Submitted:
20 April 2026
Posted:
21 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Experimental Animals and Foods
2.2. Procedures
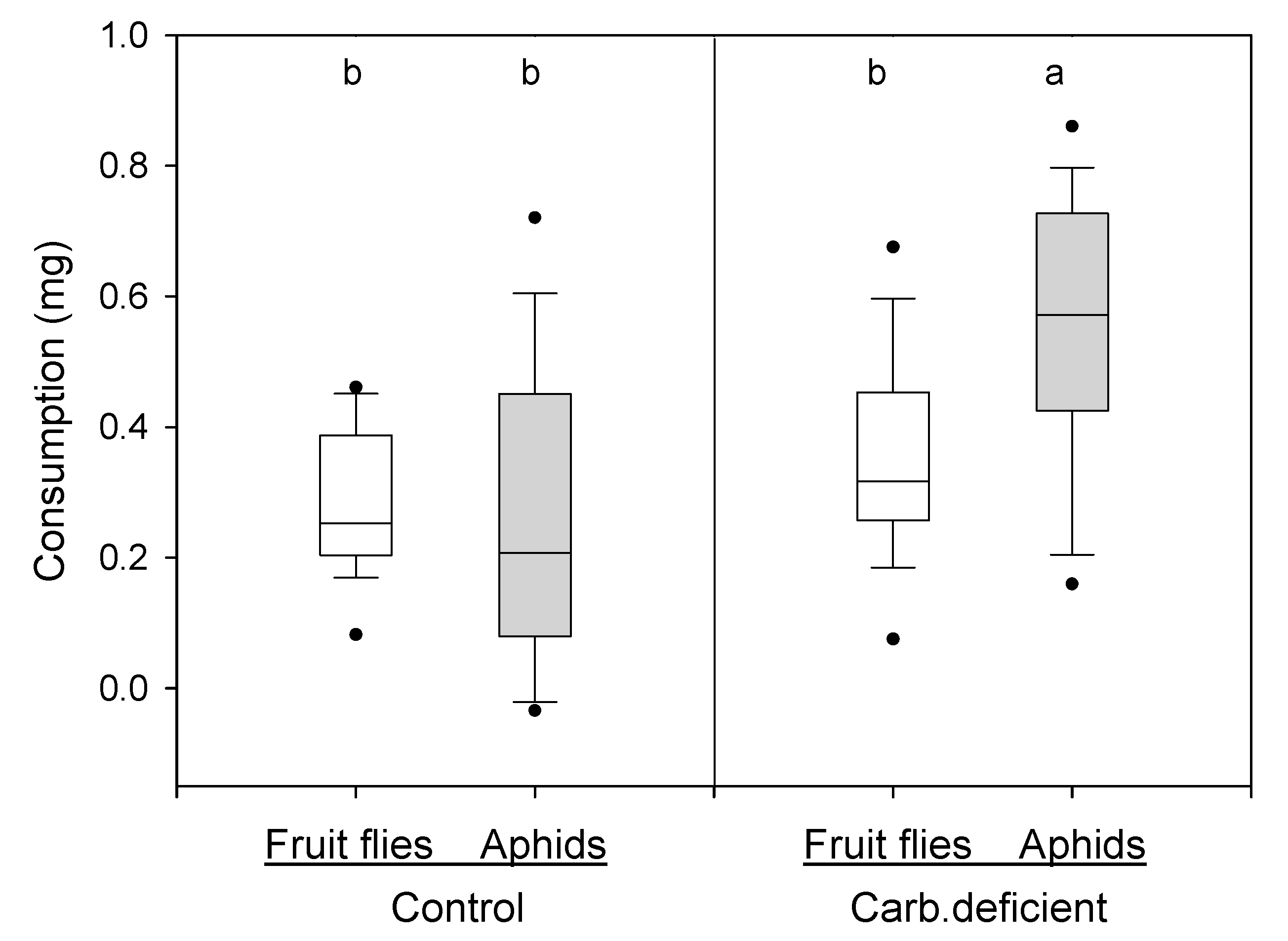
2.2.1. Experiment 1. Does Aphid Consumption Increase in Carbohydrate Deficient Harvestmen?
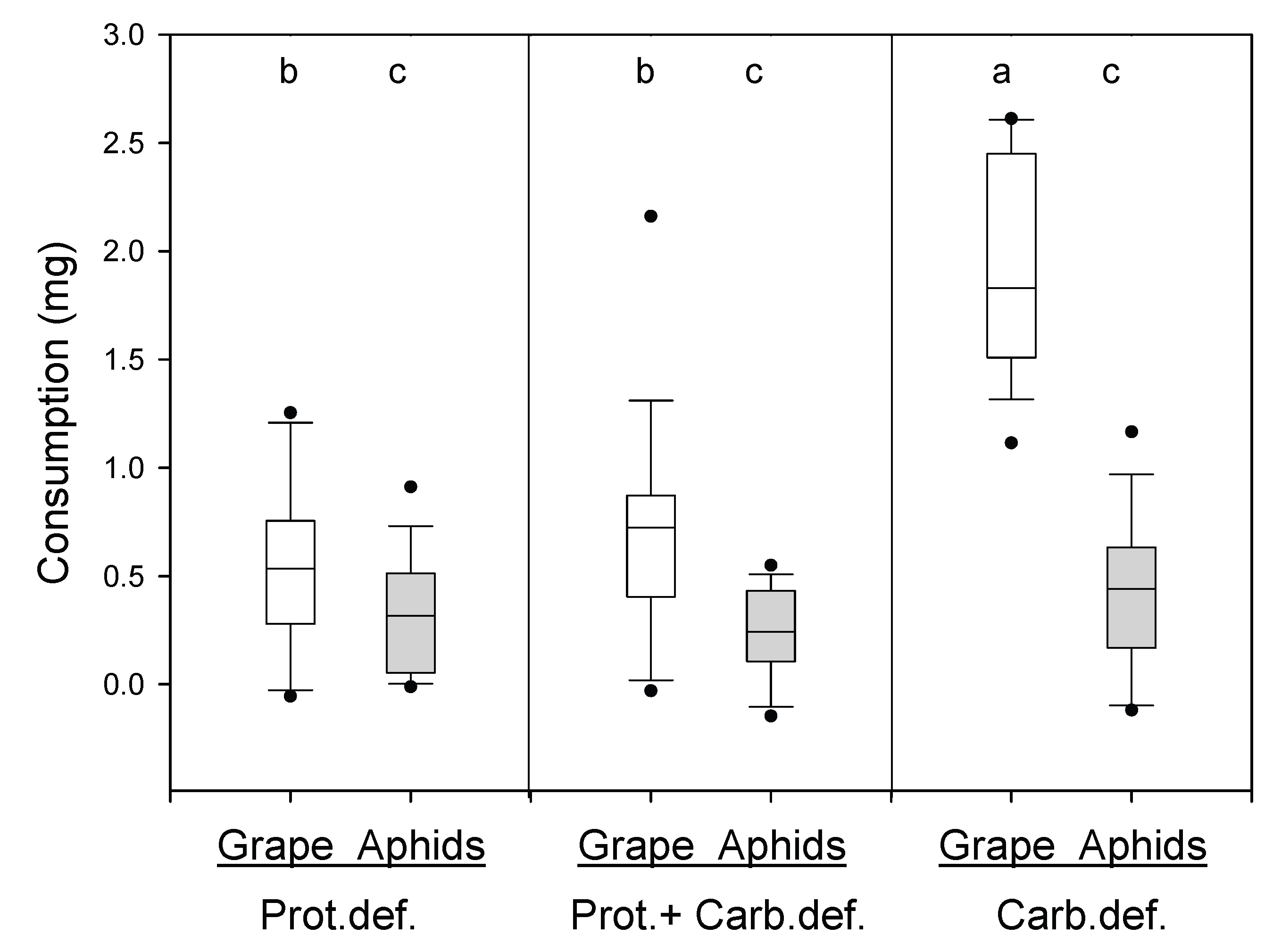
2.2.2. Experiment 2. Does Aphid Consumption Increase in Protein Deficient Harvestmen?
2.3. Statistical Analysis
3. Results
3.1. Experiment 1
3.2. Experiment 2
4. Discussion
5. Implications
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Behmer, S.T. Insect herbivore nutrient regulation. Ann. Rev. Entomol. 2009, 54, 165–187. [Google Scholar] [CrossRef]
- Mayntz, D.; Raubenheimer, D.; Salomon, M.; Toft, S.; Simpson, S.J. Nutrient-specific foraging in invertebrate predators. Science 2005, 307(5706), 111–113. [Google Scholar] [CrossRef] [PubMed]
- Simpson, S.J.; Raubenheimer, D. The nature of nutrition: a unifying framework; Princeton University Press: Princeton, N.J, 2012. [Google Scholar]
- Wilder, S.M. Spider nutrition: an integrative perspective. Adv. Insect Physiol. 2011, 40, 87–136. [Google Scholar] [CrossRef]
- Toft, S. Nutritional aspects of spider feeding. In Spider Ecophysiology; Nentwig, W., Ed.; Springer Verlag: Berlin, 2013; pp. 373–384. [Google Scholar] [CrossRef]
- Mayntz, D.; Toft, S. Nutrient composition of the prey’s diet affects fitness of a generalist predator. Oecologia 2001, 127, 207–213. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, S.M.B.; Bilde, T.; Toft, S. Macronutrient niches and field limitation in a woodland assemblage of harvestmen. J. Anim. Ecol. 2022, 91, 593–603. [Google Scholar] [CrossRef]
