0001 |
| Plasma cortisol (day 14:00–16:00), µg/dL | 11.9 (9.9–13.4) | 15.1 (12.8–17.6) | <0.0001 |
| Plasma cortisol (night 02:00–04:00), µg/dL | 6.5 (5.3–7.7) | 4.1 (3.2–4.8) | <0.0001 |
| Microbiome Shannon diversity index | 2.83 (2.27–3.38) | 5.07 (4.61–5.41) | <0.0001 |
| Firmicutes/Bacteroidetes ratio | 0.58 (0.47–0.75) | 2.02 (1.81–2.16) | <0.0001 |
| Gordonibacter (relative abundance, %) | 0.090 (0.050–0.120) | 0.540 (0.440–0.630) | <0.0001 |
| Ellagibacter (relative abundance, %) | 0.040 (0.030–0.060) | 0.320 (0.250–0.410) | <0.0001 |
| Plasma urolithin A, ng/mL | 2.40 (1.60–3.10) | 25.20 (22.65–27.25) | <0.0001 |
3.3. Microbiome and Metabolic Markers
3.4. Systemic Stress, Immune, and Neuroimaging Markers
| Marker | Patients (median [IQR]) | Controls (median [IQR]) | p-value |
|---|---|---|---|
| BNP, pg/mL | 294 (193–362) | 55 (38–70) | <0.0001 |
| CD4/CD8 ratio | 0.81 (0.48–1.20) | 1.77 (1.51–2.10) | <0.0001 |
| Perivascular spaces score (0–3) | 2 (2–3) | 0 (0–1) | <0.0001 |
| WMH volume, mL | 9.99 (8.09–12.22) | 1.54 (1.15–2.08) | <0.0001 |
| DTI-ALPS index | 1.05 (0.86–1.25) | 1.70 (1.64–1.79) | <0.0001 |
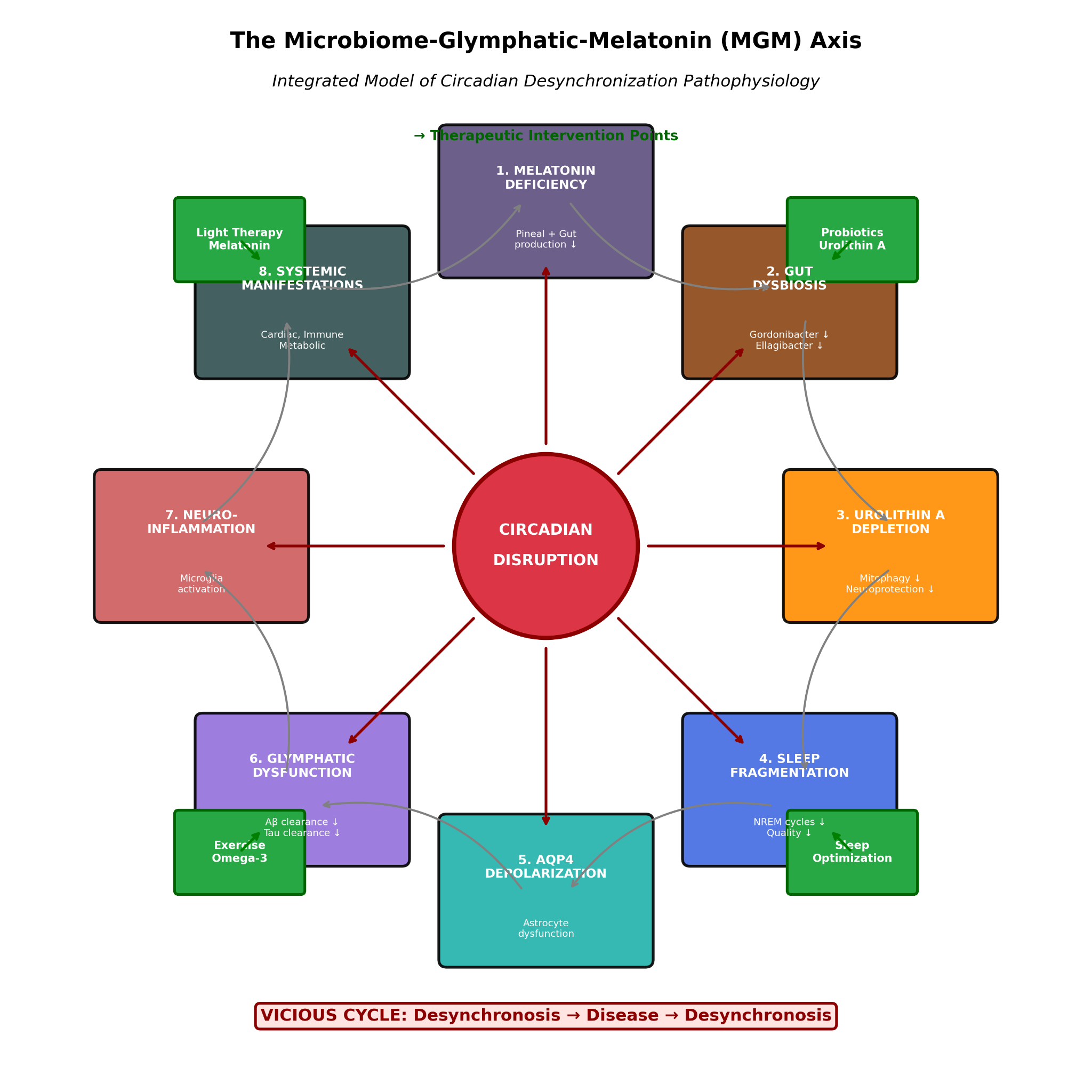
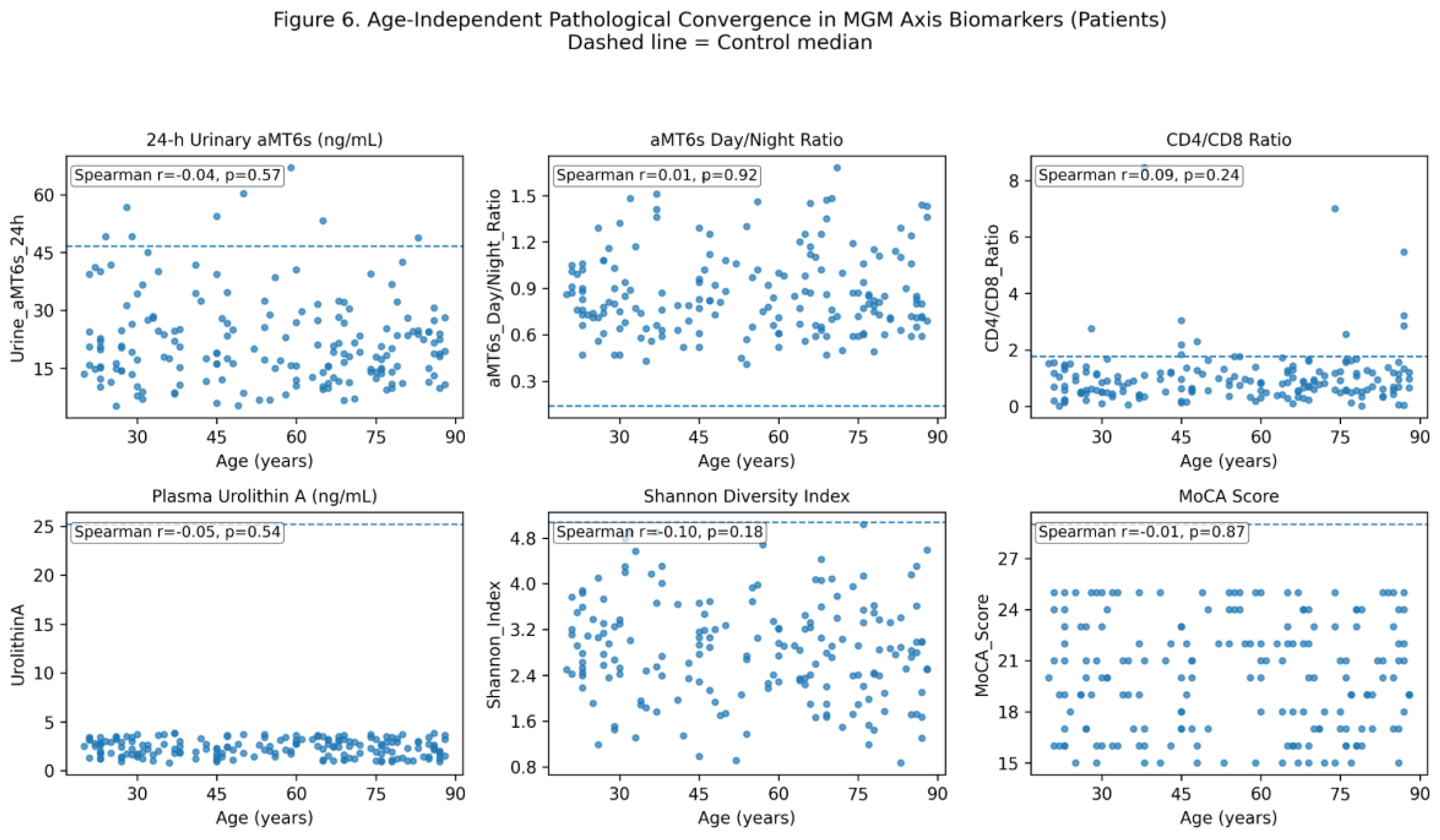
3.5. Age-Independent Convergence of MGM-Axis Biomarkers
