Submitted:
20 April 2026
Posted:
22 April 2026
You are already at the latest version
Abstract
Keywords:
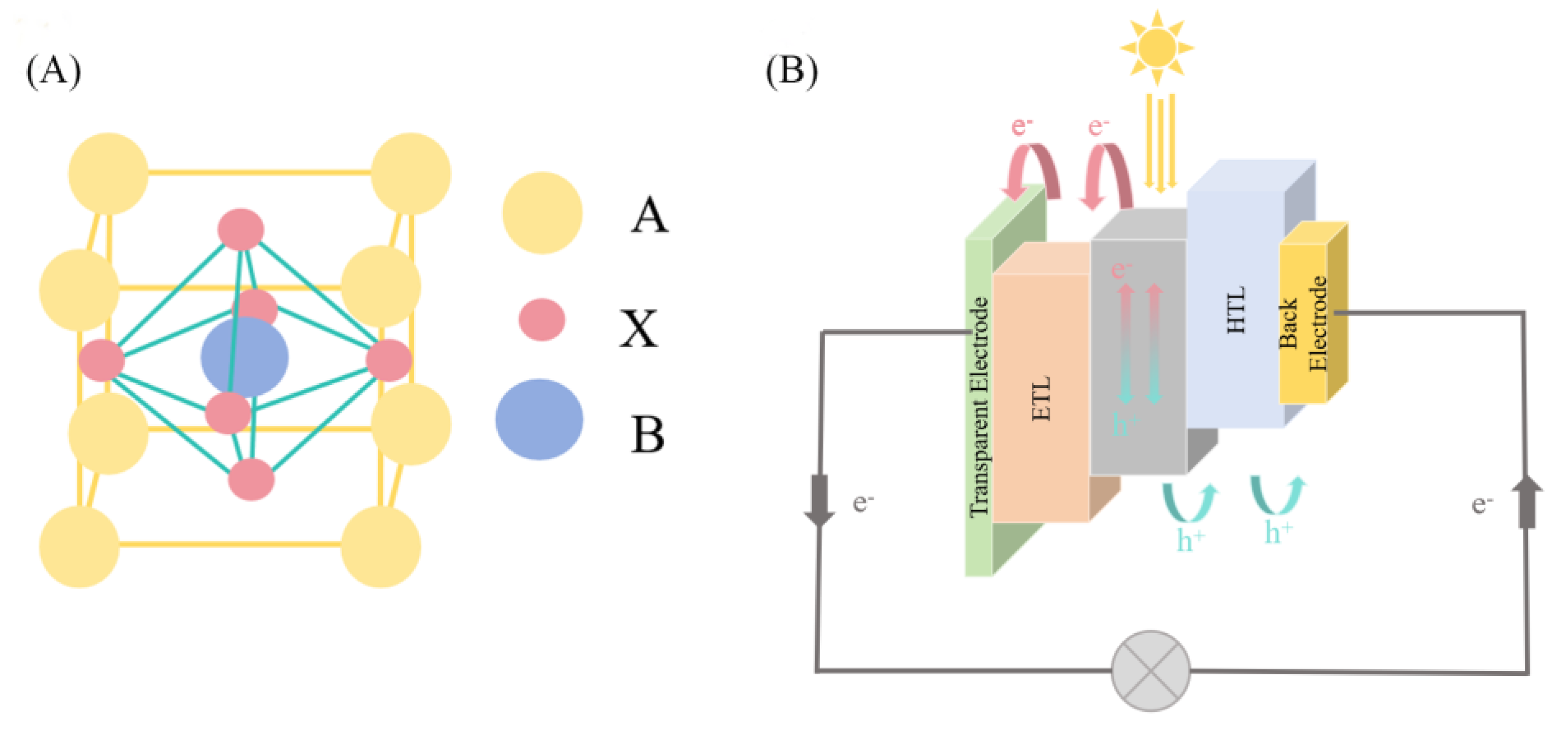
1. Introduction
2. Materials and Methods
3. Carbon nanomaterials
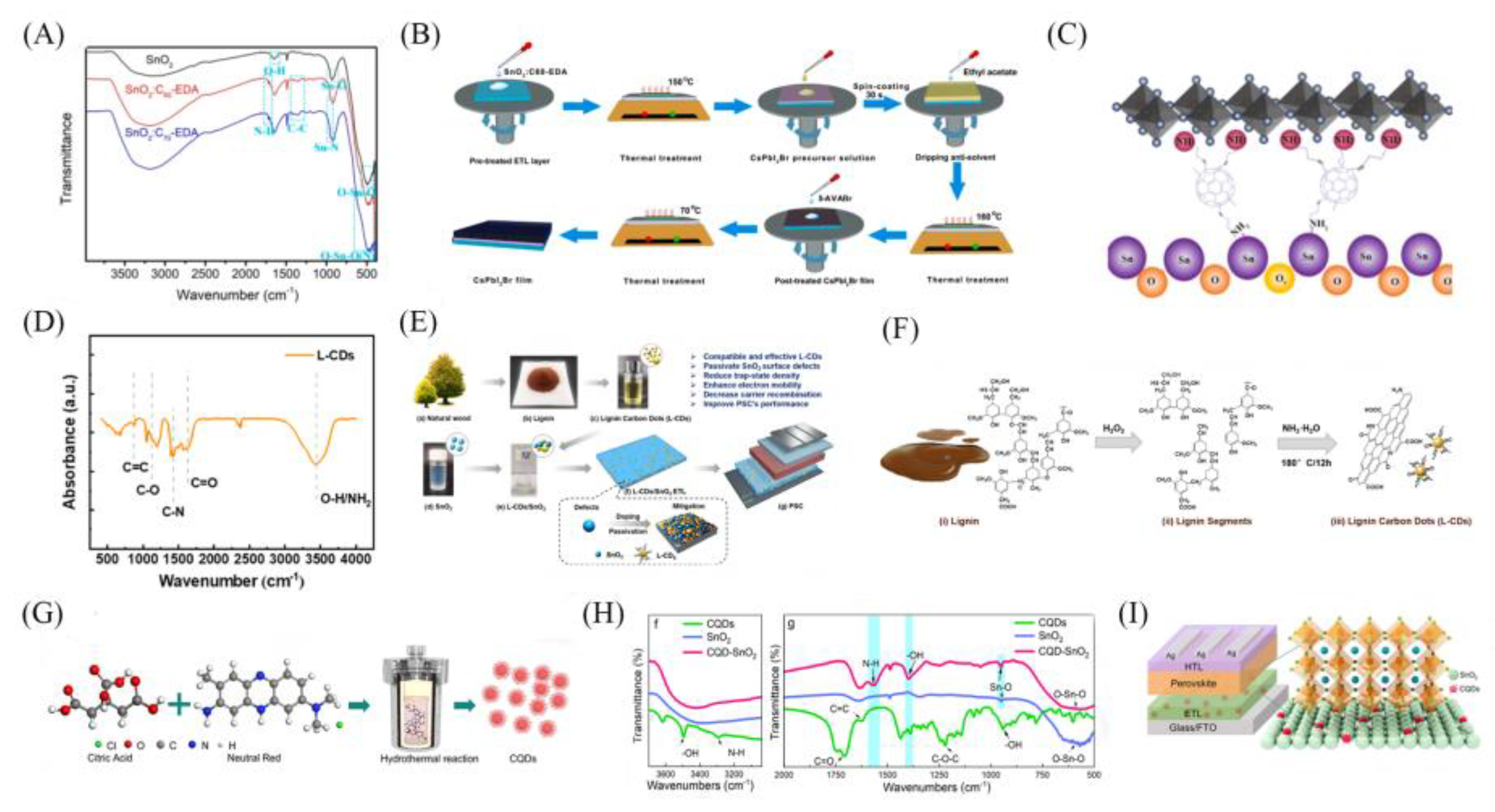
3.1. Common Carbon Materials Applied in Perovskite Solar Cells
3.2. Common Methods for Preparing Carbon Materials
3.2.1. Solid-Liquid Reaction
3.2.2. Hydrothermal Method
3.2.3. Hummer's Method
3.2.4. Microwave-assisted Method
3.2.5. Solvthermal Process
3.2.6. Electrospinning
3. Modifying PSCs with Carbon Materials
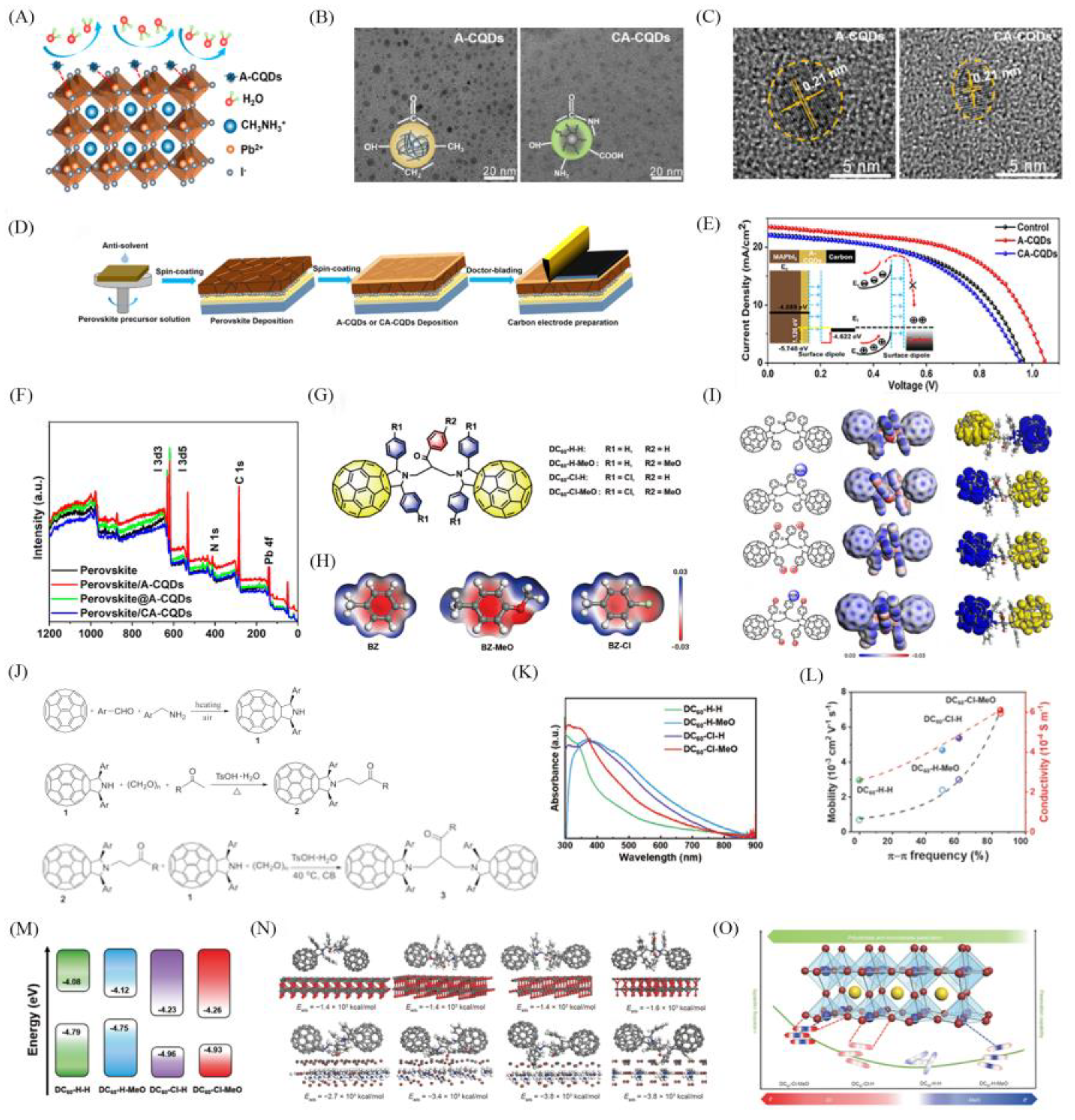
4.1. Modify the Charge Transport Layer
4.3. Carbon Materials as Surface Modifiers
3. Summary and Prospectives
3. Appendix Table
| Full name | Abbreviation |
|---|---|
| Perovskite solar cells | PSCs |
| Photoelectric conversion efficiency | PCE |
| Carbon nanotubes | CNTs |
| Electron transport layers | ETL |
| Hole transport layers | HTL |
| Silver | Ag |
| Gold | Au |
| Methylammonium | MA+ |
| Formamidinium | FA+ |
| Rubidium | Rb+ |
| Lead | Pb2+ |
| Chloride | Cl- |
| Bromide | Br- |
| Iodide | I- |
| 0-dimensional | 0D |
| 1-dimensional | 1D |
| 2-dimensional | 2D |
| 3-dimensional | 3D |
| Carbon quantum dot | CQD |
| Carbon nanofiber | CNF |
| Lithium | Li |
| Copper | Cu |
| External Quantum Efficiency | EQE |
| Time-Resolved Photoluminescence Spectroscopy | TRPL |
| Graphene quantum dots | GQDs |
| Fullerene Indanones | TBu-FIDO |
| Carbon dots | CDs |
| Multi-Walled Carbon Nanotube | MWCNT |
| Lignin carbon dots | L-CDs |
| Black phosphor quantum dots | BPQDs |
| Fourier transform infrared spectra | FTIR |
| CQDs containing bromine functional groups | Br-CQDs |
| Carbon nanodots | CNDs |
| Graphene Oxide | GO |
| Brominated graphene oxide | Br-GO |
| Nanodot | ND |
| Imidazole bromide functionalized graphene quantum dots | I-GQDs |
| Lead formamidine iodide | FAPbI3 |
| N, N-methylenebisacrylamide-functionalized CQDs | MBA/CQDs |
| Carbon quanta with functional groups | A-CQDs |
| X-ray Photoelectron Spectroscopy | XPS |
| Photoluminescence Spectroscopy | PL |
| Scanning electron microscope | SEM |
| Transmission Electron Microscope | TEM |
| Density Functional Theory | DFT |
| Single-Walled Carbon Nanotube | SWCNT |
| Hydrochloric acid | HCl |
| Sodium hydroxide | NaOH |
| 5-aminopentanoic acid hydrobromide | 5-AVABr |
| Citric acid | CA |
| Neutral red | NR |
| Infrared Spectroscopy | IR |
| X-ray Diffraction | XRD |
7. Patents
Author Contributions
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhang, Y.; Yu, B.; Wei, X.; Yu, H. Using post-treatment additives for crystal modulation and interface passivation enables the fabrication of efficient and stable perovskite solar cells in air. Adv. Energy Mater. 2024, 15, 2402990. [Google Scholar] [CrossRef]
- Dong, Y.; Gao, Y.; Luo, D.; Huang, P.; Liu, S.; Ren, M.; Duan, J.; Li, Y.; Zhu, R.; Jia, Y. Suppression of ion migration and improvement of electronic properties for CsPbI₂Br via organic amine substitution. Sol. Energy 2024, 275, 112639. [Google Scholar] [CrossRef]
- Bi, D.; Li, X.; Yi, C.; Décoppet, J.-D.; Zakeeruddin, S.M.; Hagfeldt, A.; Grätzel, M.; Luo, J. A vacuum flash–assisted solution process for high-efficiency large-area perovskite solar cells. Science 2016, 353, 58. [Google Scholar]
- Doherty, T.A.S.; Nagane, S.; Kubicki, D.J.; Jung, Y.-K.; Johnstone, D.N.; Iqbal, A.N.; Guo, D.; Frohna, K.; Danaie, M.; Tennyson, E.M.; Macpherson, S.; Abfalterer, A.; Anaya, M.; Chiang, Y.-H.; Crout, P.; Ruggeri, F.S.; Collins, S.M.; Grey, C.P.; Walsh, A.; Midgley, P.A.; Stranks, S.D. Stabilized tilted-octahedra halide perovskites inhibit local formation of performance-limiting phases. Science 2021, 374, 1598–1605. [Google Scholar] [CrossRef]
- Kagan, C.R.; Mitzi, D.B.; Dimitrakopoulos, C.D. Organic-inorganic hybrid materials as semiconducting channels in thin-film field-effect transistors. Science 1999, 286, 945–947. [Google Scholar] [CrossRef]
- Akihiro, K.; Shirai, Y.; Miyasaka, T. Organometal halide perovskites as visible-light sensitizers for photovoltaic cells. J. Am. Chem. Soc. 2009, 131, 6050–6051. [Google Scholar] [CrossRef]
- Li, Y.; Liao, J.-F.; Pan, H.; Xing, G. Interfacial engineering for high-performance PTAA-based inverted 3D perovskite solar cells. Solar RRL 2022, 6, 2200647. [Google Scholar] [CrossRef]
- Chen, H.; Ye, F.; Tang, W.; He, J.; Yin, M.; Wang, Y.; Xie, F.; Bi, E.; Yang, X.; Grätzel, M.; Han, L. A solvent- and vacuum-free route to large-area perovskite films for efficient solar modules. Nature 2017, 550, 92–95. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Gao, Y.; Guo, J.; Wu, M. Synthesis of VS₄ and VO on carbon nanofibers for back interface modification in HTL free CsPbBr₃-based perovskite solar cells. Carbon 2024, 230, 119566. [Google Scholar] [CrossRef]
- Huang, X.; Chen, R.; Deng, G.; Han, F.; Ruan, P.; Cheng, F.; Yin, J.; Wu, B.; Zheng, N. Methylamine-dimer-induced phase transition toward MAPbI₃ films and high-efficiency perovskite solar modules. J. Am. Chem. Soc. 2020, 142, 6149–6157. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Guo, J.; Li, Z.; Han, W.; Ren, G.; Liu, C.; Shen, L.; Guo, W. Incorporating self-assembled silane-crosslinked carbon dots into perovskite solar cells to improve efficiency and stability. J. Mater. Chem. A 2020, 8, 5629–5637. [Google Scholar] [CrossRef]