- Toft, S.; Cuende, E.; Olesen, A.L.; Mathiesen, A.; Larsen, M.M.; Jensen, K. Food and specific macronutrient limitation in an assemblage of predatory beetles. Oikos 2019, 128, 1467–1477. [Google Scholar] [CrossRef]
- Toft, S.; Pavón-Peláez, C.; Martinez-Villar, M.; Rengifo, L.; Arroyave, A.; Pompozzi, G.; Franco, V.; Albo, M.J. Contrasting patterns of food and macronutrient limitation in the field among co-existing omnivorous carnivores. Ecol. Entomol. 2021, 46, 898–909. [Google Scholar] [CrossRef]
- Christensen, J.; Nielsen, S.M.B.; Toft, S. The three-dimensional macronutrient niche of an invasive generalist predator. Ecol. Entomol. 2020, 45, 644–651. [Google Scholar] [CrossRef]
- Acosta, L.E.; Machado, G. Diet and foraging. In Harvestmen: the biology of Opiliones; Pinto-da-Rocha, R., Machado, G., Giribet, G., Eds.; Harvard University Press: Cambridge, 2007; pp. 309–338. [Google Scholar]
- Nyffeler, M.; Machado, G.; Hartmann, A.; Keiser, C.N. Fungus and fruit consumption by harvestmen and spiders (Opiliones, Araneae): The vegetarian side of two predominantly predaceous arachnid groups. J. Arachnol. 2023, 51, 1–18. [Google Scholar] [CrossRef]
- Jordano, P. Fruits and frugivory. In The ecology of regeneration in plant communities, 3rd ed.; Gallagher, R.S., Ed.; CABI: Wallingford, UK, 2014; pp. 18–61. [Google Scholar]
- Srivastava, P.N. Nutritional physiology. In Aphids, their biology, natural enemies and control; Minks, A.K., Harrewijn, P., Eds.; Elsevier: Amsterdam, 1987; Volume A, pp. 99–121. [Google Scholar]
- Lv, N.; Wang, L.; Sang, W.; Liu, C.-Z.; Qiu, B.-L. Effects of endosymbiont disruption on the nutritional dynamics of the pea aphid Acyrthosiphon pisum. Insects 2018, 9, 161. [Google Scholar] [CrossRef]