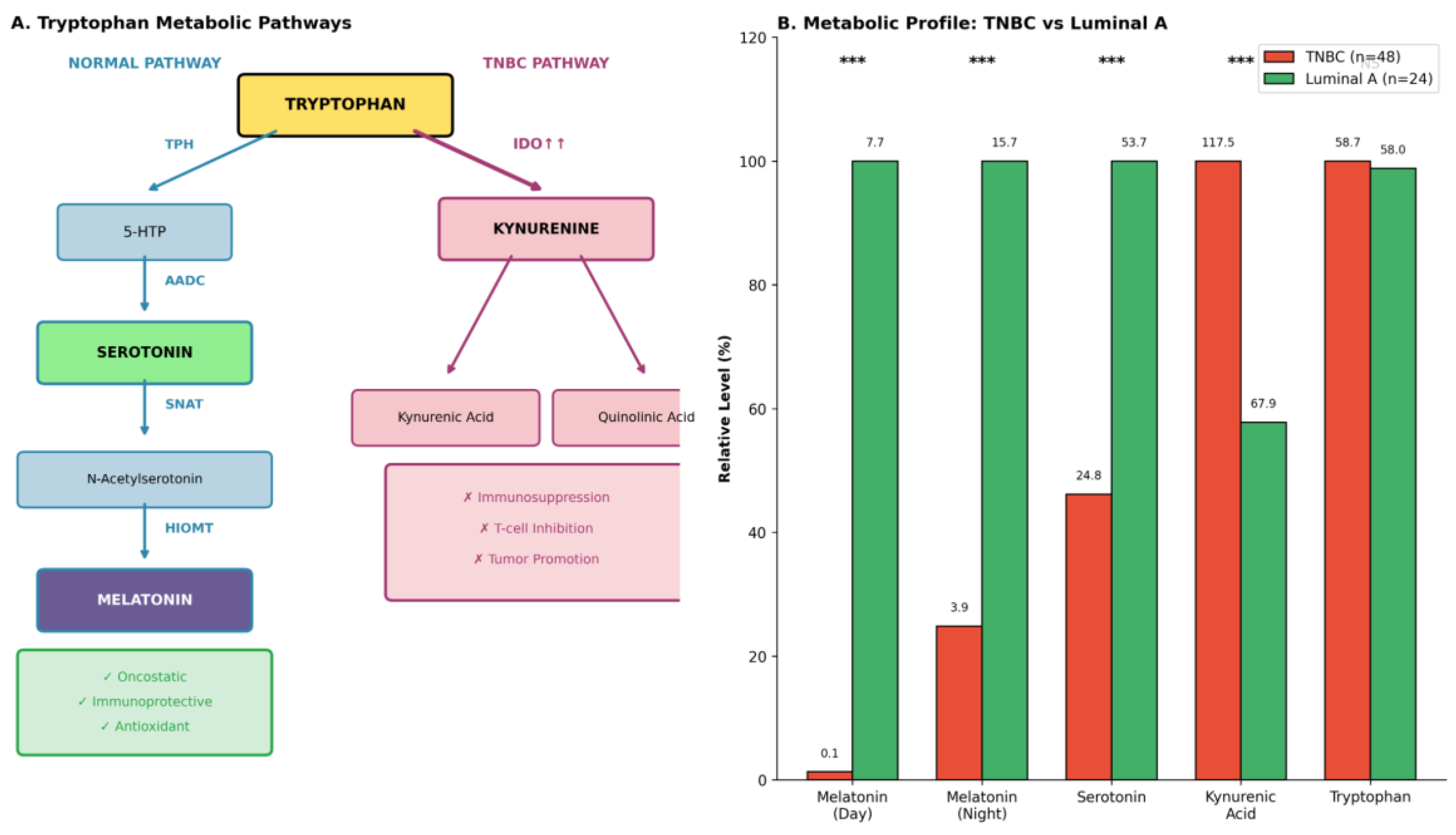
3.6. Translational Context: Tumour Metabolism and Chronotherapy
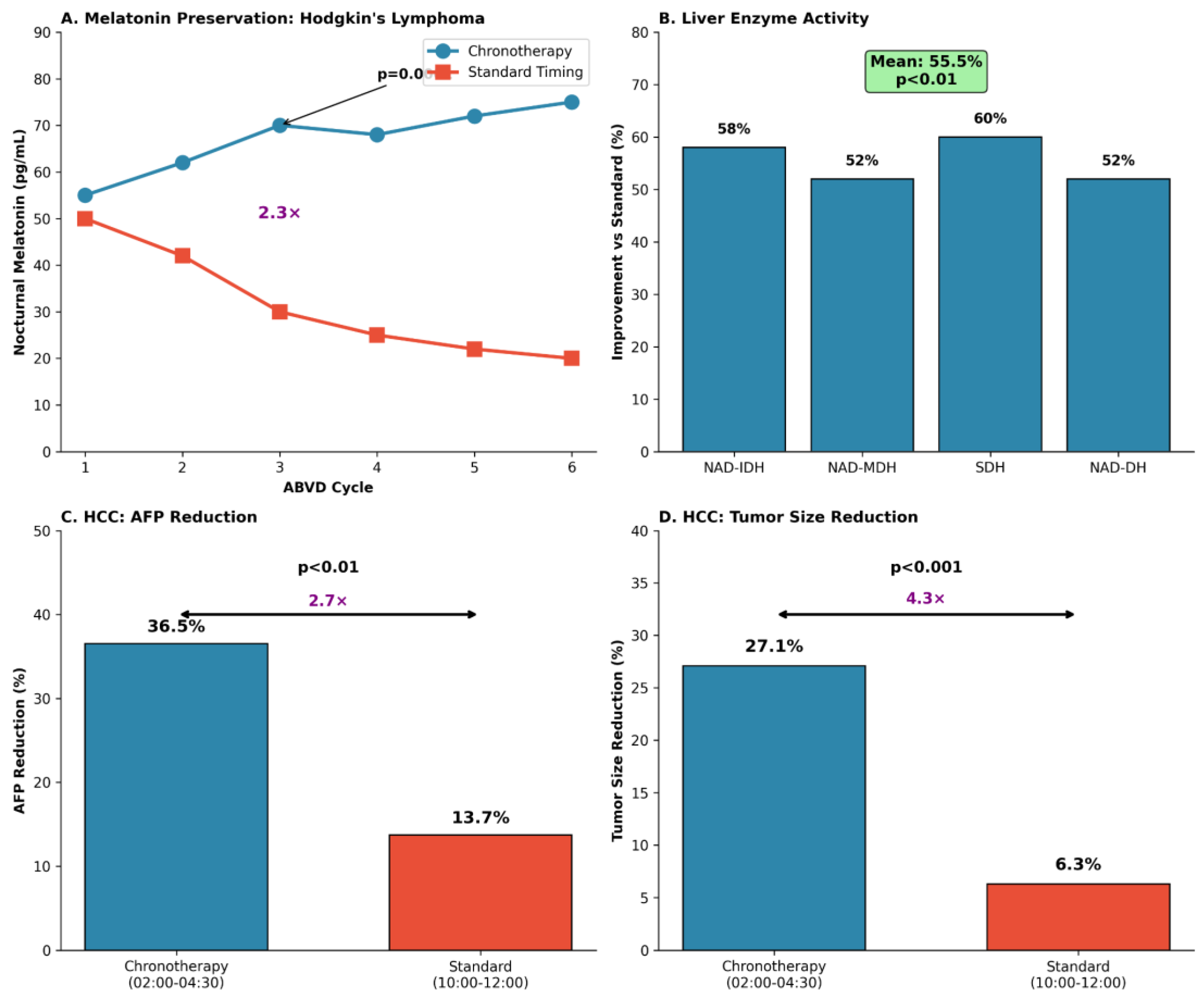
| Cohort / Study | Comparison (n) | Key outcomes |
|---|---|---|
| Breast cancer metabolic profile | TNBC (n=48) vs Luminal A (n=24) | Melatonin day: 0.1 vs 7.7; melatonin night: 3.9 vs 15.7; serotonin: 24.8 vs 53.7; kynurenic acid: 117.5 vs 67.9 (p<0.001 all). |
| Hodgkin lymphoma chronotherapy | Chronotherapy vs standard timing (n=38) | Nocturnal melatonin preserved across 6 ABVD cycles; Cycle 3: 70 vs 30 pg/mL (≈2.3×; p=0.0001). |