- Shi, Y.; Zhang, L.; Hu, S.; Wang, X.; Han, J.; Huang, J.; Chen, J.; Zhang, Y.; Zhang, X.; He, J.; Zuo, H.; Ju, J.; Wu, Z.; Zhao, W.; Zeng, Y.; Zou, Y.; Liao, K.; Yang, R.; Ye, W.; Gu, Y.; Gong, L.; Fan, S.; Peng, Z.; Chen, J. Bridging the buried interface with conjugated molecule for highly efficient carbon-based inorganic CsPbI₂Br perovskite solar cells fabricated in air. Chem. Eng. J. 2024, 492, 152210. [Google Scholar] [CrossRef]
- Shin, S.; Seo, S.; Jeong, S.; Sharbirin, A.S.; Kim, J.; Ahn, H.; Park, N.G.; Shin, H. Kinetic-controlled crystallization of α-FAPbI₃ inducing preferred crystallographic orientation enhances photovoltaic performance. Adv. Sci. 2023, 10, 2300798. [Google Scholar] [CrossRef]
- Xiong, Z.; Chen, X.; Zhang, B.; Odunmbaku, G.O.; Ou, Z.; Guo, B.; Yang, K.; Kan, Z.; Lu, S.; Chen, S.; Ouedraogo, N.A.N.; Cho, Y.; Yang, C.; Chen, J.; Sun, K. Simultaneous interfacial modification and crystallization control by biguanide hydrochloride for stable perovskite solar cells with PCE of 24.4%. Adv. Mater. 2022, 34, 2106118. [Google Scholar] [CrossRef]
- Xu, W.; Gao, Y.; Ming, W.; He, F.; Li, J.; Zhu, X.-H.; Kang, F.; Li, J.; Wei, G. Suppressing defects-induced nonradiative recombination for efficient perovskite solar cells through green antisolvent engineering. Adv. Mater. 2020, 32, 2003965. [Google Scholar] [CrossRef] [PubMed]
- Seo, S.; Jeon, I.; Xiang, R.; Lee, C.; Zhang, H.; Tanaka, T.; Lee, J.-W.; Suh, D.; Ogamoto, T.; Nishikubo, R.; Saeki, A.; Chiashi, S.; Shiomi, J.; Kataura, H.; Lee, H.M.; Yang, Y.; Matsuo, Y.; Maruyama, S. Semiconducting carbon nanotubes as crystal growth templates and grain bridges in perovskite solar cells. J. Mater. Chem. A 2019, 7, 12987–12992. [Google Scholar] [CrossRef]
- Xu, Z.; Gong, Y.; Wang, J.; Ma, Z.; Yu, R.; Yang, J.; Liu, Y.; Guo, Q.; Zhou, E.; Tan, Z. Carbon nanofibers fabricated via electrospinning to guide crystalline orientation for stable perovskite solar cells with efficiency over 24%. Chem. Eng. J. 2023, 453, 139961. [Google Scholar] [CrossRef]
- Huo, B.; Jia, S.; Li, Y.; Zhang, H.; Hao, C.; Wang, H.; Hu, S. Unveiling the effect of carbon dots on the TiO₂-involved electron transport in perovskite solar cells. J. Alloys Compd. 2023, 951, 169958. [Google Scholar] [CrossRef]
- Chen, H.; Zheng, X.; Li, Q.; Yang, Y.; Xiao, S.; Hu, C.; Bai, Y.; Zhang, T.; Wong, K.S.; Yang, S. An amorphous precursor route to the conformable oriented crystallization of CH₃NH₃PbBr₃ in mesoporous scaffolds: toward efficient and thermally stable carbon-based perovskite solar cells. J. Mater. Chem. A 2016, 4, 12897–12912. [Google Scholar] [CrossRef]
- Yang, G.; Liu, H.; Li, X.; Liu, C.; Li, Z.; Li, X. Carboxylated carbon nanotubes as efficient additive to enhance the performance of different types of perovskite solar cells. Surf. Interfaces 2023, 38, 102768. [Google Scholar] [CrossRef]
- Wang, M.; Sun, H.; Meng, L.; Wang, M.; Li, L. A universal strategy of intermolecular exchange to stabilize α-fapbi3 and manage crystal orientation for high-performance humid-air-processed perovskite solar cells. Adv. Mater. 2022, 34, 200041–200051. [Google Scholar] [CrossRef]
- Rahighi, R.; Bakhshayesh, A.; Nezamoddinykachooye, N.; Hosseini, S.M.; Heydari, M.; Gholipour, S. Sequential spin-coating method in enhancing crystal morphology of ambient air-processed perovskite solar cells. Appl. Phys. A 2023, 130, 604–617. [Google Scholar] [CrossRef]
- Li, R.; Liu, X.; Chen, J. opportunities and challenges of hole transport materials for high-performance inverted hybrid-perovskite solar cells. Exploration 2023, 3, 20220027. [Google Scholar] [CrossRef]
- Suo, J.; Yang, B.; Mosconi, E.; Bogachuk, D.; Doherty, T.A.S.; Frohna, K.; Kubicki, D.J.; Fu, F.; Kim, Y.; Er-Raji, O.; Zhang, T.; Baldinelli, L.; Wagner, L.; Tiwari, A.N.; Gao, F.; Hinsch, A.; Stranks, S.D.; Angelis, F.D.; Hagfeldt, A. Multifunctional sulfonium-based treatment for perovskite solar cells with less than 1% efficiency loss over 4,500-h operational stability tests. Nat. Energy 2024, 9, 172–183. [Google Scholar] [CrossRef]
- Chen, M.; Kapil, G.; Li, Y.; Kamarudin, M.A.; Baranwal, A.K.; Nishimura, K.; Sahamir, S.R.; Sanehira, Y.; Li, H.; Ding, C.; Zhang, Z.; Shen, Q.; Hayase, S. Large synergy effects of doping, a site substitution, and surface passivation in wide bandgap Pb-free ASnI2Br perovskite solar cells on efficiency and stability enhancement. J. Power Sources 2022, 520, 230848. [Google Scholar] [CrossRef]
- McAndrews, G.R.; Guo, B.; Kaczaral, S.C.; Fukuda, K.; Poma, M.R.S.; Belisle, R.A.; Amassian, A.; McGehee, M.D. Moisture uptake relaxes stress in metal halide perovskites at the expense of stability. ACS Energy Letters 2024, 9, 4153–4161. [Google Scholar] [CrossRef]
- Guo, S.; Fan, B.; Yao, S.; Rao, L.; Zhang, S.; Hu, X.; Chen, Y. The effect of interfacial humidity on the printing of highly reproducible perovskite solar cells in the air. Adv. Funct. Mater. 2024, 34, 2313715–2313724. [Google Scholar] [CrossRef]
- Xu, M.; Liu, Y.; Yang, K.; Li, S.; Wang, M.; Wang, J.; Yang, D.; Shkunov, M.; Silva, S.R.P.; Castro, F.A.; Zhao, Y. Minimally invasive power sources for implantable electronics. Exploration 2023, 4, 20220106. [Google Scholar] [CrossRef]
- Alias, N.; Umar, A.A.; Sadikin, S.N.; Ridwan, J.; Hamzah, A.A.; Umar, M.I.A.; Ehsan, A.A.; Nurdin, M.; Zhan, Y. Air-processable perovskite solar cells by hexamine molecule phase stabilization. ACS Omega 2023, 8, 18874–18881. [Google Scholar] [CrossRef]
- Gao, L.; Tu, Y.; Li, R.; Liu, F.; Qiu, X.; Xu, Y.; Jiang, D.; Du, Z.; Liu, Y.; Wu, J.; Huang, M.; Lan, Z. High efficiency carbon-based CsPbI2Br solar cells achieved by bidirectional passivation of cadmium p-aminobenzoate. J. Power Sources 2024, 623, 235420. [Google Scholar] [CrossRef]