- Liu, H.-P.; Yang, Q.-Y.; Liu, J.-X.; Haq, I.U.; Li, Y.; Zhang, Q.-Y.; Attia, K.A.; Abushady, A.M.; Liu, C.-Z.; Lv, N. Host plant mediated effects on Buchnera symbiont: implications for biological characteristics and nutritional metabolism of pea aphids (Acyrthosiphon pisum). Front. Plant Sci. 2023, 14, 1288997. [Google Scholar] [CrossRef]
- Cuff, J.P.; Tercel, M.P.T.G.; Vaughan, I.P.; Drake, L.E.; Wilder, S.M.; Bell, J.R.; Müller, C.T.; Orozco-terWengel, P.; Symondson, W.O.C. Prey nutrient content is associated with the trophic interactions of spiders and their prey selection under field conditions. Oikos 2024a, e10712. [Google Scholar] [CrossRef]
- Cuff, J.P.; Tercel, M.P.T.G.; Vaughan, I.P.; Drake, L.E.; Wilder, S.M.; Bell, J.R.; Müller, C.T.; Orozco-terWengel, P.; Symondson, W.O.C. Prey nutrient content is associated with the trophic interactions of spiders and their prey selection under field conditions [Dataset]. Zenodo 2024b. [Google Scholar] [CrossRef]
- Jensen, K.; Mayntz, D.; Toft, S.; Clissold, F.; Raubenheimer, D.; Simpson, S.J. Optimal foraging for specific nutrients in predatory beetles. Proc. R. Soc. B 2012, 279, 2212–2218. [Google Scholar] [CrossRef]
- Noreika, N.; Madsen, N.E.L.; Jensen, K.; Toft, S. Balancing of lipid, protein, and carbohydrate intake in a predatory beetle following hibernation, and consequences for lipid restoration. J. Insect Physiol. 2016, 88, 1–9. [Google Scholar] [CrossRef]
- Phillipson, J. A contribution to the feeding biology of Mitopus morio (F.) (Phalangida). J. Anim. Ecol. 1960, 29, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Posit team. RStudio: Integrated Development Environment for R. Posit Software, PBC, Boston, MA. 2024. Available online: http://www.posit.co/.