| Hodgkin lymphoma (enzyme activity) | Chronotherapy vs standard timing | Liver enzyme activity improvement vs standard: mean 55.5% (p<0.01). |
| Hepatocellular carcinoma | Cisplatin 02:00–04:30 vs 10:00–12:00 | AFP reduction: 36.5% vs 13.7% (p<0.01); tumor size reduction: 27.1% vs 6.3% (p<0.001). |
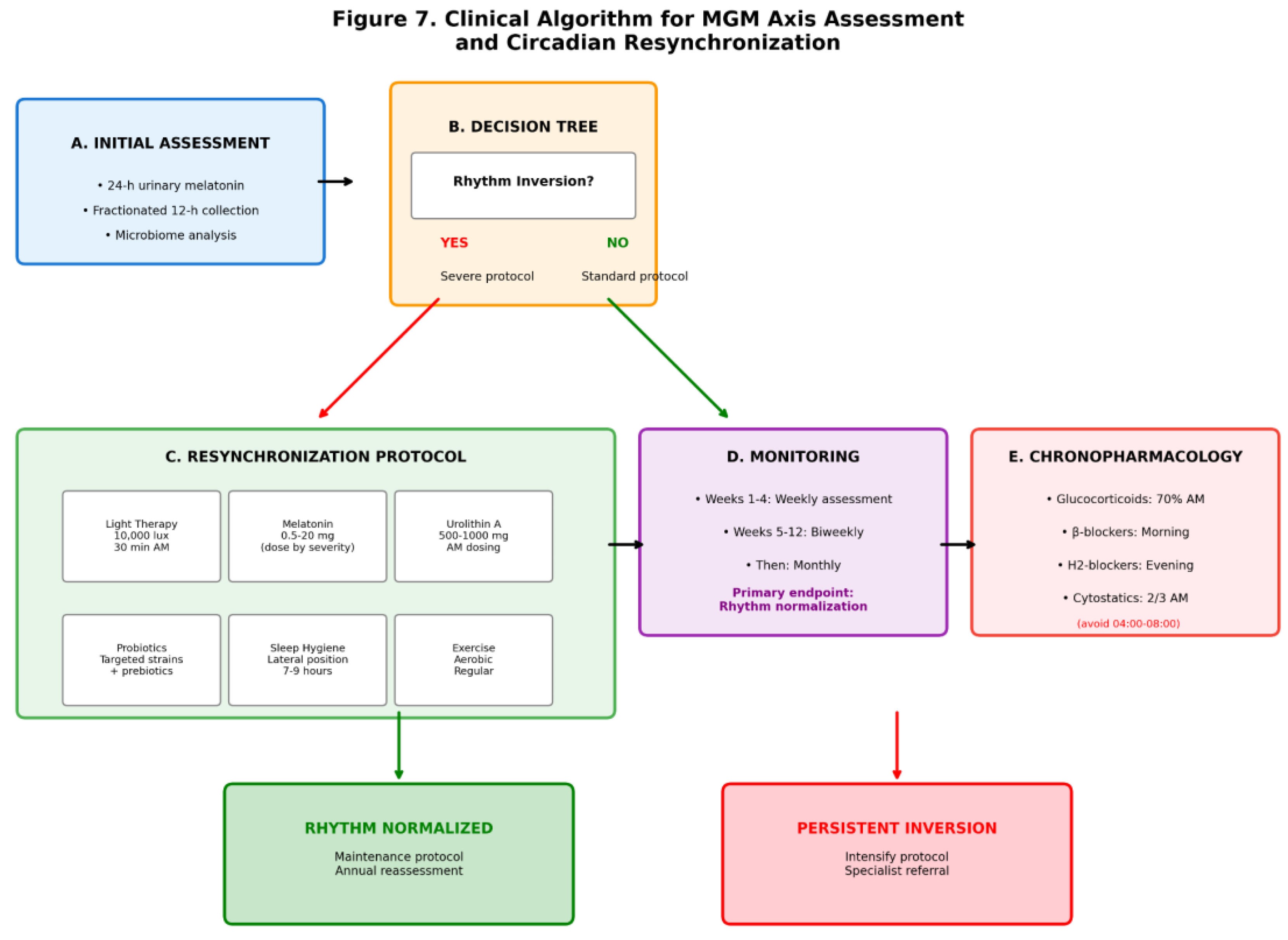
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Reppert SM, Weaver DR. Coordination of circadian timing in mammals. Nature. 2002;418(6901):935–941. [CrossRef]
- Takahashi JS. Transcriptional architecture of the mammalian circadian clock. Nat Rev Genet. 2017;18(3):164–179. [CrossRef]