- Martinez, V.C.; Xie, H.; Mingorance, A.; Pereyra, C.; Narymany, A.; Gómez, M.M. Carbon-based perovskite solar cells by screen printing with preheating. J. Phys.: Conf. Ser. 2020, 1433, 012009. [Google Scholar] [CrossRef]
- Kim, M.; Cortecchia, D.; Borzda, T.; Mróz, W.; Leoncino, L.; Dellasega, D.; Lee, S.-H.; Petrozza, A. Coordinating solvent-assisted synthesis of phase-stable perovskite nanocrystals with high yield production for optoelectronic applications. Chem. Mater. 2021, 33, 547–553. [Google Scholar] [CrossRef]
- Zhao, W.; Guo, P.; Wu, J.; Lin, D.; Jia, N.; Fang, Z.; Liu, C.; Ye, Q.; Zou, J.; Zhou, Y.; Wang, H. TiO2 electron transport layer with p–n homojunctions for efficient and stable perovskite solar cells. Nano-Micro Lett. 2024, 16, 191. [Google Scholar] [CrossRef]
- Yun, T.; Cai, H.; Lyu, W.; Lu, X.; Gao, X.; Liu, J.-M.; Wu, S. Regulation of the buried interface to achieve efficient HTL-free all-inorganic CsPbI2Br-based perovskite solar cells. ACS Appl. Mater. Interfaces 2024, 16, 57412–57420. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Yao, D.; Dong, P.; Tang, Z.; Li, Y.; Chen, B.; Tian, N.; Zheng, G.; Peng, Y.; Long, F. Synergetic modification on buried and upper surfaces of perovskites with nitrogen-doped carbon quantum dots for efficient and stable solar cells. Sppl. Surf. Sci. 2024, 658, 159848. [Google Scholar] [CrossRef]
- Iqbal, Z.; Félix, R.; Musiienko, A.; Thiesbrummel, J.; Köbler, H.; Gutierrez-Partida, E.; Gries, T.W.; Husam, E.; Saleh, A.; Wilks, R.G.; Zhang, J.; Stolterfoht, M.; Neher, D.; Albrecht, S.; Bär, M.; Abate, A.; Wang, Q. Unveiling the potential of ambient air annealing for highly efficient inorganic CsPbI3 perovskite solar cells. J. Am. Chem. Soc. 2024, 146, 4642–4651. [Google Scholar] [CrossRef] [PubMed]
- Xie, Z.; Luo, H.; Jiang, Q.-S.; Zhao, Y.; Peng, Y.; Yuan, L.; Yan, K.; Abdi-Jalebi, M. A universal reverse-cool annealing strategy makes two-dimensional ruddlesden-popper perovskite solar cells stable and highly efficient with Voc exceeding 1.2 v. Ecomat 2024, e12501, 327327. [Google Scholar] [CrossRef]
- Liu, J.; Zhao, Z.; Qian, J.; Liang, Z.; Wu, C.; Wang, K.; Liu, S.F.; Yang, D. Thermal radiation annealing for overcoming processing temperature limitation of flexible perovskite solar cells. Adv. Mater. 2024, 36, 2401236–2401248. [Google Scholar] [CrossRef]
- Yu, Y.; Liu, R.; Liu, C.; Shi, X.-L.; Yu, H.; Chen, Z.-G. Synergetic regulation of oriented crystallization and interfacial passivation enables 19.1% efficient wide-bandgap perovskite solar cells. Adv. Energy Mater. 2022, 12, 2201509–2201518. [Google Scholar] [CrossRef]
- Yang, Y.; Huang, H.; Yan, L.; Cui, P.; Lan, Z.; Sun, C.; Du, S.; Wang, X.; Yao, C.; Qu, S.; Zhang, Q.; Wang, M.; Zhao, X.; Li, M. Compatible soft-templated deposition and surface molecular bridge construction of SnO2 enable air-fabricated perovskite solar cells with efficiency exceeding 25.7%. Adv. Energy Mater. 2024, 14, 2400416–2400425. [Google Scholar] [CrossRef]
- Wang, H.; Liu, H.; Zhu, C.; Bai, Y.; Dong, Z.; Wei, X.; Li, W.; Chen, H. Moisture is not always bad: H2O accelerates the conversion of DMAPbI3 intermediate to CsPbI3 for boosting the efficiency of carbon-based perovskite solar cells to over 16 %. Fundam. Res. 2024, 4, 1110–1117. [Google Scholar] [CrossRef] [PubMed]
- Mohammed, M.K.A.; Abualsayed, M.I.; Alshehri, A.M.; Kumar, A.; Dehghanipour, M.; Alnayli, R.S.; Aftab, S.; Akman, E. Synergistic Effects of Energy Level Alignment and Trap Passivation via 3,4-Dihydroxyphenethylamine Hydrochloride for Efficient and Air-Stable Perovskite Solar Cells. ACS Appl. Energy Mater. 2024, 7, 1358–1368. [Google Scholar] [CrossRef]
- Liu, H.; Yan, N.; Bai, H.; Kwok, R.T.K.; Tang, B.Z. Aggregation-induced emission luminogens for augmented photosynthesis. Exploration 2022, 2, 20210053. [Google Scholar] [CrossRef]
- Astakala, A.K.; Lee, S.-Y.; Gautam, J.; Thapa, K.B.; In, I.; Lee, S.J.; Park, S.-J. Engineering inorganic perovskite solar cells: Overcoming efficiency and stability barriers for next-generation photovoltaics. Adv. Powder Mater. 2025, 100354. [Google Scholar] [CrossRef]
- Asghar, U.; Qamar, M.A.; Hakami, O.; Farhan, A.; Parveen, H.; Sharma, M. Recent Advances in Carbon Nanotube Utilization in Perovskite Solar Cells: A Review. Micromachines 2024, 15, 529–564. [Google Scholar] [CrossRef] [PubMed]
- Peng, J.; Wu, Q.; Hou, H.; Hu, T.; Huang, Y.; Cai, X.; Luo, W.; Chen, X.; Yu, H. Interlayer engineering via alkaline hypophosphates for efficient and air-stable perovskite solar cells. Mater. Chem. Front. 2023, 7, 2426–2435. [Google Scholar] [CrossRef]
- Liu, P.; Sun, Y.; Wang, S.; Zhang, H.; Gong, Y.; Li, F.; Shi, Y.; Du, Y.; Li, X.; Guo, S.; Tai, Q.; Wang, C.; Zhao, X. Two dimensional graphitic carbon nitride quantum dots modified perovskite solar cells and photodetectors with high performances. J. Power Sources 2020, 451, 227825. [Google Scholar] [CrossRef]
- Berhe, T.A.; Su, W.; Chen, C.; Pan, C.; Cheng, J.; Chen, H.; Tsai, M.; Chen, L.; Dubale, A.A.; Hwang, B. Organometal halide perovskite solar cells: degradation and stability. Energy Environ Sci. 2016, 9, 323–356. [Google Scholar] [CrossRef]
- Shui, Q.; Shan, S.; Zhai, Y.; Aoyagi, S.; Izawa, S.; Huda, M.; Yu, C.; Zuo, L.; Chen, H.; Lin, H.; Matsuo, Y. Evaporable Fullerene Indanones with Controlled Amorphous Morphology as Electron Transport Layers for Inverted Perovskite Solar Cells. J. Am. Chem. Soc. 2023, 145, 27307–27315. [Google Scholar] [CrossRef]