- R Core Team. R: A language and environment for statistical computing; R Foundation for Statistical Computing: Vienna, Austria, 2024; Available online: https://www.R-project.org/.
- Escalante, I.; O’Brien, S.L. Robustness to leg loss in Opiliones: a review and framework considerations for future research. Integr. Comp. Biol. 2024, 64, 1338–1353. [Google Scholar] [CrossRef]
- Elton, C.S. The pattern of animal communities; Methuen & Co, London: London, 1966. [Google Scholar]
- Coll, M.; Guershon, M. Omnivory in terrestrial arthropods: mixing plant and prey diets. Ann. Rev. Entomol. 2002, 47, 267–297. [Google Scholar] [CrossRef]
- Lefchek, J.S.; Whalen, M.A.; Davenport, T.M.; Stone, J.P.; Duffy, J.E. Physiological effects of diet mixing on consumer fitness: a meta-analysis. Ecology 2013, 94, 565–572. [Google Scholar] [CrossRef]
- Marques, R.V.; Sarmento; Lemos, F.; Pedro-Neto, M.; Sabelis, M.W.; Venzon, M.; Pallini, A.; Janssen, A. Active prey mixing as an explanation for polyphagy in predatory arthropods: synergistic dietary effects on egg production despite a behavioural cost. Funct. Ecol. 2015, 29, 1317–1324. [Google Scholar] [CrossRef]
- Kohl, K.D.; Coogan, S.C.P.; Raubenheimer, D. Do wild carnivores forage for prey or for nutrients?: Evidence for nutrient-specific foraging in vertebrate predators. BioEssays 2015, 37, 701–709. [Google Scholar] [CrossRef] [PubMed]
- Bilde, T.; Toft, S. Evaluation of prey for the spider Dicymbium brevisetosum Locket (Araneae: Linyphiidae) in single-species and mixed-species diets. Ekológia (Bratislava) 2000, 19, 9–18. Available online: https://www.european-arachnology.org/esa/wp-content/uploads/2015/08/009-018_Bilde.pdf.
- Symondson, W.O.C.; Sunderland, K.D.; Greenstone, M.H. Can generalist predators be effective biocontrol agents? Ann. Rev. Entomol. 2002, 47, 561–594. [Google Scholar] [CrossRef]
- Dixon, P.L.; Mckinlay, R.G. Aphid predation by harvestmen in potato fields in Scotland. J. Arachnol. 1989, 17, 253–255. Available online: http://www.jstor.org/stable/3705637.
- Madsen, M.; Terkildsen, S.; Toft, S. Microcosm studies on control of aphids by generalist arthropod predators: effects of alternative prey. BioControl 2004, 49, 483–504. [Google Scholar] [CrossRef]
| Ingredients by weight | Macronutrient content by energy |
||||||
| %grasshopper | %casein | %lard | %sucrose | %protein | %lipid | %carb. | |
| Exp. 1 | |||||||
| C | 40.0 | 28.0 | 12.0 | 20.0 | 47.2 | 35.7 | 17.2 |
| CD | 50.0 | 35.0 | 15.0 | 0.0 | 74.0 | 24.5 | 1.5 |
| Exp. 2 | |||||||
| PD | 44.4 | 0.0 | 22.2 | 35.5 | 24.4 | 48.4 | 27.2 |
| CD | 51.2 | 35.8 | 15.4 | 0.0 | 57.4 | 41.3 | 1.2 |
| PCD | 57.7 | 0.0 | 30.2 | 12.1 | 30.4 | 60.2 | 9.4 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