- Hastings MH, Maywood ES, Brancaccio M. Generation of circadian rhythms in the suprachiasmatic nucleus. Nat Rev Neurosci. 2018;19(8):453–469. [CrossRef]
- Stevens RG, Hansen J, Costa G, et al. Considerations of circadian impact for defining 'shift work' in cancer studies. Occup Environ Med. 2011;68(2):154–162.
- Bass J, Lazar MA. Circadian time signatures of fitness and disease. Science. 2016;354(6315):994–999. [CrossRef]
- Panda S. Circadian physiology of metabolism. Science. 2016;354(6315):1008–1015.
- Sulli G, Lam MTY, Bhaskara S. Interplay between circadian clock and cancer: New frontiers for cancer treatment. Trends Cancer. 2019;5(8):475–494. [CrossRef]
- Dubocovich ML, Delagrange P, Krause DN, et al. International Union of Basic and Clinical Pharmacology. LXXV. Nomenclature, classification, and pharmacology of G protein-coupled melatonin receptors. Pharmacol Rev. 2010;62(3):343–380. [CrossRef]
- Liu C, Weaver DR, Jin X, et al. Molecular dissection of two distinct actions of melatonin on the suprachiasmatic circadian clock. Neuron. 1997;19(1):91–102. [CrossRef]
- Reiter RJ, Tan DX, Rosales-Corral S, Manchester LC. The universal nature, unequal distribution and antioxidant functions of melatonin and its derivatives. Mini Rev Med Chem. 2013;13(3):373–384.