- Wang, Z.; He, B.; Wei, M.; Liu, W.; Li, X.; Zhu, J.; Chen, H.; Tang, Q. Enhanced charge extraction enabled by amide-functionalized carbon quantum dots modifier for efficient carbon-based perovskite solar cells. Chem. Eng. J. 2024, 479, 147736. [Google Scholar] [CrossRef]
- Liu, C.; Sun, J.; Jiang, X.; Huang, L.; Lou, Q.; Cheng, Y.; Song, S.; Ge, Z. A universal tactic of using Lewis-base polymer-CNTs composites as additives for high performance cm2-sized and flexible perovskite solar cells. Sci. China Chem. 2020, 64, 281–292. [Google Scholar] [CrossRef]
- Fang, X.; Ding, J.; Yuan, N.; Sun, P.; Lv, M.; Ding, G.; Zhu, C. Graphene quantum dot incorporated perovskite films: passivating grain boundaries and facilitating electron extraction. Phys. Chem. Chem. Phys. 2017, 19, 6057–6063. [Google Scholar] [CrossRef]
- Zhou, Q.; Tang, S.; Yuan, G.; Zhu, W.; Huang, Y.; Li, S.; Lin, M. Tailored graphene quantum dots to passivate defects and accelerate charge extraction for all-inorganic CsPbIBr2 perovskite solar cells. J. Alloys Compd. 2022, 895, 162529. [Google Scholar] [CrossRef]
- Han, G.S.; Song, Y.H.; Jin, Y.U.; Lee, J.; Park, N.; Kang, B.K.; Lee, J.; Cho, I.S.; Yoon, D.H.; Jung, H.S. Reduced Graphene Oxide/Mesoporous TiO2 Nanocomposite Based Perovskite Solar Cells. ACS Appl. Mater. Interfaces 2015, 7, 23521–23526. [Google Scholar] [CrossRef]
- Li, H.; Tao, L.; Huang, F.; Sun, Q.; Zhao, X.; Han, J.; Shen, Y.; Wang, M. Enhancing Efficiency of Perovskite Solar Cells via Surface Passivation with Graphene Oxide Interlayer. ACS Appl. Mater. Interfaces 2017, 9, 38967–38976. [Google Scholar] [CrossRef]
- Xie, J.; Huang, K.; Yu, X.; Yang, Z.; Xiao, K.; Qiang, Y.; Zhu, X.; Xu, L.; Wang, P.; Cui, C.; Yang, D. Enhanced Electronic Properties of SnO2 via Electron Transfer from Graphene Quantum Dots for Efficient Perovskite Solar Cells. ACS Nano 2017, 11, 9176–9182. [Google Scholar] [CrossRef]
- Ryu, J.; Yoon, S.; Park, J.; Jeong, S.M.; Kang, D.-W. Fabrication of nickel oxide composites with carbon nanotubes for enhanced charge transport in planar perovskite solar cells. Appl. Surf. Sci. 2020, 516, 146116. [Google Scholar] [CrossRef]
- Al-gamal, A.G.; Elseman, A.M.; Chowdhury, T.H.; Kabel, K.I.; Farag, A.A.; Rabie, A.M.; El-sattar, N.E.A.A.; Islam, A. Promising Nitrogen-Doped Graphene Derivatives; A Case Study for Preparations, Fabrication Mechanisms, and Applications in Perovskite Solar Cells. Top. Curr. Chem. 2022, 6, 381–412. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.-G.; Monfreid, T.D.; Kim, J.-H.; Goubard, F.; Berry, J.J.; Zhu, K.; Bui, T.-T.; Park, N.-G. Fabrication of nickel oxide composites with carbon nanotubes for enhanced charge transport in planar perovskite solar cells. ACS Energy Letters 2023, 8, 2267–2275. [Google Scholar] [CrossRef]
- Liu, J.; Yin, Y.; He, B.; Wang, P.; Wang, M.; Cai, W.; Han, Y.; Su, Z.; Guo, J.; Cai, R.; Jin, S.; Gao, X.; Bian, J.; Shi, Y. Focusing on the bottom contact: Carbon quantum dots embedded SnO2 electron transport layer for high-performance and stable perovskite solar cells. Mater. Today Phys. 2023, 33, 101041. [Google Scholar] [CrossRef]
- Zhu, J.; He, B.; Wang, M.; Yao, X.; Huang, H.; Chen, C.; Chen, H.; Duan, Y.; Tang, Q. Elimination of defect and strain by functionalized CQDs dual-engineering for all-inorganic HTMs-free perovskite solar cells with an ultrahigh voltage of 1.651 V. Nano Energy 2022, 104, 107920. [Google Scholar] [CrossRef]
- Ma, X.; Zhang, T.; Zhao, B.; Liu, C.; Li, X.; Liu, H.; Yang, G.; Chen, Y.; Jiang, L.; Li, X. Functionalized CNTs as Effective Additives to Improve the Efficiency of Perovskite Solar Cells. ACS Appl. Energy Mater. 2020, 3, 11674–11680. [Google Scholar] [CrossRef]
- Zhang, G.; Zhang, J.; Pan, Z.; Rao, H.; Zhong, X. Enhancing hole extraction via carbon nanotubes/poly(3-hexylthiophene) composite for carbon-based CsPbI2Br solar cells with a new record efficiency. Sci. China Mater. 2023, 66, 1727–1735. [Google Scholar] [CrossRef]
- Liu, K.; Yang, S.; Wu, J.; Zhang, H.; Qin, M.; Lu, X.; Tu, Y.; Meng, Q.; Zhan, X. Fullerene derivative anchored SnO2 for high-performance perovskite solar cells. Energy Environ Sci. 2018, 11, 3463–3471. [Google Scholar] [CrossRef]
- Zhang, J.; Tong, T.; Zhang, L.; Li, X.; Zou, H.; Yu, J. Enhanced Performance of Planar Perovskite Solar Cell by Graphene Quantum Dot Modification. ACS Sustain. Chem. Eng. 2018, 6, 8631–8640. [Google Scholar] [CrossRef]
- Shawky, A.; Nam, J.-S.; Kim, K.; Han, J.; Yoon, J.; Seo, S.; Lee, C.S.; Xiang, R.; Matsuo, Y.; Lee, H.M.; Maruyama, S.; Jeon, I. Controlled Removal of Surfactants from Double-Walled Carbon Nanotubes for Stronger p-Doping Effect and Its Demonstration in Perovskite Solar Cells. Small Methods 2021, 5, 2100080–2100090. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Tian, C.; Sun, C.; Yang, P.; Feng, W.; Zheng, L.; Yang, L.; Hou, E.; Luo, J.; Xie, L.; Wei, Z. Chlorofullerene C60Cl6 Enables Efficient and Stable Tin-Based Perovskite Solar Cells. Energy & Environ. Mater. 2022, 7, e12529–e12537. [Google Scholar]
- Ma, Q.; Chu, W.; Wu, S.; Wei, Q.; Cheng, Z.; Wu, J.; Liu, W.; Ma, S.; Ma, X.; Chen, J.; Dong, J. Design and performance optimization of carbon-based all-inorganic CsPbIBr2 perovskite battery with C60 buffer layer. Solar Energy 2022, 246, 245–255. [Google Scholar] [CrossRef]
- Wu, C.; Wang, Z.; Liang, L.; Gui, T.; Zhong, W.; Du, R.; Xie, C.; Wang, L.; Luo, L. Graphene-Assisted Growth of Patterned Perovskite Films for Sensitive Light Detector and Optical Image Sensor Application. Small 2019, 15, 1900730–1900739. [Google Scholar] [CrossRef]
- Duan, C.; Li, J.; Liu, Z.; Wen, Q.; Tang, H.; Yan, K. Highly electroluminescent and stable inorganic CsPbI2Br perovskite solar cell enabled by balanced charge transfer. Chem. Eng. J. 2021, 417, 128053. [Google Scholar] [CrossRef]
- Fang, D.; Niu, T.; Chen, Z.; Zhang, J.; Zhang, Z.; Zhou, S.; Liu, H.; Chen, G.; Fu, N.; Xue, Q.; Tao, J. Lignin carbon dots as effective dopants and passivators for SnO₂ electron transport layers to achieve high-performance perovskite solar cells. J. Power Sources 2025, 634, 236497. [Google Scholar] [CrossRef]
- Arjun, V.; Muthukumaran, K.P.; Nithya, A.; Prabhakaran, R.; Yoshimura, M.; Karuppuchamy, S. NiO/MWCNT incorporated methyl ammonium lead iodide for an efficient perovskite solar cells. Ceram. Int. 2024, 50, 54306–54319. [Google Scholar] [CrossRef]
- Zhu, M.; Liu, W.; Ke, W.; Xie, L.; Dong, P.; Hao, F. Graphene-Modified Tin Dioxide for Efficient Planar Perovskite Solar Cells with Enhanced Electron Extraction and Reduced Hysteresis. ACS Appl. Mater. Interfaces 2018, 11, 666–673. [Google Scholar] [CrossRef]
- Yao, X.; Qi, Z.; Yang, P.; Li, J.; Yang, W. Two-Dimensional Ti3C2Tx-Patched-GO heterojunction as charge booster and defect passivator for Stable, Carbon-Based inorganic perovskite solar cell. Chem. Eng. J. 2023, 470, 144315. [Google Scholar] [CrossRef]
- Sun, X.; He, B.; Zhu, J.; Zhu, R.; Chen, H.; Duan, Y.; Tang, Q. Multifunctional brominated graphene oxide boosted charge extraction for high-efficiency and stable all-inorganic CsPbBr3 perovskite solar cells. Chem. Eng. J. 2021, 41, 128727. [Google Scholar] [CrossRef]
- Wang, Y.; Xie, C.; Yao, X.; Chen, Q.; Liu, W.; Fu, Y.; Liu, Q.; Li, J.; Li, Y.; He, D. Improved comprehensive performance of CsPbI2Br perovskite solar cells by modifying the photoactive layers with carbon nanodots. J. Materiomics 2022, 8, 358–365. [Google Scholar] [CrossRef]
- Lin, S.; Cheng, Y.; Lin, C.; Fang, J.; Xiang, W.; Liang, X. Carbon nanodots with intense emission from green to red and their multifunctional applications. J. Alloys Compd. 2018, 742, 212–219. [Google Scholar] [CrossRef]
- Guo, X.; Zhao, B.; Xu, K.; Yang, S.; Liu, Z.; Han, Y.; Xu, J.; Xu, D.; Tan, Z.; Liu, S. P-Type Carbon Dots for Effective Surface Optimization for Near-Record-Efficiency CsPbI2Br Solar Cells. Small 2021, 17, 2102272–2102281. [Google Scholar] [CrossRef]
- Batmunkh, M.; Macdonald, T.J.; Shearer, C.J.; Bat-Erdene, M.; Wang, Y.; Biggs, M.J.; Parkin, I.P.; Nann, T.J.G.; Shapter. Carbon Nanotubes in TiO2 Nanofiber Photoelectrodes for High-Performance Perovskite Solar Cells. Adv. Sci. 2017, 4, 1600504–1600515. [Google Scholar] [CrossRef]
- Hui, W.; Yang, Y.; Xu, Q.; Gu, H.; Feng, S.; Su, Z.; Zhang, M.; Wang, J.; Li, X.; Fang, J.; Xia, F.; Xia, Y.; Chen, Y.; Gao, X.; Huang, W. Red-Carbon-Quantum-Dot-Doped SnO2 Composite with Enhanced Electron Mobility for Efficient and Stable Perovskite Solar Cells. Adv. Mater. 2019, 32, 1906374–1906383. [Google Scholar] [CrossRef]
- Luo, T.; Ye, G.; Chen, X.; Ding, M.; Ye, T.; Zhao, C.; Zhang, W.; Chang, H. Interface and Grain Boundary Passivation by PEA-SCN Double Ions via One-Step Crystal Engineering for All Air-Processed, Stable Perovskite Solar Cells. J. ACS Appl. Energy Mater. 2021, 4, 12290–12297. [Google Scholar] [CrossRef]
- Kang, D.-H.; Lee, S.-U.; Park, N.-G. Effect of Residual Chloride in FAPbI3 Film on Photovoltaic Performance and Stability of Perovskite Solar Cell. ACS Energy Lett. 2023, 8, 2122–2129. [Google Scholar] [CrossRef]
- Kartikay, P.; Sadhukhan, D.; Yella, A.; Mallick, S. Enhanced charge transport in low temperature carbon-based n-i-p perovskite solar cells with NiOx-CNT hole transport material. Sol. Energy Mater. Sol. Cells 2021, 230, 111241. [Google Scholar] [CrossRef]
- Ye, J.Y.; Tong, J.; Hu, J.; Xiao, C.; Lu, H.; Dunfield, S.P.; Kim, D.H.; Chen, X.; Larson, B.W.; Hao, J.; Wang, K.; Zhao, Q.; Chen, Z.; Hu, H.; You, W.; Berry, J.J.; Zhang, F.; Zhu, K. Enhancing Charge Transport of 2D Perovskite Passivation Agent for Wide-Bandgap Perovskite Solar Cells Beyond 21 %. Solar RRL 2020, 4, 2000082–2000090. [Google Scholar] [CrossRef]
- Zong, Z.; He, B.; Zhu, J.; Ding, Y.; Zhang, W.; Duan, J.; Zhao, Y.; Chen, H.; Tang, Q. Boosted hole extraction in all-inorganic CsPbBr3 perovskite solar cells by interface engineering using MoO2/N-doped carbon nanospheres composite. Sol. Energy Mater. Sol. Cells 2020, 209, 110460. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, J.; Chen, S.; Zhang, H.; Li, L.; Fu, Z. Surface passivation with nitrogen-doped carbon dots for improved perovskite solar cell performance. J. Mater. Sci. 2018, 53, 9180–9190. [Google Scholar] [CrossRef]
- Chen, C.; Wu, C.; Ding, X.; Tian, Y.; Zheng, M.; Cheng, M.; Xu, H.; Jin, Z.; Ding, L. Constructing binary electron transport layer with cascade energy level alignment for efficient CsPbI2Br solar cells. Nano Energy 2020, 71, 104604. [Google Scholar] [CrossRef]
- Zhu, W.; Chen, Q.; Yamaguchi, Y.; Zhao, F.; Hao, D.; Liu, X.; Dou, X. Perovskite solar cells prepared under infrared irradiation during fabrication process in air ambience. J. Mater. Sci.: Mater. Electron. 2020, 31, 9535–9542. [Google Scholar] [CrossRef]
- Jeong, M.J.; Jeon, S.W.; Kim, S.Y.; Noh, J.H. High Fill Factor CsPbI2Br Perovskite Solar Cells Via Crystallization Management. Adv. Energy Mater. 2023, 13, 2300698–2300707. [Google Scholar] [CrossRef]
- He, J.; Fang, W.-H.; Long, R.; Prezhdo, O.V. Superoxide/Peroxide Chemistry Extends Charge Carriers Lifetime but Undermines Chemical Stability of CH3NH3PbI3 Exposed to Oxygen: Time-Domain ab Initio Analysis. J. Am. Chem. Soc. 2019, 141, 5798–5807. [Google Scholar] [CrossRef]