- Reiter RJ, Rosales-Corral S, Tan DX, et al. Melatonin, a full service anti-cancer agent: inhibition of initiation, progression and metastasis. Int J Mol Sci. 2017;18(4):843. [CrossRef]
- Hill SM, Belancio VP, Dauchy RT, et al. Melatonin: an inhibitor of breast cancer. Endocr Relat Cancer. 2015;22(3):R183–R204.
- Chen CQ, Fichna J, Bashashati M, et al. Distribution, function and physiological role of melatonin in the lower gut. World J Gastroenterol. 2011;17(34):3888–3898. [CrossRef]
- Iliff JJ, Wang M, Liao Y, et al. A paravascular pathway facilitates CSF flow through the brain parenchyma and the clearance of interstitial solutes. Sci Transl Med. 2012;4(147):147ra111.
- Xie L, Kang H, Xu Q, et al. Sleep drives metabolite clearance from the adult brain. Science. 2013;342(6156):373–377.
- Jessen NA, Munk AS, Lundgaard I, Nedergaard M. The glymphatic system: a beginners guide. Neurochem Res. 2015;40(12):2583–2599. [CrossRef]
- Nedergaard M, Goldman SA. Glymphatic failure as a final common pathway to dementia. Science. 2020;370(6512):50–56. [CrossRef]
- Hablitz LM, Plá V, Giannetto M, et al. Circadian control of brain glymphatic and lymphatic fluid flow. Nat Commun. 2020;11(1):4411.
- Mestre H, Tithof J, Du T, et al. Flow of cerebrospinal fluid is driven by arterial pulsations and is reduced in hypertension. Nat Commun. 2018;9(1):4878. [CrossRef]
- Wardlaw JM, Benveniste H, Nedergaard M, Zlokovic BV, Mestre H, Lee H, et al. Perivascular spaces in the brain: anatomy, physiology and pathology. Nat Rev Neurol. 2020;16(3):137–153. [CrossRef]
- Taoka T, Masutani Y, Kawai H, et al. Evaluation of glymphatic system activity with the diffusion MR technique: diffusion tensor image analysis along the perivascular space (DTI-ALPS) in Alzheimer's disease cases. Jpn J Radiol. 2017;35(4):172–178.
- Thaiss CA, Zeevi D, Levy M, et al. Transkingdom control of microbiota diurnal oscillations promotes metabolic homeostasis. Cell. 2014;159(3):514–529. [CrossRef]
- Ryu D, Mouchiroud L, Andreux PA, et al. Urolithin A induces mitophagy and prolongs lifespan in C. elegans and increases muscle function in rodents. Nat Med. 2016;22(8):879–888. [CrossRef]
- Selma MV, González-Sarrías A, Salas-Salvadó J, et al. The gut microbiota metabolism of pomegranate or walnut ellagitannins yields two urolithin-metabotypes that correlate with cardiometabolic risk biomarkers. Mol Nutr Food Res. 2018;62(3):1800066. [CrossRef]
- Innominato PF, Lévi FA, Bjarnason GA. Chronotherapy and the molecular clock: Clinical implications in oncology. Adv Drug Deliv Rev. 2010;62(9-10):979–1001. [CrossRef]
- Potter GM, Doubal FN, Jackson CA, Chappell FM, Sudlow CL, Dennis MS, Wardlaw JM. Cerebral perivascular spaces visible on magnetic resonance imaging: development of a qualitative rating scale and its observer reliability. Cerebrovasc Dis. 2015;39:224–231.
- Tavartkiladze A, et al. Chronotherapy in hematological malignancies: preservation of nocturnal melatonin during ABVD chemotherapy. [Internal cohort data, 2019a].
| Characteristic | Patients (n=179) | Controls (n=107) | p-value |
|---|---|---|---|
| Age, years (median [IQR]) | 57 (34–74) | 47 (35–59) | 0.003 |
| Sex, female n (%) | 87 (48.6) | 55 (51.4) | 0.737 |
| Sex, male n (%) | 92 (51.4) | 52 (48.6) | |
| BMI, kg/m² (median [IQR]) | 28.0 (24.8–30.5) | 23.2 (21.8–24.9) | <0.0001 |
| MoCA score (median [IQR]) | 20 (17–23) | 29 (28–30) | <0.0001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