- Ansari, F.; Shirzadi, E.; Salavati-Niasari, M.; LaGrange, T.; Nonomura, K.; Yum, J.-H.; Sivula, K.; Zakeeruddin, S.M.; Nazeeruddin, M.K.; Grätzel, M.; Dyson, P.J.; Hagfeldt, A. Passivation Mechanism Exploiting Surface Dipoles Affords High-Performance Perovskite Solar Cells. J. Am. Chem. Soc. 2020, 142, 11428–11433. [Google Scholar] [CrossRef] [PubMed]
- Mali, S.S.; Patil, J.V.; Steele, J.A.; Jung, Y.H.; Nazeeruddin, M.K.; Hong, C.K. Controlled crystallization and surface engineering of mixed-halide γ-CsPbI2Br inorganic perovskites via guanidinium iodide additive in air-processed perovskite solar cells. Mater. Today 2023, 67, 33–45. [Google Scholar] [CrossRef]
- Lee, K.; Chan, S.; Chiu, W.; Ahn, S.; Ting, C.; Chang, Y.; Suryanarayanan, V.; Wu, M.; Liu, C. Reducing Defects in Organic-Lead Halide Perovskite Film by Delayed Thermal Annealing Combined with KI/I2 for Efficient Perovskite Solar Cells. Nanomaterials 2021, 11, 1607. [Google Scholar] [CrossRef]
- Lu, C.; Zhang, W.; Jiang, Z.; Zhang, Y.; Ni, C. Graphene quantum dots doping SnO2 for improving carrier transport of perovskite solar cells. Ceram. Int. 2021, 47, 29712–29721. [Google Scholar] [CrossRef]
- Khorshidi, E.; Rezaei, B.; Irannejad, N.; Adhami, S.; Ebrahimi, M.; Kermanpur, A.; Ensafi, A.A. The role of GQDs additive in TiO2 nanorods as an electron transfer layer on performance improvement of the perovskite solar cells. Electrochim. Acta 2020, 337, 135822. [Google Scholar] [CrossRef]
- Liu, Z.; Siekmann, J.; Klingebiel, B.; Rau, U.; Kirchartz, T. Interface Optimization via Fullerene Blends Enables Open-Circuit Voltages of 1.35 V in CH3NH3Pb(I0.8Br0.2)3 Solar Cells. Adv. Energy Mater. 2021, 11, 2003386–2003399. [Google Scholar] [CrossRef]
- Younes, E.M.; Gurung, A.; Bahrami, B.; El-Maghraby, E.M.; Qiao, Q. Enhancing efficiency and stability of inverted structure perovskite solar cells with fullerene C60 doped PC61BM electron transport layer. Carbon 2021, 180, 226–236. [Google Scholar] [CrossRef]
- Sidhik, S.; Velusamy, J.; Rosa, E.D.I.; Pérez-García, S.A.; Ramos-Ortiz, G.; López-Luke, T. Role of carbon nanodots in defect passivation and photo-sensitization of mesoscopic-TiO2 for perovskite solar cells. Carbon 2019, 146, 388–398. [Google Scholar] [CrossRef]
- Zhao, X.; Tao, L.; Li, H.; Huang, W.; Sun, P.; Liu, J.; Liu, S.; Sun, Q.; Cui, Z.; Sun, L.; Shen, Y.; Yang, Y.; Wang, M. Efficient Planar Perovskite Solar Cells with Improved Fill Factor via Interface Engineering with Graphene. Nano Lett. 2018, 18, 2442–2449. [Google Scholar] [CrossRef]
- Kim, J.K.; Nguyen, D.N.; Lee, J.; Kang, S.; Kim, Y.; Kim, S.; Kim, H. Carbon quantum dot-incorporated nickel oxide for planar p-i-n type perovskite solar cells with enhanced efficiency and stability. J. Alloys Compd. 2020, 818, 152887. [Google Scholar] [CrossRef]
- Mohammed, M. K. A. 21.4 % efficiency of perovskite solar cells using BMImI additive in the lead iodide precursor based on carbon nanotubes/TiO2 electron transfer layer. Ceram. Int. 2020, 46, 27647–27654. [Google Scholar] [CrossRef]
- Zheng, T.; Fan, L.; Jin, B.; Peng, R. Concise synthesis of low-cost fullerene derivatives as electron transport materials for efficient air-processed invert perovskite solar cells. J. Colloid Interface Sci. 2023, 642, 497–504. [Google Scholar] [CrossRef]
- Cheng, N.; Liu, P.; Qi, F.; Xiao, Y.; Yu, W.; Yu, Z.; Liu, W.; Guo, S.; Zhao, X. Multi-walled carbon nanotubes act as charge transport channel to boost the efficiency of hole transport material free perovskite solar cells. J. Power Sources 2016, 332, 24–29. [Google Scholar] [CrossRef]
- Fuente, M.S.; Kaur, S.; Hu, Q.; Barnard, E.S.; Dudenas, P.; Kusoglu, A.; Russell, T.P.; Urban, J.J.; Prasher, R. Enhanced Charge Carrier Transport in 2D Perovskites by Incorporating Single-Walled Carbon Nanotubes or Graphene. ACS Energy Lett. 2019, 5, 109–116. [Google Scholar] [CrossRef]
- Hsu, H.L.; Hsiao, H.T.; Juang, T.; Jiang, B.; Chen, S.; Jeng, R.; Chen, C. Carbon Nanodot Additives Realize High-Performance Air-Stable p–i–n Perovskite Solar Cells Providing Efficiencies of up to 20.2 %. Adv. Energy Mater. 2018, 8, 1802323–1802332. [Google Scholar] [CrossRef]
- Bahadur, J.; Ryu, J.; Lee, D.; Hong, J.; Hayase, S.; Cho, J.S.; Jeong, S.M.; Kang, D. In-situ surface defects passivation with small carbon chain molecules for highly efficient, air-processed inorganic CsPbI2Br perovskite photovoltaics. Appl. Surf. Sci. 2023, 614, 156229. [Google Scholar] [CrossRef]
- Wei, X.; Liu, X.; Liu, H.; Yang, S.; Zeng, H.; Meng, F.; Lei, X.; Liu, J. Exfoliated graphitic carbon nitride self-recognizing CH3NH3PbI3 grain boundaries by hydrogen bonding interaction for improved perovskite solar cells. Solar Energy 2019, 181, 161–168. [Google Scholar] [CrossRef]
- Gong, X.; Guan, L.; Li, Q.; Li, Y.; Zhang, T.; Pan, H.; Sun, Q.; Shen, Y.; Grätzel, C.; Zakeeruddin, S.M.; Grätzel, M.; Wang, M. Black phosphorus quantum dots in inorganic perovskite thin films for efficient photovoltaic application. Sci. Adv. 2020, 6, 5661. [Google Scholar] [CrossRef]
- Lin, H.-S.; Okawa, S.; Ma, Y.; Yotsumoto, S.; Lee, C.; Tan, S.; Manzhos, S.; Yoshizawa, M.; Chiashi, S.; Lee, H.M.; Tanaka, T.; Kataura, H.; Jeon, I.; Matsuo, Y.; Maruyama, S. Polyaromatic Nanotweezers on Semiconducting Carbon Nanotubes for the Growth and Interfacing of Lead Halide Perovskite Crystal Grains in Solar Cells. Chem. Mater. 2020, 32, 5125–5133. [Google Scholar] [CrossRef]
- Agbolaghi, S. Efficacy beyond 17 % via engineering the length and quality of grafts in organic halide perovskite/CNT photovoltaics. New J. Chem. 2019, 43, 10567–10574. [Google Scholar] [CrossRef]
- Bag, M.; Renna, L.A.; Jeong, S.P.; Han, X.; Cutting, C.L.; Maroudas, D.; Venkataraman, D. Evidence for reduced charge recombination in carbon nanotube/perovskite-based active layers. Chem. Phys. Lett. 2016, 662, 35–41. [Google Scholar] [CrossRef]
- Tiong, V.T.; Pham, N.D.; Wang, T.; Zhu, T.; Zhao, X.; Zhang, Y.; Shen, Q.; Bell, J.; Hu, L.; Dai, S.; Wang, H. Octadecylamine-Functionalized Single-Walled Carbon Nanotubes for Facilitating the Formation of a Monolithic Perovskite Layer and Stable Solar Cells. Adv. Func. Mater. 2018, 28, 201705545. [Google Scholar] [CrossRef]
- Zhou, J.; Wu, J.; Li, N.; Li, X.; Zheng, Y.; Tao, X. Efficient all-air processed mixed cation carbon-based perovskite solar cells with ultra-high stability. J. Mater. Chem. A 2019, 7, 17594–17603. [Google Scholar] [CrossRef]
- Wang, Y.; Li, J.; Yao, X.; Xie, C.; Chen, Q.; Liu, W.; Gao, Z.; Fu, Y.; Liu, Q.; He, D.; Li, Y. Improved Comprehensive Photovoltaic Performance and Mechanisms by Additive Engineering of Ti3C2Tx MXene into CsPbI2Br. ACS Appl. Mater. Interfaces 2022, 14, 40930–40938. [Google Scholar] [CrossRef]
- Chen, D.; Tian, B.; Fan, G.; Wang, Y.; Zhu, W.; Ren, Z.; Xi, H.; Su, K.; Zhang, J.; Zhang, C.; Zhang, J.; Hao, Y. Simple and Convenient Interface Modification by Nanosized Diamond for Carbon Based All-Inorganic CsPbIBr2 Solar Cells. ACS Appl. Energy Mater. 2021, 4, 5661–5667. [Google Scholar] [CrossRef]
- Zhou, S.; Tang, R.; Yin, L. Slow-Photon-Effect-Induced Photoelectrical-Conversion Efficiency Enhancement for Carbon-Quantum-Dot-Sensitized Inorganic CsPbBr3 Inverse Opal Perovskite Solar Cells. Adv. Mater. 2017, 29, 1703682–1703691. [Google Scholar] [CrossRef]
- Jia, L.; Ma, X.; Xiang, W.; Jiang, X.; Ding, H.; Li, X.; Shang, Y.; Zhu, J.; Li, Z.; Qiu, Y.; Chen, M.; Chen, J.; Yang, S. Lowering the dielectric mismatch for efficient inverted perovskite solar cells through incorporating cyano-functionalized fullerene additive. Sci. China Mater. 2023, 66, 2146–2158. [Google Scholar] [CrossRef]
- Tang, H.; Xu, T.; Qin, X.; Zou, K.; Lv, S.; Fan, J.; Huang, T.; Chen, L.; Huang, W. Carbon Quantum Dot-Passivated Perovskite/Carbon Electrodes for Stable Solar Cells. ACS Appl. Nano Mater. 2021, 4, 13339–13351. [Google Scholar] [CrossRef]
- Zhang, Z.; Wang, X.; Yan, Q.; Yuan, X.; Lu, Y.; Cao, H.; He, D.; Jiang, Z.; Xu, R.; Chen, T.; Ma, Z.; Song, H.; Hong, F.; Xu, F. Dual-Doping Strategy of Metal Chlorides in Ambient Air with High Humidity for Achieving Highly Air-Stable All-Inorganic Perovskite Solar Cells. Solar RRL 2024, 8, 2400216–2400228. [Google Scholar] [CrossRef]
- Zeng, Q.; Xiao, H.; Ma, Q.; Huang, R.; Pan, Y.; Li, L.; Liao, X.; Liu, S.; Zhang, W.; Liu, F. Highly Layer-Oriented PbI2 Films Enabling All-Air Processed Perovskite Solar Cells. Adv. Energy Mater. 2024, 14, 2401279–2401288. [Google Scholar] [CrossRef]
- Fooladvand, P.; Eskandari, M.; Fathi, D.; Das, N. Single-walled carbon nanotube as hole transport layer in perovskite solar cell: Efficiency enhancement. Energy Rep. 2023, 10, 3652–3664. [Google Scholar] [CrossRef]
- Gao, Z.W.; Wang, Y.; Liu, H.; Sun, J.; Kim, J.; Li, Y.; Xu, B.; Choy, W.C.H. Tailoring the Interface in FAPbI3 Planar Perovskite Solar Cells by Imidazole-Graphene-Quantum-Dots. Adv. Funct. Mater. 2021, 31, 2101438–2101445. [Google Scholar] [CrossRef]
- Meng, F.; Li, Y.; Gao, L.; Liu, A.; Li, Y.; Wang, T.; Zhang, C.; Fan, M.; Wei, G.; Ma, T. Intermediate-Controlled Interfacial Engineering for Stable and Highly Efficient Carbon-Based PSCs. ACS Appl. Mater. Interfaces 2020, 12, 34479–34486. [Google Scholar] [CrossRef]
- Wang, H.; Guo, C.; Li, F.; Zeng, S.; Li, X.; Fu, H.; Wang, T.; Liu, D. Push–pull substituent design of fullerene dimer at the buried interface toward stable and efficient perovskite solar cells. Sci. China Mater. 2023, 67, 58–66. [Google Scholar] [CrossRef]
- Li, Z.; Liu, C.; Ren, G.; Han, W.; Shen, L.; Guo, W. Cations Functionalized Carbon Nano-Dots Enabling Interfacial Passivation and Crystallization Control for Inverted Perovskite Solar Cells. Solar RRL 2019, 4, 1900369–1900377. [Google Scholar] [CrossRef]
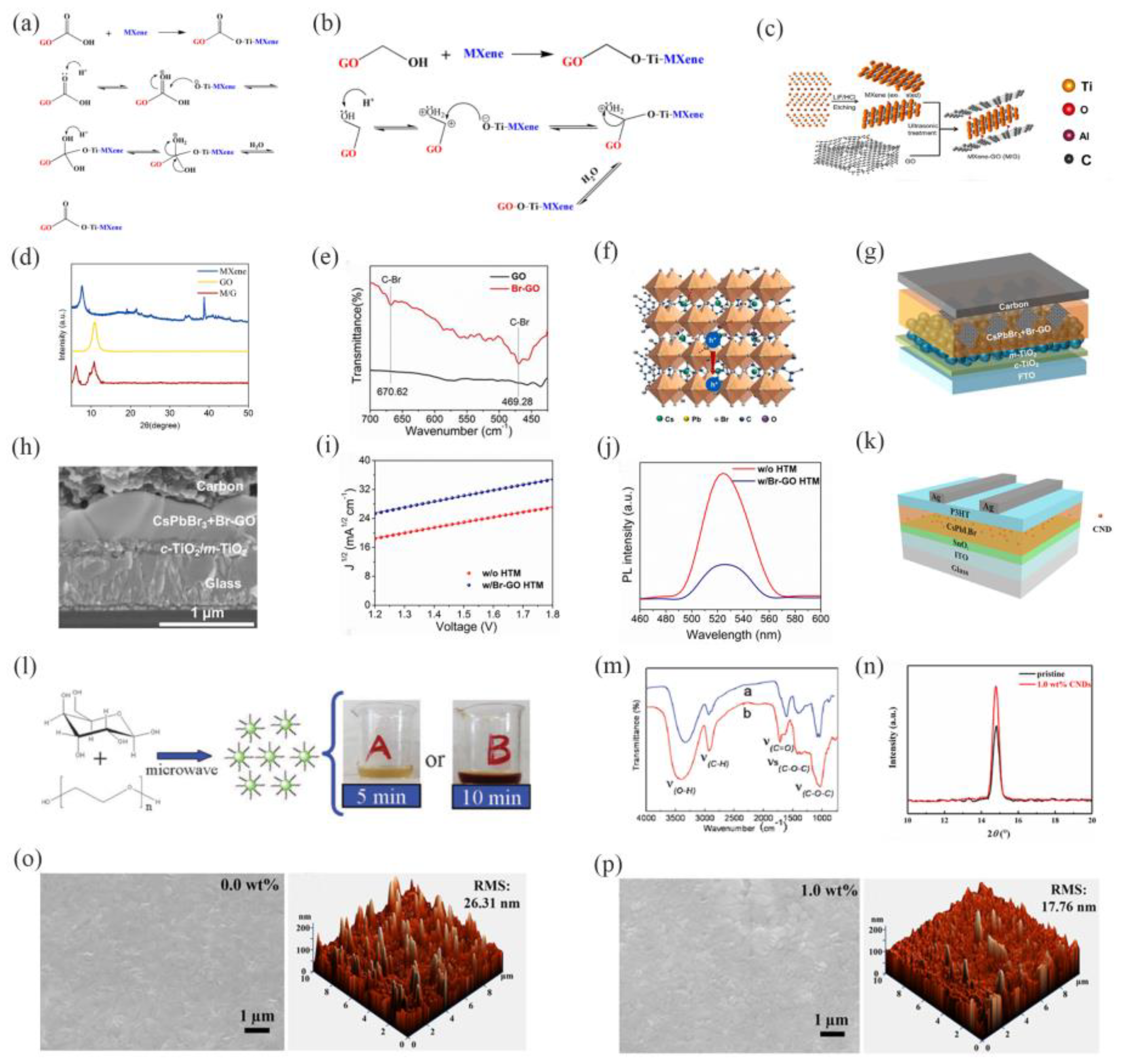
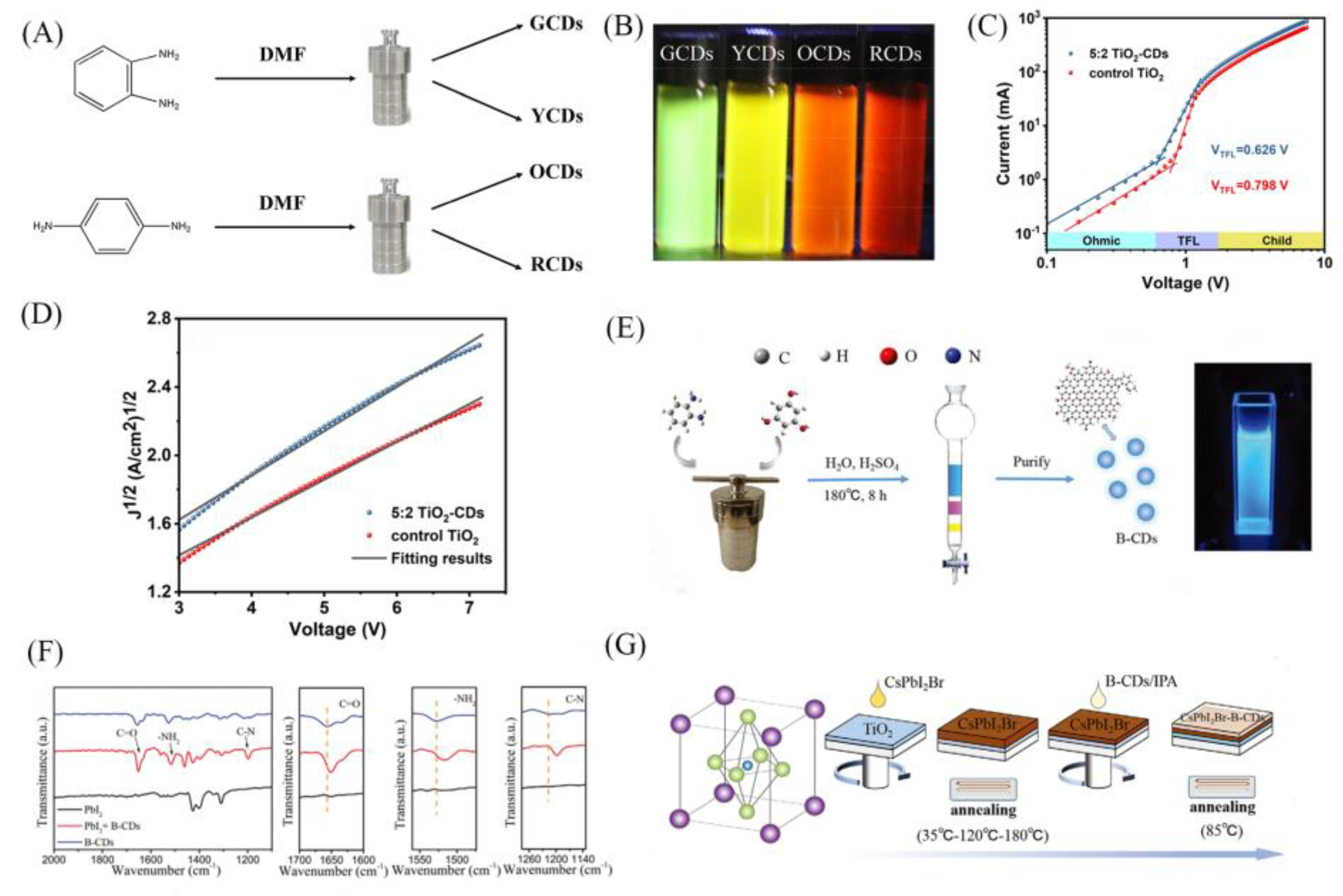
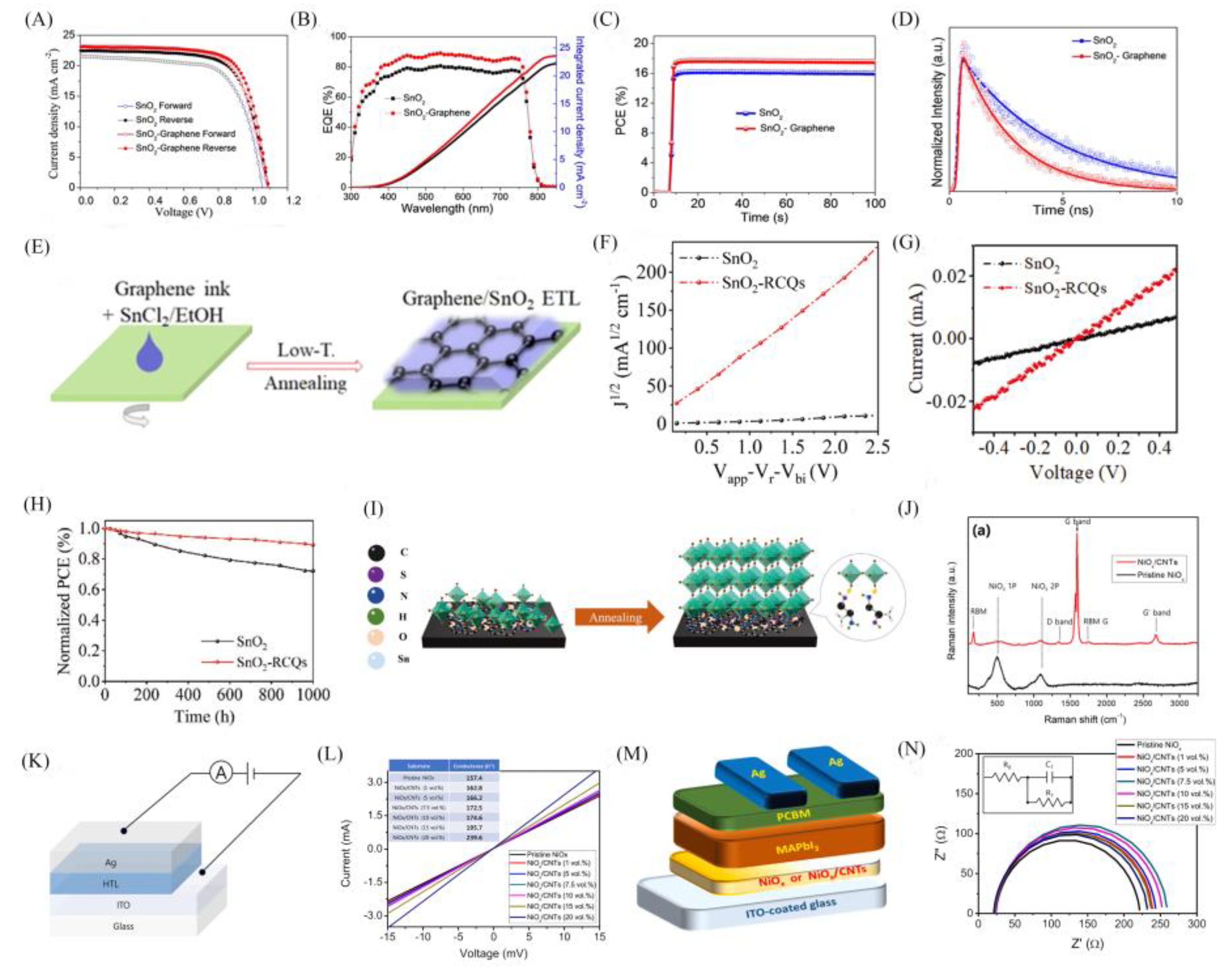
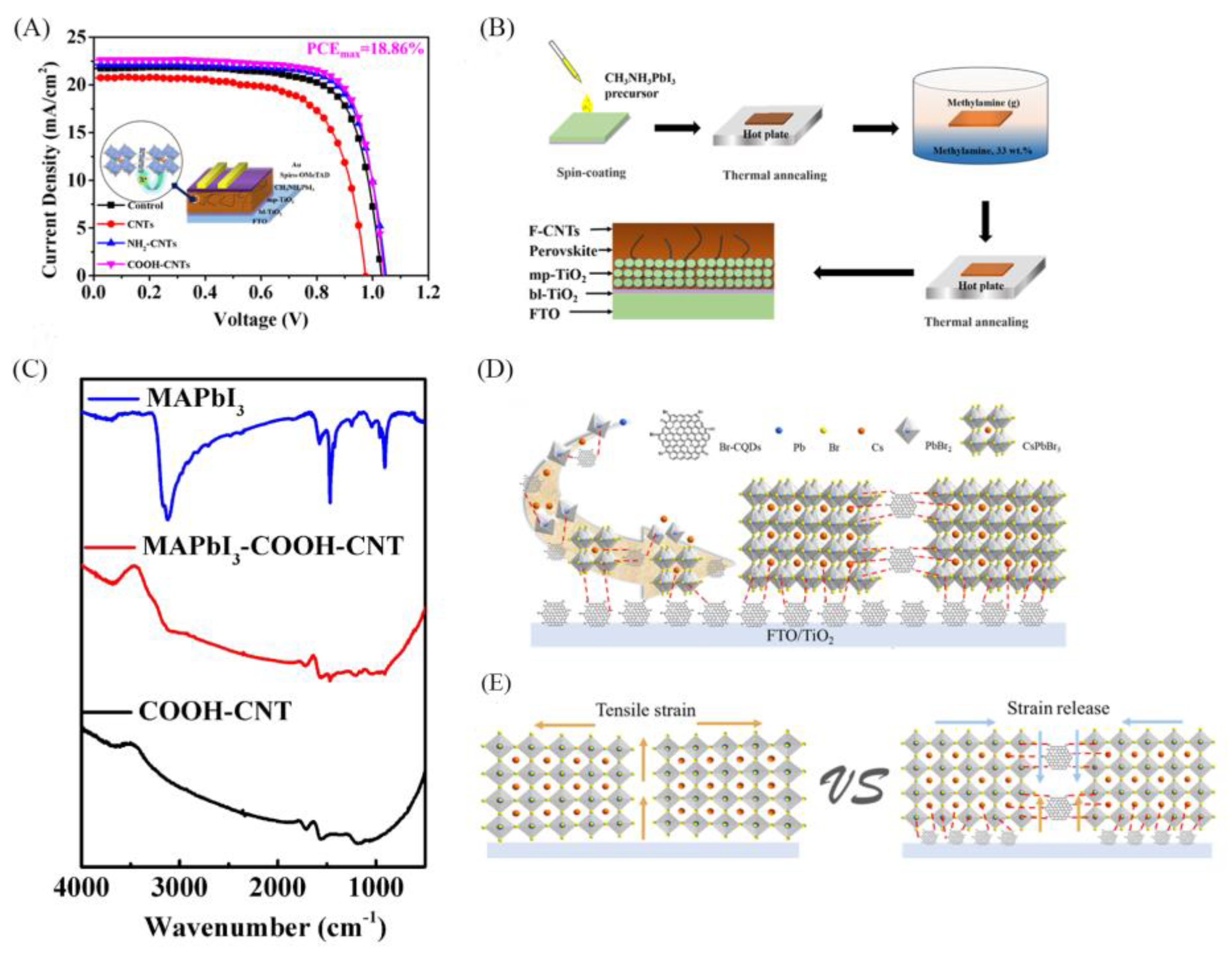
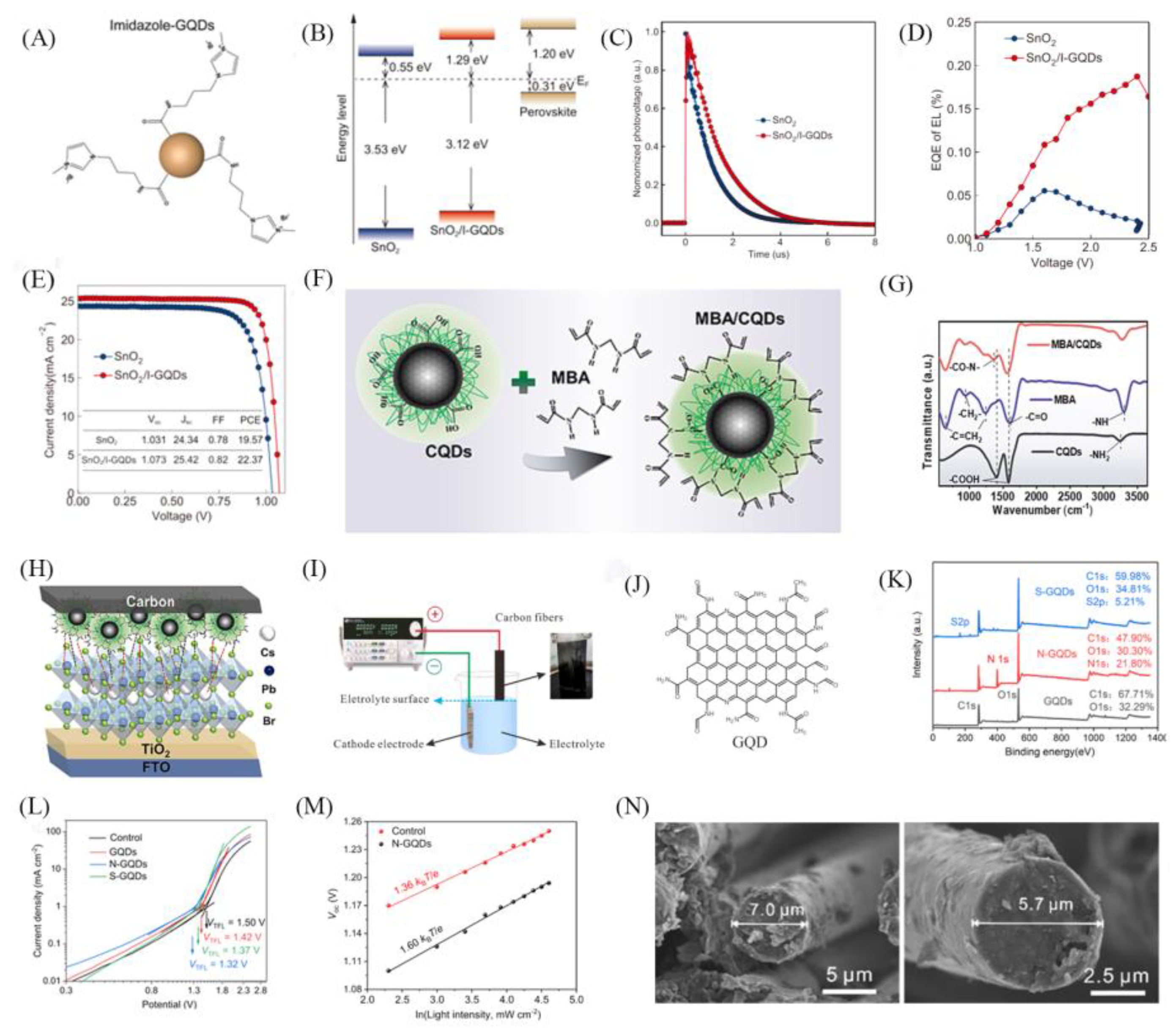
| Structure | JSC (mA/cm2) |
VOC (V) |
FF (%) |
PCE (%) |
Type | Refs. |
|---|---|---|---|---|---|---|
| FTO/compact-TiO2/TiO2-CDs/FA0.85MA0.15Pb(I0.85Br0.15)3 /PCBM/Ag | 26.0 | 1.2 | 70.0 | 21.1 | (carbon dots) CDs | 18 |
| ITO/NiOx/Me-4PCAZ/Cs0.04(FA0.95MA0.05)0.96Pb(I0.95Br0.05)3/tBu -FIDO/BCP/Ag | 24.8 | 1.2 | 78.3 | 22.1 | Indanones (tBu-FIDO) |
49 |
| FTO/b-TiO2/TiO2@r-GO/CH3NH3PbI3/Spiro-OMeTAD /Ag | 22.0 | 0.9 | 70.7 | 14.5 | GO/Mesoporous TiO2 | 54 |
| ITO/SnO2@GQDs/CH3NH3PbI3/Spiro-OMeTAD /Au | 23.0 | 1.1 | 75.4 | 19.7 | GQDs | 56 |
| ITO/NiOx@CNTs/MAPbI3/PCBM/Ag | 22.0 | 1.0 | 75.9 | 16.9 | CNT | 57 |
| FTO/SnO2/CQD-SnO2/FAPbI3/Spiro-OMeTAD /Ag | 25.6 | 1.2 | 81.2 | 24.1 | CQDs | 60 |
| FTO/TiO2/CsPbI2Br/P3HT/CNT/C | 14.6 | 1.4 | 78.5 | 15.6 | CNTs/poly(3-hexylthiophene) | 63 |
| ITO/SnO2@C60-EDA/CsPbI2Br/5-AVABr/ Spiro-OMeTAD /MoO3/Ag | 15.4 | 1.3 | 80.3 | 16.6 | C60-ethylenediamine derivatives (C60-EDA) | 70 |
| Ag/L-CDs@SnO2/Perovskite/Spiro-OMeTAD/Ag | 23.8 | 1.1 | 81.3 | 20.9 | Lignin carbon dots(L-CDs) | 71 |
| FTO/Graphene@SnO2/MAPbI3/Spiro-OMeTAD/Au | 23.1 | 1.1 | 72.0 | 18.1 | graphene− SnO2 |
73 |
| FTO/cp-TiO2@SWCNTs/CH3NH3PbI3/Spiro-OMeTAD/Au | 21.4 | 1.0 | 67.0 | 14.0 | Single-Walled Single-Walled(SWCNT)/TiO2 | 79 |
| ITO/SnO2@RCQs/Cs0.05FA0.81MA0.14PbI2.55Br0.45/Spiro-OMeTAD/MoOX/Au | 24.1 | 1.1 | 82.9 | 22.8 | red- CQDs (RCQs) functionalized NiOx |
80 |
| FTO/TiO2/FA0.83Cs0.17PbI2.5Br0.5/f-NiOx+CNT/C | 18.3 | 0.6 | 64.0 | 11.4 | (f-NiOx) and CNT (f-NiOx + CNT) | 83 |
| FTO/c-TiO2/m-TiO2/CsPbBr3/MoO2@ /N-doped carbon nanospheres composites)/carbon |
7.2 | 1.5 | 85.2 | 9.4 | MoO2/N- doped NC |
85 |
| FTO/SnO2/@CQDs/FAMA0.2Pb1.1I2.2Br0.6/Sprio-OMeTAD/Ag | 21.6 | 1.1 | 78.0 | 18.6 | SnO2: GQDs | 94 |
| FTO/GQDs@TiO2NRs/Csx(MA0.17FA0.83)(1−x)Pb(I0.83Br0.17)3/Spiro-OMeTAD /Au | 22.1 | 0.8 | 64.0 | 11.7 | GQDs- TiO2 NRs | 95 |
| ITO/PTAA/MAPb(I0.8Br0.2)3/PCBM@ICBA/BCP/Ag | 17.6 | 1.4 | 81.2 | 18.9 | ICBA | 96 |
| FTO/PEDOT:PSS/CH3NH3PbI3/PC61BM@C60/Ag | 23.0 | 1.0 | 78.0 | 17.5 | C60 doped PC61BM | 97 |
| FTO/TiO2 :CNDs/CH3NH3PbI3/Spiro-OMeTAD /Au | 23.3 | 1.1 | 75.2 | 19.5 | TiO2:carbon nanodots (CNDs) | 98 |
| ITO/SnO2@NDI-Graphene/FA0.75MA0.15Cs0.1PbI2.65Br0.35/Spiro-OMeTAD/Au | 22.7 | 1.1 | 82.1 | 20.2 | 2D naphthalene diimide- | 99 |
| ITO/NiO@CQD/CH3NH3PbI3/PCBM/BCP/Ag | 20.2 | 1.1 | 77.2 | 16.9 | incorporating CQDs into a NiOx | 100 |
| FTO/MWCNT/TiO2/CH3NH3PbI3/Spiro-OMeTAD/Au | 28.0 | 1.1 | 73.3 | 21.4 | Multi-Walled Carbon Nanotube (MWCNT) | 101 |
| FTO/NiOX/MAPbI3/C60@PMME/BCP/Ag | 22.2 | 1.0 | 79.6 | 18.4 | Fullerene-bis (pyridin-2-ylmethyl) malonate | 102 |
| Structure | JSC (mA/cm2) |
VOC (V) |
FF (%) |
PCE (%) |
Type | Refs. |
|---|---|---|---|---|---|---|
| ITO/SnO2/FAPbI3@N-CNFs/Spiro-OMeTA D/Ag | 25.5 | 1.2 | 81.5 | 24.1 | nitrogen-doped CNFs (N-CNFs) |
17 |
| ITO/SnO2/(FAPbI3)0.97(MAPbBr3)0.03@COO H-CNTs/TPA-PEABr/Spiro-OMeTAD/Au | 23.9 | 1.1 | 76.0 | 18.9 | COOH–CNTs | 20 |
| FTO/TiO2/FAMA0.2Cs0.065Pb1.32I2.165Br0.64@ CNTs/Sprio-OMeTAD/Au | 23.3 | 1.2 | 79.8 | 21.7 | Lewis-base polymer-CNTs |
51 |
| FTO/TiO2/CsPbBr3@Br-CQDs/C | 7.84 | 1.7 | 83.4 | 10.8 | Br-CQDs | 61 |
| FTO/PEDOT:PSS/CH3NH3PbI3@COOH-C NTs/Spiro-OMeTAD/Au | 21.9 | 1.1 | 76.8 | 17.1 | COOH-CNTs | 62 |
| ITO/SnO2/PEA0.15FA0.85SnI3@C60Cl/PCBM/BCP/Ag |
20.3 |
0.9 |
76.0 |
13.3 |
Six chlorine attached to the fullerene cage brominated |
67 |
| FTO/c-TiO2/m-TiO2/CsPbBr3@Br-GO/C | 7.9 | 1.6 | 80.0 | 10.1 | graphene oxide (Br-GO) | 75 |
| ITO/SnO2/CDNS@CsPbI2Br/P3HT/Ag | 15.1 | 1.2 | 77.0 | 13.9 | CNDs | 76 |
| FTO/c-TiO2/SiO2/CH3NH3PbI3@MWCNT/C | 21.3 | 0.9 | 62.3 | 11.6 | MWCNTs | 103 |
| ITO/PEDOT:PSS/BA2MA3Pb4I13@SWCNT/PCBM/Al | 14.4 | 1.0 | 63.0 | 9.0 | MWCNTs | 104 |
| ITO/NiOx/CH3NH3PbI3(MAPbI3)@CNDs/ PC61BM/BCP/Ag. |
23.2 | 1.1 | 79.2 | 19.5 | CNDs | 105 |
| ITO/SnO2/ZnO/CsPbI2Br@DIM/P3HT/Au | 16.1 | 1.2 | 82.5 | 16.4 | diiodo methane | 106 |
| ITO/SnO2/CsPbI2Br@BPQDs/Spiro-OMeTAD/Au | 15.9 | 1.3 | 78.0 | 15.5 | BPQDs | 108 |
| ITO/SnO2/MAPbI3@DPD+SWCNT/Spiro-OMeTAD/Au | 24.0 | 1.1 | 75.9 | 19.7 | DOC-SWCNTs | 109 |
| ITO/SnO2/MAPbI3@SWCNT+DPB/Spiro-OMeTAD/Au |
24.1 | 1.1 | 78.2 | 20.7 | DPB-SWNTs P3HT+ |
109 |
| ITO/c-TiO2/m-TiO2/MAPbI3@P3HT+MWCNT/Spiro-OMeTAD/Ag |
23.6 | 1.0 | 76.0 | 17.4 | MWCHTs HBC-PMMA- |
110 |
| ITO/PEDOT:PSS/MAxFA1-xPbI3@MWCNT/PCBM/Ca/Al | 18.2 | 1.0 | 72.8 | 12.9 | MWCNTs | 111 |
| FTO/SnO2/(FA0.83MA0.17)0.95Cs0.05Pb(I0.83Br0.17)3@SWCNT/Spiro-OMeTAD/Au | 20.8 | 1.1 | 69.0 | 16.1 | ODA-SWCNTs | 112 |
| ITO/SnO2/FAxMA1-xPbIyBr3-y@MWCNTs/C | 20.5 | 1.0 | 59.7 | 12.3 | MWCNTs | 113 |
| ITO/SnO2/CsPbI2Br@Ti3C2TX/P3HT/Ag | 15.0 | 1.2 | 78.0 | 15.1 | Ti3C2Tx MXene nanosheets | 114 |
| FTO/c-TiO2/CsPbIBr2@ND/Carbon | 11.3 | 1.3 | 62.0 | 11.3 | Nanodot (ND) | 115 |
| FTO/TiO2/CQD-IP@CsPbBr3/Spiro-OMeTAD/Ag | 11.3 | 1.1 | 69.0 | 8.3 | CQDs inverse opal | 116 |
|
FTO/NiOX/MAPbI3@C60-Rhd-CN/PC61BM/BCP/Ag |
23.4 |
1.1 |
83.0 |
20.8 |
cyano-functionl ized fullerene in which two cyano groups incorporated a rhodanine moiety |
117 |
| Structure | JSC (mA/cm2) |
VOC (V) |
FF (%) |
PCE (%) |
Type | Refs. |
| FTO/c-TiO2/m-TiO2/CsPbBr3/MBA@CQDs/C |
7.82 |
1.6 |
82.5 |
10.4 |
MBA/CQDs |
50 |
| FTO/c-TiO2/CsPbIBr2/N-GQDs/C |
11.9 |
1.3 |
65.4 |
9.8 |
N- GQDs |
53 |
|
ITO/TiO2/MAPbIXCl3−x/rGO-4FPH/Sprio-OMeTAD/Au |
21.5 | 1.1 | 78.6 | 18.8 | GO Interlayer with 4-fluorophenyl-hydrazi ne hydrochloride | 55 |
|
ITO/SnO2/C9/(FAPbI3)x(MAPbBr3)1-x/Sprio-OMeTAD/Au |
24.1 |
1.1 |
78.9 |
21.3 |
9-(1-(6-(3,5-bis(hydrx ymethyl)phenoxy)-1-hexyl)-1H-1,2,3-triazol-4-yl)-1-nonyl fluoroacetate (C9) |
64 |
|
FTO/TiO2/CsPbI2Br/Mxene+Go/C |
14.9 |
1.3 |
78.3 |
15.0 |
Ti3C2Tx-Patched-GO |
74 |
| FTO/TiO2/CsPbI2Br/B-CDs/Spiro-OMeTAD/Au |
15.8 | 1.3 | 80.0 | 16.8 | p type blue CDS | 78 |
| FTO/c-TiO2/m-TiO2/MAPbI3/A-CQDs/C |
23.5 |
1.1 |
56.7 |
14.0 |
A-CQ |
118 |
| ITO/TiO2/CH3NH3PbI3/SWCNTS/PbS-QDs/C |
17.5 |
1.0 |
87.0 |
14.5 |
SWCNTs |
121 |
| ITO/SnO2/I-GQDs/FAPbI3/PCBM:C60/Ag |
25.4 |
1.1 |
82.0 |
22.4 |
I-GQDs |
122 |
| FTO/TiO2/Cs0.05(MA0.15FA0.85)0.95Pb(I0.86Br0.14)3/IIE/C |
23.3 |
1.0 |
71.0 |
16.4 |
acetylene black |
123 |
| ITO/SnO2/DC60-H-H/(FAPbI3)x(MAPbBr3)1−x/ Spiro-OMeTAD /Ag |
24.7 |
1.1 |
74.0 |
21.1 |
DC60-H-H |
124 |
| ITO/SnO2/DC60-H-MeO/(FAPbI3)x(MAPbBr3)1−x/Spiro-OMeTAD/Ag | 25.0 | 1.1 | 79.0 | 21.6 | DC60-H-MeO | 124 |
| ITO/SnO2/DC60-Cl-H/(FAPbI3)x(MAPbBr3)1−x/ Spiro-OMeTAD /Ag | 25.2 | 1.1 | 80.0 | 22.8 | DC60-Cl-H | 124 |
| ITO/PTAA/CNDs@Na/CH3NH3PbI3/PCBM/BPHEHE/Ag |
23.1 |
1.1 |
80.7 |
20.6 |
Natrium ion-functionalized CNDS |
125 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




